Abstract
Objective
To identify risk factors and develop predictive web-based nomograms for the early death of patients with bone metastasis of lung adenocarcinoma (LUAD).
Methods
Patients in the Surveillance, Epidemiology, and End Results database diagnosed with bone metastasis of LUAD between 2010 and 2016 were included and randomly divided into training and validation sets. Early death-related risk factors (survival time ≤7 months) were evaluated by logistic regression. Two predictive nomograms were established and validated by calibration curves, receiver operating characteristic curves, and decision curve analysis.
Results
A total of 9189 patients (56.59%) died from all causes within 7 months of being diagnosed, including 8585 patients (56.67%) who died from cancer-specific causes. Age >65 years, sex (men), T stage (T3 and T4), N stage (N2 and N3), brain metastasis, and liver metastasis were risk factors for all-cause and cancer-specific early death. The area under the curves of the nomograms for all-cause and cancer-specific early death prediction were 0.754 and 0.753 (training set) and 0.747 and 0.754 (validation set), respectively. Further analysis showed that the two nomograms performed well.
Conclusions
Our two web-based nomograms for all-cause and cancer-specific early death provide valuable tools for predicting early death in these patients.
Keywords
Introduction
Lung cancer (LC) is a serious threat to human survival and health, with the second highest incidence (11.4% of all cancer types) and mortality rate (18.0% of total cancer deaths) worldwide. 1 The most common type of LC is lung adenocarcinoma (LUAD), which accounts for 40% of all LC cases. 2 Approximately 55% of patients with LC develop distant metastasis, and bone metastasis is the most common type (30%–40% of all patients).3,4 Gaining a further understanding of the physiological and pathological status of LC and the implementation of biomarker-based therapies have improved the survival rates of patients.5–7 However, approximately 30% to 40% of patients with LC have bone metastasis, and the median survival time of these patients is less than 7 months. 8 Therefore, it is important to identify high-risk factors associated with early death and provide treatment options for patients with LUAD with bone metastasis. Studies on high-risk factors have been conducted in gastrointestinal tumors and endometrial cancers, but few have been reported in LUAD.9–11 Furthermore, risk factors for early death in patients with bone metastasis of LUAD have not been well documented. Therefore, analyzing risk factors of early death (survival time ≤7 months) in patients with LUAD and bone metastasis is clinically significant. 8 As a new tool, nomograms have been widely used to predict the occurrence and prognosis of diseases, especially various tumors.12,13 Web-based nomograms have a more user-friendly operation interface, and accurate evaluation values can be obtained according to the different disease characteristics of each patient.14,15 Therefore, based on the Surveillance, Epidemiology, and End Results (SEER) database, this study aimed to identify risk factors and establish two web-based nomograms for predicting early death in patients with LUAD and bone metastasis.
Materials and methods
Data collection
All patients diagnosed with LUAD and bone metastasis between 2010 and 2016 were selected from the SEER database. Patients who met the following criteria were included in the study: (I) adenocarcinomas of the lung (C34.9, 8140/3), (II) year of diagnosis between 2010 and 2016, and (III) bone metastasis (SEER Combined Mets at DX-bone (2010+)). Patients with incomplete or missing information were excluded. Patient characteristics included the following: race, sex, age, surgery, radiotherapy, chemotherapy, T stage, N stage, brain metastasis, liver metastasis, lung metastasis, cause of death, survival duration (months), and vital status record. All-cause early death and cancer-specific early death were defined as a survival time ≤7 months from diagnosis to all causes of death and cancer-specific causes of death, respectively. Because public and anonymous data were used, the need for informed consent and ethics committee approval was waived. The reporting of this study conforms to STROBE guidelines. 16
Nomogram construction and statistical analysis
The primary outcomes of this study were all-cause early death and cancer-specific early death. X-tile software was used to obtain the optimal cut-off point for age. 17 All data were randomly divided into training and validation sets at a ratio of 7:3 using the “caret” package.18,19 The chi-square test was used for the comparison of categorical variables between the training set and validation set. The odds ratio (OR) was calculated to quantify the effect of each predictor on all-cause early death and cancer-specific early death. Based on univariate logistic regression analysis (p-value <0.05), a multivariate logistic regression model was used to further analyze the variables. The variables with p-values <0.05 in the multivariate regression analysis were considered significant risk factors. Based on the significant variables associated with all-cause and cancer-specific early death, two sets of forest maps were established. Significant variables were also included in establishing two nomograms. Receiver operating characteristic (ROC) curves, calibration curve analysis, and decision curve analysis (DCA) were used to evaluate the performance of the nomograms. The “DynNom” software package was used to build two network-based nomograms that could accurately calculate the probability of early death. All statistical analyses were conducted using R (www.r-project.org).
Results
Patient characteristics
In our study, 16,239 patients with LUAD with bone metastasis were included and divided into training (n = 11,368) and validation (n = 4871) sets. A total of 9189 (56.59%) patients were defined as all-cause early death. For cancer-specific survival or death, 15,149 patients with bone metastasis of LUAD were included and divided into training (n = 10,605) and validation (n = 4544) sets. Specific inclusion and exclusion criteria are shown in Figure 1. Of these, 8185 (56.67%) patients died early from cancer-specific causes. The optimal age cut-off (Figure 2) of patients with LUAD with bone metastasis was 65 and 75 years old for the all-cause death group and cancer-specific death group, respectively. Therefore, we converted age from a continuous variable to a categorical variable (≤65, 66–75, and >75 years old). Most patients were ≤65 years (n = 7397, 45.55%) and men (n = 8656, 53.30%). The main primary race was white (n = 12,509, 77.03%), followed by black (n = 1953, 12.03%), and others (American Indian/Alaska Native, Asian/Pacific Islander, n = 1777, 10.94%). The most common T and N stages were T4 (n = 5404, 33.28%) and N2 (n = 7599, 46.79%), respectively. Among these patients, 4302 (26.49%) patients had brain metastases, 3660 (22.54%) had liver metastases, and 5127 (31.57%) had intrapulmonary metastasis. Regarding treatment plans, 9342 (57.53%), 10,651 (65.59%), and 284 (1.75%) patients underwent radiotherapy, chemotherapy, and surgery, respectively. Additional details are shown in Table 1 (all-cause early death) and Table 2 (cancer-specific early death).

Patient selection and study design flow chart.

The appropriate cut-off values for age. The cut-off values were assessed by X-tile analysis. The frequency distribution histogram (a) with appropriate cut-off values of age (65 and 75 years old) and survival curve of the three groups (b) for the all-cause early death group. The frequency distribution histogram (c) with appropriate cut-off values of age (65 and 75 years old) and survival curve of the three groups (d) for the cancer-specific early death group.
Demographic and clinicopathological variables for patients with lung adenocarcinoma in the all-cause early death group.

#Chi-square test was performed to compare the training set and validation set.
Demographic and clinicopathological variables for patients with lung adenocarcinoma in the cancer-specific early death group.

#Chi-square test was performed to compare the training set and validation set.
Risk factors for early death
The univariate logistic analysis results of 16,239 patients with LUAD (all-cause death group) showed that age, sex, race, T stage, N stage, brain metastasis, liver metastasis, lung metastasis, chemotherapy, and surgery influenced the onset of all-cause early death (p < 0.001). The results of the multivariate logistic analysis showed that age (65–75 and >75), T stage (T2, T3, and T4), N stage (N2 and N3), brain metastasis, and liver metastasis were associated with higher all-cause early death (p < 0.001). Chemotherapy, surgery, and race (others) were protective factors and associated with survival longer than seven months (p < 0.01). The univariate logistic analysis results of 15,149 patients with LUAD (cancer-specific death group) showed that age, sex, race, T stage, N stage, brain metastasis, liver metastasis, lung metastasis, chemotherapy, and surgery influenced the onset of cancer-specific early death (p < 0.01). For cancer-specific death, age (65–75 and >75), T stage (T2, T3, and T4), N stage (N2 and N3), brain metastasis, and liver metastasis were risk factors (p < 0.01). Chemotherapy, surgery, and race (others) were protective factors (p < 0.01). Additional details are shown in Table 3 (all-cause early death) and Table 4 (cancer-specific early death).
Univariate and multivariate logistic analysis based on all variables for all-cause early death.
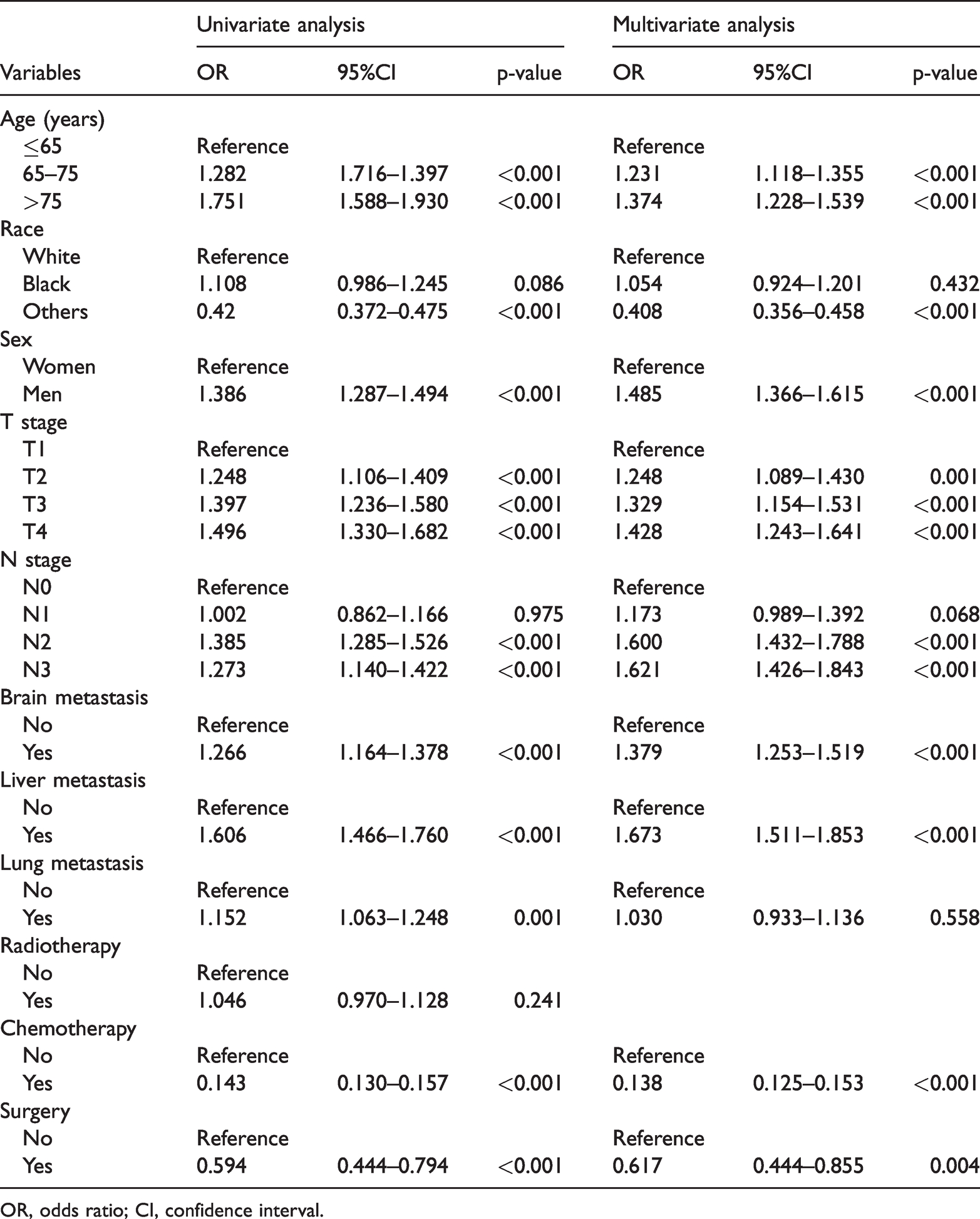
OR, odds ratio; CI, confidence interval.
Univariate and multivariate logistic analysis based on all variables for cancer-specific early death.

OR, odds ratio; CI, confidence interval.
Forest plots and nomogram construction
We established two sets of forest plots for all-cause (Figure 3a–b) and cancer-specific (Figure 3c–d) early death. Based on the results of multivariate logistic analysis, we constructed two prognostic nomograms, including all significant independent factors of all-cause (Figure 4a) and cancer-specific (Figure 4b) early death. Using these nomograms, the early death probability of each patient can be determined by adding the specific numerical value of each predictive variable.

Forest plots. Forest plots depicting the efficacy of different prognostic factors for predicting all-cause early death by univariate logistic regression analysis (a) and multivariate logistic regression analysis (b). Forest plots depicting the effects of different prognostic factors on cancer-specific early death by univariate logistic regression analysis (c) and multivariate logistic regression analysis (d).
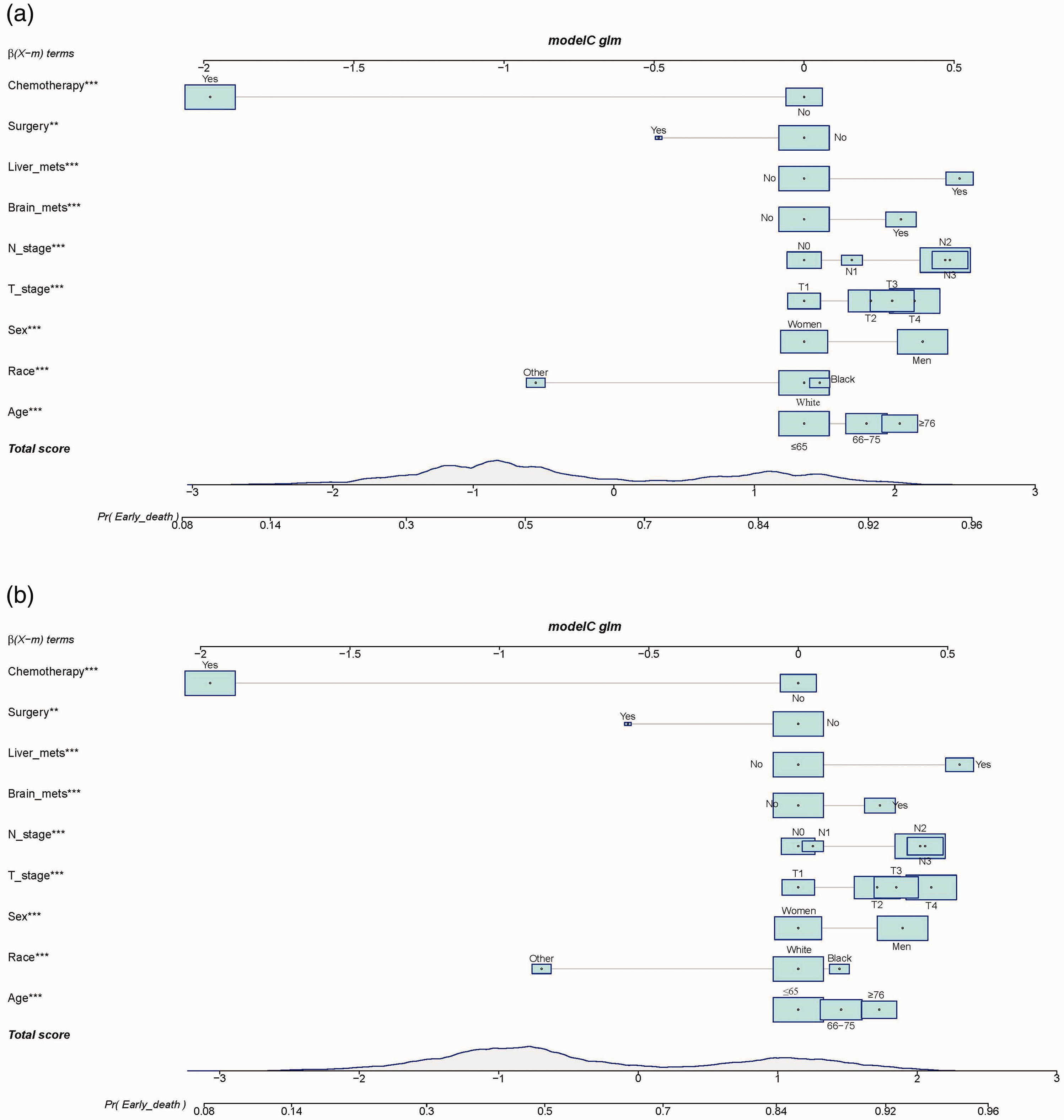
Nomograms developed in this study. Nomograms for predicting all-cause (a) and cancer-specific (b) early death (**p < 0.01 and ***p < 0.001).
Performance of nomograms
In the ROC curve analysis, the area under the curve of the nomograms (Figure 5) for all-causes and cancer-specific early death prediction was 0.754 and 0.753 in the training set and 0.747 and 0.754 in the validation set, respectively. In addition, calibration curves showed the robust calibration of all-cause early death nomograms and cancer-specific early death nomograms (Figure 6). DCA showed that the nomograms provide excellent tools to assess the risk of early death after bone metastasis in patients with LUAD (Figure 7).

ROC curves. The ROC curves for predicting all-cause early death in the training set (a Blue line, AUC = 0.754) and validation set (a Red line, AUC = 0.747). ROC curves for cancer-specific early death in the training set (b Blue line, AUC = 0.753) and validation set (b Red line, AUC = 0.754).

Calibration curves. The calibration curves for predicting all-cause early death in the training set (a) and validation set (b). Calibration curves for cancer-specific early death in the training set (c) and validation set (d).
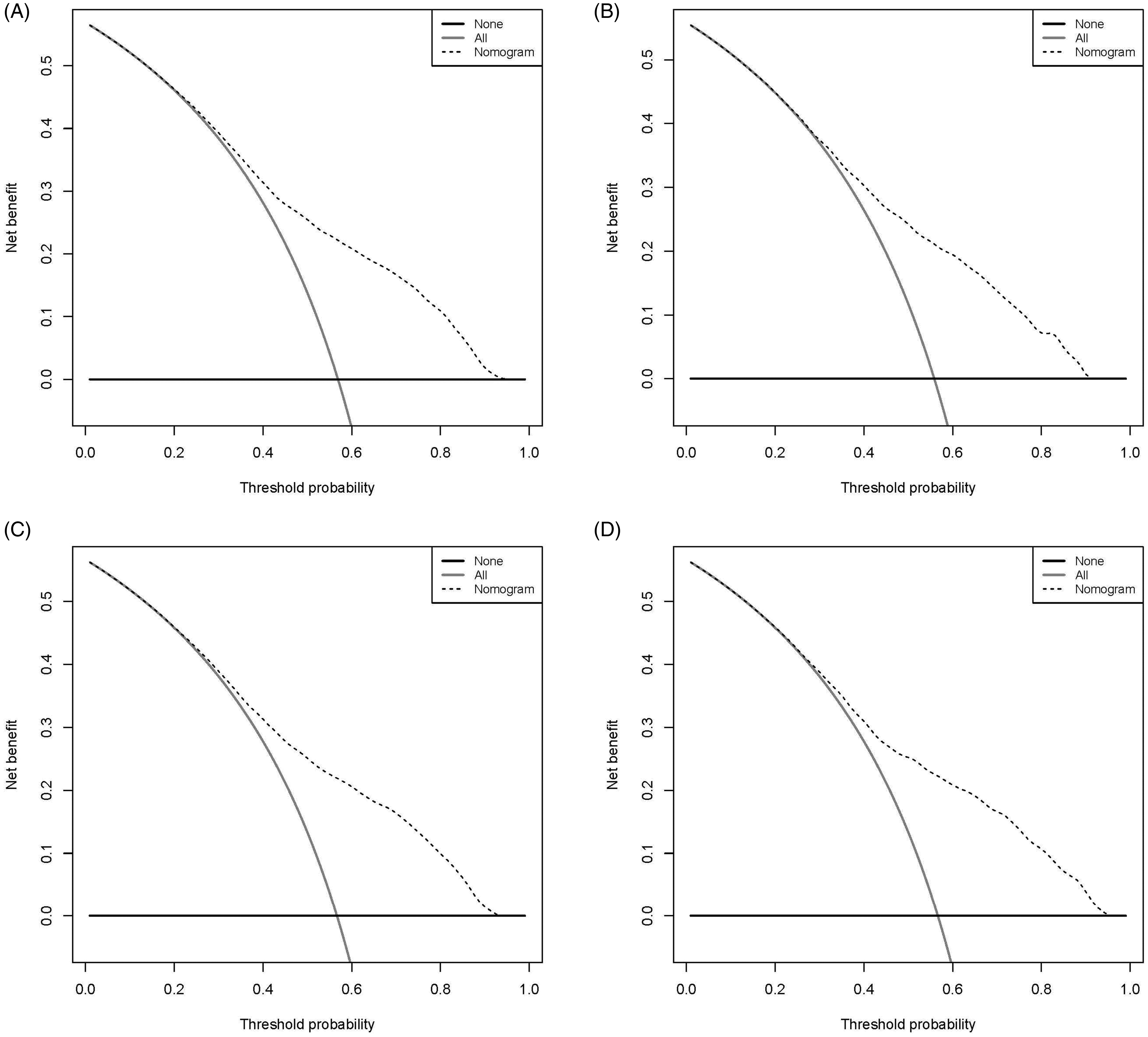
DCA curves. The DCA curve for predicting all-cause early death in the training set (a) and validation set (b) and cancer-specific early death in the training set (c) and validation set (d).
Web-based nomogram
Two web-based nomograms were constructed to predict the all-cause (https://wenn23.shinyapps.io/pmedlabm/) and cancer-specific (https: //wenn23.shinyapps.io/pmedlabmcss/) early death of patients with bone metastasis of LUAD. The nomogram forecast is shown on the left side of the page for all-cause early death (Figure 8a). As an example for calculating the risk of all-cause early death, we included the case of a white 63-year-old female patient with LUAD. Her clinicopathological features were T1 stage, N3 stage, and no brain or liver metastasis. Chemotherapy and surgery were not performed. The patient characteristics are shown on the left side of the web-based nomogram. The graphical summary (Figure 8b) and numerical summary (Figure 8c) showed the probability of all-cause early death by line and precise numerical values [early death prediction = 0.753, 95% confidence interval (CI) = 0.719–0.784], respectively. For cancer-specific death, the nomogram forecast is shown on the left side of the page (Figure 8d), and the results are displayed on the right side. The graphical summary (Figure 8e) and numerical summary (Figure 8f) showed the probability of early death by line and precise numerical values (early death prediction = 0.749, 95%CI=0.713–0.782), respectively.

Web-based nomogram. The nomogram forecast is shown on the left side of the page for all-cause early death (a). As an example of calculating the risk of all-cause early death, we included the case of a 63-year-old white female patient with LUAD. Her clinicopathological features were T1 stage, N3 stage, and no brain or liver metastasis. Chemotherapy and surgery were not performed. The line segment of the graphical summary shows the approximate range of all-cause early death risk (b). The numerical summary shows the precise numerical value of all-cause early death risk (c). The nomogram forecast is included on the left side of the page for cancer-specific early death (d). The line segment of the graphical summary shows the approximate range of cancer-specific early death risk (e). The numerical summary shows the precise numerical value of cancer-specific early death risk (f).
Discussion
Previous epidemiological studies revealed that the prognosis of patients with LC and bone metastasis is poor.1,20 Cho et al. showed that the median time to bone metastasis after LC diagnosis was 2.5 months, and the median survival time of patients with bone metastasis was 7 years.8,21 As reported in the study by Klikovits et al., approximately 75% of patients with LUAD developed bone metastasis within 1 month after being diagnosed. 22 Coleman reported that the incidence of bone metastases in patients with advanced LC was 30% to 40% at first diagnosis. 23 Sandler et al. concluded that the median survival times after the diagnosis of bone metastases was 6 to 12 months. 24 In conclusion, patients with LUAD are prone to bone metastasis during the early stage, and they usually have a poor prognosis.
Studies of metastatic LUAD have usually focused on predicting patient survival.25,26 For malignant and advanced tumors, only a few studies have investigated early death.9–11 Based on different diseases and risk factors, many studies have defined early death.27–29 Using a definition of early death that is too long or short is not conducive for clinicians to provide effective treatment. Therefore, we defined early death as a survival time ≤7 months. 8 For patients with more aggressive tumors, the risk of early death is higher. 30 Therefore, for patients with LUAD and bone metastasis, it is particularly important to calculate the risk of early death to select the most effective treatment. Calculating the risk of early death will facilitate personalized diagnosis and treatment strategies. For patients at high risk, the benefits of treatment in hospitals do not outweigh the associated side effects. In addition, clinicians often overestimate overall survival, which sometimes leads to overtreatment.31,32
In our study, 9189 (56.59%) patients with LUAD and bone metastasis were defined as all-cause early death, and 8585 (56.67%) patients died early from cancer-specific causes. Therefore, we constructed two web-based nomograms for the prediction of early death in these patients. To the best of our knowledge, this is the first study to establish an early death risk prediction model with internal validation for patients with LUAD and bone metastasis. In addition, two web-based nomograms derived from the SEER database of patients with bone metastasis of LUAD were established. These web-based nomograms can be used to help physicians formulate personalized treatment and follow-up plans in patients with LUAD and bone metastasis. 33
Retrospective analysis based on the SEER database has shown that radiotherapy is closely related to the prognosis of patients with LUAD with bone metastasis.25,26 However, our results showed that radiotherapy did not affect cancer-specific early death. Therefore, according to the different needs of the same patient, the type of nomogram should be adjusted. Furthermore, we found that patients with bone metastasis of LUAD rarely underwent surgery (n = 284, 1.75%), but surgical treatment of the primary tumor improved the prognosis of these patients (OR = 0.617, 95%CI = 0.444–0.855, p-value = 0.004). From a clinical perspective, this phenomenon may be related to specific patient conditions. Our study showed that the simultaneous presence of liver metastasis and brain metastasis with bone metastasis was a risk factor for all-cause early death and cancer-specific early death. In contrast, multivariate logistic regression analysis showed that intrapulmonary metastasis was not a risk factor (OR = 1.030, 95%CI = 0.933–1.136, p-value = 0.558). Similarly, Nakajima et al. confirmed that the prognostic impact of intrapulmonary metastases in non-small cell lung cancer was less than that of extrapulmonary metastases. 34 It is well-established that age, T stage, and N stage influence LC prognosis. Based on our results, we further confirmed that age, T stage, and N stage affected early death after bone metastasis in LUAD. Surprisingly, we identified sex (male) as a risk factor for early death in patients with LUAD and bone metastases, and this was possibly closely related to smoking.35,36
There are still some limitations to our research. First, the nomograms did not include potentially relevant factors. For example, the level of smoking is associated with the prognosis of LC. 37 Second, the included indicators lacked a more detailed classification, especially the specific type of surgery, radiation dose, and chemotherapy drug. Third, due to the lack of data from other databases, our nomograms were not verified externally. Finally, this is a retrospective study, and only patients with complete data were included. Excluding patients with incomplete data from the study inevitably led to bias.
Conclusion
We constructed two nomograms and corresponding network-based nomograms to identify all-cause and cancer-specific early death in patients with LUAD and bone metastasis. The early death prediction models can help oncologists develop better treatment strategies.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211047771 - Supplemental material for A predictive web-based nomogram for the early death of patients with lung adenocarcinoma and bone metastasis: a population-based study
Supplemental material, sj-pdf-1-imr-10.1177_03000605211047771 for A predictive web-based nomogram for the early death of patients with lung adenocarcinoma and bone metastasis: a population-based study by Zhehong Li, Junqiang Wei, Haiying Cao, Mingze Song, Yafang Zhang and Yu Jin in Journal of International Medical Research
Footnotes
Acknowledgements
We are grateful for the contributions of the SEER database and the 18 registration agencies that provided information on cancer research. We also thank all colleagues involved in the study.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contributions
YJ designed this study. ZL, JW, HC, MS, and YZ conducted this study and analyzed the data. ZL and JW drafted the manuscript. YJ revised this study. All authors have read and approved the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
