Abstract
Objective
The objective of this study was to evaluate the effects of dexmedetomidine on respiratory and hemodynamic parameters in patients receiving high-flow nasal cannula therapy.
Methods
This retrospective observational study included 206 adult patients who received dexmedetomidine during high-flow nasal cannula therapy between November 2019 and September 2023. Patients in whom dexmedetomidine was initiated during mechanical ventilation and those with incomplete data were excluded. Dexmedetomidine was administered intravenously at a median dose of 0.2 (0.2–0.3) mcg/kg/h for 3.0 (2.0–5.0) days. Primary outcomes were changes in respiratory rate and the partial pressures of carbon dioxide and oxygen. Secondary outcomes included hemodynamic changes, adverse events, and discontinuation of dexmedetomidine.
Results
Respiratory rate and the partial pressure of carbon dioxide remained stable over 24 h, whereas the partial pressure of oxygen significantly increased at 3 h (p = 0.007). Dexmedetomidine led to significant reductions in systolic blood pressure and heart rate (p < 0.001). Hypotension occurred in 31.6% of patients, and vasopressors were required in 28.2%. Dexmedetomidine was discontinued in 26.7% of patients because of adverse events.
Conclusions
Dexmedetomidine administration was associated with improved oxygenation without compromising respiratory drive in patients receiving high-flow nasal cannula, although careful monitoring is warranted because of potential hemodynamic effects.
Introduction
High-flow nasal cannula (HFNC) has emerged as an important noninvasive respiratory support modality for patients with acute respiratory failure.1,2 By delivering heated, humidified oxygen at high flow rates, HFNC offers several physiological benefits, including improved oxygenation, reduced inspiratory effort, and increased patient comfort.3,4 Additionally, HFNC provides a modest positive end-expiratory pressure effect, promoting alveolar recruitment and optimizing gas exchange in compromised lung regions. 5 Despite these advantages, some patients experience difficulty tolerating HFNC therapy because of discomfort, agitation, or anxiety. 6 Poor compliance can result in HFNC failure, necessitating escalation to more invasive respiratory support modalities, such as noninvasive ventilation (NIV) or invasive mechanical ventilation (IMV). Such an escalation increases healthcare costs and may be associated with increased morbidity and mortality rates.7,8
To address these challenges, various sedation strategies have been proposed to improve HFNC tolerance and preserve spontaneous breathing patterns.9,10 Among these, dexmedetomidine (Dex), a selective α2-adrenergic agonist, has gained attention for its unique pharmacological profile. 11 It is known to provide effective anxiolysis and mild sedation with minimal suppression of respiratory drive, making it theoretically suitable for nonintubated patients requiring respiratory support.10,12 Dex is well established in patients receiving mechanical ventilation, where it has been shown to reduce the incidence of delirium compared with other sedatives.13,14 However, its use in nonintubated patients receiving HFNC therapy remains poorly characterized. Although the potential benefits of Dex in this context include improved HFNC compliance and tolerability,15,16 concerns persist regarding its hemodynamic effects, particularly its potential to induce hypotension and bradycardia. 17
Existing literature has predominantly focused on the use of Dex in critically ill patients undergoing invasive ventilation. In contrast, limited data are available on its effects in patients receiving HFNC therapy. A better understanding of how Dex influences respiratory function, oxygenation, and hemodynamic stability in this population is essential for optimizing clinical protocols. Therefore, this study aimed to evaluate the effects of Dex on respiratory and hemodynamic parameters in patients receiving HFNC, with particular focus on changes in gas exchange efficiency and cardiovascular responses to its administration.
Methods
Study design and setting
This retrospective observational study was conducted at a tertiary care center in Daejeon, Republic of Korea, from November 2019 to September 2023. The study protocol was reviewed and approved by the Institutional Review Board of Chungnam National University Hospital (IRB Number: 2021-07-041). Given the retrospective design, the requirement for written informed consent was waived. All research procedures adhered to the ethical principles outlined in the Declaration of Helsinki (1975, as revised in 2024).
All patient data were deidentified prior to analysis in accordance with institutional privacy guidelines. As this was a retrospective study, the sample size was determined by the number of patients who received Dex during HFNC therapy within the study period. No formal calculation of the sample size was performed. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 18
Study population
The inclusion criteria were as follows: (a) age ≥19 years; (b) receipt of HFNC therapy for acute respiratory failure; and (c) administration of Dex during HFNC treatment. Patients were excluded if they received Dex during IMV, had incomplete medical records, or did not receive HFNC therapy (Figure 1). HFNC therapy was delivered using the Optiflow™ Airvo™ 2 system (Fisher & Paykel Healthcare; Auckland, New Zealand).

Study flowchart. HFNC: high-flow nasal cannula; MV: mechanical ventilation.
Dex administration
Dex (Pfizer Inc.; New York, NY, USA) was administered via continuous intravenous infusion according to institutional sedation protocols. The indication for Dex administration in patients receiving HFNC was agitation, restlessness, or discomfort that interfered with tolerance of HFNC therapy. The initial infusion rate, maintenance dose adjustments, and total duration were determined at the discretion of the attending physician based on each patient’s sedation requirements. Sedation was titrated to achieve a target Richmond Agitation–Sedation Scale (RASS) score between −2 and +1, aiming to ensure patient comfort without inducing respiratory depression or hemodynamic instability.
As part of routine clinical monitoring at our center for patients receiving HFNC with sedation, arterial blood gas analyses (ABGAs) were performed at baseline and at 3 and 24 h after Dex administration to evaluate changes in gas exchange.
Data collection
To minimize potential sources of bias, we included all consecutive patients who met the eligibility criteria throughout the study period. We extracted data using standardized electronic medical records and defined variables prior to analysis. Multivariable analyses were used to adjust for confounding factors. The data included demographic characteristics, body mass index, comorbidities, Acute Physiology and Chronic Health Evaluation II (APACHE II) scores, Sequential Organ Failure Assessment (SOFA) scores, Clinical Frailty Scale scores, and HFNC indications. Respiratory parameters, including respiratory rate (RR), oxygen saturation, and arterial blood gas analysis values such as pH, partial pressure of carbon dioxide (pCO2), partial pressure of oxygen (pO2), and the partial pressure of arterial oxygen (PaO2)/fraction of inspired oxygen (FiO2) (P/F) ratio, were recorded at baseline, 3 h, and 24 h after Dex initiation. Hemodynamic parameters, including systolic blood pressure (SBP), diastolic blood pressure (DBP), and heart rate (HR), were assessed at the same time points. Adverse events, including hypotension (SBP <90 mmHg or mean arterial pressure (MAP) <65 mmHg), bradycardia (HR < 50 bpm), need for vasopressors, and Dex discontinuation due to adverse effects, were also analyzed.
The data used in this study were originally collected from the Patient-Centered Clinical Research Coordinating Center research (Grant Number: HC19C0226) funded by the Ministry of Health & Welfare, Republic of Korea.
Statistical analysis
Continuous variables were summarized as mean ± SD or median with interquartile range (IQR), depending on normality as assessed using the Shapiro–Wilk test. Categorical variables were presented as frequencies and percentages. To evaluate changes before and after Dex administration, paired t-tests were used for normally distributed continuous variables, and Wilcoxon signed-rank tests were used for non-normally distributed variables. Categorical variables were compared using chi-squared or Fisher’s exact tests, as appropriate. A p-value of <0.05 was considered statistically significant. Missing data were not imputed, and all analyses were conducted using complete-case analysis. The extent of missingness was minimal and was deemed unlikely to affect the results.
All analyses were performed using Statistical Package for Social Sciences (SPSS) for Windows, version 25.0 (IBM Corp.; Armonk, NY, USA) and GraphPad Prism, version 9.0 (GraphPad Software; San Diego, CA, USA).
Results
Baseline characteristics of the study population
Table 1 presents the baseline characteristics of the study population. The mean patient age was 69.5 ± 14.5 years, and 67.5% of the patients were men. Disease severity was assessed using the APACHE II score (mean, 22.8 ± 6.9), whereas frailty was evaluated using the Clinical Frailty Scale score (mean, 4.4 ± 1.4). HFNC therapy was primarily used for postextubation support (69.4%), followed by acute hypoxemic respiratory failure (26.7%) and mild hypercapnic respiratory failure with preserved consciousness (3.9%). Of the 143 patients who received HFNC as postextubation respiratory support, four patients (2.8%) required reintubation during their intensive care unit (ICU) stay.
Baseline characteristics.

APACHE: Acute Physiology and Chronic Health Evaluation, COPD: chronic obstructive pulmonary disease; HFNC: high-flow nasal cannula; SOFA: Sequential Organ Failure Assessment.
Data are presented as mean ± SD; bdata are presented as n (%).
Dex administration and patient outcomes
Table 2 summarizes Dex administration and patient outcomes. The median ICU length of stay was 13.5 (8.0–19.3) days, whereas the median hospital length of stay was 33.0 (18.8–63.0) days. In-hospital mortality was observed in 52 patients (25.2%). Prior use of mechanical ventilation was documented in 152 patients (73.8%), with a median ventilation duration of 7.5 (4.0–12.8) days. The median initial Dex infusion rate was 0.2 (0.2–0.3) mcg/kg/h, and the median infusion duration was 3.0 (2.0–5.0) days. Additionally, 45 patients (21.8%) experienced delirium prior to Dex administration.
Patients outcomes.
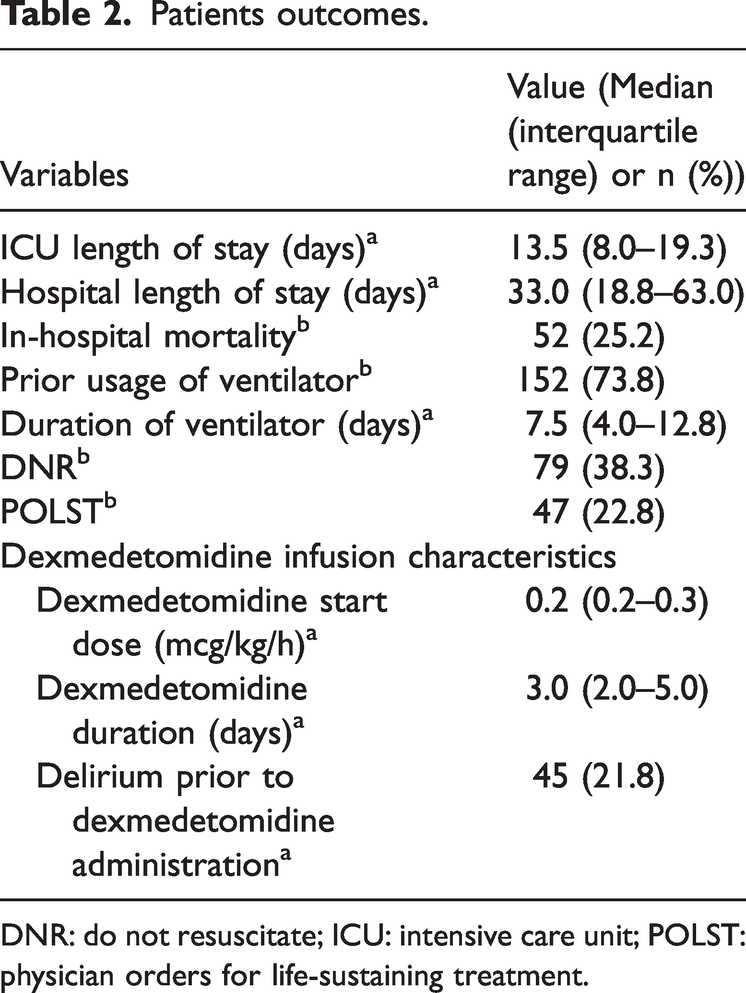
DNR: do not resuscitate; ICU: intensive care unit; POLST: physician orders for life-sustaining treatment.
Respiratory and hemodynamic changes
Table 3 and Figure 2 illustrate the respiratory and hemodynamic changes before and after Dex administration. SBP significantly decreased from 132.9 ± 23.4 mmHg to 127.0 ± 23.2 mmHg at 3 h (p < 0.001) and remained lower at 1 day (p = 0.002). Similarly, HR significantly decreased from 96.6 ± 20.9 bpm to 92.7 ± 18.8 bpm at 3 h (p = 0.001) and remained lower at 1 day (p < 0.001). Moreover, RR exhibited a slight but significant increase at 3 h (p = 0.021); however, this effect was not maintained at 1 day (p = 0.172).
Respiratory and hemodynamic changes before and after dexmedetomidine administration.

BP: blood pressure; Dex: dexmedetomidine; P/F ratio: partial pressure of arterial oxygen/fraction of inspired oxygen ratio; pCO2: partial pressure of carbon dioxide; pO2: partial pressure of oxygen; SpO2: peripheral capillary oxygen saturation.
Data are presented as mean ± SD.

Changes in respiratory and hemodynamic parameters before and after dexmedetomidine (Dex) administration. (a) Hemodynamic changes, including systolic blood pressure (SBP), diastolic blood pressure (DBP), heart rate, and respiratory rate, were measured before Dex administration, 3 h after initiation, and 1 day after administration and (b) arterial blood gas analysis parameters, including partial pressure of carbon dioxide (pCO2), partial pressure of oxygen (pO2), and the partial pressure of arterial oxygen (PaO2)/fraction of inspired oxygen (FiO2) (P/F) ratio, were measured at the same time points. Error bars represent SDs.
Regarding oxygenation and gas exchange, pO2 levels increased at 3 h (p = 0.007) but did not remain elevated at 1 day (p = 0.118); pCO2 remained stable, with no significant changes after Dex administration (p = 0.756). Importantly, the P/F ratio improved over time, increasing from 232.3 ± 99.6 to 243.9 ± 91.3 at 3 h (not significant, p = 0.388) and to 247.5 ± 105.3 at 1 day (p = 0.042).
Adverse events and safety outcomes
Table 4 summarizes hemodynamic adverse events associated with Dex administration. Hypotension, defined as SBP <90 mmHg or MAP <65 mmHg, occurred in 65 patients (31.6%). Bradycardia, defined as HR <50 bpm, occurred in 24 patients (11.7%). In addition, 58 patients (28.2%) required vasopressor support following Dex administration. Dex was discontinued in 55 patients (26.7%) due to adverse events.
Adverse events following dexmedetomidine administration.
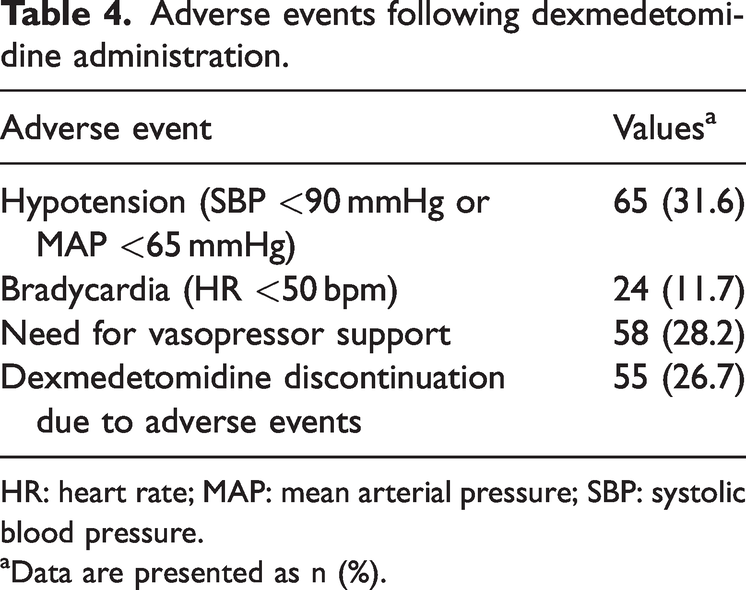
HR: heart rate; MAP: mean arterial pressure; SBP: systolic blood pressure.
Data are presented as n (%).
Discussion
In this retrospective study, we evaluated the effects of Dex on respiratory and hemodynamic parameters in patients receiving HFNC therapy. Our findings demonstrated that RR and pCO2 levels remained stable following Dex administration, whereas the P/F ratio showed a modest but statistically significant improvement. However, Dex administration was also associated with hemodynamic changes, including hypotension and bradycardia, leading to treatment discontinuation in a subset of patients. These findings provide preliminary insights into the application of Dex in spontaneously breathing patients receiving HFNC and highlight the importance of carefully weighing potential benefits against hemodynamic risks in this population.
Previous research has primarily focused on Dex use in mechanically ventilated patients, demonstrating its effectiveness in providing sedation and preserving respiratory drive. 19 However, data on its use in nonintubated patients receiving HFNC therapy remain limited. Our findings align with existing evidence suggesting that Dex does not contribute to respiratory depression, making it a viable sedation option for patients requiring noninvasive respiratory support.9,20
In the present study, the observed improvement in the P/F ratio following Dex administration suggests a potential association with enhanced oxygenation; however, this finding must be interpreted cautiously, given the retrospective design of the study and potential confounding factors. The concurrent use of HFNC, variations in disease etiology (e.g. pneumonia, acute respiratory distress syndrome (ARDS), and postextubation status), and spontaneous clinical improvement may have contributed to changes in oxygenation status. The anxiolytic and sympatholytic properties of Dex may reduce respiratory effort and relieve dyspnea-related distress, potentially improving patient compliance with HFNC therapy. By promoting a calm yet cooperative state, Dex may facilitate more stable breathing patterns that support effective gas exchange without compromising respiratory drive. Our study demonstrated a statistically significant increase in the P/F ratio from 232.3 ± 99.6 to 247.5 ± 105.3 (p = 0.042). Similar improvements have been reported in other clinical contexts. Simioli et al. 9 found that patients with moderate-to-severe coronavirus disease 2019 (COVID-19)–related ARDS receiving Dex had a significantly higher P/F ratio than controls (125 mmHg vs. 94 mmHg, p < 0.0001). Lee et al. 21 observed a significant increase in the P/F ratio with Dex administration in patients with moderate chronic obstructive pulmonary disease (COPD) undergoing lung cancer surgery (27.9 ± 5.8 vs. 22.5 ± 8.4 kPa, p < 0.05). Similarly, Uusalo et al. 22 reported a significant improvement in the P/F ratio (p < 0.001) in patients with COVID-19 following Dex administration. Although these findings collectively suggest a beneficial effect of Dex on oxygenation, further prospective studies are warranted to elucidate the underlying mechanisms and confirm this association in controlled settings.
Despite its potential respiratory benefits, the hemodynamic risks associated with Dex administration must be carefully considered. In our study, 31.6% of patients experienced hypotension, and 28.2% required vasopressor support. Additionally, bradycardia occurred in 11.7% of cases and contributed to Dex discontinuation in 26.7% of patients. Although these are well-documented side effects of Dex, their occurrence in spontaneously breathing patients receiving HFNC therapy underscores the need for heightened vigilance in this clinical context. These findings are consistent with previous studies documenting the dose-dependent hemodynamic effects of Dex in critically ill populations. 23 Although our study used a median infusion rate of 0.2 (0.2–0.3) μg/kg/h, the results underscore the importance of balancing sedation efficacy with cardiovascular stability in nonintubated patients. Individualized titration strategies, such as initiating Dex at lower doses and closely monitoring hemodynamics, may help optimize safety and tolerability in this population. Future studies should explore whether lower initial doses could provide comparable benefits and minimize cardiovascular complications in patients receiving HFNC.
Although our study did not include a control group for direct comparison, previous literature suggests that Dex may help prevent progression to IMV. For example, one study reported a 62% reduction in endotracheal intubation rates (odds ratio, 0.38; p < 0.008) in Dex-treated patients compared to those in controls. 9 This suggests that Dex may play a role in reducing the need for more invasive forms of respiratory support. Dex administration is also associated with a reduction in NIV duration; a decrease from 13 (10–17) days in the control group to 10 (7–16) days in the Dex group (p < 0.02) has been reported. 9 Prolonged NIV use is associated with complications such as mask-related pressure ulcers, ventilator-associated pneumonia, and delayed mobilization. Therefore, improving patient tolerance and spontaneous ventilation with Dex may facilitate earlier weaning from respiratory support, potentially accelerating recovery and reducing complications. However, the effects of Dex may vary according to underlying patient characteristics. Patients with preexisting cardiovascular disease may be more susceptible to Dex-induced hypotension and bradycardia, potentially limiting its use in this population. In contrast, younger or hemodynamically stable patients may tolerate Dex better and benefit from its sedative properties without excessive cardiovascular instability. Therefore, future studies should focus on identifying patient populations that derive the greatest benefit from Dex and minimize adverse effects. This approach could help refine patient selection criteria and optimize Dex use in HFNC therapy.
Our study had several limitations. First, as a single-center study, our findings may have limited generalizability to other clinical settings. Second, the retrospective design may have introduced selection bias, as treatment decisions, including Dex administration, were made at the physicians’ discretion rather than according to standardized protocols. Third, the absence of a control group that received HFNC without Dex prevents a direct comparative analysis of its true efficacy and safety profile. These limitations underscore the need for randomized controlled trials to determine optimal Dex dosing strategies and assess whether the observed improvements in oxygenation translate into meaningful clinical outcomes for patients receiving HFNC.
Conclusions
Our results suggest that Dex administration during HFNC therapy may be associated with improved oxygenation, as reflected by the observed improvement in the P/F ratio. However, associated hemodynamic effects, particularly hypotension and bradycardia, indicate the need for vigilant monitoring during treatment. Given the limitations of our retrospective, single-center study without a control group, prospective studies are necessary to better define optimal Dex dosing regimens and clarify its potential role in supporting HFNC tolerance in spontaneously breathing patients.
Footnotes
Acknowledgments
Language editing and formatting of the manuscript was provided by Editage, Cactus Communications.
Authors’ contributions
MJK conceptualized the study, performed the investigation, curated and analyzed the data, and contributed to the writing of the manuscript. SYK conceptualized the study, performed the investigation, curated and analyzed the data, and contributed to the writing of the manuscript. DHK conceptualized the study, analyzed the data, and contributed to the writing of the manuscript. GRH conceptualized the study, analyzed the data, and contributed to the writing of the manuscript. SIL conceptualized the study, performed the investigation, curated and analyzed the data, acquired funding, and contributed to the writing of the manuscript. MJK, SYK, and SIL had full access to all the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis. All authors reviewed the results and approved the final version of the manuscript.
Consent for publication
Not applicable.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare that they have no competing interests.
Ethical approval and consent statements
The study protocol was approved by the Institutional Review Board of Chungnam National University Hospital (approval number: 2021-07-041). Written informed consent was waived by the Institutional Review Board of Chungnam National University Hospital due to the retrospective nature of the study. This study was conducted in accordance with the Declaration of Helsinki.
Funding
This research was supported by a grant of Patient-Centered Clinical Research Coordinating Center (PACEN, ![]() ) funded by the Ministry of Health & Welfare, Republic of Korea (grant number: RS-2025-02233056). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
) funded by the Ministry of Health & Welfare, Republic of Korea (grant number: RS-2025-02233056). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
