Abstract
Objectives
This randomized, double-blind study compared the antiemetic efficacy of ramosetron with that of ramosetron combined with midazolam, and investigated whether the timing of midazolam administration affected the incidence of postoperative nausea and vomiting (PONV).
Methods
Nonsmoking female patients undergoing laparoscopic gynaecological surgery were randomized to three groups: group R received intravenous (i.v.) normal saline at induction of anaesthesia and 30 min before the end of surgery; group RM1 received midazolam i.v. at induction of anaesthesia and normal saline i.v. 30 min before the end of surgery; group RM2 received normal saline i.v. at induction of anaesthesia and midazolam i.v. 30 min before the end of the surgery. All patients received 0.3 mg ramosetron i.v. at the end of surgery. Incidence of PONV and need for rescue antiemetics were assessed during the 48-h postoperative period.
Results
A total of 126 patients were included in the analyses. There was no significant difference in the incidence of severe nausea, emetic episodes or use of antiemetics among the three groups. The incidence of complete response (no PONV and no rescue antiemetics) was significantly higher in the RM1 (30/41; 73%) and RM2 (30/42; 71%) groups compared with group R (19/43; 44%).
Conclusions
Midazolam given at induction of anaesthesia or at the end of the surgery, combined with ramosetron, was more effective than ramosetron alone in reducing the incidence of PONV.
Introduction
Postoperative nausea and vomiting (PONV) is one of the most unpleasant complications associated with anaesthesia and surgery, causing not only patient discomfort but also increased postoperative pain. The incidence of PONV is nearly 80% in high-risk patients. 1 Many patients state that PONV is an undesirable postoperative outcome with a higher priority than incisional pain. 2
Although antiemetic prophylaxis can significantly reduce the incidence of PONV, its incidence remains high for patients with multiple risk factors. It has been reported that the incidence of PONV is 66.7% in patients given ondansetron after laparoscopic gynaecological surgery and a postoperative opioid. 3 Therefore, PONV prophylaxis should be considered for patients at moderate-to-high risk for PONV. 4 Furthermore, combining two or more antiemetics with different mechanisms of action has been shown to be more effective than using a single agent, in high-risk patients.4–7
The 5-hydroxytryptamine (5-HT3) receptor antagonists are the recommended first- and second-line pharmacological antiemetics for PONV prophylaxis and they have favourable side-effect profiles. 4 Ramosetron has a higher affinity for the serotonin 5-HT3 receptor than ondansetron, granisetron or tropisetron, and its efficacy is well maintained during a 48-h period. 8 The efficacy of 0.3 mg ramosetron to reduce the incidence of PONV in female patients after gynaecological surgery was found to be similar to that of 8 mg ondansetron. 9
Midazolam is a commonly used drug for sedation, premedication and co-induction of anaesthesia. Besides its known anxiolytic effects, it has been reported that midazolam plays a role in the prevention10–14 and treatment15,16 of PONV. Midazolam administered after the induction of anaesthesia, with or without dexamethasone, reduced the incidence of PONV in children following strabismus repair. 11 However, several studies have reported controversial findings associated with the use of midazolam in combination with antiemetics.17,18 Furthermore, it has been reported that midazolam given 30 min before the end of surgery was more effective than premedication with midazolam at decreasing the incidence of PONV. 12
We hypothesized that ramosetron combined with midazolam would be more effective in reducing the incidence of PONV than ramosetron administered alone, following laparoscopic gynaecological surgery in patients at high risk of PONV. The primary purpose of this randomized, double-blind study was to compare the antiemetic efficacy of ramosetron alone with that of ramosetron and midazolam in combination. This study also investigated whether the timing of midazolam administration affected the incidence of PONV in patients at high risk of PONV, who were undergoing laparoscopic gynaecological surgery.
Patients and methods
Study population
This randomized, double-blind study enrolled nonsmoking female patients who were classified as American Society of Anesthesiologists physical status I or II (i.e. normal/ healthy or with mild systemic disease; http://www.asahq.org), aged 19–64 years, and scheduled to undergo laparoscopic gynaecological surgery of <2 h in duration under general anaesthesia at the Department of Obstetrics and Gynaecology, Hallym University Sacred Heart Hospital, College of Medicine, Hallym University, Anyang, Republic of Korea, between August 2012 and January 2013. The patients underwent laparoscopic ovarian cystectomy, ovarian wedge resection, myomectomy, salpingo-oophorectomy or hysterectomy. According to the simplified risk scores for predicting PONV, 1 there are four predictors: female sex; history of motion sickness or PONV; nonsmoking; use of postoperative opioids. As all female patients enrolled in the study were scheduled to use postoperative opioid-based intravenous (i.v.) patient-controlled analgesia (PCA) and were nonsmokers, they all had at least three risk factors for PONV. The minimum anticipated incidence of PONV was 61% in patients with at least three risk factors. 1 Exclusion criteria included any of the following: pregnancy or breast-feeding; obesity (body mass index >30 kg/m2), psychological disease; use of nasogastric tube; administration of antiemetics or systemic steroids within 24 h; vomiting within 24 h; alcohol or drug abuse; allergy to drugs used in the study.
This study was approved by the Institutional Review Board of Hallym University Sacred Heart Hospital (reference numbers: IORG0004993, IRB00005964). Written informed consent was obtained from each patient.
Study design and anaesthesia protocol
Patients were premedicated with 0.004 mg/kg glycopyrrolate, administered intramuscularly. While in the operating room, all patients were monitored by standard limb lead electrocardiography and noninvasive blood pressure measurement, pulse oximetry (CARESCAPE Monitor B650; GE Healthcare, Helsinki, Finland) and the bispectral index (BIS VISTA™ Monitoring System; Aspect Medical Systems, Norwood, MA, USA). General anaesthesia was induced with 4–5 mg/kg of thiopental sodium i.v. and 0.15–0.3 µg/kg per min of remifentanil i.v.; endotracheal intubation was facilitated with 0.6 mg/kg of rocuronium bromide i.v; anaesthesia was maintained with inhaled 1.5–2.5% sevoflurane, continuous i.v. infusion of 0.05–0.2 µg/kg/min remifentanil and air in oxygen (fraction of inspired oxygen, 0.5). The infusion rate of remifentanil and the concentration of sevoflurane were adjusted to maintain blood pressure and heart rate within a 20% deviation from values measured before anaesthesia, and a BIS between 40 and 60. Mechanical ventilation was adjusted to maintain a partial pressure of end-tidal carbon dioxide of 35–40 mmHg throughout the procedure. Rocuronium bromide 0.1–0.15 mg/kg i.v. was administered for muscle relaxation as required, and all patients received 1 µg/kg fentanyl i.v. ∼20 min before the end of surgery.
Study drugs were prepared in 5-ml syringes: normal saline 5 ml or 50 µg/kg midazolam (maximum 5 mg) in a total volume of 5 ml normal saline. Patients were randomly allocated into one of three groups (R, RM1, RM2) by the sealed-envelope method. Patients in group R received normal saline i.v. at induction of anaesthesia and 30 min before the end of surgery. Patients in group RM1 received midazolam i.v. at induction of anaesthesia and normal saline i.v. 30 min before the end of surgery. Patients in group RM2 received normal saline i.v. at induction of anaesthesia and midazolam i.v. 30 min before the end of surgery.
At the end of surgery, sevoflurane and remifentanil administration were stopped and 0.3 mg ramosetron i.v. was administered. Antagonism of muscle relaxation was achieved with i.v. administration of 0.04 mg/kg neostigmine and 0.008 mg/kg glycopyrrolate. The endotracheal tube was removed when adequate ventilation was recovered and the patient was awake. For postoperative pain control, a PCA device, consisting of an i.v. infusion of 16–17 µg/kg fentanyl in a total volume of 100 ml normal saline, was initiated at the completion of surgery; administration of PCA continued for 48 h.
During the postoperative period, patients received 30 mg ketorolac i.v. by their request or when they reported a pain score >6 on an 11-point verbal rating scale (VRS; 0, no pain; 10, worst imaginable pain).
Study assessments
Demographic data, duration of surgery, duration of anaesthesia and history of motion sickness or PONV were recorded for each patient. All episodes of PONV (nausea, retching and vomiting) were recorded during three time periods: 0–2 h, 2–24 h, and 24–48 h after surgery. An investigator (Y. S. K.), who did not know which drug was administered to which patient, assessed all episodes of PONV.
The primary efficacy variable of this study was the incidence of complete response during the 48-h period after surgery. The secondary efficacy variables were the incidence of severe nausea, emetic episodes, and need for rescue antiemetics.
Nausea was defined as a subjectively unpleasant sensation associated with an inclination to vomit. Retching was defined as an involuntary effort to vomit that does not result in ejection of gastric contents. Vomiting was defined as the forceful expulsion of gastrointestinal contents from the stomach through the mouth. Retching and vomiting were defined as emetic episodes. The severity of nausea was evaluated on a VRS (0, no nausea; 10, worst imaginable nausea) divided into mild (1–3), moderate (4–6) and severe (7–10) nausea. Complete response was defined as the absence of PONV with no need for rescue antiemetic therapy during the 48-h period after surgery. The rescue antiemetic, metoclopramide (i.v.), was given as a 10-mg dose in cases of severe nausea or two or more emetic episodes, or in response to a patient's request. If the first-line rescue therapy with metoclopramide was ineffective, the second-line rescue antiemetic, ondansetron (i.v.) was given as a 4-mg dose. When both metoclopramide and ondansetron treatments were ineffective, PCA was stopped for 3 h. Adverse events (including headache, dizziness, drowsiness and general weakness) were also recorded.
Statistical analyses
In a preliminary study conducted in 10 patients who were given 0.3 mg ramosetron i.v. alone, five patients (50%) who underwent laparoscopic gynaecological surgery (using fentanyl-based PCA) showed a complete response during the 48-h period after surgery. The sample-size calculation was made using power analysis (α = 0.05, β = 0.8) to detect a 30% increase in the rate of complete response (from 50% to 80%), and was found to require 40 patients per group. Assuming a potential dropout rate of 10%, the final sample size was set at 44 patients per group.
The data were presented as mean ± SD or n (%) of patients. All statistical analyses were performed using the IBM® SPSS® statistical software package, version 20.0 (IBM Corporation, Somers, NY, USA). Statistical analyses were undertaken using the Student's t-test, χ2-test and Fisher's exact test. A P-value < 0.05 was considered statistically significant.
Results
Demographic and clinical data for patients undergoing laparoscopic gynaecological surgery, who received ramosetron alone (group R) or combined with midazolam, given intravenously (i.v.) at induction of anaesthesia (group RM1) or 30 min before the end of the surgery (group RM2); normal saline 5 ml was given as a control.

Data presented as mean ± SD or n (%) of patients.
No statistically significant between-group differences (P ≥ 0.05); Student's t-test and Fisher's exact test.
All patients received ramosetron 0.3. mg i.v. at the end of surgery.
Group R, patients received normal saline at induction of anaesthesia and 30 min before the end of the surgery; group RM1, patients received midazolam 50 µg/kg i.v. at induction of anaesthesia and normal saline 30 min before the end of the surgery; Group RM2, patients received normal saline at induction of anaesthesia and midazolam 30 min before the end of the surgery; PONV, postoperative nausea and vomiting.
Incidence of postoperative nausea and vomiting, use of rescue antiemetics and complete response in patients undergoing laparoscopic gynaecological surgery who received ramosetron alone (group R) or ramosetron combined with midazolam, given intravenously (i.v.) at induction of anaesthesia (group RM1) or 30 min before the end of the surgery (group RM2).
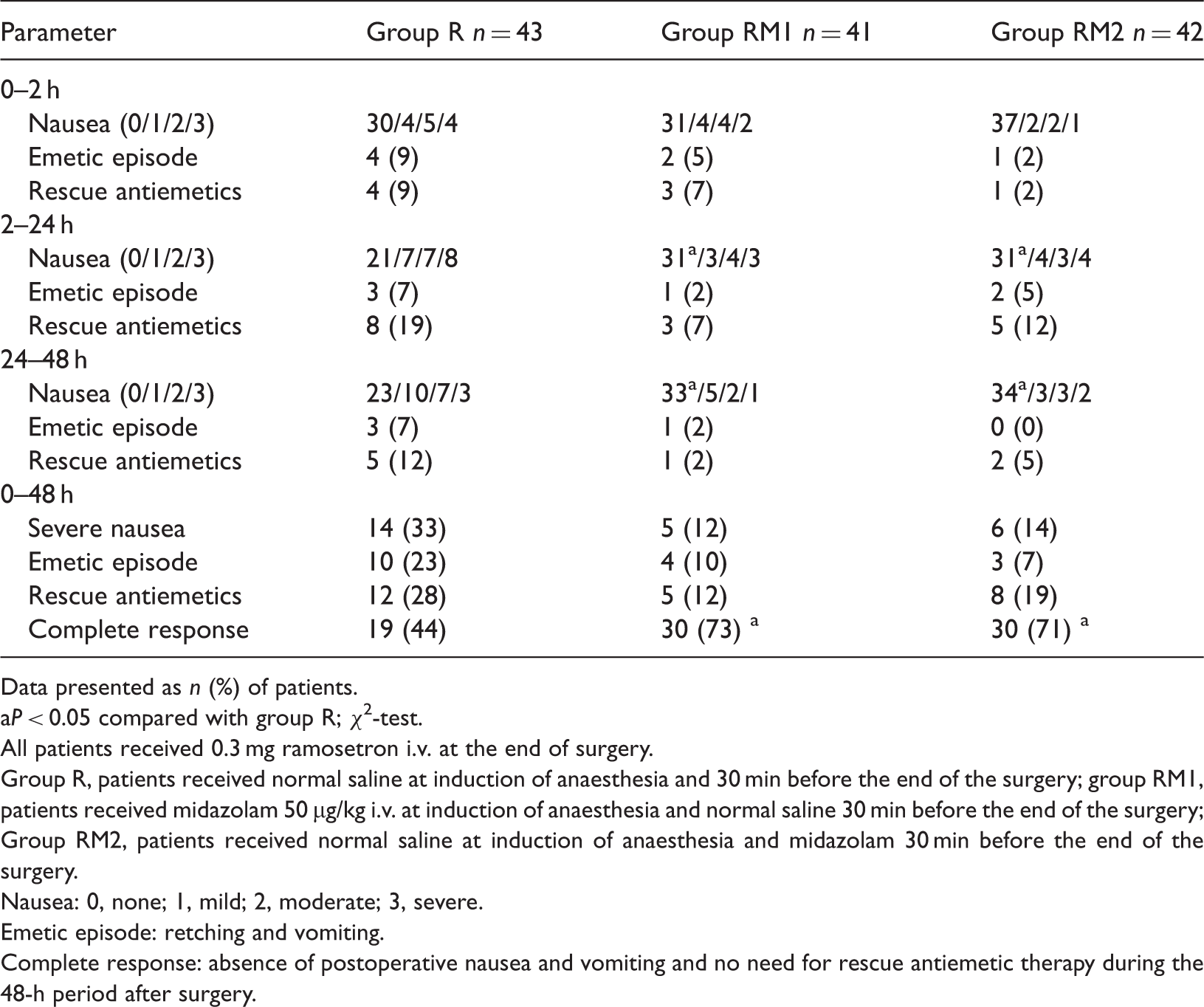
Data presented as n (%) of patients.
P < 0.05 compared with group R; χ2-test.
All patients received 0.3 mg ramosetron i.v. at the end of surgery.
Group R, patients received normal saline at induction of anaesthesia and 30 min before the end of the surgery; group RM1, patients received midazolam 50 µg/kg i.v. at induction of anaesthesia and normal saline 30 min before the end of the surgery; Group RM2, patients received normal saline at induction of anaesthesia and midazolam 30 min before the end of the surgery.
Nausea: 0, none; 1, mild; 2, moderate; 3, severe.
Emetic episode: retching and vomiting.
Complete response: absence of postoperative nausea and vomiting and no need for rescue antiemetic therapy during the 48-h period after surgery.
Incidence of adverse events in patients undergoing laparoscopic gynaecological surgery who received ramosetron alone (group R) or ramosetron combined with midazolam given intravenously (i.v.) at induction of anaesthesia (group RM1) or 30 min before the end of surgery (group RM2).

Data presented as n patients.
No statistically significant between-group differences (P ≥ 0.05); χ2-test.
All patients received 0.3 mg ramosetron intravenously at the end of surgery.
Group R, patients received normal saline at induction of anaesthesia and 30 min before the end of the surgery; group RM1, patients received midazolam 50 µg/kg i.v. at induction of anaesthesia and normal saline 30 min before the end of the surgery; Group RM2, patients received normal saline at induction of anaesthesia and midazolam 30 min before the end of the surgery.
Discussion
This current randomized, double-blind study compared the antiemetic efficacy of ramosetron administered alone with that of ramosetron administered in combination with midazolam, in female nonsmoking patients at high risk for PONV undergoing laparoscopic gynaecological surgery. This current study found that the combination of ramosetron and midazolam was more effective than ramosetron alone in reducing the incidence of postoperative PONV. In addition, the incidence of postoperative adverse events was not significantly different among the three treatment groups. In the present study, midazolam was equally effective at reducing PONV when given either at induction of anaesthesia or 30 min before the end of the surgery.
The efficacy of ramosetron is maintained in terms of nausea and vomiting 6–48 h after treatment. 8 Ramosetron was as effective as ondansetron in reducing the incidence of PONV in female patients after gynaecological surgery 9 and more effective than ondansetron in preventing vomiting and reducing the severity of nausea related to opioid-based i.v. PCA, in highly susceptible patients. 19
It has been reported that midazolam has an effect on the prevention10–14 and treatment15,16 of PONV. A continuous infusion of midazolam was reported to be a more effective antiemetic than ondansetron for the prevention of PONV after cardiac surgery, 10 and low-dose midazolam infusion is an effective treatment for patients with PONV resistant to standard antiemetics. 15 Midazolam administered after the induction of anaesthesia, with or without dexamethasone, reduced the incidence of PONV in children undergoing strabismus repair. 11 However, several studies reported controversial results associated with the use of midazolam in combination with antiemetics. The combination of dexamethasone and midazolam reduced the incidence of vomiting and the rescue antiemetic requirements, but did not decrease the total incidence of nausea and vomiting, compared with the use of dexamethasone alone, in female patients undergoing middle-ear surgery. 17 Combined midazolam and ramosetron had no advantages compared with ramosetron alone in reducing the incidence of PONV in children undergoing strabismus surgery. 18 Several important risk factors associated with PONV are female sex, nonsmoking status, history of PONV or motion sickness, age, duration of anaesthesia and use of postoperative opioids.20,21 Longer procedures (which are associated with longer exposure to volatile anaesthetics) are associated with an increased incidence of PONV. 22 Intraoperative use of opioids 23 and laparoscopic, strabismus, and middle-ear surgery 21 may increase the risk of PONV. Patients who participated in this current study had a relatively high risk for PONV because of the presence of several of these known risk factors (including female sex, nonsmoking status, laparoscopy, and intra- and postoperative opioid use). Other risk factors (such as a history of PONV or motion sickness, age and duration of anaesthesia) did not differ among the three groups. Although the combination of ramosetron and midazolam did not significantly decrease the incidence of severe nausea, emetic episodes or the use of rescue antiemetics, the overall incidence of complete responses was higher in patients given the combination therapy compared with ramosetron alone, demonstrating the prophylactic efficacy of the combination therapy.
Midazolam has been shown to be an effective antiemetic when administered continuously,10,15 before14,18,24 and after 17 induction of anaesthesia, or before the end of the surgery. 25 It was reported that midazolam administered 30 min before the end of surgery was more effective in decreasing the incidence of PONV than midazolam given 15 min before induction of anaesthesia, in patients who had undergone lower abdominal surgery lasting 1–2 h. 12 In previous studies, doses of midazolam for preventing PONV were 35–75 µg/kg.11,12,17,24 Watcha and White 21 recommended the use of minimally effective doses of antiemetic drugs to reduce the incidence of sedation and other deleterious side-effects, and potent nonopioid analgesics such as ketorolac can be used to avoid some opioid-related adverse effects. The present study used 50 µg/kg of midazolam and (as a rescue analgesic) ketorolac; the incidence of adverse events did not differ significantly among the three treatment groups. Furthermore, midazolam given at anaesthesia induction or 30 min before the end of the surgery was effective for preventing PONV, regardless of the time of administration in this current study. However, this result may not apply to other types of surgery, such as those with durations longer than those of procedures in the present study.
Although the precise mechanism of action of the antiemetic effect of midazolam has not been fully elucidated, it has been suggested that benzodiazepines reduce the psychic input to the vomiting centre; blocking the re-uptake of adenosine may result in an adenosine-mediated inhibition of dopamine synthesis, release and action in the chemoreceptor trigger zone. 26 Adenosine receptor agonists have been shown to inhibit nigrostriatal release of dopamine, 27 and benzodiazepines have been shown to inhibit the uptake of adenosine into rat brain cortical synaptosomes. 28 Midazolam may also affect striatal dopamine release 29 and the anaesthetic actions of midazolam are partially related to inhibition of dopamine neuron A1 activity. 30 In our opinion, these studies support the hypothesis of Di Florio, 26 and demonstrate that midazolam combined with ramosetron could reduce PONV more effectively than ramosetron alone.
This current study had several limitations. First, the concentrations of sevoflurane and the infusion rates of remifentanil were adjusted according to BIS and vital signs, but the amounts were not measured exactly. However, the amount of anaesthetic used was probably a minor factor in terms of causing PONV, because the durations of anaesthesia did not differ significantly among the three treatment groups. Secondly, it was difficult to determine which time of midazolam administration was more effective in reducing PONV, because the present study was not sufficiently powered to compare such differences. Further studies are required to determine the appropriate dose and administration time of midazolam in different types and durations of surgery.
In conclusion, midazolam given at induction of anaesthesia or 30 min before the end of the surgery combined with ramosetron was more effective for the prevention of PONV than ramosetron alone in patients at high risk for PONV following laparoscopic gynaecological surgery.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
