Abstract
Objective
To develop a prognostic model for Chinese patients with relapsed diffuse large B-cell lymphoma (DLBCL) after initial R-CHOP therapy.
Methods
We retrospectively analyzed the characteristics and survival outcomes of 79 patients with relapsed DLBCL initially treated with R-CHOP at Peking Union Medical College Hospital from February 2012 to September 2016. We used the data to develop a novel prognostic model.
Results
The median age at the start of salvage therapy was 59 (17–85) years and median time from diagnosis to relapse was 319 (49–1018) days. Multivariate analysis identified short time to relapse (TTR) and B symptoms as independent prognostic factors for reduced progression-free survival (PFS) and overall survival (OS). We created a new prognostic scoring system including TTR, lactate dehydrogenase, absolute lymphocyte count at relapse, and B symptoms, referred to as the TLLB model, which could separate patients into three risk groups with 2-year PFS and OS rates of 70.7%, 40.0%, and 11.1%, and 87.5%, 53.7%, and 29.4%, respectively.
Conclusion
TTR and B symptoms can be used as important predictors of survival in patients with DLBCL. The TLLB system provides a useful prognostic model compared with the previous TTL system.
Keywords
Introduction
Diffuse large B-cell lymphoma (DLBCL) is a heterogeneous and invasive entity with a highly variable prognosis. 1 Rituximab with cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) has been established as the standard initial therapy for this disease, with about 60% of patients being cured.2,3 However, many patients still experience chemoresistance or relapse, and there is currently no standard second-line therapy. The outcome of DLBCL is also diverse and there are no well-established prognostic models.4,5 It is therefore difficult for clinicians to inform patients about their prognosis, and to identify patients with anticipated poor outcomes to receive more aggressive or innovative treatments.
Former studies have identified certain factors which could predict outcome. For example, the International Prognostic Index (IPI) at relapse (IPI-R) was associated with clinical response in relapsed/refractory (R/R) patients in the PARMA trial. 6 Moreover, a short time to relapse (TTR) and low absolute lymphocyte count (ALC) at relapse (ALC-R) have been also associated with poorer clinical outcomes in DLBCL patients at first relapse.7,8 However, these studies were conducted before the rituximab era, and contradictory results have been obtained after the introduction of rituximab-containing treatments. The National Comprehensive Cancer Network (NCCN) IPI was reported as an independent valid predictor in R/R patients. 9 However, another study found that IPI-R failed to predict the objective response rate to second-line treatment. 10 In 2017, Yamamoto et al. 11 proposed a simple prognostic model combining lactate dehydrogenase (LDH) ratio, TTR, and ALC-R, referred to as the TLL model, which could separate patients into three risk groups with different progression-free survival (PFS) rates. However, this model was based on a limited number of patients and thus required verification. We therefore aimed to validate the TLL model in a larger cohort and to explore possible more suitable prognostic models for Chinese patients.
Patients and methods
This was a single-center retrospective analysis of patients with DLBCL undergoing salvage chemotherapy after initial treatment with R-CHOP at Peking Union Medical College Hospital. The study was approved by Peking Union Medical College Hospital Institutional Review Board (approval number ZS-2181). All patients provided written informed consent for participation in the study.
Patients
We retrospectively evaluated patients diagnosed with relapsed DLBCL who were treated at Peking Union Medical College Hospital from February 2012 to September 2016. The inclusion criteria were patients with biopsies classified as DLBCL (according to the World Health Organization classification); patients in first relapse after initial treatment with R-CHOP regimen; and patients with intact medical records. Patients with meningeal or central nervous system involvement were excluded. All patients were re-staged by positron-emission tomography combined with computed tomography or computed tomography. We re-assessed the laboratory data and prognostic factors at the start of the first salvage therapy. All patients were followed up until 30 September 2017.
Treatment and response assessment
Patients with relapsed DLBCL mainly received DICE (dexamethasone, ifosfamide, cisplatin, etoposide)/DHAP (cisplatin, cytarabine, dexamethasone) (31.5%) with/without rituximab. Other regimens included MA (methotrexate, cytarabine)/ESHAP (etoposide, cisplatin, methylprednisolone, cytarabine), MINE (ifosfamide, mesna, mitoxantrone, etoposide), and GDP (gemcitabine, dexamethasone, cisplatin)/GEMOX (gemcitabine, oxaliplatin). Some patients received autologous stem cell transplantation after high-dose chemotherapy. According to the International Working Group criteria, 12 the disease status at the start of salvage therapy was classified as relapse, progression, or primary refractory. Response to salvage therapy was assessed by conventional diagnostic methods after the second or third chemotherapy course.
Statistical analysis
PFS was defined as the time from the start of salvage therapy to disease progression or death. Overall survival (OS) was defined as the time from the start of salvage therapy to death or date of last follow-up for patients who were alive and censored. Statistical analysis was performed using SPSS Statistics for Windows, Version 20.0 (SPSS Inc., Armonk, NY: IBM Corp). The Kaplan–Meier method coupled with the log-rank test was used for univariate analysis and to generate survival curves. All factors with a P value <0.10 in univariate analysis were included in the multivariate analysis using the Cox proportional hazards model. All reported P-values were two-sided, and a P-value <0.05 was considered significant. Receiver operating characteristic (ROC) and area under the curve (AUC) analyses were used to determine the ALC-R cutoff value.
Results
Patient characteristics
Seventy-nine patients were included in this study, of whom 76 received salvage therapy. The clinical characteristics of the patients at diagnosis and at first relapse are summarized in Table 1. Most patients were stage III/IV and nearly half of all patients were in the high-intermediate or high-risk groups according to the NCCN-IPI.
Patient characteristics.

GCB, germinal center B-cell like; ALC, absolute lymphocyte count; AMC, absolute monocyte count; LDH, lactate dehydrogenase; TTR, time from diagnosis to relapse; IPI, International Prognostic Index; HI, high-intermediate; H, high; ASCT, autologous stem cell transplantation; CR, complete response; PR, partial response; MA, methotrexate, cytarabine; ESHAP, etoposide, cisplatin, methylprednisolone, cytarabine; MINE, ifosfamide, mesna, mitoxantrone, etoposide; DICE, dexamethasone, ifosfamide, cisplatin, etoposide; DHAP, cisplatin, cytarabine, dexamethasone; GDP, gemcitabine, dexamethasone, cisplatin; GEMOX, gemcitabine, oxaliplatin; R-CHOP, rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone; R, rituximab; MTX, methotrexate; EA, etoposide, cytarabine.
Response to salvage therapy
Most patients with relapsed DLBCL in our center received DICE/DHAP (34.2%) or MA/ESHAP (19.0%) as salvage therapy, while other regimens included MINE (15.2%) and GDP/GEMOX (12.7%) (Table 1). The choice of regimen was based on comprehensive consideration and depended on the individual patient’s status. The patients’ overall responses to salvage therapy are summarized in Table 2. Three patients were lost during follow-up, leaving 76 patients for analysis.
Patient responses to salvage therapy.

CR, complete response; PR, partial response; ECOG, Eastern Cooperative Oncology Group; PS, performance status; ALC-R, absolute lymphocyte count at relapse; TTR, time from diagnosis to relapse; LMR-R: lymphocyte/monocyte ratio at relapse; IPI-R, IPI at relapse. Extranodal specific site: bone marrow, central nervous system, liver/gastrointestinal tract, or lung.
The overall response rate (i.e. complete response plus partial response) for the whole cohort was 61.8%. Forty-seven patients responded to salvage treatment and 29 did not. We classified the whole population into two groups based on response, and analyzed the associations between prognostic factors and clinical outcome. The factors that significantly affected the overall response rate included specific extranodal sites, such as the bone marrow, liver/gastrointestinal tract, or lung (involved vs. not involved, P=0.020), B symptoms (yes vs. no, P=0.024), TTR (≤12 vs. >12 months, P=0.018), and ALC-R (≤985 vs. >985/µL, P=0.039). Refined LDH categorization failed to add any extra benefit for predicting prognosis compared with dichotomized LDH, with similar P-values for both factors. Notably, ALC-R was significantly lower in unresponsive patients (P=0.039). The cutoff values of ALC and lymphocyte–monocyte ratio (LMR) were determined by ROC and AUC analyses.
Survival analysis based on therapeutic response
The whole cohort was followed until 30 September 2017. After a median follow-up of 386 (19–1840) days, 35 (44.3%) patients were still alive and 36 patients had died (45.6%). Among all 79 patients, 53 (67.1%) patients experienced disease progression or relapse. The median time from the start of salvage therapy to relapse was 134 (13–1339) days. The calculated 2-year PFS and 2-year OS rates were 12.7% and 22.8%, respectively. The Kaplan–Meier survival curves showed that patients who responded to salvage therapy had better prognoses than those who failed to respond, with significant differences between the groups in terms of 1-year PFS (57.4% vs. 0%) and OS (76.6% vs. 27.6%) (both P=0.001) (Figure 1). It was expected that a better response to salvage therapy would be associated with a better prognosis.
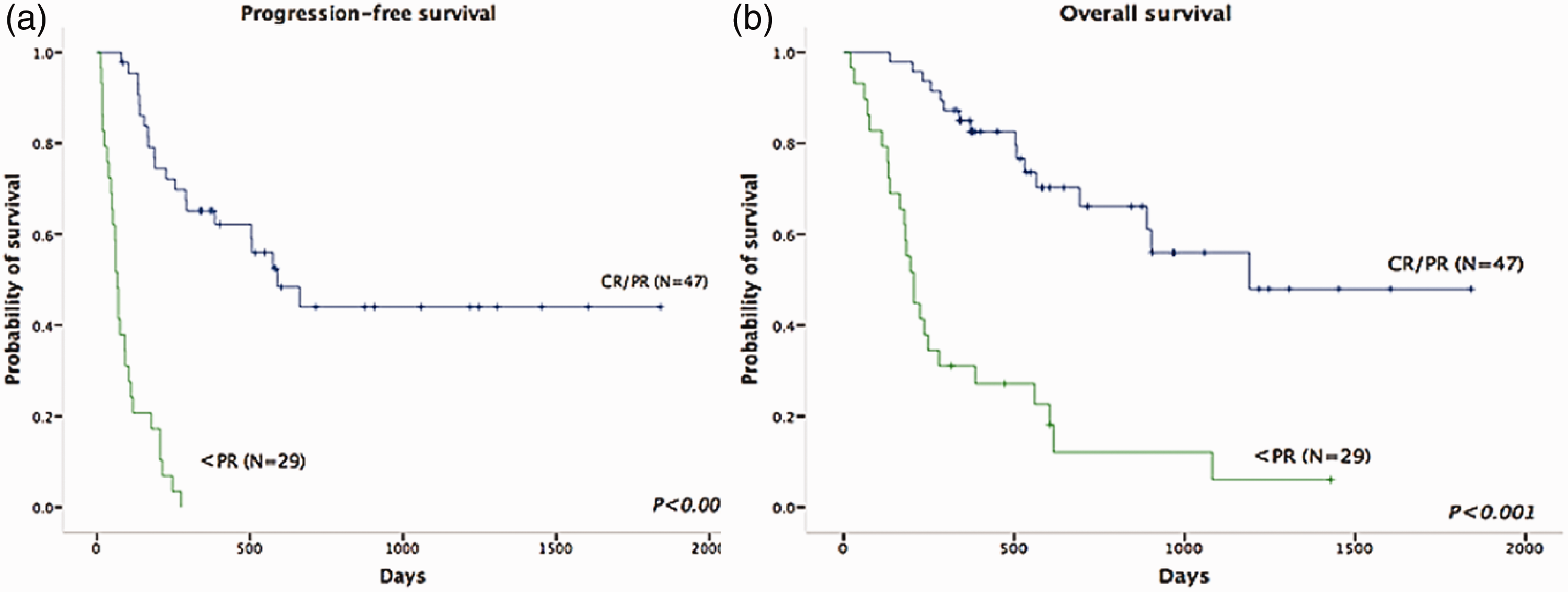
Impact of first salvage therapy on response in patients with relapsed diffuse large B-cell lymphoma. Impacts of first salvage therapy on (a) progression-free survival (PFS) and (b) overall survival (OS). The 1-year PFS (57.4% vs. 0%) and OS (76.6% vs. 27.6%) rates were significantly higher in patients who responded to salvage therapy (P=0.001).
Survival analysis based on prognostic factors
We conducted a Cox proportional hazards regression analysis to determine how the prognostic factors could predict the clinical outcome. We conducted univariate analysis and generated survival curves using the Kaplan–Meier method coupled with the log-rank test. Univariate analysis (Table S1) identified TTR, LDH level, B symptoms, Eastern Cooperative Oncology Group (ECOG) performance status (PS), NCCN-IPI, and extranodal specific sites as significantly associated with PFS, while multivariate analysis (Table S2) confirmed a short TTR and B symptoms as significant predictors of reduced PFS. Regarding OS, ECOG PS, B symptoms, extranodal specific sites, LDH level, ALC-R, LMR at relapse (LMR-R), and TTR significantly affected OS in the univariate analysis, and short TTR and B symptoms also predicted reduced OS in multivariate analysis (Table S2).
Validation of TLL prognostic model in our patients
Yamamoto et al.’s, 11 TLL model included three prognostic factors: TTR, LDH categorized ratio, and ALC-R. This model included a maximum of 5 points: TTR >12 months=0 points, ≤12 months=1 point; categorized LDH ratio ≤1=0 points, >1–3=1 point, >3=2 points; and ALC-R ≥1100/µL=0 points, <1100/µL=1 point. This model separated patients in to low- (0 points), intermediate- (1 point), and high- (≥2 points) risk groups with 2-year PFS rates after first salvage therapy of 100%, 68.6%, and 4.8%, respectively (P<0.0001). However, their study only included 31 patients, and the model therefore required validation. We therefore tested this model in our group of 79 patients. Twelve patients were classified as low-risk (0 points), 21 as intermediate-risk (1 point), and 43 as high-risk (≥2 points). The 2-year PFS rates of these three subgroups were 72.9%, 38.1%, and 10.4%, (P<0.001) and the 2-year OS rates were 88.9%, 48.3% and 29.0% (P<0.001), respectively (Figure S1). These results confirmed that the TLL model could efficiently discriminate between low-risk and high-risk populations. Considering that some patients failed to respond to salvage treatment, which would affect their survival rates, we also applied this model in responding patients (Figure S2). Responding patients had obviously better PFS and OS. For responding patients, a higher TLL score was also associated with poorer PFS and OS outcomes.
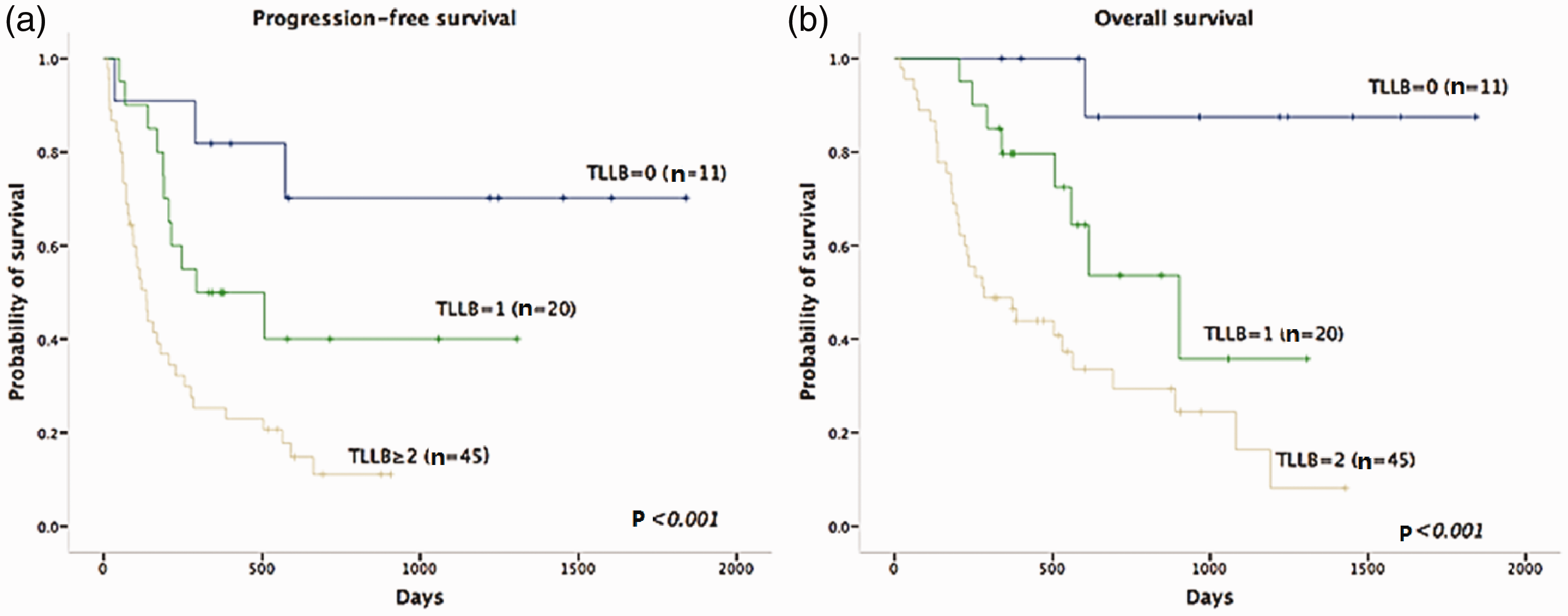
Survival probabilities of patients with relapsed diffuse large B-cell lymphoma (DLBCL) based on TLLB scoring system. (a) Progression-free survival (PFS) and (b) overall survival (OS) of first-relapse DLBCL patients using TLLB scoring system: low-risk (TLLB=0), intermediate-risk (TLLB=1), and high-risk (TLLB ≥2). The 2-year PFS and OS rates after salvage therapy were 70.7%, 40%, and 11.1% and 87.5%, 53.7%, and 29.4%, respectively (all P<0.001).
Proposed new prognostic model
The current study identified B symptoms as an independent prognostic factor for both PFS and OS. In addition, although LDH level was significantly associated with PFS and OS in univariate analysis, its function was not confirmed in multivariate analysis, and a refined categorization of LDH value offered no prognostic advantage over mere LDH abnormality. We therefore developed the TLLB prognostic model combing TTR (>12 months=0 points; ≤12 months=1 point), ALC-R (≥985/µL=0 points; <985/µL=1 point), LDH abnormality (≤ upper limit of normal (ULN)=0 points, >ULN=1 point), and B symptoms (no=0 points; yes=1 point), with a maximum of 4 points. We tested this model by classifying patients into three groups based on the scores using Kaplan–Meier curves: low-risk (0 points, n=11), intermediate-risk (1 point, n=20), and high-risk (≥2 points, n=45). After 2 years of the first salvage therapy, the PFS rates for these three subgroups were 70.7%, 40.0%, and 11.1% (P<0.001) and the OS rates were 87.5%, 53.7%, and 29.4% (P<0.001), respectively (Figure 2.). The TLLB model could efficiently discriminate between low-risk and high-risk populations, similar to the TLL model. However, because we used LDH abnormality instead of more detailed LDH categorization, the TLLB model was easier to apply. Also, as an independent prognostic factor, B symptoms were included as an eligible factor for predicting outcome.
Discussion
The current study validated the TLL model proposed by Yamamoto et al. 11 in a larger cohort and confirmed its utility for predicting the outcome of patients with R/R DLBCLs after initial treatment with R-CHOP. We also proposed a new model, referred to as the TLLB model. This model was easier to use, by including LDH as simply normal or abnormal. We also included B symptoms as an independent prognostic factor (P=0.004) for outcome, as reported in a previous Chinese study. 13 We interpreted this as indicating that B symptoms reflected typical characteristics of Chinese patients with DLBCL, suggesting that the TLLB model was more suitable for Chinese patients.
Despite progress in the upfront treatment of DLBCL, patients still experience relapses. There are currently no optimized salvage chemotherapy regimens for R/R DLBCL, and the outcomes of these patients thus vary. Patients who respond to second-line regimens followed by transplantation could achieve long-term survival, while patients resistant to salvage therapy have poor outcomes. 4 The values of new therapies, such as novel agents or chimeric antigen receptor T-cell therapy for patients with R/R DLBCL need further study.14–16 It is therefore necessary to identify patients likely to be resistant to chemotherapy before making a treatment plan.
Some previous studies addressed the possible prognostic factors for R/R DLBCLs. IPI has been relatively well-studied. IPI was first established in 1993 for CHOP and CHOP-like regimens before the rituximab era, and is currently used because of its validity and convenience. 17 However, its use has been questioned, given that most trials were conducted before the introduction of rituximab. Ziepert et al. 18 reported that IPI was still valid in 1037 untreated patients, but Panizo et al. 10 found that IPI-R failed to predict the overall response. 10 . In accordance with this report, our study showed that IPI-R was not significantly associated with PFS or OS. NCCN-IPI was better able to discriminate between low- and high-risk subgroups of untreated DLBCL patients in the rituximab era, 9 but has not been tested in relapsed DLBCL patients. The current study showed that a high NCCN-IPI score was significantly associated with shorter PFS but not OS. TTR has also been identified as an indicator of outcome, and patients with a TTR >1 year had good outcomes. 7
In addition to DLBCL, TTR has proved effective in many kinds of non-Hodgkin’s lymphomas, and could be used to stratify patients at time of first relapse of intermediate to high-grade non-Hodgkin’s lymphoma in the PARMA trial. 19 Moreover, disease progression within 2 years was associated with poorer outcomes in patients with follicular lymphoma, 20 and early relapse remained a marker of poor prognosis in patients with myeloma, despite use of novel agents. 21 In the current study, TTR was a strong predictor of both PFS and OS, and could reflect aggressive tumor biological characteristics as well as chemosensitivity. Much attention has also been paid to the tumor microenvironment. ALC-R is used as a surrogate marker of host immunity and tumor growth, and several studies have addressed the prognostic value of ALC-R in relapsed DLBCL patients. Panizo et al. 10 reported that low ALC-R, rather than IPI, was the only prognostic factor that could predict OS after second-line treatment, and Porrata et al. 8 also found that ALC-R predicted survival. 8 The LMR has also been evaluated, and lower LMR-R was found to be an adverse prognostic factor for PFS and OS in patients with relapsed DLBCL.22,23 In accordance with these studies, the current study showed that ALC-R and LMR-R significantly affected OS but not PFS.
Based on the above findings, IPI-R/TTR and IPI-R/ALC-R have been proposed as prognostic models.7,8 However, these models were not developed in the rituximab era, which limits their clinical use. The TLL model was developed for R/R DLBCL after initial therapy with R-CHOP, and could effectively classify patients into three risk groups with different outcomes; however, it was only tested in 31 patients, and we confirmed the efficacy of the TLL model in our cohort of 79 patients. Given that B symptoms were also strongly related to PFS and OS in our study, we added B symptoms to the TLL model to create the TLLB model. In addition, because LDH categorization was not superior to simple dichotomization in our study, we used normal or abnormal LDH levels in our model, making it easier for clinicians to use.
This study had some limitations. The patients received different salvage regimens, which was also the case in Yamamoto et al.’s 11 study. Neither study compared these treatment in uni- or multivariate analyses, because no evidence has indicated the superiority of any specific second-line therapy. For instance, the most widely used salvage therapies, R-DHAP and R-ICE, had similar impacts on DLBCL at first relapse, according to the CORAL study. 24
The other limitations of the current study were principally those typically associated with observational and retrospective studies. Because the information was collected retrospectively, the intactness and homogeneity of the information could not be ensured. The results were also limited by the relatively small sample size. Moreover, increasing evidence25,26 has shown that tumor biological characteristics affect the prognosis, and our study did not include information on immunohistochemical or molecular markers, such as Bcl-2, Bcl-6, Myc, and p53, and we did not evaluate the effect of double-expressor or double-hit lymphomas. 27
Despite these limitations, this study summarized the reliable prognostic factors that might aid the early identification of patients at high risk of poorer outcomes, who should be targeted for clinical trials. The results suggest that future clinical trials might identify novel agents with greater efficacy in patients with R/R DLBCL.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211002971 - Supplemental material for Novel model predicts prognosis for patients with diffuse large B-cell lymphoma in first relapse after initial R-CHOP therapy: a single-institution study in China
Supplemental material, sj-pdf-1-imr-10.1177_03000605211002971 for Novel model predicts prognosis for patients with diffuse large B-cell lymphoma in first relapse after initial R-CHOP therapy: a single-institution study in China by Wei Wang, Jingjing Yin, Wei Zhang, Yan Zhang, Daobin Zhou, Danqing Zhao and Chong Wei in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605211002971 - Supplemental material for Novel model predicts prognosis for patients with diffuse large B-cell lymphoma in first relapse after initial R-CHOP therapy: a single-institution study in China
Supplemental material, sj-pdf-2-imr-10.1177_03000605211002971 for Novel model predicts prognosis for patients with diffuse large B-cell lymphoma in first relapse after initial R-CHOP therapy: a single-institution study in China by Wei Wang, Jingjing Yin, Wei Zhang, Yan Zhang, Daobin Zhou, Danqing Zhao and Chong Wei in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605211002971 - Supplemental material for Novel model predicts prognosis for patients with diffuse large B-cell lymphoma in first relapse after initial R-CHOP therapy: a single-institution study in China
Supplemental material, sj-pdf-3-imr-10.1177_03000605211002971 for Novel model predicts prognosis for patients with diffuse large B-cell lymphoma in first relapse after initial R-CHOP therapy: a single-institution study in China by Wei Wang, Jingjing Yin, Wei Zhang, Yan Zhang, Daobin Zhou, Danqing Zhao and Chong Wei in Journal of International Medical Research
Footnotes
Acknowledgements
We thank all the patients who participated in this study.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Beijing Municipal Natural Science Foundation [grant number 7202154].
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
