Abstract
Primary ovarian sarcoma is a rare malignancy, with primary ovarian leiomyosarcoma being even rarer because of the lack of smooth muscle in the ovaries. We herein report a case of primary ovarian leiomyosarcoma in a woman in her late 50s who presented with a 6-month history of abdominal pain. Imaging revealed a pelvic mass. The patient underwent surgery and was diagnosed with ovarian leiomyosarcoma. One month postoperatively, she began gemcitabine and docetaxel chemotherapy and continued this treatment for 6 months. Eight months postoperatively, however, recurrence was detected in the pelvic cavity. This case is reported with the aim of raising awareness about this rare disease.
Introduction
Primary ovarian sarcoma is a rare gynecologic tumor, accounting for less than 3% of all ovarian tumors. 1 Primary ovarian leiomyosarcoma (POLMS) is even rarer, representing less than 0.1% of all ovarian sarcomas. 2 This tumor is highly malignant and lacks specific clinical manifestations, making it difficult to diagnose. 3 Because of its rarity, there is limited literature on POLMS, and optimal treatment guidelines have not been established. To enhance understanding of this disease, we analyzed the diagnosis and treatment of a case of POLMS in our hospital, integrating it with existing literature.
Case presentation
A woman in her late 50s was admitted to our hospital because of a 6-month history of lower abdominal discomfort as well as a pelvic mass that had been discovered 2 days earlier. During the previous 6 months, she had experienced sporadic lower abdominal swelling and pain but had not sought medical attention. Two days before admission, an examination at an outside medical facility revealed a low-echo growth on the left side of her pelvis, and a computed tomography scan showed a lesion occupying the pelvic area. Her menstrual history was unremarkable, with menopause occurring at 52 years of age (G3P1). She had a 1-year history of well-managed hypertension.
Clinical assessment revealed slight anemia, normal vulvar findings, a smooth vaginal texture, an unobstructed cervix, and uterine shrinkage. A pelvic mass was palpable; it was approximately 11 cm in size and extended to a point immediately below the umbilicus. The mass had indistinct edges, limited mobility, and was immovable, with clear tenderness in the neighboring left region. Tumor marker results showed normal levels of carcinoembryonic antigen, alpha-fetoprotein, and CA199, whereas CA125 was slightly elevated at 25.8 U/mL (reference range: 0.0–25.0 U/mL), and human epididymal protein-4 was elevated at 132.2 pmol/L (reference range: 0.0–74.3 pmol/L).
An enhanced pelvic magnetic resonance imaging (MRI) scan revealed a cystic solid mass in the pelvis, measuring approximately 13.1 × 11.4 × 14.5 cm, with mixed signals and distinct boundaries (Figure 1). Irregular hyperintensity was present in the solid portion on the diffusion-weighted imaging sequence, with significant uneven enhancement of the parenchymal section after contrast injection, areas of unenhanced liquefaction necrosis at various stages, and displacement of adjacent structures.
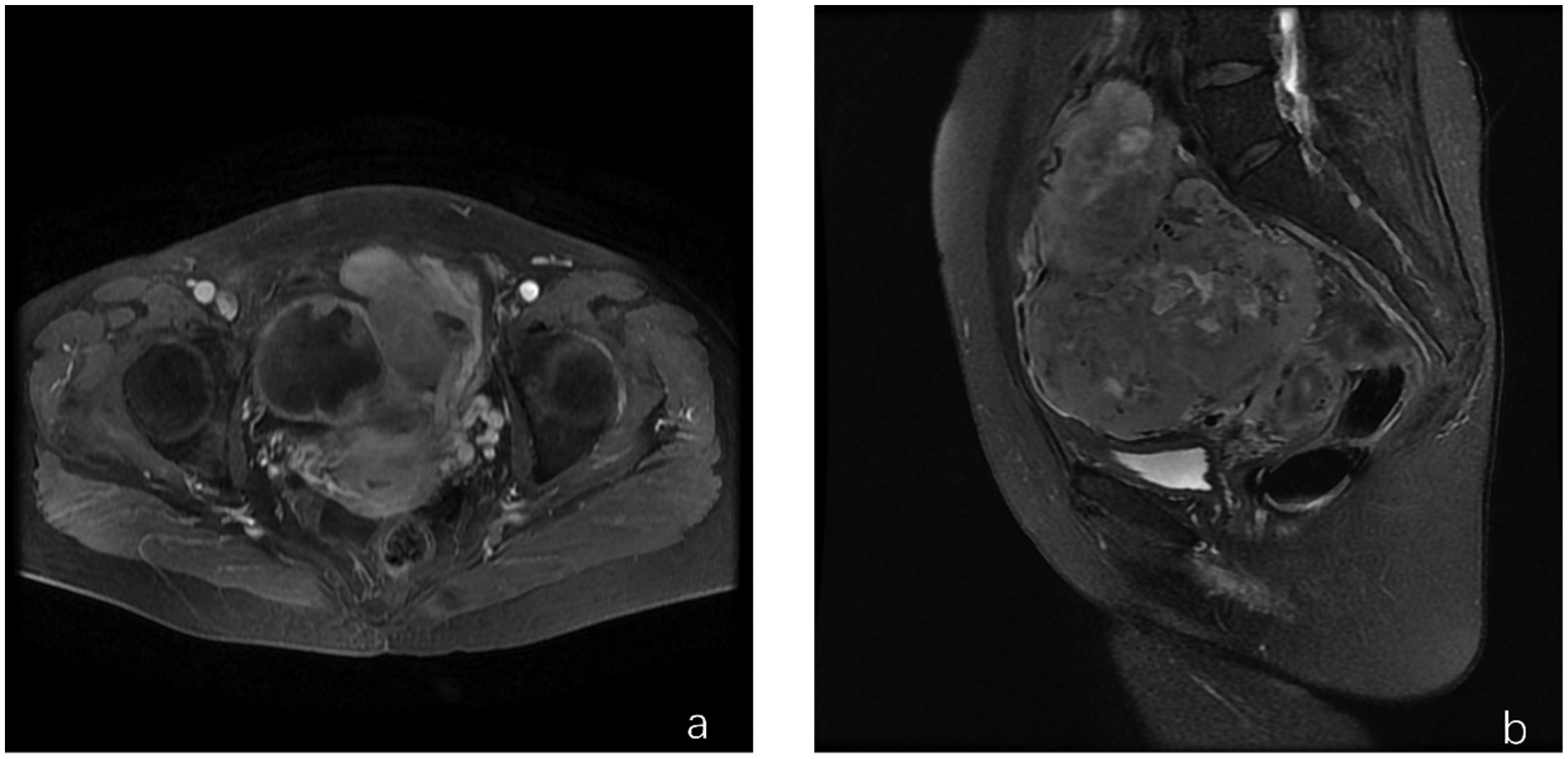
Pelvic enhanced magnetic resonance imaging scan.
Following the examination, an exploratory laparotomy was performed. Surgery revealed a left ovarian mass, approximately 15 × 14 cm in size, with a fractured surface and fragile texture. The mass was attached to the intestine, although the intestine itself was not invaded. The uterus was atrophic, and the right adnexa appeared normal. Intraoperative frozen section analysis suggested a malignant tumor of mesenchymal origin with widespread tumor cell distribution.
The surgical procedure involved extensive ligation of the infundibulopelvic ligament, total hysterectomy, bilateral adnexectomy, para-aortic lymph node biopsy, pelvic lymph node dissection, intestinal adhesiolysis, and abdominal wall incision and drainage. Peritoneal washings showed no tumor cells (Figure 2(a)). Paraffin sections confirmed a malignant mesenchymal tumor, with significant tumor cell atypia and extensive areas of coagulative necrosis (Figure 2(b), (c)), consistent with leiomyosarcoma (FNCLCC grade 3). Immunohistochemical analysis showed vimentin (+), CD10 (−), smooth muscle actin (+), desmin (−), epithelial membrane antigen (−), and Ki-67 (index 30%) (Figure 2(d)–(f)). There was no lymph node metastasis. The final diagnosis was stage IIB leiomyosarcoma of the left ovary.

Histological examination of the tumor. (a) No tumor cells were detected in the intraoperative peritoneal irrigation fluid. (b, c) Tumor cells exhibited high atypia and large areas of coagulative necrosis. Immunohistochemical analysis showed (d) vimentin positivity and (e) epithelial membrane antigen positivity, and (f) 30% Ki-67 positivity.
The patient recovered well postoperatively. One month later, she received six courses of gemcitabine and docetaxel chemotherapy. Regular follow-ups were conducted, but the patient developed recurrence 8 months postoperatively. Transvaginal color Doppler ultrasound showed multiple mixed echoes in the pelvic cavity, with the largest lesion measuring approximately 4.6 × 3.3 cm and displaying irregular boundaries and blood flow signals. Pelvic MRI revealed multiple abnormal signals in the pelvic cavity. The patient ultimately opted for conservative treatment.
The patient provided verbal informed consent for the reporting of this case. She provided written informed consent for all treatments. The reporting of this study is in line with the CARE guidelines. 4
Discussion
POLMS is most commonly found in postmenopausal women 5 at an average age of 52.9 years, 6 but it has also been reported in younger women7,8 and even around menstruation. 9 Clinical symptoms are nonspecific and often include abdominal pain, abdominal distention, palpable abdominal lumps, loss of appetite, and urination difficulties,10,11 leading to late-state diagnosis in many cases. The tissue origin is unclear because the ovary does not contain smooth muscle. However, POLMS is hypothesized to arise from the corpus luteum, follicle, surrounding smooth muscle, blood vessels, remnants of Wolffian ducts, or mesenchymal stem cells. 12 The tumor typically presents as a large unilateral mass. 13
Early diagnosis of ovarian leiomyosarcoma is challenging and often occurs only when clinical symptoms or signs are evident. Laboratory tests may reveal anemia, while tumor markers may show normal or slightly elevated levels, which are of limited specificity.14,15 Imaging tests can determine the location, size, morphology, and internal structure of the tumor. One study described histopathological imaging findings of POLMS; in that case, contrast-enhanced computed tomography revealed a poly-lobulated pelvic mass with heterogeneous enhancement and associated intraluminal effusion. 16 Coronal maximum intensity projection reconstruction demonstrated the blood supply and tumor origin. On MRI, the mass exhibited a muscle-consistent signal on T1-weighted images, an uneven high signal on T2-weighted images, and restricted diffusion on diffusion-weighted images and apparent diffusion coefficient maps. 16 A definitive diagnosis also requires pathologic examination. Grossly, ovarian leiomyosarcomas vary in size and usually present as a large single mass. 17 The mass often has a fish-like appearance and may be accompanied by bleeding, necrosis, and cystic changes. Microscopically, the presence of at least two of the following three criteria is required for diagnosis: coagulative necrosis, cellular atypia, and a mitotic index of ≥10/HPF. 18 Some authors have suggested that POLMS can also be diagnosed without necrosis if significant atypia is present, even with a mitotic index of ≤10/HPF.19,20 Immunohistochemical staining further aids in the diagnosis, with positive h-caldesmon expression in vascular tumors and negative expression in non-vascular tumors. 21 Other immunohistochemical features include smooth muscle actin (+), desmin (+), and vimentin (+).
Because of the rarity of ovarian leiomyosarcoma, no clear treatment guideline has been established. 22 Surgery typically involves sterilization, total abdominal hysterectomy, bilateral salpingo-oophorectomy, and sarcomata lumpectomy.23,24 For staging, the International Federation of Gynecology and Obstetrics recommends using the epithelial ovarian cancer classification system to assess leiomyosarcoma. 25 Radiotherapy and chemotherapy are used as adjuvant therapies, although their efficacy is not definitively proven, 23 and chemotherapy regimens vary. Our patient received six courses of chemotherapy with a gemcitabine and docetaxel regimen but unfortunately relapsed 8 months after surgery. At the time of this writing, she was under observation.
Ovarian leiomyosarcoma has a poor prognosis because of its nonspecific symptoms, with most cases diagnosed at a late stage. Recurrence is most common within the first year after diagnosis, and most patients die within 2 years. The 5-year survival rate is less than 20%. 26 In a study of 113 patients with POLMS by Mandato et al., 13 an advanced stage and a high mitotic index negatively impacted survival. Treatment type may also influence survival, with lymph node removal and chemotherapy improving outcomes in some patients with POLMS.
In summary, ovarian leiomyosarcoma is a rare but highly aggressive tumor. Most patients are diagnosed at a late stage, and there is ongoing debate about the use of adjuvant radiotherapy and chemotherapy in advanced cases. Further research is needed to delay recurrence and extend patient survival. For patients with postoperative recurrence, reoperation along with rational radiotherapy, chemotherapy, and comprehensive treatment may be considered. Enhancing early diagnosis and treatment requires the collaborative efforts of gynecologists, sonographers, radiologists, and pathologists.
Footnotes
Acknowledgements
The authors would like to extend their appreciation to the pathology department faculty for their valuable technical guidance on the pathology images.
Author contributions
Qingli Zhang collected the data and wrote the initial draft. Kexin Wang conducted the literature search and obtained informed consent for manuscript publication. Bin Wang reviewed and approved the final manuscript. All authors have read and approved the final manuscript.
Data availability statement
The data are available from the authors upon reasonable request.
Declaration of conflicting interest
The authors declare that they have no conflict of interest regarding the publication of this research.
Ethics
In this study, patient identification information was anonymized, and informed consent was obtained. The case report was approved by the Ethics Committee of the Second Clinical Medical School of Inner Mongolia University for Nationalities and was exempt from further ethical review.
Funding
This research received no specific grant from funding agency in the public, commercial, or not-for-profit sectors.
