Abstract
Objective
De novo CD5-positive (CD5+) diffuse large B-cell lymphoma (DLBCL) has different clinical characteristics compared with CD5-negative (CD5−) DLBCL. However, few studies have been reported in Chinese cohorts. We investigated the clinical features and prognosis of patients with CD5+ DLBCL and summarized the related literature.
Methods
Data from 245 patients with newly diagnosed DLBCL were retrospectively assessed.
Results
Thirty-one and 214 patients were diagnosed with CD5+ DLBCL or CD5− DLBCL, respectively. In the CD5+ DLBCL group, there were significantly higher proportions of patients with older age (≥60 years), International Prognostic Index (IPI) ≥3, Eastern Cooperative Oncology Group (ECOG) scores ≥ 2, bone marrow involvement, positive B-cell lymphoma 2 expression, and positive MYC expression. Survival analysis showed that CD5+ DLBCL had a markedly poorer 2-year progression-free survival than CD5− DLBCL (18.2% vs. 56.2%). Univariate analysis indicated that age ≥60 years, ECOG score ≥ 2, IPI ≥ 3, B symptoms, and no rituximab-based treatment were poor predictive factors for overall survival (OS). Multivariate analysis revealed that B symptoms and no rituximab-based treatment, but not positive CD5 expression, were independent factors for OS.
Conclusions
Patients with CD5+ DLBCL had heterogeneous clinical characteristics and poor survival. The development of more targeted and effective therapies is needed.
Introduction
As the most common type of non-Hodgkin lymphoma (NHL) with a highly heterogeneous clinical course, diffuse large B-cell lymphoma (DLBCL) is a biologically distinct disorder with specific morphological, immunohistochemical, and molecular variants.1,2 CD5, a cell surface glycoprotein that mediates signal transduction, is mainly expressed in T cells and expressed at low levels in B cells. 3 In general, CD5-positive (CD5+) B-cell NHL is observed in chronic lymphocytic leukemia, mantle cell lymphoma, marginal zone B-cell lymphoma, Burkitt lymphoma, and 5% to 22% of DLBCL cases.4–8 CD5+ DLBCL is a specific subgroup with aggressive biological characteristics, such as a higher lactic dehydrogenase (LDH) level, bone marrow (BM) involvement, and central nervous system (CNS) involvement.9,10 With the current front-line chemotherapy regimen R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone), patients with CD5+ DLBCL have an inferior survival rate, with a 5-year overall survival (OS) of only 35.5% compared with 64.8% in patients with CD5− DLBCL. 4 Moreover, there are still no standard therapies for patients with CD5+ DLBCL. Some studies have reported that even intensive chemotherapy, such as the dose-adjusted chemotherapy regimen EPOCH (etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin) plus rituximab (R-DA-EPOCH) or autologous hematopoietic stem cell transplantation (auto-HSCT), may not achieve promising OS in patients with CD5+ DLBCL.11–13
Currently, large-scale studies on de novo CD5+ DLBCL are relatively rare because of the limited cases.14–16 Moreover, most reports are from the Western world, with few studies in Chinese cohorts.5,17 Recently, novel treatments, such as chimeric antigen receptor T-cell therapy or targeted drugs, have been reported to be effective for patients with CD5+ DLBCL.18–20 In the present study, we aimed to explore the clinical characteristics and prognostic factors of patients with de novo CD5+ DLBCL in our center. Moreover, we summarized the related literature to further improve the recognition and understanding of this heterogeneous subtype of DLBCL.
Patients and methods
Patients and tests
We collected and retrospectively analyzed the relevant information from patients with newly diagnosed DLBCL who were admitted to the First Affiliated Hospital of Nanchang University from January 2014 to January 2019. Patients with unspecified DLBCL were included in our study. Patients with primary mediastinal (thymic) B-cell lymphoma, anaplastic lymphoma kinase-positive large B-cell lymphoma, plasma cell lymphoma, lymphoma tumorous granulomatous disease, and intravascular large B-cell lymphoma were excluded. Patients were diagnosed with CD5+ DLBCL according to the positive expression of CD5 by immunohistochemistry and flow cytometry.
Pretreatment evaluation consisted of a history and physical examination, and laboratory tests included peripheral blood examination, renal and liver function, LDH level, serum beta 2-microglobulin level, serum immunoglobulin level, and virus inspection. Patients underwent BM aspiration for immunophenotyping and metaphase karyotyping, BM biopsy, and computed tomography (CT) scans of the chest, abdomen, and pelvis or positron emission tomography/CT. DLBCL-related immunohistochemistry indexes, including B-cell lymphoma 2 (BCL-2), MYC, BCL-6, Multiple myeloma oncogene-1 (MUM-1), CD10, and Ki-67, were routinely detected. No ethical approval was required because this study did not involve ethical issues. This study was a retrospective analysis and did not require ethics committee approval. The patients participating in the study all provided verbal informed consent. The article has been prepared according to the STROBE checklist. 21
Treatment regimens
Patients received rituximab-based chemoimmunotherapy (R-CHOP [rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone]), CHOP, or R-DA-EPOCH according to standard doses. Rituximab was administered at 375 mg/m2 on day 0 per cycle. Dexamethasone (10 mg) and promethazine hydrochloride (25 mg) were administered before rituximab for each course. Treatments, including hydration, alkalization, and protection of the liver, heart, and stomach, were routinely given. Myeloid growth factors were only administered if patients experienced grade 3 or 4 neutropenia. Suspensions of red blood cells or platelets were infused for patients with hemoglobin <70 g/L or platelet counts <20 × 109/L. Courses were repeated every 4 weeks depending on the recovery of neutrophil or platelet counts. The therapeutic effects were evaluated every two courses. The tumor was evaluated every 3 months for 2 years until relapse or progression when all induction therapies were completed.
Response criteria
Treatment response was evaluated when at least two cycles of therapy were completed according to the NHL international therapeutic evaluation standard. 22 Complete response (CR) was defined as the complete disappearance of all detectable sites and symptoms of the disease. Partial response (PR) was defined as a 50% or greater improvement in the disease localization. The overall response rate (ORR) was defined as CR plus PR. The rest of patients were defined as not consistent with the criteria of CR and PR.
Statistical analysis
Progression-free survival (PFS) was calculated from the date of treatment initiation until disease progression or death, and OS was calculated from the date of treatment initiation to death. Survival curves were graphed by the Kaplan–Meier method, and differences between curves were analyzed for statistical significance using the log-rank test. Categorical variables were compared using Pearson’s chi-square test. Multivariate analysis was performed using the cox-regression method. A P-value of <0.05 was considered statistically significant. All data analyses were performed using IBM SPSS Statistics for Windows, Version 22.0 (IBM Corp., Armonk, NY, USA).
Results
Clinical characteristics of patients newly diagnosed with CD5+ DLBCL
Among all 245 patients with DLBCL, there were 31 CD5+ DLBCL cases and 214 CD5− DLBCL cases. Table 1 shows the baseline characteristics of these patients. Among 31 patients with CD5+ DLBCL, 54.8% were women, and 45.2% were men. Their age ranged from 35 to 84 years old, and the median age was 61 years old. The CD5+ DLBCL group had significantly higher proportions of cases with International Prognostic Index (IPI) ≥3 (51.6% vs. 32.7%, P = 0.045), Eastern Cooperative Oncology Group (ECOG) scores ≥ 2 (54.8% vs. 25.7%, P = 0.001), and BM involvement (19.4% vs. 7.5%, P = 0.031) than the CD5− DLBCL group. There was no significant difference in sex, serum LDH levels, Ann Arbor stage, B symptoms, and extranodal invasion areas. Among those with CD5 + DLBCL, the primary site was the lymph nodes in seven patients, and the extranodal sites of the disease included the BM (two patients), abdominal organs (six patients), tonsil (five patients), facial organs (one patient), CNS (four patients), bone (two patients), testicles (two patients), and chest organs (two patients) (Figure 1). Importantly, in addition to four patients with primary CD5+ DLBCL of the CNS, five patients with CD5+ DLBCL were admitted for headache and dizziness, but their disease did not involve the CNS, and one patient presented with acute cerebral infarction during hospitalization.
Characteristics of 245 patients.

CD5+, CD5 positive; DLBCL, diffuse large B-cell lymphoma; CD5−, CD5 negative; IPI, international prognostic index; LDH, lactate dehydrogenase; ECOG, Eastern Cooperative Oncology Group; BM, bone marrow.

Organ involvement in de novo CD5+ and CD5− DLBCL. The disease site at diagnosis was mainly divided into lymph node involvement and extranodal involvement. Sites of extranodal involvement mainly included abdominal organs, tonsils, BM, facial organs, CNS, bone, testicles, and chest organs.
Immunohistochemical characteristics
Table 2 summarizes the immunohistochemical findings. Based on the cell of origin, 35.5% (11/31) of patients with CD5 + DLBCL were assigned to the germinal center B-cell-like (GCB) subgroup, and 64.5% (20/31) of patients were assigned to the non-GCB subgroup. In addition, 42.1% (90/214) of cases in the GCB subgroup and 49.1% (105/214) of cases in the non-GCB group had CD5− DLBCL, but this difference was not significant. Importantly, we found that the expression levels of MYC (54.8% vs. 33.5%, P = 0.035) and BCL-2 (90.3% vs. 73%, P = 0.038) were significantly higher in patients with CD5+ DLBCL in our cohort. Other indexes, including the proliferation marker Ki-67, CD10, BCL-6, MUM-1, and MYC/BCL-2 double expression, were not significantly different between CD5+ DLBCL and CD5− DLBCL cases.
Immunophenotypic features according to immunohistochemistry.

Notes: CD5+, CD5 positive; DLBCL, diffuse large B-cell lymphoma; CD5−, CD5 negative; COO, cell of origin; GCB, germinal center B-cell-like; BCL, B-cell lymphoma; MUM-1, multiple myeloma oncogene-1.
Therapeutic response and outcomes
A total of 111 newly diagnosed patients (22 CD5+ DLBCL; 89 CD5− DLBCL) received at least 6 to 8 courses of R-based chemotherapy (R-CHOP for patients with IPI <3, R-DA-EPOCH for patients with IPI ≥3), 73 patients (four CD5+ DLBCL; 69 CD5− DLBCL) received at least 6 to 8 courses of CHOP, 46 patients (five CD5+ DLBCL; 41 CD5− DLBCL) received only 2 to 3 courses of R-CHOP or CHOP, and the remaining patients with CD5− DLBCL (n = 15) received modified CHOP-like chemotherapy, including CVP (cyclophosphamide, vincristine, and prednisolone) (n = 5), CHP (cyclophosphamide, epirubicin, and prednisolone) (n = 5), CHO (cyclophosphamide, vincristine, and epirubicin) (n = 4), and EPOCH (n = 1). No patients underwent HSCT. All patients had complete follow-up data.
Rituximab-based therapy is particularly important for the treatment of DLBCL. Therefore, we further analyzed the therapeutic response in 111 patients receiving at least 6 to 8 courses of R-based chemotherapy. The CR rate and ORR were 59.6% and 71.9% in patients with CD5− DLBCL and 45.5% and 63.6% in patients with CD5+ DLBCL, respectively. There was no statistical difference in the CR rate or ORR between the two groups. We further analyzed the survival data of these patients. The median follow-up time from the initial treatment was 24 months (range: 6–68 months). The 2-year PFS was significantly shorter in patients with CD5 + DLBCL than in those with CD5− DLBCL (18.2% vs. 56.2%, P = 0.038). However, OS (31.8% vs. 56.2%) was not statistically different between the two groups (Figure 2).

Kaplan–Meier survival curves in patients with de novo CD5+ and CD5− DLBCL treated with rituximab combination therapy. (a) Patients with CD5+ DLBCL showed an obviously shorter 2-year PFS compared with patients with CD5− DLBCL (18.2% vs. 56.2%, P = 0.038) and (b) There was no difference in the 2-year OS rate between the de novo CD5+ and CD5− DLBCL groups (31.8% vs. 56.2%).
Univariate and multivariate survival analyses for OS
We further analyzed the risk factors that might affect OS in the 184 patients who received at least 6 to 8 courses of R-CHOP or CHOP. Based on the results of univariate and multivariate analyses shown in Table 3, patients with age ≥60 years old (P = 0.010), ECOG scores ≥ 2 (P = 0.005), IPI ≥3 (P = 0.006), B symptoms (P < 0.001), and no rituximab-based therapy (P = 0.007) had inferior survival compared with the control group. In the multivariate analysis, when the above-mentioned prognostic variables were adjusted, B symptoms (P = 0.007) and no rituximab-based therapy (P = 0.005) remained independent factors for OS. However, the positive expression of CD5 was not an independent prognostic factor for OS. Finally, Table 4 summarizes previously reported cases of de novo CD5+ DLBCL.
Multivariate analysis of parameters for OS.
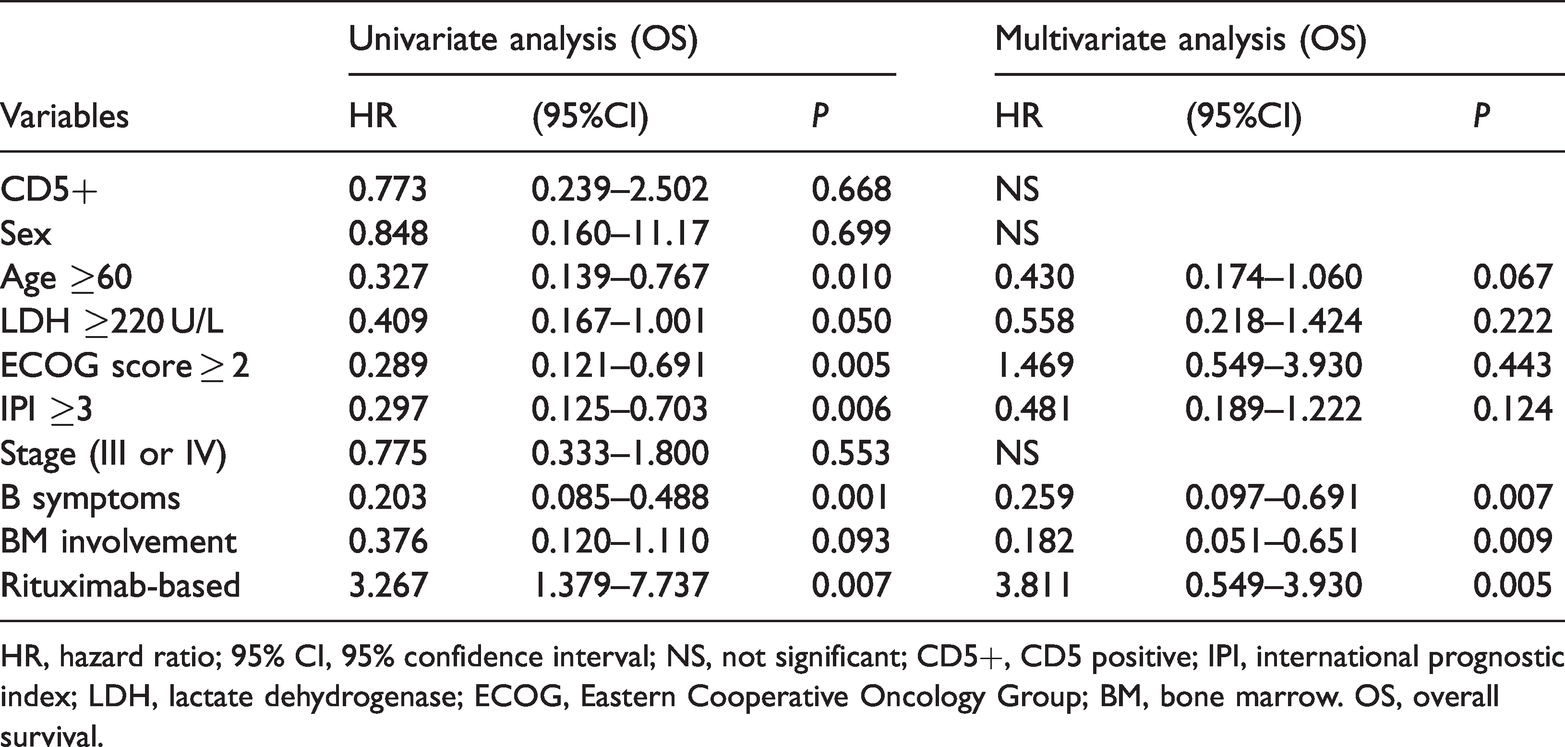
HR, hazard ratio; 95% CI, 95% confidence interval; NS, not significant; CD5+, CD5 positive; IPI, international prognostic index; LDH, lactate dehydrogenase; ECOG, Eastern Cooperative Oncology Group; BM, bone marrow. OS, overall survival.
Summary of previously reported cases of de novo CD5+ DLBCL.

M, men; W, women; GCB, germinal center B-cell-like; CHOP, cyclophosphamide, doxorubicin, vincristine, prednisone.; RCHOP, rituximab, cyclophosphamide, doxorubicin, vincristine, prednisone; R-EPOCH, rituximab, etoposide, cyclophosphamide, doxorubicin, vincristine, prednisone; DA-EPOCH, dose-adjusted etoposide, cyclophosphamide, doxorubicin, vincristine, prednisone; R-Hyper CVAD, rituximab, cyclophosphamide, vincristine, doxorubicin, dexamethasone alternating, methotrexate, cytarabine; CR, complete response; ORR, overall response rate; PFS, progression-free survival; OS, overall survival; m, month. *, patients with CD5+ DLBCL /all patients.
Discussion
As a poor prognostic subtype, de novo CD5+ DLBCL has gradually attracted increasing attention in recent years. The mechanism underlying the poor prognosis of CD5+ DLBCL remains largely unclear. According to currently available reports, there may be two main potential mechanisms involved. 23 One mechanism is the CD5-mediated B-cell receptor (BCR)-dependent pathway. As an immunoreceptor tyrosine-based inhibitory motif, CD5 functionally inhibits BCR signaling-mediated apoptosis, likely by recruiting the SH2 domain-containing protein tyrosine phosphatase-1 after being phosphorylated by Lyn.24,25 Another mechanism is the CD5-mediated BCR-independent pathway, which involves the overexpression of interleukin-10, BCL-2, cyclin D2, and C-X-C chemokine receptor 4 and activates the phosphorylation of ERK, signal transducer and activator of transcription 3, and nuclear factor of activated T cells.26–30 Both promote tumor cell survival and extranodal infiltration, resulting in poor prognosis in de novo CD5+ DLBCL.
CD5+ DLBCL is identified according to the positive expression of CD5 in DLBCL tumor cells via immunohistochemistry or flow cytometry. 13 However, the cut-off value of positive CD5 expression by immunohistochemistry has no standard threshold, ranging from 10% to 30% in different reports. 6 In our present Chinese cohort, immunohistochemistry was used to identify the positive expression of CD5 with a cut-off value of 10%. According to the limited reports, patients with CD5+ DLBCL appear to be predominately older (>60 years old) and women (women/men >1).9,10,31 Moreover, several studies have reported a higher LDH level in CD5+ DLBCL populations and an increased tendency of developing B symptoms. 32 Additionally, CD5+ DLBCL has been found to have more than one extranodal site involvement (26%–45%), including the BM (28%) and CNS (13%). Therefore, CD5+ DLBCL possesses a higher probability of ECOG performance status scores > 1, and 40% of cases belong to the IPI high-risk group.10,31 In our present research, the CD5+ DLBCL group also had a higher proportion of elderly patients and those with higher IPI scores, ECOG scores, and BM involvement, which was consistent with the literature reports.
Immunohistochemically, the majority of CD5+ DLBCL cases are CD10-, BCL-2+, and MUM-1+, belonging to the activated B-cell/non-GCB subtype of DLBCL. Moreover, the phenotype of CD19+, CD20+, CD21−, CD23− and a predominance of surface IgMκ have been found in CD5+ DLBCL. 33 In our present study, 64.5% and 35.5% of the 31 patients with CD5+ DLBCL were assigned to the non-GCB subgroup and GCB subgroup, respectively, using the Hans typing method (based on CD10, BCL-6, and MUM-1). However, the difference was not statistically significant between CD5+ DLBCL and CD5− DLBCL cases. Double-expression DLBCL has been reported in 27.6% of patients with CD5+ DLBCL, which predicts poor survival, but only in 3% of those with CD5− DLBCL. 17 In our present study, we found that 90.3% and 54.8% of patients with CD5+ DLBCL positively expressed BCL-2 and MYC, respectively, which were higher than the percentages for patients with CD5− DLBCL (P = 0.038, 0.035). However, no difference in double expression was found between CD5+ DLBCL and CD5− DLBCL (25.9% vs. 23.5%).
Currently, few studies have compared the therapeutic effect between de novo CD5+ DLBCL and CD5− DLBCL. Alinari et al. reported an ORR of 85% in 102 patients with CD5+ DLBCL receiving rituximab-related treatment. More importantly, compared with CD5− DLBCL, CD5+ DLBCL has a significantly worse prognosis and higher relapse rate (14.2%). 11 In our present study, the ORR was only 63.6% in patients with CD5+ DLBCL, which was lower than that reported in the study by Alinari et al., potentially because of ethnic differences. Moreover, we found a higher proportion of patients with GCB DLCBL using rituximab-based therapy at our center, which may be the most important reason for the lower ORR in our study. The 5-year OS for CD5+ DLBCL is only 35.5%, whereas that for CD5− DLBCL is 64.8%. 11 Table 4 summarizes the previously reported cases of de novo CD5+ DLBCL. The PFS of patients with CD5+ DLBCL was remarkably shorter compared with that of patients with CD5− DLBCL, supporting the conclusion of a poorer prognosis in those with CD5+ DLBCL. However, there was no statistical difference in OS because of the short follow-up time and small sample size in our present study.
The current standard first-line therapy R-CHOP is insufficient for the CD5+ DLBCL cohort, and their long-term OS is not improved even with high-intensity chemotherapy, such as R-DA-EPOCH, R-Hyper CVAD, or autologous peripheral blood stem cell transplantation (auto-PBSCT).11,16,34 Thakral et al. reported that 56% of patients with CD5+ DLBCL receiving R-EPOCH reach CR, and the median OS is only 28.1 months, which is worse compared with that of patients with CD5− DLBCL with a CR rate of 69%. 12 Moreover, Alinari et al. reported that among 102 patients with CD5+ DLBCL at nine USA institutions, 28 patients received auto-PBSCT, 20 patients relapsed, six patients died, and the median OS from HSCT to relapse or death was only 4.9 months, suggesting that auto-PBSCT fails to salvage the majority of these patients. 11 However, some studies have reported that R-DA-EPOCH in combination with high-dose methotrexate decreases CNS involvement in patients with CD5+ DLBCL. 35 A clinical phase II study of R-DA-EPOCH in combination with high-dose methotrexate in patients with CD5+ DLBCL reported 2-year PFS and OS rates of 77% and 87%, respectively, which are significantly improved compared with those for the conventional R-chemotherapy control. 36
The optimal treatments for patients with CD5+ DLBCL remain largely unclear. Therefore, it is necessary to develop novel drugs for these patients. Because most patients with CD5+ DLBCL have the non-GCB/ABC subtype, 37 new drugs that can improve the outcomes of non-GCB/ABC DLBCL may theoretically be suitable for those with CD5+ DLBCL. Lenalidomide in combination with R-CHOP (R2-CHOP) was reported to significantly increase the 2-year OS of patients with non-GCB DLBCL from 46% to 83% in a phase II study. 38 Ibrutinib in combination with lenalidomide (>15 mg) and rituximab or R-CHOP yields a response rate of 48% and 100% in non-GCB DLBCL, respectively.39,40
Despite the higher occurrence of BCL-2 overexpression in patients with CD5+ DLBCL, venetoclax alone does not show promising results (ORR: 18%; CR: 12%; PFS: 1 month) because of the highly variable BCL-2 expression in DLBCL.18,41 However, it shows synergistic effects in combination with ibrutinib, R-CHOP, or radiotherapy.19,42,43 The expression of programmed death-ligand 1 (PD-L1) is higher in the non-GCB subtype, 44 and sporadic cases of CD5+ DLBCL have also been found to express PD-L1. 20 A study reported that 90% of patients with DLBCL respond to PD-L1 inhibitors in combination with R-CHOP, with a 2-year OS rate of 84%, indicating that PR-CHOP is a potentially effective method for these patients. 45 However, very few studies have reported the effects of PD-1 antibodies alone or in combination for patients with CD5+ DLBCL.
As a retrospective study, our study also has certain limitations. First, this was a single-center study with a limited number of patients. Unfortunately, among patients treated with CHOP, there were only four cases with CD5 + DLBCL; therefore, a meaningful statistical analysis could not be performed. Second, we did not obtain sufficient fluorescence in situ hybridization results from patients. Third, the follow-up time was limited.
Conclusions
Collectively, CD5+ DLBCL is a subtype of DLBCL with a poor prognosis, and its biology, mechanisms, and optimal therapeutic options need to be further explored. Lenalidomide, Bruton tyrosine kinase inhibitors, BCL-2 inhibitors, and immune therapies alone or in combination presented promising results for patients with non-GCB/ABC DLBCL and might improve the outcomes of those with CD5+ DLBCL. Further clinical trials are required to test the effectiveness of these novel drugs alone or in combination for patients with CD5+ DLBCL.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221110075 - Supplemental material for CD5+ diffuse large B-cell lymphoma has heterogeneous clinical features and poor prognosis: a single-center retrospective study in China
Supplemental material, sj-pdf-1-imr-10.1177_03000605221110075 for CD5+ diffuse large B-cell lymphoma has heterogeneous clinical features and poor prognosis: a single-center retrospective study in China by Ting Yin, Ling Qi, Yulan Zhou, Fancong Kong, Shixuan Wang, Min Yu and Fei Li in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors have stated that they have no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by grants from the National Natural Science Foundation of China (81960043) and the National Natural Science Foundation of Jiangxi Province (20192ACB20030).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
