Abstract
Fungal orbital cellulitis is usually seen in immunocompromised individuals, and opportunistic pathogens are the main etiology. We herein report a case of fungal orbital cellulitis due to Aspergillus in a patient with no history of trauma. A 48-year-old man presented to the emergency room of our hospital with a 2-week history of periorbital swelling, conjunctival hyperemia, and chemosis of his right eye. The visual acuity of his right eye was 6/20, and the intraocular pressure was 44 mmHg. The main clinical findings were proptosis of the right ocular globe with conjunctival hyperemia and a palpable infratemporal orbital mass. Laboratory testing failed to detect the presence of a pathogenic infection, and the lesions on computed tomography images resembled those of a malignant tumor of the orbit. The diagnosis was finally confirmed by postoperative pathological examination, and the patient responded favorably to debridement combined with antifungal therapy. Histopathological examination may help to reveal the nature of this disease. Surgical removal of inflammatory lesions can serve as an important diagnostic and treatment method for fungal orbital cellulitis.
Keywords
Introduction
Orbital cellulitis is an inflammatory process that involves the muscle and fat located posterior to the orbital septum within the bony orbit, but the term is generally used to describe infectious inflammation.1,2 Orbital infections always occur secondary to other conditions, such as paranasal sinusitis, trauma, surgery, infection of the eyelids or face, or even hematogenous spread from distant locations.1,3 The causative organisms of orbital cellulitis are usually bacterial but can also be polymicrobial or fungal. 4 Fungal orbital cellulitis is always caused by opportunistic pathogens and is associated with a high mortality rate. 5 It generally occurs in patients with immunodeficiencies. The clinical presentation and imaging findings of fungal orbital cellulitis are nonspecific, and misdiagnosis readily occurs. The management of fungal orbital cellulitis is also challenging, and no consensus guidelines have yet been established. We herein present a rare case of fungal orbital cellulitis due to Aspergillus in an adult with no history of trauma. The diagnosis was confirmed through pathological examination, and the patient responded favorably to debridement combined with antifungal therapy.
Case report
The reporting of this study conforms to the CARE guidelines. 6 The patient and his parent provided written informed consent for both treatment and publication of this case report, including any accompanying images. Because no identifiable health information was included in this case report, the requirement for ethics approval was waived.
A 48-year-old man presented to the emergency room of our hospital with a 2-week history of periorbital swelling, conjunctival hyperemia, and chemosis of his right eye. He also noted decreased vision in his right eye. On presentation, his body temperature was 36.6°C. The visual acuity of his right eye was 6/20, and the intraocular pressure was 44 mmHg. The main clinical findings were proptosis of the right ocular globe with conjunctival hyperemia and a palpable infratemporal orbital mass. Notably, the patient had been diagnosed with type 2 diabetes mellitus (DM) 1 month prior and was being treated with insulin, but his blood glucose level was not being monitored. The patient denied any other prior systemic medical history or ocular history, including trauma. His family history was unremarkable.
Laboratory tests revealed an elevated blood glucose level (11.08 mmol/L) and HbA1c level (15.1%). Serum (1,3)-beta-D glucan and Aspergillus galactomannan tests were negative. Routine blood screening showed no abnormality. Both bacterial and fungal cultures of tracheal secretions were negative. Chest X-ray results were normal. Computed tomography (CT) revealed a 3.5- × 2.1-cm soft tissue mass occupying the infratemporal orbital area along with evidence of bony erosion (Figure 1(a), (b)). The mass was compressing the right eyeball and inferior rectus muscle. CT also revealed right maxillary sinusitis (Figure 1(c)). Based on the patient’s clinical and imaging findings, the initial clinical diagnosis was a malignant tumor of the orbit.
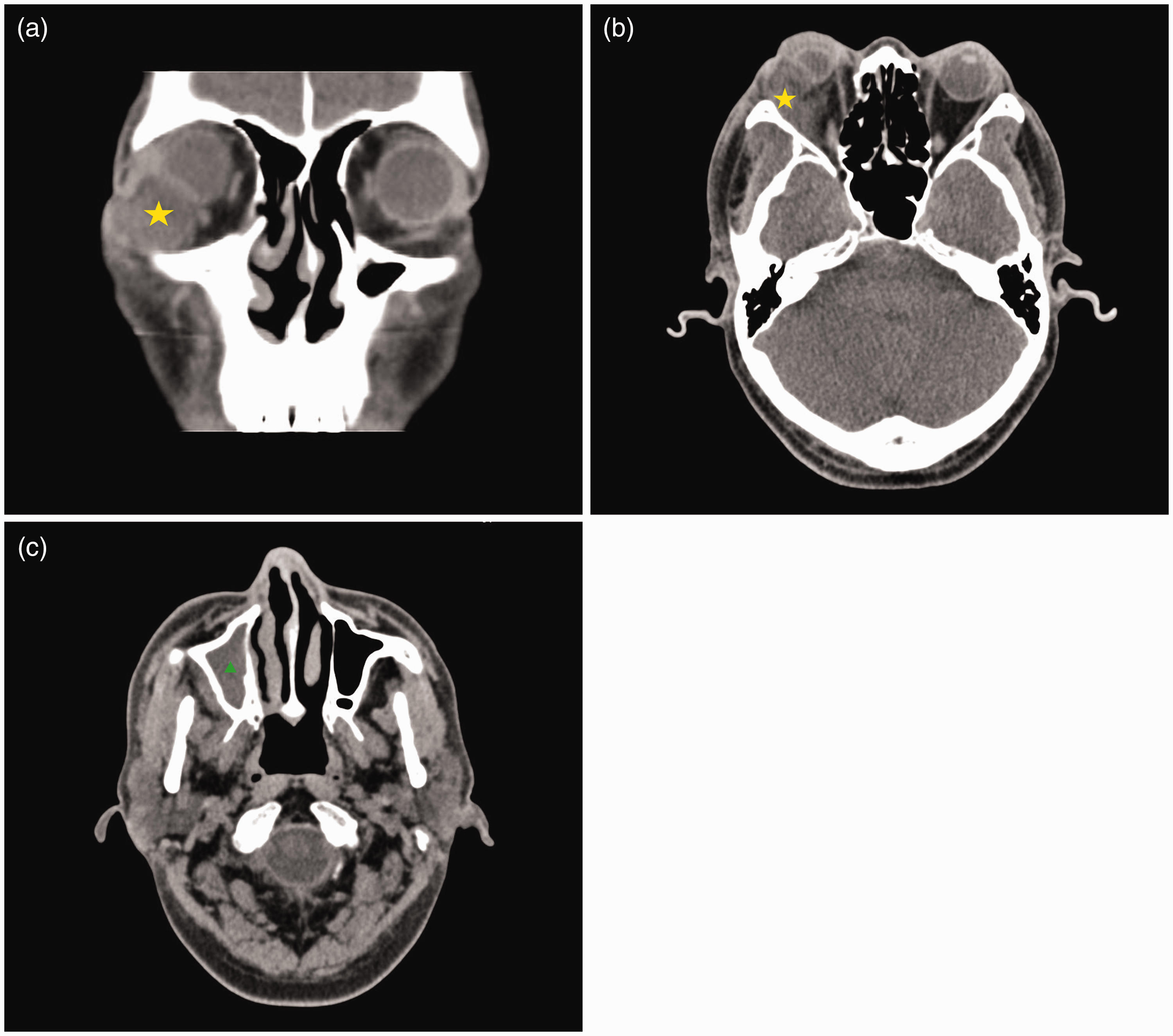
Computed tomography images of the patient in the present case. (a, b) The orbital mass (yellow asterisk) was located in the infratemporal orbital area. The mass compressed the right eyeball and inferior rectus muscle and (c) right maxillary sinusitis (green arrowhead) was also evident.
Surgery was performed to remove the lesion and clarify the diagnosis. Intraoperative examination revealed a mass with unclear boundaries (Figure 2). We also observed necrotic and purulent material inside the mass, and it was carefully debrided. Histopathological examination was performed postoperatively. Hematoxylin–eosin staining showed the presence of septate fungal hyphae (Figure 3(a)) and tissue necrosis. Immunohistochemical staining also showed positive staining of fungal hyphae using both periodic acid–Schiff (Figure 3(b)) and Gomori methenamine silver (Figure 3(c)). Histopathological examination showed a ribbon-like fungus with some acute-angle branching. Orbital aspergillosis was diagnosed, and antifungal treatment (voriconazole) was initiated. After adjusting the insulin dosage, the patient’s blood glucose level was effectively controlled. The patient recovered well, and the visual acuity of his right eye improved to 20/20. He was recommended to continue systemic antifungal treatment in the infectious disease department and ear, nose, and throat department. The patient underwent regular follow-up for 3 years and developed no recurrence or fungal infections in other parts of the body.

Resected mass. Gross observation revealed a red–yellow surface and unclear boundaries.

Microscopic examination. Hematoxylin–eosin staining showed (a) fungal hyphae (original magnification, ×200) as well as (b) necrosis, bleeding, and infiltration of inflammatory cells (original magnification, ×100). The fungal hyphae showed positive immunohistochemical staining using (c) periodic acid–Schiff (original magnification, ×200) and (d) Gomori methenamine silver (original magnification, ×200).
Discussion
In this report, we have described a case of acute orbital aspergillosis manifesting in a 48-year-old patient. The increase in opportunistic fungal infections is due to the rising prevalence of immunocompromised patients. Opportunistic pathogens are the main cause of fungal orbital cellulitis, which is usually seen in immunocompromised individuals. The pathogens involved in invasive fungal orbital cellulitis include Mucorales, which can cause mucormycosis, and Aspergillus, which causes life-threatening invasive orbital infections. 4 Fungal orbital cellulitis can result in blindness as well as venous and intracranial extension, which is associated with a high mortality rate.5,7 Fungal orbital cellulitis is quite rare in patients without a history of trauma. Our patient had been diagnosed with type 2 DM 1 month before presentation, and his blood glucose level had not been monitored. In adult patients with no history of ocular trauma, uncontrolled DM is the condition most commonly associated with fungal orbital cellulitis.5,8,9 Even when the patient’s blood glucose level is not markedly high, prolonged and uncontrolled hyperglycemia can still be an important cause of fungal infection. Orbital aspergillosis is a rare but serious infection that usually arises from the paranasal sinuses through bony erosion and/or vascular involvement. 10 In our patient, the orbital aspergillosis might have originated from the right maxillary sinusitis, and the negative results of the serum (1,3)-beta-D glucan and Aspergillus galactomannan tests ruled out involvement of vascular pathways.
Fungal infections are also an important cause of mortality in patients with a broad range of clinical conditions. 7 Early diagnosis and appropriate therapy are crucial in the management of fungal infection and preservation of vision. Orbital aspergillosis exhibits an insidious course, and its presentation is variable. 10 It may be confused with a variety of other orbital processes, and the diagnosis is often challenging because of its nonspecific clinical and radiologic appearance. Many other orbital pathologies may exhibit similar symptoms, such as periorbital swelling, proptosis, visual impairment, and conjunctival reaction. 11 Especially when an orbital mass is the main clinical finding, achieving an early and accurate diagnosis is difficult. The most relevant differential diagnoses for aspergillosis are vascular, inflammatory, neoplastic, or infectious orbital diseases. 11 Radiologic examinations may be useful for the differential diagnosis, and CT can reveal calcifications and bony erosion, which can be seen in orbital aspergillosis; however, the signs are nonspecific. Many orbital disease processes have imaging findings in common with orbital infection. Indolent growth of a solitary, discrete mass is usually suggestive of a tumor. 12 In our case, laboratory testing failed to detect the presence of pathogenic infection, and the lesions on CT images were very similar to a malignant tumor. Because of the similarity of the clinical and imaging manifestations between orbital aspergillosis and an orbital tumor in this case, misdiagnosis occurred even after comprehensive imaging and etiological examination. When serological and imaging examinations cannot provide enough diagnostic information, histopathological examination may play a crucial role in the diagnosis of fungal orbital cellulitis. This highlights the importance of obtaining biopsies in the appropriate clinical setting. In our patient, the nature of the lesion was finally determined by postoperative histopathology.
Although there are no established guidelines specific to the management of orbital cellulitis, multidisciplinary involvement is often encouraged. This is especially true considering that fungal orbital cellulitis may occur after recovery from COVID-19 infection. 13 Immunocompromised patients, such as those with uncontrolled DM, are more vulnerable to fungal infection. 14 Patients on long-term steroid medication or mechanical ventilation may also be prone to opportunistic fungal infections. 15 We reviewed recent studies on the management of fungal orbital cellulitis secondary to COVID-19 infection. Combinations of surgery and systemic antifungal therapy played an important role in treatment. 15 Early surgical intervention is recommended because of its importance in diagnosis and treatment.10,16 Debridement can help to remove lesions, including necrotic tissue and fungi, thus decreasing the total fungal burden. Moreover, postoperative histopathological examination can help clarify the diagnosis. Once a clear diagnosis has been achieved, targeted antifungal therapy can be used. Systemic antifungal therapy is also important in the treatment of fungal orbital cellulitis, especially for patients who are not fit for surgery. Studies have shown that voriconazole is effective and well tolerated in the treatment of aspergillosis.17,18 Voriconazole with debridement was useful treatment for orbital aspergillosis in our case.
In summary, we have presented a rare case of orbital aspergillosis in an immunocompromised man with no history of trauma. We reported this patient’s clinical data to improve the understanding and diagnostic accuracy of this disease. Fungal orbital cellulitis can have clinical and imaging manifestations similar to those of orbital malignant tumors. When laboratory testing and imaging fail to provide enough diagnostic information, histopathological examination may help to reveal the nature of this disease. Surgical removal of inflammatory lesions can serve as an important diagnostic and treatment method for fungal orbital cellulitis.
Footnotes
Author contributions
Both authors participated in the diagnosis and treatment of the patient. YZ drafted the manuscript, and WH reviewed the manuscript. Both authors provided critical feedback and approved the submitted version.
Data availability statement
All the data supporting our findings are contained within the manuscript.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Natural Science Foundation of Science and Technology Department of Sichuan Province (No. 2023NSFSC1665).
