Abstract
Objective
We developed and validated a nomogram for overall survival (OS) and cancer-specific survival (CSS) prediction in patients with primary testicular diffuse large B-cell lymphoma (PT-DLBCL).
Methods
Patients diagnosed with PT-DLBCL were selected from the Surveillance, Epidemiology, and End Results (SEER) database. Independent prognostic factors were analyzed to establish a nomogram of OS and CSS. Patients were reclassified into high- and low-risk groups; survival was compared using Kaplan–Meier curves and log-rank tests.
Results
We collected 1099 PT-DLBCL cases (2000–2019) from SEER and randomized into training (n = 771) and validation (n = 328) cohorts. In univariate and multivariate Cox regression analyses, five prognostic indicators (age, treatment modality, diagnosis year, Ann Arbor stage, laterality) were used to establish a nomogram of OS and CSS. The nomogram demonstrated excellent discrimination and calibration, with concordance indices in the training and validation cohorts of 0.702 (95% confidence interval [CI], 0.677–0.727) and 0.705 (95% CI 0.67–0.74) for OS and 0.694 (95% CI 0.663–0.725) and 0.680 (95% CI 0.63–0.72) for CSS. The calibration curve and ROC analysis indicated good predictive capability of the nomogram.
Conclusions
The constructed prognostic model showed good predictive value for PT-DLBCL to assist clinicians in developing individualized treatment strategies.
Introduction
Primary testicular lymphoma (PTL) accounts for 1% to 2% of all cases of non-Hodgkin lymphoma and 3% to 9% of all testicular malignancies. However, PTL is the most pervasive malignant testicular neoplasm in individuals aged more than 60 years and increases with age. 1 , 2 The most common pathological type of PTL is primary testicular diffuse large B-cell lymphoma (PT-DLBCL), which comprises 80% to 90% of all primary lymphomas of the testis. Although the addition of a combined regimen has greatly improved the prognosis of patients with PT-DLBCL in recent decades, this type of PTL demonstrates substantial extranodal tropism, and its recurrence often involves the central nervous system (CNS) and contralateral testis. PT-DLBCL has a poor prognosis and a higher risk of late disease-related death than nodal DLBCL. 3 , 4
Developing an optimal method for accurately predicting prognosis and determining individualized treatment in PT-DLBCL remains an urgent clinical problem to be solved. The International Prognostic Index (IPI) has become the main clinical tool for risk stratification of lymphoma in planning treatment and surveillance. However, some studies report that approximately 70% of patients with PTL are in stage I/II at the first diagnosis, and their IPI score (always less than 2) has limited prognostic value. 5 Although positron emission tomography–computed tomography (PET-CT) is the main criterion used in staging and response evaluation, several studies have revealed that treatment measures based on PET-CT have not improved patient prognosis. 4 , 6 Therefore, further investigation is needed to develop an effective and accurate prediction model to screen patients with PT-DLBCL and formulate individualized therapeutic regimens.
The nomogram is a useful tool in oncological medicine that shows constructed proportional hazards regression models in a visual form to estimate patient prognosis. Because of its rarity, efforts to clarify the clinical characteristics and prognostic outcomes of PT-DLBCL have mostly relied on individual cases or small series studies. 3 ,7–9 Therefore, to improve prognosis, it is clinically important to collect detailed information for individual patients to develop a novel predictive nomogram for this population in a large real-world setting. Surveillance, Epidemiology, and End Results (SEER) is one of the most representative large tumor databases in North America and an excellent resource for the study of malignant and rare tumors.
We conducted the present research using the SEER database to explore the baseline characteristics and prognostic factors of PT-DLBCL and establish a prognostic nomogram to assist clinicians in accurately estimating patient prognosis and to provide guidance and assistance in clinical treatment.
Methods
Patients and data collection
We extracted data for patients with PT-DLBCL diagnosed between 2000 and 2019 from the SEER database using SEER*STAT software (version 8.4.0.1). DLBCL was identified using International Classification of Diseases for Oncology histology codes (9680/3, 9675/3, 9684/3). 5 DLBCL originating from the testis was identified using lesion numbers C62.0, C62.1, and C62.9. 10
The inclusion criteria were defined as follows: histologically proven DLBCL; the primary site was the testis; and complete follow-up information. The exclusion criteria were defined as unknown Ann Arbor staging and missing survival information.
We collected the following clinicopathological features for included patients: age, race and ethnicity, marital status, laterality, year of diagnosis, Ann Arbor stage, therapy modality (surgery, surgery + chemotherapy, surgery + chemotherapy + radiation, surgery + radiation, and other), and survival outcomes (survival months, vital status, cause of death). Patients were randomly assigned to the training set and validation set at a ratio of 7:3.
Overall survival (OS) and cancer-specific survival (CSS) were the endpoints of interest, which were defined as duration of survival from the diagnosis of PT-DLBCL to the last follow-up/death from any cause or PT-DLBCL, respectively.
Owing to the nature of this retrospective study and the use of publicly accessible data from the SEER database, there was no requirement for ethics approval and patient informed consent in the current study.
Construction and validation of the nomogram
We conducted univariate and multivariate Cox regression analyses to determine the independent prognostic variables and significant factors associated with OS or CSS. On this basis, we developed a nomogram to predict OS and CSS at 3 years and 5 years. Performance of the nomogram was assessed using Harrell’s concordance index (C-index), calibration curves, and the area under the receiver operator characteristic (ROC) curve (AUC). Discriminative ability was quantitatively assessed using the C-index, and calibration was assessed with calibration curves.
Statistical analysis
We used R statistical software, version 4.2.1 (The R Project for Statistical Computing, Vienna, Austria) for all statistical analyses. The results of univariate and multivariate Cox risk regression analyses are expressed as the hazard ratio (HR) and corresponding 95% confidence interval (CI). The Cox regression model, nomogram, calibration curve, and ROC curve were analyzed using the R packages “survival” and “foreign,” “rms,” “ggplot2,” and “survivalROC,” respectively. The function “stdca. R” was used to perform DCA. The calibration reflected the consistency between the prediction probability of the model and the actual observation results. DCA was carried out to assess the nomogram’s clinical usefulness. Kaplan–Meier (KM) survival curves were drawn by dividing the mortality rates into high-risk and low-risk groups using the maximum Jordan index as the best cutoff value. Log-rank tests were used to compare differences between high-risk and low-risk groups. 11 All statistical tests were two-sided, and p < 0.05 indicated a significant difference.
Results
Baseline characteristics of patients
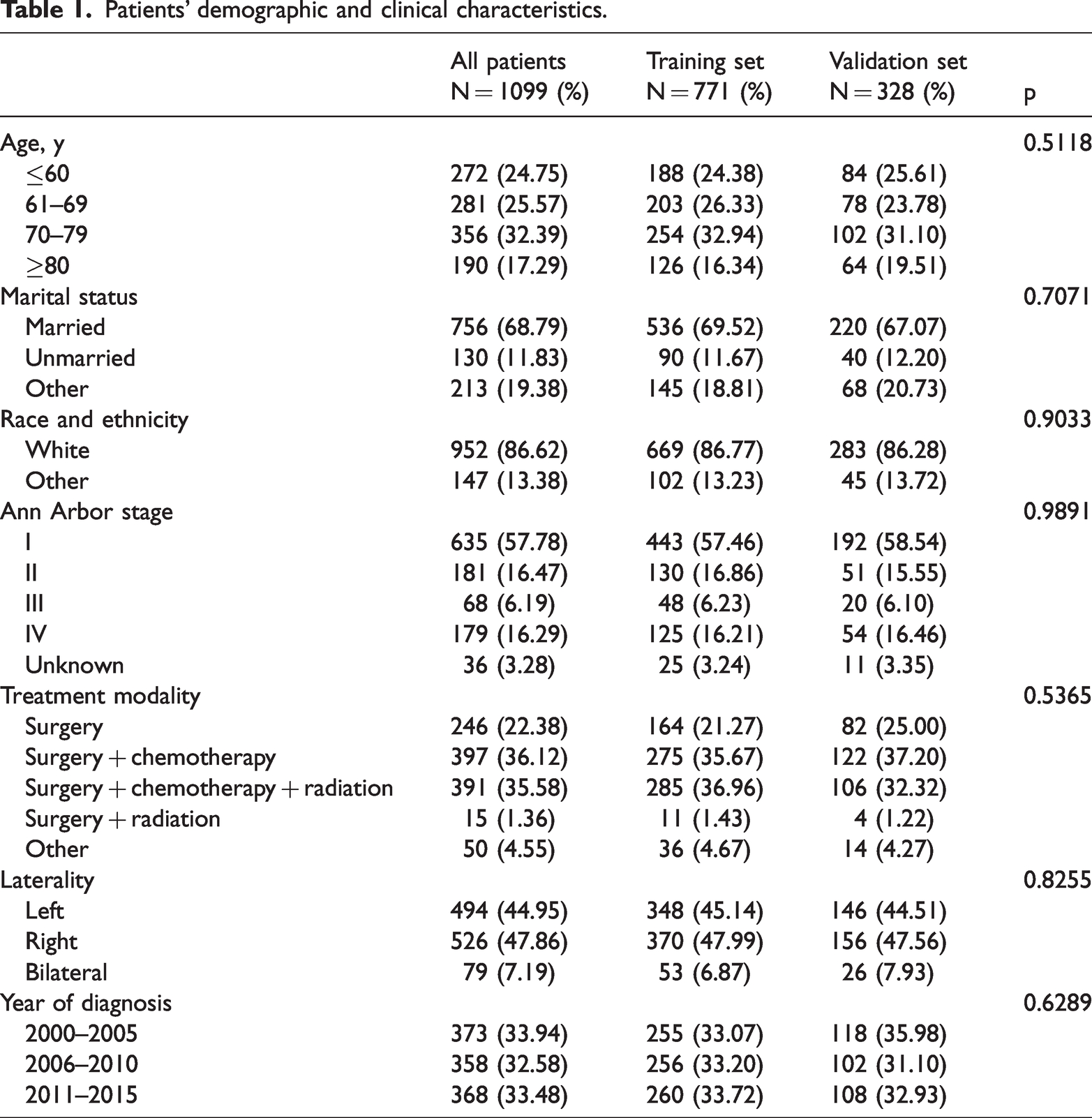
Data for a total of 1099 eligible patients diagnosed with PT-DLBCL from 2000 to 2019 were collected from the SEER database. These patients were then randomly assigned to training (n = 771) and validation (n = 328) cohorts at a ratio of 7:3. The demographic and clinicopathological features are compiled in Table 1. Patients’ year of diagnosis was divided into three groups: 2000 to 2005, 2006 to 2010, and 2011 to 2015. Most patients were White (n = 952, 86.62%); the remaining 147 patients (13.38%) were of other/unknown race and ethnicity. Patients’ ages were as follows: 272 (24.75%) patients were aged ≤60 years, 281 (25.57%) were aged 61 to 69 years, 356 (32.39%) were aged 70 to 79 years, and 190 (17.29%) patients were aged ≥80 years. Unilateral testicular involvement was found in 1020 (92.81%) patients and bilateral testicular involvement was observed in only 79 (7.19%) patients. More than half of patients (n = 756, 68.79%) were married. Most patients had Ann Arbor stage I (n = 635, 57.78%), and 16.47%, 6.19%, 16.29%, and 3.28% of patients had stage II, III, IV, and unknown, respectively. Over 95.4% (n = 789) of patients received surgery whereas 65.9% and 70.5% of patients underwent radiation therapy and chemotherapy, respectively.
Patients’ demographic and clinical characteristics.
Identifying independent prognostic factors for OS and CSS
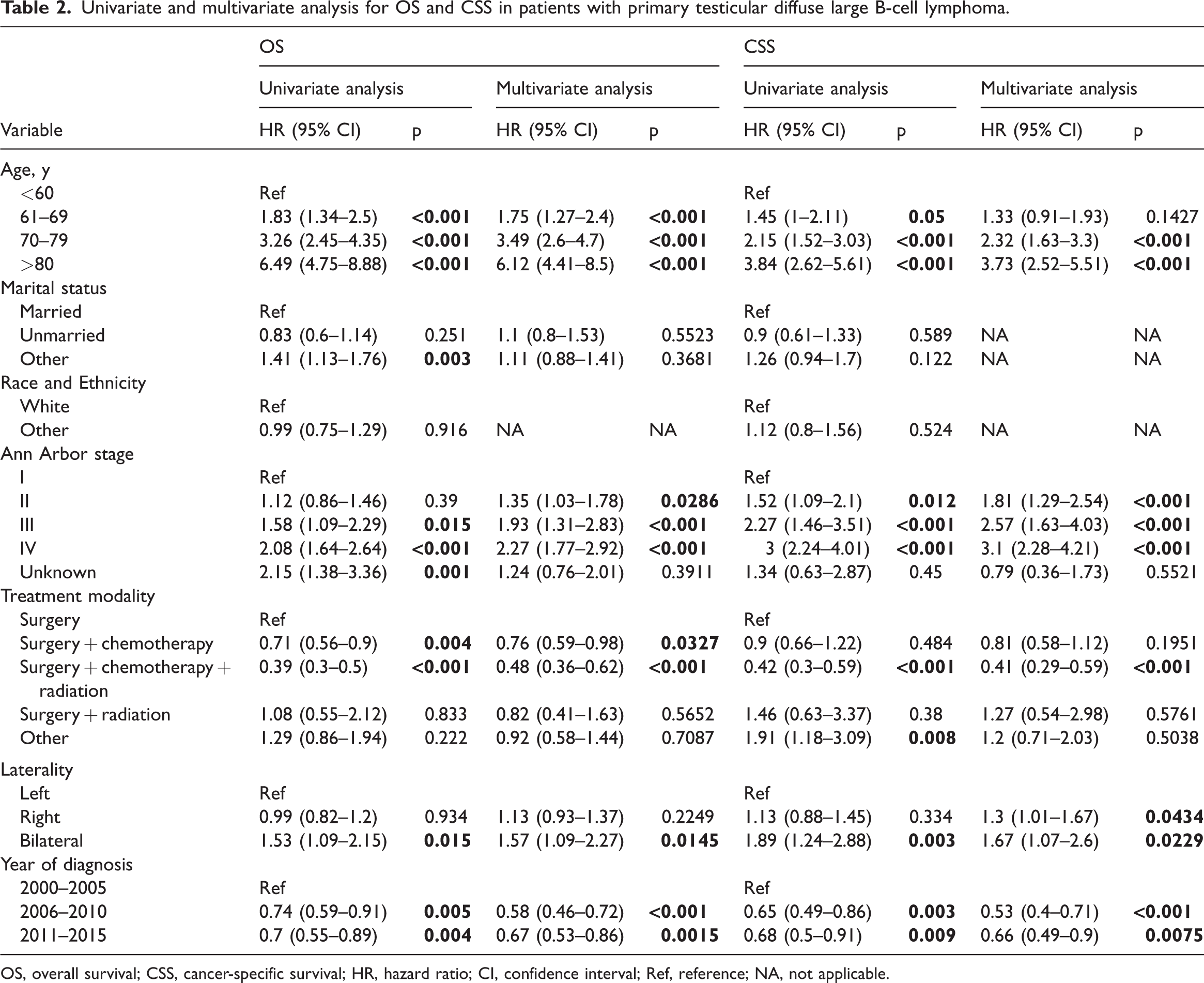
The results of univariate and multivariate analyses are presented in Table 2. Cox regression analysis was performed for seven potential parameters in the training cohort for the purpose of determining independent prognostic factors for survival in patients with PT-DLBCL.
Univariate and multivariate analysis for OS and CSS in patients with primary testicular diffuse large B-cell lymphoma.
OS, overall survival; CSS, cancer-specific survival; HR, hazard ratio; CI, confidence interval; Ref, reference; NA, not applicable.
Finally, age, year of diagnosis, Ann Arbor stage, treatment modality, and laterality were identified as independent prognostic factors for OS and CSS.
Construction of the nomogram
The prognostic nomogram for 3- and 5-year OS and CSS are shown in Figure 1. By adding a score on the point scale of each selected variable, a patient's probability of individual survival can be easily calculated. The OS was better for younger patients, patients with early Ann Arbor stage, and patients who were diagnosed from 2006 to 2010. After surgery, patients who received chemotherapy and radiation therapy also exhibited better OS. We found that left-side involvement was associated with better outcomes. Additionally, the prognostic nomogram for 3- and 5-year CSS of PT-DLBCL was similar to that for OS, in general.

Prognostic nomogram of patients with primary testicular diffuse large B-cell lymphoma based on five risk factors for overall survival and cancer-specific survival.
Validation of the nomogram
The nomogram was validated internally. The C-indices in the training and validation cohorts of the OS nomogram were 0.702 (95% CI 0.677–0.727) and 0.705 (95% CI 0.67–0.74), and those of the CSS nomogram were 0.694 (95% CI 0.663–0.725) and 0.680 (95% CI 0.63–0.72), respectively, indicating considerably good discrimination of the newly established nomogram (Figure 2).

Calibration plot of the prognostic nomogram for each cohort. (a) The 3- and 5-year OS calibration plots for the development set; (b) 3- and 5-year CSS calibration plots for the development set; (c) 3- and 5-year OS calibration plots for the validation set and (d) 3- and 5-year CSS calibration plots for the validation set.
DCA curves in the training cohort for the OS nomogram and CSS nomogram separately demonstrated great consistency between actual and predicted survival. Analogously, calibration curves in the validation cohort and all patients demonstrated a consistent pattern (Figure 3).

Prognostic DCA. (a) The 3-year and 5- year OS survival DCA for the development set; (b) 3- and 5-year CSS survival DCA for the development set; (c) 3- and 5-year OS survival DCA for the validation set and (d) 3- and 5-year CSS survival DCA for the validation set.
We drew ROC curves to verify the ability of the nomogram to accurately predict survival. In the training cohort, the 3- and 5-year AUCs of the OS nomogram were 0.77 and 0.779 (Figure 4a) and those of the CSS nomogram were 0.789 and 0.779 (Figure 4b), respectively, showing good sensitivity and specificity for the nomogram. In the validation cohort, the 3- and 5-year AUCs of the OS nomogram were 0.764 and 0.78 (Figure 4c), and those of the of the CSS nomogram were 0.752 and 0.742 (Figure 4d). Therefore, the nomogram performed well.

ROC curves. (a) The 3- and 5-year ROC curves of the development set for OS; (b) 3- and 5-year ROC curves of the development set for CSS; (c) 3- and 5-year ROC curves of the validation set for OS and (d) 3- and 5-year ROC curves of the validation set for CSS.
Survival analysis
Patients with mortality lower than the cutoff point were defined as the low-risk group. The remaining patients were defined as the high-risk group, and a log-rank test was used for comparison (Figure 5). The optimal cutoff points for 3- and 5-year OS mortality in the development cohort were 0.263 and 0.362, respectively. Figure 5a shows the development cohort probability of survival at 3 years in the low-risk group (0.84, 95% CI 0.81 to 0.88; p < 0.0001) and high-risk group (0.53, 95% CI 0.48–0.58; p < 0.0001). Figure 5b shows the development cohort probability of survival at 5 years in the low-risk group (0.78, 95% CI 0.74–0.82; p < 0.0001) and high-risk group (0.40, 95% CI 0.35–0.45; p < 0.0001). The optimal cutoff points for 3- and 5-year CSS mortality in the development cohort were 0.257 and 0.338, respectively. In the development cohort, the 3-year survival was 0.85 (95% CI 0.82–0.88; p < 0.0001) and 0.61 (95% CI 0.55–0.67; p < 0.0001) in the low-risk and high-risk groups, respectively (Figure 5c). The 5-year survival rates were 0.80 (95% CI 0.76–0.84; p < 0.0001) and 0.50 (95% CI 0.45–0.57; p < 0.0001) in the low-risk and high-risk groups, respectively (Figure 5d).

KM survival curves. (a) Development set: KM survival curve based on 3-year OS mortality. (b) Development set: KM survival curve based on 5-year OS mortality. (c) Development set: KM survival curve based on 3-year CSS mortality and (d) Development set: KM survival curve based on 3-year CSS mortality.
Discussion
PT-DLBCL is a rare and aggressive type of lymphoma that mainly affects older men. 5 , 12 As such, PT-DLBCL research has been limited to case reports and small-sample retrospective studies.13–15 Some clinical features such as patient age at diagnosis, stage, and absolute lymphocyte counts in the peripheral blood have been shown to be prognostic factors in PT-DLBCL. No specific prognostic score has yet been developed for PT-DLBCL. 16 , 17
Our study was the first to develop and validate a clinical prognostic model for PT-DLBCL using the SEER database. We included the information of 1099 patients diagnosed with PT-DLBCL from SEER. Using Cox multivariate analysis, we found that age, treatment modality, year of diagnosis, Ann Arbor stage, and laterality were risk factors associated with survival. To the best of our knowledge, this is the largest study to construct a visual nomogram to predict the 3- and 5-year OS and CSS in patients with PT-DLBCL. The good predictive and clinical net benefits of the developed nomogram were demonstrated using the ROC, calibration, and DCA curves. The developed nomogram can help clinicians predict survival in individual patients and make treatment decisions.
According to previous studies, the median age at diagnosis in patients with PT-DLBCL is 68 years. In our study cohort, patients were slightly older (70 years) than those in past studies. Gundrum et al. reported that approximately 70% of patients with PT-DLBCL were in stage I or II at the first diagnosis. 5 In our study, most patients (74.52%) were diagnosed in the early stage of the disease (stage I–II), which is in accordance with previous reports. Our prognostic nomogram for patients with PT-DLBCL also confirmed that compared with patients aged ≤60 years, those older than age 80 years had a 3 to 7 times higher risk of death in terms of OS and CSS. Additionally, patients in earlier stages had longer OS and CSS than those in stages III and IV, which is consistent with previous findings that advanced age and stage are prognostic factors of adverse outcomes. 2 , 10 , 18
The relevance of laterality in the prognosis of PTL is controversial. A 2009 study including 769 cases in the SEER database showed that lymphoma involvement of the right testis is associated with improved CSS. 5 Another study from China including 32 cases conducted in 2011 showed that left testicular involvement was a negative prognostic factor for OS. 3 Unlike previous studies, our results showed that bilateral involvement was significantly associated with worse outcomes. Bilateral testicular swelling was observed in 7.19% of patients in our study; this low proportion may have influenced the results. Another possible explanation was that the low incidence of PTL and the population analyzed were distinct, which may represent an unknown bias in the selection. In general, a larger amount of research evidence from real-world settings is needed.
A large retrospective study including 1169 patients reported that a later period at diagnosis was a good prognostic factor of survival in PTL. 10 We also found that the OS and CSS of patients diagnosed during 2006 to 2010 and 2011 to 2015 were significantly improved compared with those of patients diagnosed in 2000 to 2005. This improvement in patients’ survival may be attributable to advancement in treatment strategies and increased access to therapy for patients. In 2017, Kridel et al. indicated that rituximab was associated with significantly improved progression-free survival (PFS) and OS. 19
The selection of therapeutics is an important factor in survival of PT-DLBCL. Owing to the lack of randomized phase III trials, no standard treatment for PT-DLBCL has yet to be established. The European Society for Medical Oncology Clinical Practice Guidelines state that orchiectomy remains a mandatory step in therapy to remove a potential sanctuary site formed by the blood–testis barrier and provide histological samples for diagnosis. In our analysis, 95.5% of patients underwent orchiectomy. A retrospective study including 769 patients demonstrated that prophylactic radiation to the contralateral testicle prevented testicular relapse and was also associated with better survival. Interestingly, however, patients in our cohort who received radiation after surgery had no significant outcomes compared with those who did not. The relatively small sample size (only 15 patients with PT-DLBCL) and older patient age (>70 years) may understate the role of radiotherapy and may introduce potential bias. The recommendations of prior phase II trials promote the use of combination therapy: surgical resection, chemotherapy associated with CNS prophylaxis, and radiotherapy of the contralateral testicle. 20 The use of multimodal therapy is associated with an increase in the 5-year survival rate from 30% to 86.6%.21–23 Xu et al. also reported that chemotherapy and radiotherapy following orchiectomy are associated with a significantly longer PFS. 10 Our study showed that patients who received combined modality therapy had a better prognosis, which is consistent with previous findings that combining radiotherapy with chemotherapy and surgery results in favorable OS. However, patients with PT-DLBCL are usually older and many may not tolerate combined regimens. In real-world settings, final decisions regarding the therapy regimen should be made by the patients.
Study limitations
This study has some limitations that should be taken into consideration. First, we only used the SEER database and did not use information from other databases for external validation. Second, some potential independent prognostic variables, such as biomarkers, B symptoms, and IPI, were lacking in our analysis; this lack of information should be addressed in future research. Third, the SEER database provides no information regarding chemotherapy and rituximab use.
Conclusion
We developed and validated a nomogram for predicting 3- and 5-year OS and CSS in patients with PT-DLBCL. The nomogram provided accurate individual survival predictions, which will help physicians make appropriate clinical decisions for individual patients.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231197052 - Supplemental material for Development and validation of a survival nomogram in patients with primary testicular diffuse large B-cell lymphoma
Supplemental material, sj-pdf-1-imr-10.1177_03000605231197052 for Development and validation of a survival nomogram in patients with primary testicular diffuse large B-cell lymphoma by Yongjin Zhi, Shuojing Bao, Jingcheng Mao, Gufan Chai, Chengjiang Liu and Jianfeng Zhu in Journal of International Medical Research
Footnotes
Authors’ contributions
All authors contributed to the article and approved the submitted version.
YZ contributed to conceiving and designing the study.
JM and SB were mainly responsible for data analysis.
GC and CL contributed to article writing.
JZ reviewed and edited the manuscript.
Data availability
The data used in this study are available from the Surveillance, Epidemiology, and End Results (SEER) research database, a public research database.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
