Abstract
Objectives
This study investigated risk factors and constructed an online tool to predict distant metastasis (DM) risk in patients with leiomyosarcoma (LMS) after surgical resection.
Methods
Data regarding patients with LMS who underwent surgical resection between 2010 and 2018 were extracted from the Surveillance, Epidemiology, and End Results (SEER) database. Data were collected regarding patients with LMS who underwent surgical resection at Tianjin Medical University Cancer Hospital and Institute (TJMUCH) between October 2010 and July 2018. Patients were randomly divided into training and validation sets. Logistic regression analyses were performed; a nomogram was established. The area under the curve (AUC) and calibration curve were used to evaluate the nomogram, which served as the basis for a web-based nomogram.
Results
This study included 4461 and 76 patients from the SEER database and TJMUCH, respectively. Age, ethnicity, grade, T stage, N stage, radiotherapy, and chemotherapy were associated with DM incidence. C-index values were 0.815 and 0.782 in the SEER and Chinese datasets, respectively; corresponding AUC values were 0.814 and 0.773, respectively. A web-based nomogram (https://weijunqiang-leimyosarcoma-seer.shinyapps.io/dynnomapp/) was established.
Conclusions
Our web-based nomogram is an accurate and user-friendly tool to predict DM risk in patients with LMS; it can aid clinical decision-making.
Keywords
Introduction
Leiomyosarcoma (LMS) represents 10% to 20% of all newly diagnosed soft tissue sarcomas (STSs). 1 LMS is derived from smooth muscle cells and can occur in various body sites; it frequently involves the abdomen, retroperitoneum, uterus, and limbs/extremities. LMS is highly aggressive; ∼90% of affected patients have moderate-to-high-grade cancer. 2 Surgical resection is the main treatment for localized STS, including LMS. However, patients with LMS have a high risk of distant metastasis (DM) and poor cancer-specific survival. 3 In LMS, hematogenous spread is a common event that often affects the lungs and liver.4,5 Malignant tumor metastases cause most cancer-related deaths worldwide.5–9 DM develops in 50% of patients with high-risk STS who undergo resection.10,11 Five-year overall survival ranges from 15% to 52% in patients with STS who exhibit DM; it is ∼65% in patients with localized STS.12–14
Histological grade, tumor size, nodal involvement, and tumor site are independent prognostic factors for LMS; they are included in the American Joint Committee on Cancer/Union for International Cancer Control (AJCC/UICC) staging system. 15 The AJCC staging system is useful for assessing STS prognosis, but it does not reflect differences among patients or consider the impact of treatment on patient prognosis. 16 Additionally, it remains difficult to evaluate long-term prognosis in patients with STS after surgical or systemic treatment. 16
A nomogram is a chart-based algorithm that integrates clinical variables to achieve accurate prediction without complex mathematical formulas. 17 Nomograms for various malignant tumors have demonstrated practical value.16,18,19 Furthermore, web-based graphical nomograms can assess prognosis in a convenient and intuitive manner. 16 Nomograms have been developed to predict STS prognosis, 18 as well as 12-year sarcoma-specific deaths. 20 Histology-specific and site-specific nomograms have been developed to predict overall survival for patients with uterine LMS or limb LMS.21–24 We recently established an internal validation web-based nomogram for LMS with lung metastasis to predict overall survival and cancer-specific survival. 16 DM is an independent risk factor for poor prognosis in patients with LMS. Therefore, early identification of patients with a high risk of DM can improve patient outcomes. To our knowledge, no studies have used nomograms to predict DM risk in patients with LMS who underwent surgery.
An accurate and convenient online model for DM prediction may enhance clinical decision-making and improve patient outcomes. This population-based study was performed to develop a web-based nomogram for predicting DM risk in patients with LMS who underwent surgery.
Methods
Patients
Patient data were extracted from the Surveillance, Epidemiology, and End Results (SEER) database, which is supported by the United States National Cancer Institute. We obtained clinicopathological information for patients diagnosed between 2010 and 2018, using SEER*Stat software (version 8.3.6). We used International Classification of Diseases for Oncology, Third Edition (ICD-O-3) histologic codes to identify patients with LMS (8890/3), epithelioid LMS (8891/3), bizarre LMS (8893/3), and myxoid LMS (8896/3). Information was collected regarding metastatic sites in bone (SEER Combined Mets at DX-bone), brain (SEER Combined Mets at DX-brain), liver (SEER Combined Mets at DX-liver), and lung (SEER Combined Mets at DX-lung) since 2010. Because there were no cancer-directed surgeries, we collected information about primary sites of resection based on RX Summ–Surg Prim Site (1998+) data. Patients were included if they were aged ≥18 years and had undergone resection of the primary site; they were excluded if their records lack information regarding demographic and clinical characteristics. The study was conducted in accordance with the Declaration of Helsinki (revised in 2013). According to the ethics guidelines, neither informed patient consent nor ethics committee approval was required for the use of public and anonymized data.
We recorded the demographic and clinical characteristics of patients with primary LMS who underwent surgical resection at Tianjin Medical University Cancer Hospital and Institute between October 2010 and July 2018. The final follow-up was conducted in January 2019. SEER cohort inclusion and exclusion criteria were also applied to patients in the Chinese cohort. All patients provided informed consent to participate in the study. This retrospective study of Chinese patients was approved by the ethics committee of Tianjin Medical University Cancer Hospital and Institute (approval no. E2019144), in accordance with the Declaration of Helsinki. The patient selection flowchart is shown in Figure 1.

Flowchart for patient selection and data analysis.
Variables
The following variables were analyzed: age (≤60 years or >60 years), sex (female or male), ethnicity [White, Black, or Others (American Indian/Alaska Native, Asian/Pacific Islander)], tumor site (retroperitoneum, corpus or uterus, soft tissue, or other), histological type (epithelioid, myxoid, or other), grade (well-differentiated, moderately differentiated, poorly differentiated, or undifferentiated), T stage (T1, T2, T3, or T4), N stage (N0 or N1), M stage (M0 or M1), radiotherapy (yes or no), chemotherapy (yes or no), and site of metastasis (lung, liver, brain, bone, or others). For consistency, the T, N, and M stages derived from the SEER database were uniformly converted to TNM stages used in the AJCC STS staging system.15,25,26
Nomogram development and statistical analysis
Statistical analyses were performed using SPSS for Windows (ver. 24.0; IBM Corp., Armonk, NY, USA). Continuous variables were converted to categorical variables. Univariate logistic regression was performed to identify variables associated with DM in the SEER cohort; variables with p < 0.1 were entered into multivariate logistic regression. Variables with p < 0.05 in multivariate logistic regression were regarded as independent risk factors for DM; these variables were used to construct the nomogram. An independent external validation cohort of Chinese patients with LMS was used to evaluate nomogram calibration and discrimination performances. Receiver operating characteristic curves and calibration curves were established to evaluate discrimination performance. The DynNom and shiny packages in R 3.6.2 software (R Foundation for Statistical Computing, Vienna, Austria) were used to establish a web-based nomogram that could dynamically predict DM risk. Differences in clinical factors between the SEER and Chinese cohorts were compared using the independent samples t-test for normally distributed variables; the Mann–Whitney U test was used to compare variables with skewed distributions. Categorical variables were compared using the χ2 test or Mann–Whitney U test, as appropriate. The threshold for statistical significance was regarded as p < 0.05.
Results
Demographic and clinical characteristics
In total, 4461 patients from the SEER database and 76 Chinese patients were included in the study. Their demographic and clinical characteristics are shown in Table 1.
Demographic and pathological characteristics of patients with leiomyosarcoma.

NOS, not otherwise specified; SEER, Surveillance, Epidemiology, and End Results.
In the SEER cohort, 51.5% of patients were aged ≤60 years. Most patients (77.9%) were White. The main primary sites were soft tissue (57.7%) and the uterus (25.0%). Only 10.4% of patients had a histological grade of well-differentiated. T1 and T2 stages were present in 31.7% and 32.6% of patients, respectively; 2.8% and 11.3% of patients experienced lymph node metastasis and DM, respectively. All patients underwent surgical resection of the primary lesion. Furthermore, 32.9% and 26.0% of patients received chemotherapy and radiotherapy after surgery, respectively.
In the Chinese cohort, 63.2% of patients were aged ≤60 years. The primary sites were the retroperitoneum (30.3%), corpus or uterus (23.7%), soft tissue (18.4%), and other sites (27.6%). Histological grades were well-differentiated, moderately differentiated, poorly differentiated, and undifferentiated in 32.9%, 18.4%, 23.7%, and 25% of patients, respectively. T1 and T2 stages were present in 30.3% and 44.7% of patients, respectively; 21.1% and 47.4% of patients experienced lymph node metastasis and DM, respectively. All patients underwent surgical resection. Finally, 30.3% and 9.2% of patients received chemotherapy and radiotherapy after surgery, respectively.
Independent risk factors for DM
Univariate and multivariate logistic regression analyses of the SEER cohort were performed to identify risk factors for DM. Univariate logistic regression showed that age, sex, ethnicity, tumor site, grade, T stage, N stage, radiotherapy, and chemotherapy were associated with DM. Multivariate logistic regression revealed that age, ethnicity, grade, T stage, N stage, radiotherapy, and chemotherapy were significantly associated with DM. Detailed information concerning risk factors for DM in patients with LMS is presented in Table 2.
Univariate and multivariate logistic regression analysis of risk factors for distant metastasis in patients with leiomyosarcoma.

CI, confidence interval; NOS, not otherwise specified; OR, odds ratio.
Nomogram construction
Based on the multivariate logistic regression results, we constructed a predictive nomogram that included all independent risk factors identified in the SEER cohort (Figure 2). The sum of scores for all variables in the nomogram could be used to predict DM risk in patients with LMS who had undergone surgery.
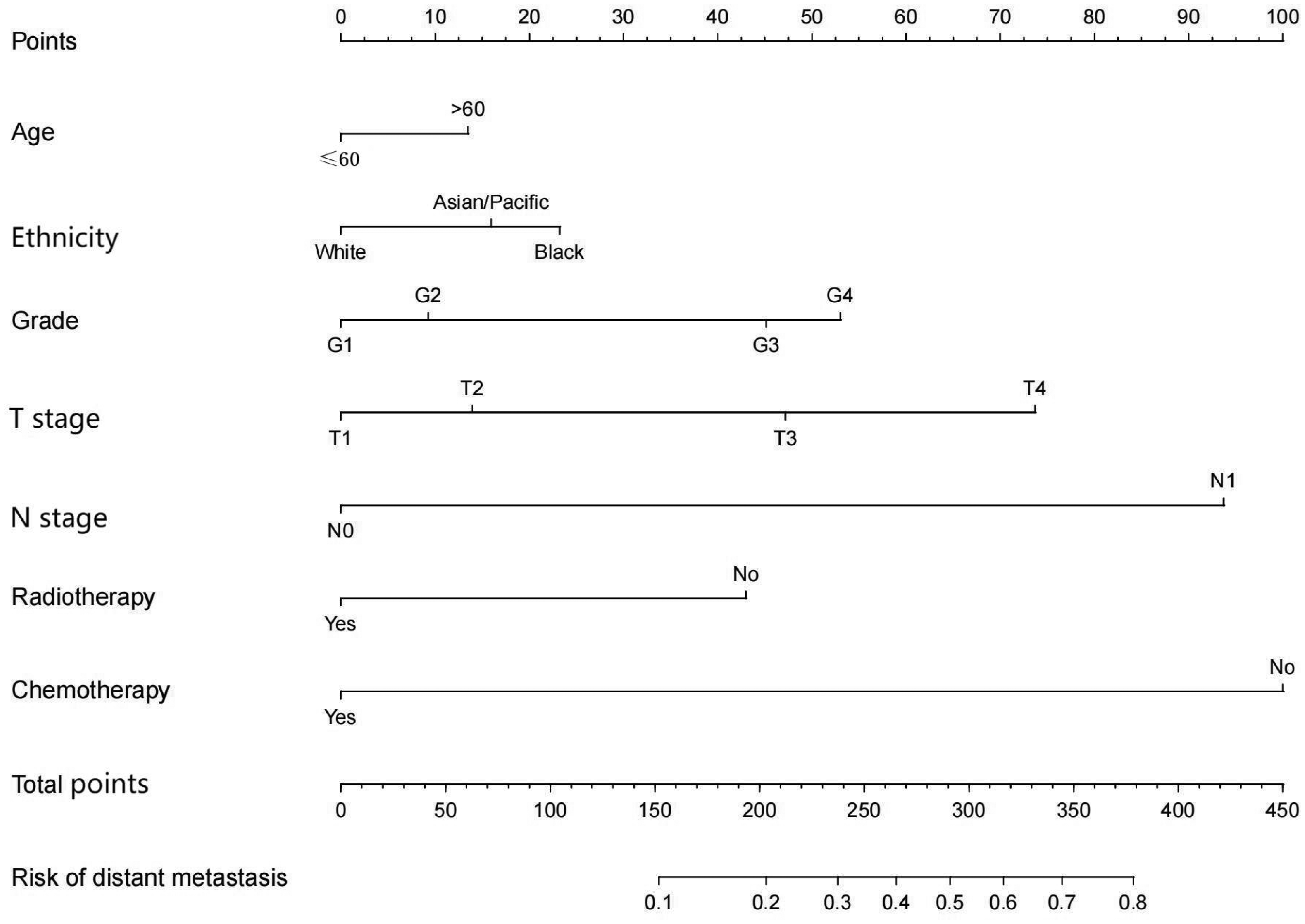
Nomogram for predicting distant metastasis risk in patients with leiomyosarcoma.
Nomogram validation
The predictive nomogram was validated in both the SEER and Chinese cohorts. The C-index was 0.815 in the SEER cohort. For the Chinese cohort, DM risk was determined using the nomogram established using SEER cohort data. The C-index was 0.782 in the Chinese cohort. Calibration plots demonstrated good performance in both the SEER and Chinese cohorts (Figure 3a, b). Furthermore, the area under the receiver operating characteristic curve values in the SEER and Chinese cohorts were 0.814 and 0.773, respectively (Figure 3c, d).

Calibration plots of the constructed nomogram in terms of predicting distant metastasis risk in the SEER cohort (a) and Chinese cohort (b). Receiver operating characteristic curves for discrimination of distant metastasis risk in the SEER (c) and Chinese (d) cohorts.
Web-based nomogram
Using the predictive nomogram described above, a graphical web-based probability calculator (https://weijunqiang-leimyosarcoma-seer.shinyapps.io/dynnomapp/) was created to predict DM risk in patients with LMS. DM risk was determined by selecting relevant clinical variables in the web interface. The prediction of DM risk in patients with LMS based on clinical parameters alone is straightforward and user-friendly. For example, DM risk was ∼79.6% (95% confidence interval: 80.6–86.3) for White patients aged >60 years with poorly differentiated or undifferentiated histological grade, T4 stage, and N1 stage without radiotherapy or chemotherapy (Figure 4a, b).
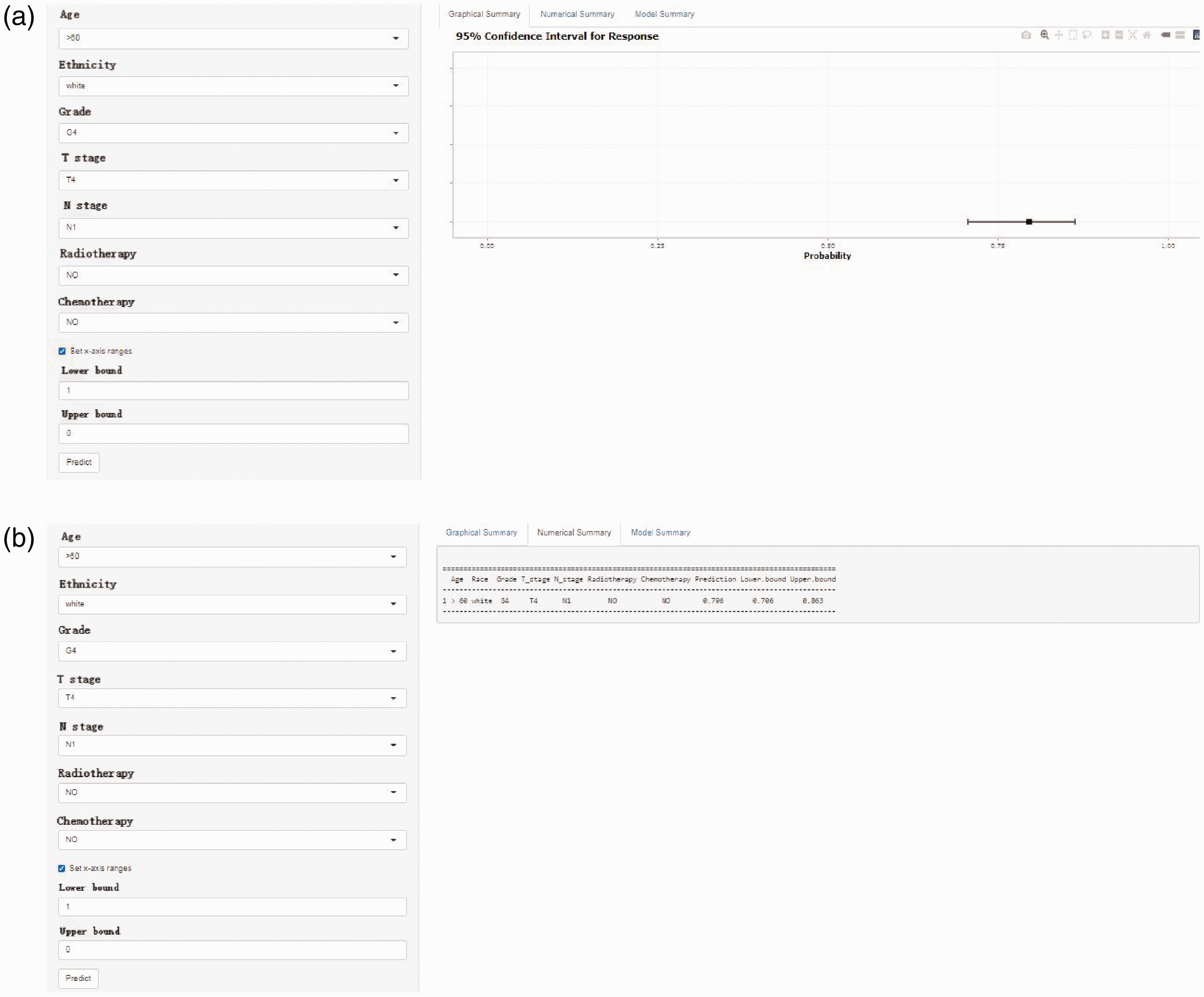
Web-based probability calculator for distant metastasis risk in patients with leiomyosarcoma. The example shows a White patient with leiomyosarcoma who underwent surgery and was aged >60 years with poorly differentiated or undifferentiated histological grade, T4 stage, and N1 stage without radiotherapy and chemotherapy. (a) Graphical summary shows the approximate range. (b) Numerical summary shows a distant metastasis risk of ∼79.6% (95% confidence interval: 80.6–86.3).
Discussion
LMS constitutes approximately 10% to 20% of all newly diagnosed STSs. 4 Surgical resection is the main treatment approach for patients with primary STS, including LMS. 27 Moreover, hematogenous metastasis is a common event in patients with LMS; it typically affects the lungs and liver. 27 To our knowledge, few studies have investigated DM risk in patients with LMS who underwent surgery; therefore, we sought to construct a practical tool to predict this risk. The present retrospective study analyzed patients with LMS from the SEER database; it revealed that age, ethnicity, grade, T stage, N stage, radiotherapy, and chemotherapy were independent risk factors for DM. These variables were used to establish a prediction model for DM risk in patients with LMS. Additionally, receiver operating characteristic curves, calibration plots, and decision curve analysis curves for internal and external validation showed that the model demonstrated good discrimination ability, robust calibration ability, and favorable clinical utility. Additionally, an easy-to-use web-based nomogram was constructed to allow calculation of a patient's DM risk by entering the values of the seven variables. This online tool will allow clinicians to predict DM risk based on a clinical assessment, facilitating personalized treatment for patients with LMS.
In this study, age, ethnicity, grade, T stage, N stage, radiotherapy, and chemotherapy were factors used in nomogram development. Age was independently associated with DM risk; similarly, previous studies have shown that the older patients with LMS have a worse prognosis. 13 There is speculation that the poor prognosis of older patients with LMS may be related to their greater susceptibility to DM; the present findings confirmed this hypothesis. Furthermore, there is evidence of ethnic disparities in terms of STS mortality.10,27 In the United States, Hispanic and Black patients have significantly worse 5-year overall survival, compared with White patients.28–30 The present study showed that Black patients with LMS were more likely to develop DM, which may explain the poor prognosis in these patients. A high histological grade is an indicator of poor prognosis in patients with LMS,31–33 and STS prognosis worsens after DM. 33 In the present study, a higher histological grade was associated with a higher probability of DM. Tumor size is an important indicator for patients with LMS32,34; tumor size ≥5 cm is associated with worse overall survival and cancer-specific survival in patients with advanced LMS. 24 The present study revealed that T stage is an independent indicator of metastasis risk. Notably, the lymph node metastasis score was high, strongly suggesting that lymph node metastasis in patients with LMS represents a high risk of DM. Radiotherapy generally can achieve good local control and relieve local symptoms in patients with STS, 35 but it is not associated with overall survival.24,35,36 We found that patients with LMS benefited from radiation therapy; their metastasis risk was reduced. Rehmani et al. showed that radiotherapy could improve survival in patients with high-grade STS. 37 The present findings confirm that radiotherapy can reduce DM risk in patients with LMS who undergo surgical resection; local radiotherapy can destroy residual lesions, thereby reducing metastasis risk. Additionally, LMS is a chemotherapy-sensitive tumor; the present study showed that systemic chemotherapy has a positive effect on DM control.
Importantly, our nomogram underwent independent external validation. It demonstrated good discrimination in the internal and external validation cohorts; it also exhibited robust calibration in the development and external validation cohorts. In contrast to previous research, this study evaluated DM risk in patients with LMS who underwent surgical resection of primary lesions. 16 The resulting model was validated by external data, then used to establish a web-based nomogram. As noted in the Introduction, malignant tumor metastases cause most cancer-related deaths worldwide.5–9 DM reportedly develops in 50% of patients with high-risk STS who undergo resection.21,23,24,31 Considering the aggressive nature of LMS, more than 36% of affected patients will experience DM and require additional surgical treatment. 38 In patients with primary STS who undergo surgical resection, the 5-year survival rate decreases from ∼60% to <15% after DM10,27; survival is similar among patients with LMS. 39 Therefore, the construction of a tool for early identification of patients with high DM risk will improve LMS prognosis. The current AJCC/UICC staging system includes multiple variables for prognostic assessment; however, it does not encompass all prognostic variables, and it is difficult to include all possible sarcoma subtypes. In the past decade, nomograms established to predict STS prognosis have demonstrated excellent results. 18 Nomograms have also been established to predict LMS prognosis; however, most lack external validation.18,21,23,24,39 In contrast, our nomogram was validated using real-world data from a tertiary cancer referral center in northern China.
Our model has multiple strengths that support its application in clinical practice. First, the nomogram was developed using a large sample of patients with LMS; validation with an external cohort revealed good performance in terms of identifying patients with high DM risk. High-risk patients should undergo more frequent follow-up to facilitate earlier detection of DM lesions; clinicians should more carefully monitor whole-body scans of these patients. In contrast, low-risk patients do not need frequent radiological investigations during follow-up. Second, the online tool offers a user-friendly interface where clinicians can enter clinicopathological characteristics in the left column; DM risk is shown in the right column. For example, in White patients with LMS who underwent surgery and were aged >60 years with poorly differentiated or undifferentiated histological grade, T4 stage, and N1 stage without radiotherapy or chemotherapy, the DM risk was ∼79.6% (95% confidence interval: 80.6–86.3) (Figure 4). Our study is unique in that it focused on DM risk in patients with LMS who underwent surgical resection of primary lesions, which is a common clinical scenario in surgical departments. Our nomogram can distinguish between patients with the same pathological type but differences in other characteristics, allowing precision clinical decisions. This approach can enhance clinician–patient communication and reduce medical risks.
This study had some limitations. First, because the nomogram was developed using retrospective information from the SEER database, selection bias may have been present. Second, the nomogram did not include other clinical variables associated with metastasis, such as site, tumor depth, mitotic rate, and tumor necrosis. Third, the external validation dataset included a small number of patients and a single ethnicity. Fourth, the SEER database does not identify the order of surgery and DM; thus, it is possible to detect DM both during and after surgery. Because of these limitations, the nomogram should be considered among other clinical data when clinicians make treatment decisions.
Conclusion
To our knowledge, we have developed the first web-based nomogram for DM risk in patients with resected LMS; development was performed using data from the SEER database, and the nomogram was externally validated with data from an independent Chinese cohort. The resulting nomogram demonstrated excellent performance in both internal and external analyses. Our user-friendly online tool can help clinicians to predict LMS prognosis after surgical resection and provide appropriate treatment recommendations.
Footnotes
Acknowledgements
We thank the United States National Cancer Institute for providing the SEER database and Tianjin Medical University Cancer Institute and Hospital for providing cancer research support. We also thank all of our colleagues who participated in the study.
Author contributions
WJQ and LLR contributed to conceptualization, formal analysis, manuscript review and editing, and project management. LZH and ZC contributed to methodology, investigation, and software. RZW, FZ and CHY contributed to validation. JY acquired funding. All authors approved the final version of this manuscript for publication.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This study was supported by the 2023 Clinical Medical Excellent Talent Cultivating Program of Hebei (grant no. ZF2023249).
