Abstract
Objective
To investigate the effect of preoperative glycopyrrolate and penehyclidine hydrochloride on postoperative nausea and vomiting in patients undergoing laparoscopic cholecystectomy.
Methods
This prospective, randomized, double-blind, controlled trial enrolled 120 patients who underwent laparoscopic cholecystectomy. Patients were randomly assigned to the glycopyrrolate group, penehyclidine hydrochloride group, or normal saline group. Postoperative nausea and vomiting, hemodynamic parameters, anesthetic consumption, duration of postanesthesia care unit stay, and other perioperative adverse events were recorded.
Results
The incidence of postoperative nausea and vomiting within 1 h in the penehyclidine hydrochloride group was significantly lower than that in the normal saline and glycopyrrolate groups. The heart rate at immediately prior to anesthesia induction, 1 min after endotracheal intubation, the start of surgery, 2 min after pneumoperitoneum, and cholecyst dissection and mean arterial pressure at 1 min after endotracheal intubation and the start of surgery in the normal saline group were significantly lower than those in the glycopyrrolate group. The heart rate at immediately prior to anesthesia induction, 1 min after endotracheal intubation, the start of surgery, and 2 min after pneumoperitoneum and mean arterial pressure at 1 min after endotracheal intubation in the penehyclidine hydrochloride group were markedly lower than those in the glycopyrrolate group. The incidence of bradycardia in the normal saline and penehyclidine hydrochloride groups was significantly higher than that in the glycopyrrolate group.
Conclusions
Penehyclidine hydrochloride is more effective than glycopyrrolate in preventing postoperative nausea and vomiting in patients undergoing laparoscopic cholecystectomy, particularly in the early postoperative period.
Keywords
Introduction
Laparoscopic cholecystectomy (LC) is a common procedure for treating symptomatic chronic cholecystitis, gallbladder stones, and other gallbladder disorders. This procedure offers advantages such as reduced surgical trauma and bleeding as well as quicker recovery.1,2 However, residual anesthetic medication, irritation from pneumoperitoneum carbon dioxide gas, and manipulation of the gastrointestinal tract can readily result in postoperative nausea and vomiting (PONV), adversely affecting the patient’s perioperative comfort and postoperative recovery. Anticholinergics are frequently administered during anesthesia and can also help prevent PONV, although their effectiveness may vary among different drug types. 3 Newer anticholinergics, such as glycopyrrolate and penehyclidine hydrochloride, significantly inhibit glandular secretion, provide a mild gastrointestinal antispasmodic effect, and cause fewer circulatory adverse effects compared with the traditional drug atropine. 4 There is a lack of studies comparing the use of glycopyrrolate and penehyclidine hydrochloride in LC.
Therefore, this study aimed to compare the effects of glycopyrrolate and penehyclidine hydrochloride on PONV and other perioperative adverse effects in patients undergoing LC, thereby providing a scientific foundation for selecting appropriate clinical anesthesia medications, optimizing the anesthetic management of LC, and improving the perioperative comfort and safety of patients.
Patients and methods
In this prospective, double-blind, randomized controlled trial, 120 patients who had undergone LC at the First Affiliated Hospital of Yangtze University were enrolled. The study was approved by the Medical Ethics Committee of the First Affiliated Hospital of Yangtze University of Hubei Province, China, on 31 December 2024 (YJ202431), and written informed consent was obtained from the patients. This manuscript adheres to the Consolidated Standards of Reporting Trials (CONSORT) guidelines. 5 The study was conducted in accordance with the principles of the Declaration of Helsinki of 1975, as revised in 2024, and registered retrospectively at the Chinese Clinical Trial Registry (ChiCTR2500107519).
Patient inclusion criteria
Patients who underwent LC at the First Affiliated Hospital of Yangtze University from January to May 2025, aged 18–64 years, with a body mass index (BMI) of 18–30 kg/m2, and an American Society of Anesthesiologists (ASA) classification of I–II were selected for this study. The exclusion criteria included patients with a history of motion sickness and PONV, allergies to the study drugs, mental unfitness or uncooperativeness, severe cardiovascular or cerebrovascular diseases, glaucoma, and pregnancy or lactation. Patients who met any of the following criteria were excluded from the study: surgical duration >2 h, additional intraoperative procedures, significant hemodynamic instability during surgery due to intraoperative bleeding or other reasons, incomplete case report forms, or failure to attend follow-up consultations.
Randomization and grouping
Eligible patients were randomly assigned to the glycopyrrolate group (group G), penehyclidine hydrochloride group (group P), or normal saline group (group C) in a 1:1:1 ratio as per a computer-generated random sequence.
Group numbers were contained in an opaque, sealed envelope. The anesthesia nurse, who was not involved in the subsequent trial process, opened the envelope prior to anesthesia induction and prepared the appropriate medications, including glycopyrrolate, penehyclidine hydrochloride, and saline; all solutions were colorless and transparent, with no distinguishable differences in appearance. Participants and investigators were blinded to group allocation. Data were collected and recorded by the investigators.
Anesthetic procedures
All patients fasted for 8 h and avoided fluid intake for 3 h prior to receiving general anesthesia. Upon arrival in the operating room, venous access was established in the left upper limb, and vital signs were routinely monitored, including electrocardiogram, pulse oxygen saturation, noninvasive blood pressure cuff readings, heart rate (HR), body temperature, and Narcotrend index. The patients inhaled oxygen via an oxygen mask at a flow rate of 4–6 L/min. Arterial blood pressure was monitored by puncturing the left radial artery under local anesthesia (the Allen test was necessary before the puncture). At 10 min prior to the induction of general anesthesia, patients in group G received 4 μg/kg glycopyrrolate intravenously (diluted to 5 mL in saline), those in group P received 10 μg/kg penehyclidine hydrochloride intravenously (diluted to 5 mL in saline), and those in group C received an equivalent volume of saline intravenously. Subsequently, an intravenous injection of 1 μg/kg dexmedetomidine (DEX), diluted in 100 mL of normal saline within 10 min, was administered. The anesthesia induction regimen was the same for the three groups; after adequate preoxygenation, a slow static injection of sufentanil (0.5 μg/kg), propofol (1–1.5 mg/kg), dexamethasone (10 mg), rocuronium (0.6 mg/kg), and flurbiprofenate (50 mg) was sequentially administered. Endotracheal intubation was performed 3 min after anesthesia induction, followed by mechanical ventilation (oxygen flow, 2 L/min and fraction of inspired oxygen, 60%) with a tidal volume (VT) of 6–8 mL/kg, a respiratory rate (RR) of 12–16 breaths per min, and an inspiratory: expiratory ratio of 1:2. Respiratory parameters were adjusted based on end-tidal carbon dioxide to maintain levels between 35 and 45 mmHg. Maintenance was achieved with continuous infusion of propofol (2–7 mg/kg/h) and remifentanil (0.2–0.5 µg/kg/min) to ensure that the Narcotrend index was maintained between 36 and 46. Additional doses of rocuronium were administered intraoperatively as needed and discontinued 30 min before the end of the procedure. Ephedrine (6–12 mg) was injected when the mean arterial pressure (MAP) was <65 mmHg or dropped to 30% from the baseline level, and atropine (0.5 mg) was administered when the HR dropped to 45 beats per min. Propofol was discontinued at the time of surgical incision closure, and remifentanil was stopped at the end of the surgery. Each incision was infiltrated layer by layer with 10 mL of 0.375% ropivacaine. The tracheal tube was removed once the patients were conscious and had regained spontaneous respiration (VT > 6 mL/kg, RR > 10 breaths/min). The patients were then transferred to the postanesthesia care unit (PACU) for monitoring and subsequently moved to the ward upon meeting the PACU discharge criteria (Aldrete score ≥ 9, stable vital signs, no bleeding, no severe pain/PONV, or other complications).
Study assessments
The incidence of PONV was documented at 0–1, 1–24, and <24 h postoperatively. Palonosetron, 5 μg/kg (upper limit 0.25 mg), was administered in case of more than two consecutive episodes of dry heaves or vomiting, or upon request. The MAP and HR were recorded at the following time points: immediately before the infusion of glycopyrrolate/penehyclidine hydrochloride/saline (T0), 5 min after DEX injection (T1), immediately prior to anesthesia induction (T2), 1 min after endotracheal intubation (T3), at the start of surgery (T4), 2 min after pneumoperitoneum (T5), during cholecyst dissection (T6), and 1 min after extubation (T7). The incidence of intraoperative hypotension (MAP < 65 mmHg or a drop of 30% from the baseline level) and bradycardia (HR < 50 beats/min), along with the frequency of administration of vasoactive drugs (ephedrine or atropine) was recorded. Additionally, the duration of PACU stay, incidence of dry mouth, postoperative delirium (using the confusion assessment method), and time to first flatus were noted.
Statistical analyses
According to the pretest results, the 24-h postoperative incidence rates in groups G, P, and C were 11%, 33%, and 44%, respectively. Assuming an α value of 0.05 and a 1-β value of 0.8, Power Analysis and Sample Size (PASS, version 2021) software was used to calculate the sample size, accounting for a 10% dropout rate. In total, 120 patients were included, with a ratio of 1:1:1, resulting in 40 patients in each group.
Data were analyzed using the Statistical Package for Social Sciences (SPSS, version 27.0) statistical software. Mean ± SD was employed for the analysis of normally distributed continuous data. One-way analysis of variance (ANOVA) was used for among-group comparisons, repeated-measures ANOVA for within-group comparisons, and the Bonferroni method for two-by-two comparisons. Non-normally distributed continuous variables were expressed as median (interquartile range, Q1–Q3) values and obtained using nonparametric tests. Count data were expressed as cases or percentages, and the χ2-test with Bonferroni correction was used for group comparisons. A p-value of <0.05 was considered statistically significant.
Results
A flow diagram of the study is presented in Figure 1. Initially, 120 patients were included in this study. One patient from group G was excluded because of modifications in the surgical procedure during the operation. Four patients from group P were excluded because of a temporary change in the surgical procedure (n = 1), surgical duration exceeding 2 h (n = 1), and incomplete data recording (n = 2). One patient from group C was excluded as the surgical duration exceeded 2 h. Finally, 114 patients were enrolled, and all procedures were completed in the three groups. There were no differences in the baseline characteristics (sex, age, BMI, ASA, and nonsmoking status) among the three groups (Table 1).

Consolidated Standards of Reporting Trials (CONSORT) diagram.
Comparison of baseline characteristics among the three groups.

Data are presented as mean ± SD or number (%).
BMI: body mass index; ASA: American Society of Anesthesiologists; Group C: normal saline group; Group G: glycopyrrolate group; Group P: penehyclidine hydrochloride group.
There were no significant differences among groups G, P, and C in terms of the intraoperative variables, including intraoperative medication consumption, surgery duration, and infusion volume (Table 2).
Comparison of intraoperative variables among the three groups.

Data are expressed as the mean ± SD or median (interquartile range).
Group C: normal saline group; Group G: glycopyrrolate group; Group P: penehyclidine hydrochloride group.
Figure 2 presents the results of hemodynamic analysis. There were statistically significant differences in the HR among the three groups at various time points, with a significant group-by-time interaction. Specifically, the HR at T2–T6 and MAP at T3–T4 in group C were significantly lower than those in group G (both p < 0.05). The HR at T2–T5 and MAP at T3 in group P were significantly lower than those in group G (both p < 0.05). However, no significant differences were observed in the MAP and HR at T0 among the three groups. The changes in HR and MAP in the patients in group G were relatively lower than those in groups P and C.
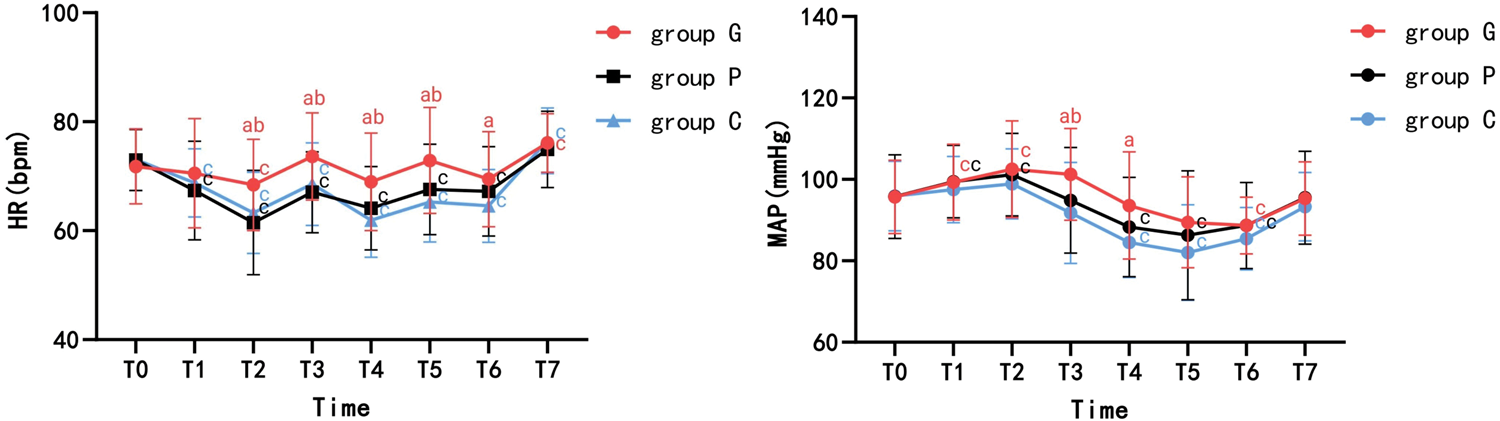
(a) Adjusted p < 0.05 compared with C. (b) Adjusted p < 0.05 compared with P. (c) Adjusted p < 0.05 compared with T0. p-values were adjusted for multiple comparisons based on the Bonferroni method. T0: immediately before the infusion of glycopyrrolate/penehyclidine hydrochloride/saline; T1: 5 min after dexmedetomidine injection; T2: immediately prior to anesthesia induction; T3: 1 min after endotracheal intubation; T4: at the start of surgery; T5: 2 min after pneumoperitoneum; T6: during cholecyst dissection; T7: 1 min after extubation; Group C: normal saline group; Group G: glycopyrrolate group; Group P: penehyclidine hydrochloride group; MAP: mean arterial pressure; HR: heart rate.
Data on intraoperative hypotension, bradycardia, and atropine use are shown in Table 3. The incidence of bradycardia in group G was significantly lower than those in groups C and P (p < 0.05). However, there were no statistically significant differences in the incidence of intraoperative hypotension or the rate of atropine use among the three groups.
Comparison of atropine, bradycardia, and hypotension across the three groups.
Data are presented as number (%).
p-values of <0.05 are indicated in bold.
Adjusted p < 0.05 compared with group C.
Adjusted p < 0.05 compared with group P.
p-values were adjusted for multiple comparisons based on the Bonferroni method.
Group C: normal saline group; Group G: glycopyrrolate group; Group P: penehyclidine hydrochloride group.
Figure 3 presents the incidence of PONV. Group P exhibited significantly lower incidence of PONV than groups C (2.8% vs. 23.1%; RR, 0.12; 95% confidence interval (CI): 0.016–0.904; adjusted p = 0.01) and G (2.8% vs. 25.6%; RR, 0.108; 95% CI: 0.015–0.805; adjusted p = 0.005). There was no statistically significant difference between groups G and C (25.6% vs. 23.1%; RR, 0.111; 95% CI: 0.508–2.433; adjusted p = 0.792). However, the differences in the incidence of PONV among the three groups at 1–24 h and within 24 h were not statistically significant.

(a) Adjusted p < 0.05 compared with C. (b) Adjusted p < 0.05 compared with G. p-values were adjusted for multiple comparisons based on the Bonferroni method. Group C: normal saline group; Group G: glycopyrrolate group; Group P: penehyclidine hydrochloride group.
Table 4 presents recovery after anesthesia. None of the patients in any group experienced delirium within 24 h following surgery. Compared with group C, the incidence of dry mouth within 24 h after operation was significantly higher in groups G and P (p < 0.05). No significant differences were observed among the groups in terms of the duration of PACU stay or the time to first flatus within 24 h.
Comparison of recovery following anesthesia in the three groups.

Data are presented as median (interquartile range) values or number (%).
p-values of <0.05 are indicated in bold
Adjusted p < 0.05 compared with group C.
p-values were adjusted for multiple comparisons based on the Bonferroni method.
Group C, normal saline group; Group G, glycopyrrolate group; Group P, penehyclidine hydrochloride group; PACU: postanesthesia care unit.
Discussion
Recently, with the popularization of and emphasis on the concept of clinical comfort medicine, LC has become widely used in clinical practice owing to its minimally invasive approach, offering advantages such as reduced postoperative pain and bleeding as well as quicker recovery than that with conventional open surgery. However, the estimated 70%–80% 6 incidence of PONV following LC poses a significant clinical challenge, with the use of artificial carbon dioxide (CO2) pneumoperitoneum believed to be the primary contributing factor. 7 Elevated CO2 pneumoperitoneal pressure, typically maintained at 12–15 mmHg, increases the intra-abdominal pressure, resulting in compression of the gastrointestinal tract and abdominal nerve plexus, while the chemosensory pull and stimulatory effects of CO2 activate the vagal reflex pathway. Moreover, residual intraoperative and postoperative CO2 can raise the partial pressure of arterial carbon dioxide during surgery, thereby increasing the risk of PONV by dilating intracranial vessels, enhancing cerebral blood flow and increasing intracranial pressure.
PONV, the second most common clinical postoperative complication, can occur peripherally, centrally, or both. It involves direct effects on the vomiting reflex system and afferent inputs from the vagus, visceral, and trigeminal nerves. 8 PONV not only increases patients’ perioperative gastrointestinal discomfort but may also lead to various complications, such as dehydration, electrolyte imbalance, surgical incision dehiscence, and reflux aspiration. In severe cases, it may prolong hospital stay, increase treatment costs, and significantly impair the patients’ quality of life.
PONV occurrence involves a multireceptor mechanism, with anticholinergic medications offering some preventive effects by blocking muscarinic cholinergic receptors (M1/M3). 9 Penehyclidine hydrochloride and glycopyrrolate are anticholinergic medications. Glycopyrrolate, a quaternary ammonium anticholinergic, predominantly acts on peripheral M1/M3 receptors and demonstrates a strong and long-lasting peripheral anticholinergic effect. Penehyclidine hydrochloride, owing to its liposolubility, can cross the blood–brain barrier and exert central and peripheral effects, with a mean elimination half-life of 10.35 h. These pharmacological differences can result in variation in prophylactic effectiveness of the two drugs against PONV. The findings of this study showed that the incidence of PONV in group P within 1 h postoperatively, which received penehyclidine hydrochloride, was significantly lower than those in groups C and G (p = 0.018), indicating the superior immediate effect of this treatment in preventing PONV during early postoperative period following LC. However, these findings are inconsistent with those of Ure et al., 10 who reported that glycopyrrolate pretreatment reduced the incidence of PONV, likely because the quaternary ammonium group in glycopyrrolate’s molecular structure limits its passage efficiency across the blood–brain barrier and weakly affects the M receptor in the vomiting center. Moreover, the inconsistencies in findings may be attributable to the relatively small sample size in the study, which could limit the statistical efficacy. Differences in baseline patient characteristics, anesthesia protocols, and types of surgeries may also influence the comparability of results.
DEX, a highly selective α2 adrenergic receptor agonist, exhibits sedative and analgesic effects and inhibits central nervous system inflammation, effectively preventing PONV. 11 However, its antisympathetic effect intensifies and disrupts the autonomic balance, resulting in perianesthesia hypotension and bradycardia. 12 The findings of this study revealed that bradycardia occurred in 12 (30.8%), 3 (7.7%), and 15 (41.7%) patients from groups C, G, and P, respectively, representing statistically significant differences (p = 0.003). Group G not only reported the lowest incidence of bradycardia but also exhibited smoother variations in HR and blood pressure compared with the other two groups. This suggests that preoperative use of glycopyrrolate reduces the vagal tone of DEX, exerts a lower impact on HR, and helps maintain stable circulatory function. This may be due to glycopyrrolate’s selectivity for M1 and M3 receptors, which is 3–5 times greater than that for M2 receptors, with a weak effect on cardiac M2 receptors. Moreover, the duration of its antivagal action is comparable to that of the antisympathetic action of DEX. 13 In contrast, penehyclidine hydrochloride exerts a negligible influence on cardiac M2 receptors. 14
Some studies15,16 have shown that penehyclidine hydrochloride and glycopyrrolate commonly cause postoperative dry mouth. In the present study, the incidence of dry mouth was significantly higher in groups P and G than in group C (p = 0.002), consistent with previous findings. Although group G exhibited a slightly higher occurrence of dry mouth compared with group P, this difference was not statistically significant, suggesting that postoperative dry mouth caused by penehyclidine hydrochloride is similar to that caused by glycopyrrolate.
This study has the following limitations: First, it was a single-center clinical study with a relatively small sample size, highlighting the need for large-sample multicenter clinical research. Second, the long-term effects of the pharmaceuticals were not evaluated, warranting further investigation. Third, the comparison of dose–response relationships among different anticholinergics requires further exploration. Finally, post-study registration may have introduced bias in our results.
In conclusion, the use of penehyclidine hydrochloride is more effective in preventing PONV, particularly during the early postoperative period in patients undergoing LC. In contrast, glycopyrrolate demonstrates superior efficacy in reducing intraoperative bradycardia and maintaining circulatory stability.
Footnotes
Acknowledgments
We are grateful to Prof Fuyuan Yang for his valuable guidance during the revision process. We acknowledge the use of Grammarly tool for assistance in improving the language of this manuscript.
Authors’ contributions
Hui Yan: This author contributed to the conception and design of the study; acquisition, analysis, and interpretation of the data; as well as drafting and critical revision of the manuscript. Renrong Ao: This author contributed to the conception and design of the study as well as drafting of the manuscript. ChengHui Fan: This author provided critical feedback on the manuscript. Rui Xia: This author contributed to the conception and design of the study as well as provided guidance and supervision throughout the research.
Data availability statement
Data are available from the first author on reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors declare that no funding was received for this study.
