Abstract
Objective
The most common site of metastasis in patients with colon cancer is the liver. This study aimed to identify patients with colon cancer at high risk of developing liver metastasis and to explore their prognosis.
Methods
The clinical characteristics, treatment methods and survival outcomes of patients diagnosed with colon cancer from 2010 to 2015 were identified from the Surveillance, Epidemiology and End Results (SEER) database. Patients were divided into two groups according to the presence of liver metastasis, and multivariate logistic and Cox regression models were used to identify risk and prognostic factors.
Results
A total of 60,018 patients with colon cancer were selected from the SEER database. The incidence of liver metastasis was 9.2%. African American ethnicity, poor differentiation, higher tumor stage, higher lymph node ratio, and lung metastases were common factors associated with both liver metastasis risk and prognosis.
Conclusions
Metastasectomy might improve survival among patients with colon cancer with resectable liver metastasis lesions and no other organ involvement.
Keywords
Introduction
Colorectal cancer is the third most common cancer and cause of cancer-related deaths worldwide in both sexes. 1 According to statistics, new cases of colon cancer accounted for about 5.6% of all cancer cases in 2018, 2 and new deaths accounted for about 8.3%. 1 At the time of diagnosis, about 20% of patients with colon cancer have already developed metastatic disease, 3 with the liver being the most common metastatic site, followed by the lungs.4–6 Studies of selected patients with colon cancer undergoing surgery to remove liver metastases showed that a cure was attainable in this population. 7 Identifying patients at high risk of liver metastasis and subsequent early intervention is thus of great importance to the prognosis of colon cancer. The treatment of colon cancer with liver metastasis includes surgical resection, chemotherapy, or radiotherapy. The range of available treatment modalities for patients with metastatic colon cancer has expanded greatly over the past 10 years, improving the median overall survival from 5 months in 1993 to more than 3 years. 8 However, its low prevalence rate and the lack of large-scale population studies mean that few studies have examined the clinical characteristics of patients with colon cancer and liver metastasis. In this study, we analyzed the risk and prognostic factors of liver metastasis from colorectal cancer using data from the Surveillance, Epidemiology and End Results (SEER) database.
Materials and methods
SEER database
Original public data were obtained from the SEER database and downloaded using SEER*Stat Software version 8.3.8 (https://seer.cancer.gov/data/) (National Cancer Institute, Bethesda, MD, USA). This was a retrospective study and all patient details were de-identified. Patients with colon cancer were identified according to the following inclusion criteria: (1) primary International Classification of Diseases for Oncology-3/American Joint Committee on Cancer 7th edition site code restricted to ‘colon’; (2) diagnosis between 1 January 2010 and 31 December 2015; (3) diagnosis confirmed by histology; and (4) age 18 years or older. Patients with an unclear TNM stage record, unknown survival time, missing cause of death, or unknown diagnostic confirmation were excluded. The detailed procedure for patient selection is outlined in Figure 1.

Flowchart of patient enrollment in this study according to the inclusion and exclusion criteria
Records from SEER between 1 January 2010 and 31 December 2015 were included in the analysis of the incidence and risk factors for liver metastases. These records were included in the survival analysis and to investigate the prognostic factors for liver metastasis.
We stratified the cohort by age, race, sex, site, histological type, pathological grade, Tumor and Node staging, lymph node ratio (LNR), surgery, radiation, chemotherapy, and the presence of other distant metastases (bone, brain, and lung).
This study complied with the Declaration of Helsinki and followed the ethical principles of the ethics committee of the Second Affiliated Hospital of Soochow University. Patients had signed written informed consent to be included in the study.
Statistical analysis
Statistical analysis was carried out using SPSS version 25.0 (IBM Corp., Armonk, NY, USA). Numerical data were expressed as percentages and compared between groups using χ2 tests. To reduce confounding bias in the included cases, all clinicopathological features that would affect the prognosis were matched by propensity scores using a 1:1 nearest neighbor matching method, and the caliper value was set to 0.001. New samples obtained after matching were used to analyze the prognostic effects. Univariate logistic regression analysis was performed to select risk factors for liver metastases, and factors with p < 0.05 were further analyzed using a multivariate logistic regression model. Cox regression analysis was applied to determine the prognostic factors. The overall and cancer-specific survival rates were analyzed using Kaplan–Meier survival curves. A p-value <0.05 was considered statistically significant.
The reporting of this study conforms to the STROBE guidelines. 9
Results
Clinical characteristics of the SEER cohort
A total of 60,018 patients diagnosed with colon cancer from 2010 to 2015 met the inclusion criteria, including 5520 patients (9.2%) with liver metastases and 54,498 patients without. The clinical characteristics of the patients with and without liver metastasis are presented in Table 1. Compared with patients without liver metastases, patients with liver metastasis were more likely to also have lymph node metastases (N1: 37.9% vs 25.4%, N2: 44.0% vs 13.1%, p < 0.001), primary tumor invasion (T3: 59.2% vs 56.1%, T4: 37.6% vs 14.7%, p < 0.001) and a lower LNR (65.3% vs 91.7%, p < 0.001). After propensity score matching, there were 5210 patients with liver metastasis of colon cancer and 5210 patients without liver metastasis. Their clinical characteristics are presented in Table 2.
Clinical characteristics of patients diagnosed with colon cancer with and without liver metastasis in the SEER database (2010–2015).
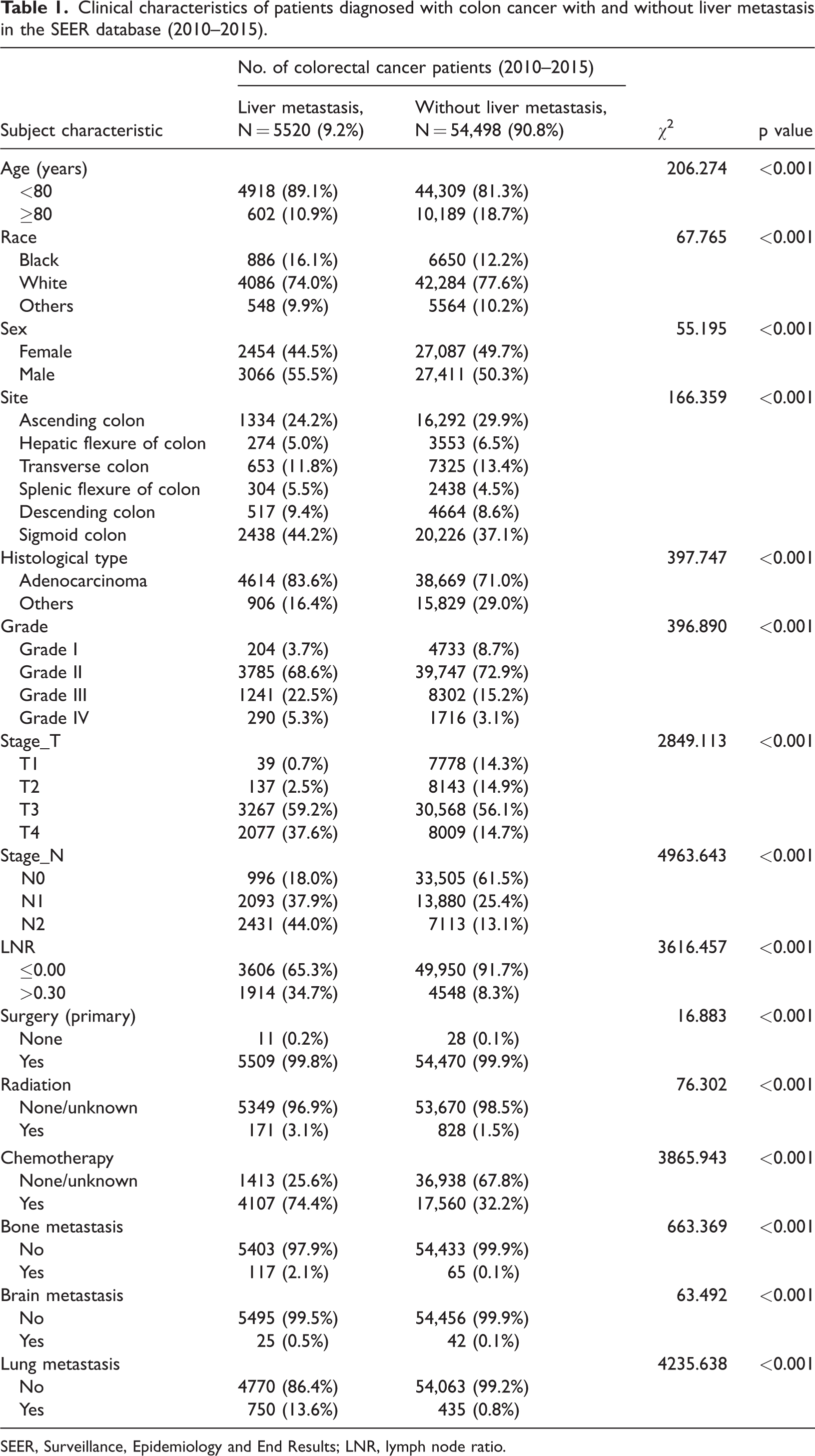
SEER, Surveillance, Epidemiology and End Results; LNR, lymph node ratio.
Clinical characteristics of patients diagnosed with colon cancer with and without liver metastasis matched by propensity scores.

LNR, lymph node ratio.
Risk factors for liver metastases in the SEER cohort
We investigated the risk factors that were significantly associated with liver metastasis in patients with colon cancer. Univariate analysis identified age <80 years, African American ethnicity, adenocarcinoma tissue type, poor differentiation, high tumor stage, high LNR, and lung metastasis as factors associated with a higher risk of liver metastasis (Table 3). In Figure 2, all the factors contained in the left circle represent risk factors for liver metastasis in patients with colon cancer. Subsequent multivariate logistic regression analysis confirmed that age <80 years, African-American ethnicity, poor differentiation, high tumor stage, high LNR, and lung metastasis were positively correlated with liver metastasis (Table 3).
Univariate and multivariate logistic regression analyses of associated risk factors for developing liver metastases in patients diagnosed with colon cancer between 2010 and 2015.
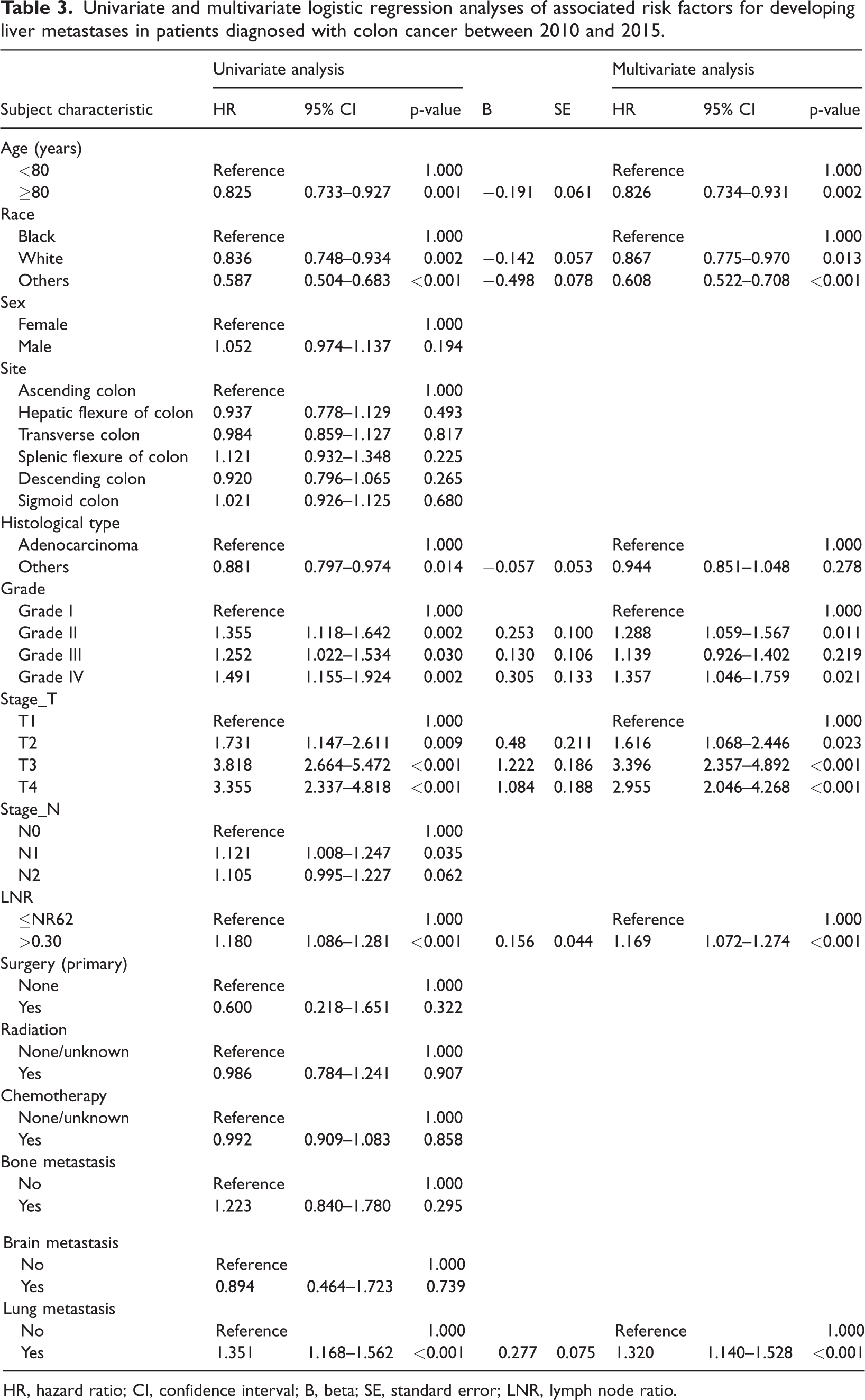
HR, hazard ratio; CI, confidence interval; B, beta; SE, standard error; LNR, lymph node ratio.

Venn diagram of risk and prognostic factors for liver metastasis of colon cancer. All factors in the left circle were identified as risk factors for developing liver metastasis and factors in the right circle as factor associated with prognosis. African American ethnicity, poor differentiation, higher tumor stage, higher lymph node ratio, and lung metastasis were all risk and prognosis factors for liver metastasis in patients with colon cancer.
Survival outcomes and prognostic factors for liver metastasis in the SEER cohort
A total of 5210 patients with colon cancer and liver metastasis were included in the survival analysis. Their median survival time was 24 months (95% confidence interval [CI]: 23.075–24.925) and the 1-, 3-, and 5-year survival rates were 70.5%, 33.5%, and 18.5%, respectively. The clinicopathological features were analyzed by univariate analysis (Table 4) and statistically significant factors were included in the multivariate analysis. Multivariate Cox regression identified advanced age, African American ethnicity, transverse colon location, non-adenocarcinoma histological type, poor differentiation, higher tumor stage, higher lymph node stage, higher LNR, no surgery of primary site, no chemotherapy, and bone, brain, and lung metastases as independent negative prognostic factors for overall survival (Table 5) and associated with shorter cancer-specific survival (Table 5). The median cancer-specific survival time was prolonged from 8.57 (95%CI: 3.351–12.649) to 26.10 (95%CI: 24.074–25.926) months in patients with surgery of the primary site. Kaplan–Meier curves for cancer-specific survival of patients with colon cancer and liver metastasis are shown in Figure 3.
Univariate Cox regression analysis of overall survival in patients with colon cancer with liver metastases in the SEER database (2010–2015).

SEER, Surveillance, Epidemiology and End Results; HR, hazard ratio; CI, confidence interval; LNR, lymph node ratio.
Multivariate Cox regression analysis of overall survival in patients with colon cancer with liver metastases in the SEER database (2010–2015).
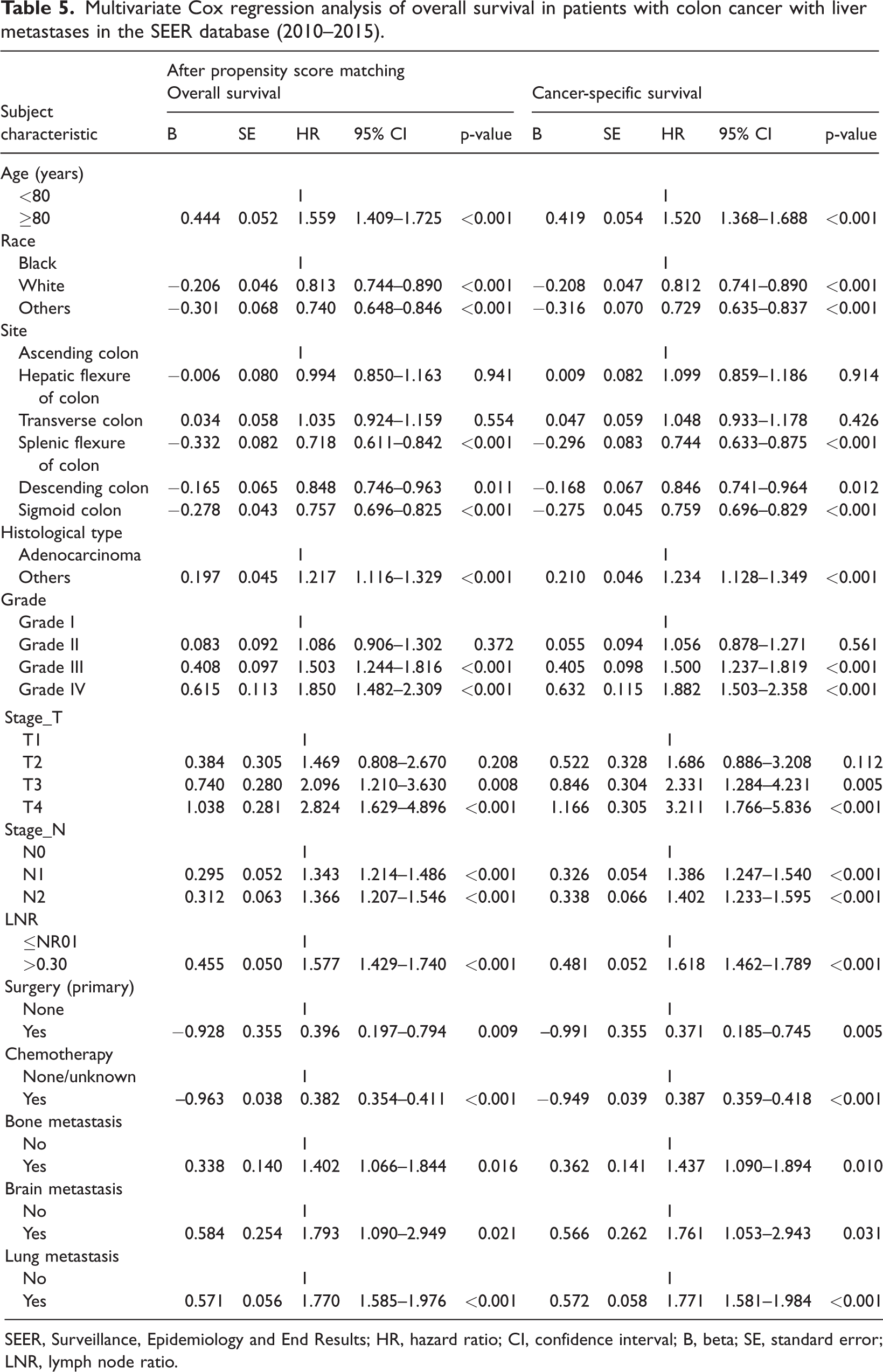
SEER, Surveillance, Epidemiology and End Results; HR, hazard ratio; CI, confidence interval; B, beta; SE, standard error; LNR, lymph node ratio.
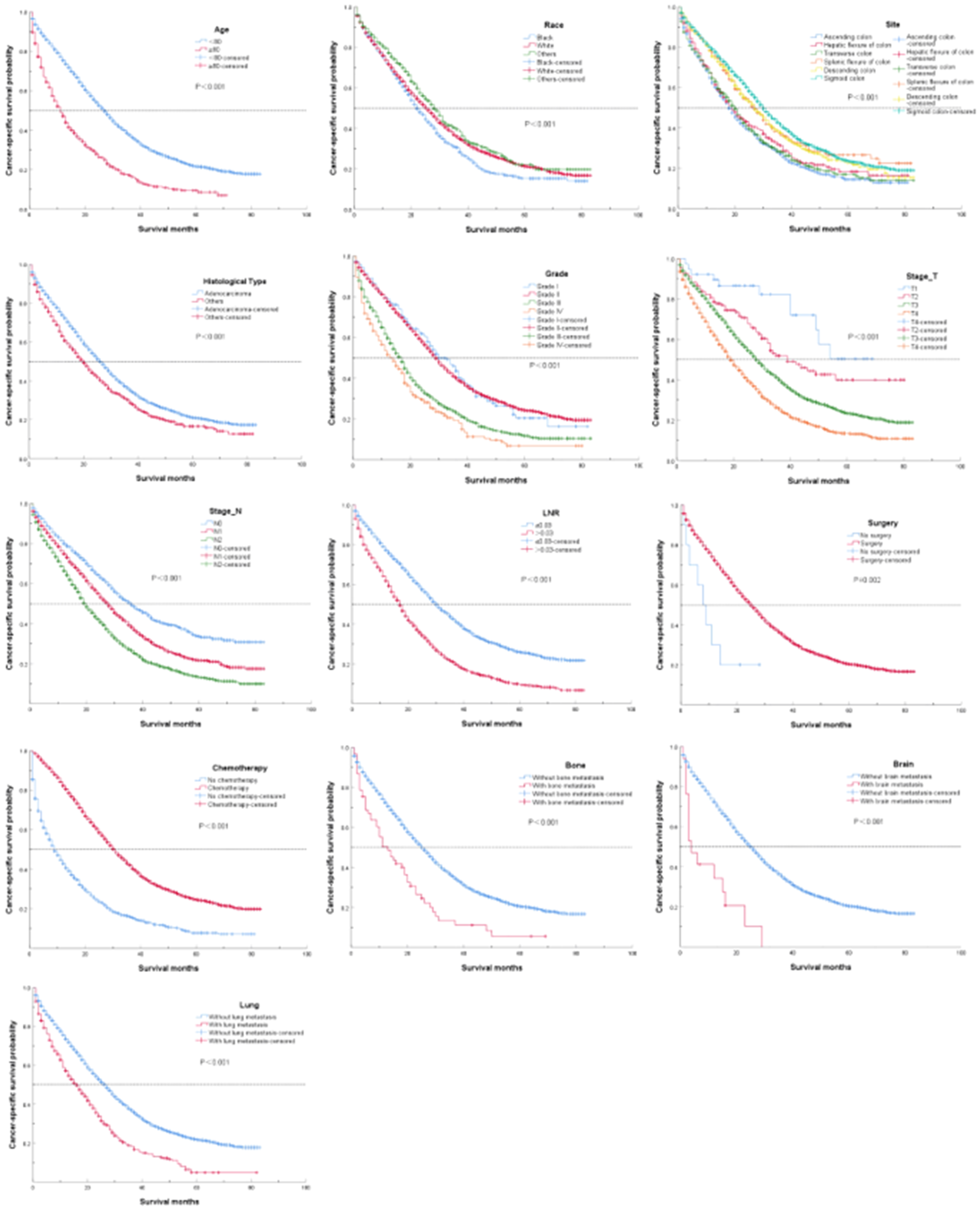
Kaplan–Meier analysis of cancer-specific survival in patients with colon cancer with liver metastasis stratified by related prognostic factors
Discussion
Colon cancer is one of the most common digestive tract tumors, with the third highest incidence and second highest fatality rates worldwide. The incidence is also currently increasing year by year. 10 The liver is the main site for the progression and recurrence of colon cancer, and about 20% of patients with colon cancer are diagnosed with liver metastasis. 11 However, the incidence of liver metastasis of colon cancer in the current study was only 9.2%, which may have been due to bias caused by the small sample size, or because some patients with metastatic tumors did not receive appropriate treatment, resulting in loss of follow-up. The prognosis of patients with liver metastasis of colon cancer is poor, with a reported median survival time for patients receiving conservative treatment of <12 months, and a 5-year survival rate close to 0%. 12 Improving the cure rate of colon cancer liver metastasis and prolonging patient survival are thus keys to improving the prognosis of patients. Based on this, this study examined the risk factors for liver metastasis of colon cancer and the relationship between clinicopathological factors and prognosis in patients with colon cancer and liver metastasis.
Liver metastases of colon cancer are usually asymptomatic but present a poor prognosis, and there is thus an urgent need to identify patients at high risk of liver metastasis. In this study, we found that patients younger than 80 years, with African American ethnicity, adenocarcinoma, late tumor stage, poor differentiation, higher LNR, and with lung metastases were more likely to develop liver metastasis. Adenocarcinoma, late tumor stage, poor differentiation, and higher LNR are unsurprising risk factors for liver metastasis and are already well known poor prognostic predictors for colon cancer. 13 The poor outcomes among African Americans may be related to epidemic-specific factors, such as pollution in the living environment, while social factors such as insufficient medical resources may also lead to a reduced survival rate. Notably, our results showed that younger patients were more likely to develop liver metastasis of colon cancer, possibly as a result of the relatively high misdiagnosis rate and relatively rapid progression of the disease in younger patients with colon cancer. In addition, colon cancer in young people is mainly poorly and moderately differentiated, and is thus more likely to metastasize.
The metastatic pathways of colon cancer can be divided into direct invasion, hematogenous, and lymphatic spread. The most common sites of colon cancer metastasis are the liver, lung, bone, and brain. The current results showed that the coexistence of liver and lung metastasis was common, given that both are caused by hematogenous spread. 14 We therefore recommend that younger patients, African Americans, and patients with late tumor stage, poor differentiation, and a higher LNR should undergo liver imaging to detect early liver metastasis.
Survival analysis showed that lung, brain, or bone metastasis might indicate a poor prognosis in patients with colon cancer and liver metastases. Our results revealed that patients with multiple organ metastases had a poorer prognosis than patients with single organ metastasis. With respect to pathological features, most authors reported that adenocarcinomas had greater propensities for lymph node and distant metastases. 15 We accordingly found that adenocarcinoma was an independent unfavorable risk variable in patients with liver metastases, as shown in previous studies. 15 The 5-year disease-free survival rate of patients with adenocarcinoma or adenosquamous carcinoma has been reported to be significantly lower than for patients with squamous cell carcinoma. 15
Modern oncology divides the colon into the left (distal) and right (proximal) colon, which have slightly different embryonic origins and blood supplies.16,17 The current results showed that the primary site of colon cancer was an independent prognostic factor affecting the survival of patients with liver metastasis, with tumors in the right colon often associated with a poor prognosis. This is similar to the results of previous studies. 18 Missiaglia et al. 19 suggested that the poor prognosis of right colon cancer might be associated with the occurrence of myxoma, high microsatellite instability, mutation of key oncogenes, expression of the BRAF proto-oncogene, and an abnormal serine/threonine kinase signaling pathway.
Multivariate Cox regression analysis showed that patients older than 80 years and African Americans had a poor prognosis. Previous studies have also reported ethnic differences in the incidence and prognosis among patients with colon cancer. 20
The long-term survival rate of patients with liver metastasis of colon cancer was also significantly lower in patients with higher histological grade, higher LNR, and later T and N stages. Increasing numbers of studies have shown that LNR is better than the number of metastatic lymph nodes for predicting the prognosis of patients with colon cancer.21,22 This is because the postoperative N staging system only considers the number of postoperative positive lymph nodes, and does not take account of the total number of postoperative lymph nodes. In contrast, LNR comprehensively considers the total number of seized lymph nodes and the number of positive lymph nodes, which can more accurately reflect the exact stage of the tumor, and thus has greater scientific and reference values for guiding the postoperative treatment of patients. In our study, the best cut-off value of LNR selected by X-tile software was 0.30, and the prognosis of patients became worse with increasing LNR. However, there is still controversy regarding the role of LNR in evaluating the survival and prognosis of patients with liver metastasis of colon cancer and how to determine the optimal cut-off value of LNR. More large-scale multicenter clinical studies are needed to determine how to increase the use of LNR in patients with liver metastasis of colon cancer.
In the current study, the prognosis of patients with liver metastasis of colon cancer was significantly improved after surgery or chemotherapy. Surgical treatment is accordingly the first choice for patients with liver metastasis of colon cancer. 23 However, the effect of surgery alone is not as good as that of surgery combined with chemotherapy, with 3- and 5-year survival rates following comprehensive treatment of 64% and 24%, respectively, and a median survival time of 52 months. 24 The implementation of surgery-based comprehensive treatment is thus key to the treatment of liver metastasis of colon cancer.
This study had some limitations. First, it was a retrospective study and it might thus have included some unavoidable bias. Second, the sample size was limited. We are currently collecting data for more patients in our center and in cooperation with another institution, and aim to update our results in the future.
In conclusion, liver metastasis from colon cancer has a poor prognosis. Younger age, African American ethnicity, poor differentiation, higher tumor stage, higher LNR, and lung metastasis are associated with an increased risk of developing liver metastasis. Liver imaging assessment is thus highly recommended in these patients within 1 year after primary treatment. Advanced age, African American ethnicity, tumor site in the transverse colon, non-adenocarcinoma histological type, poor differentiation, higher tumor stage, higher lymph node stage, higher LNR, no surgery of primary site, no chemotherapy, and bone, brain, and lung metastases are all negative prognostic factors in patients with colon cancer with liver metastasis. Metastasectomy might help to prolong survival, especially in patients with solitary liver lesions and without other organ involvement.
Footnotes
Author contributions
JG, LZ, and CH designed the research. LZ, ZZ, and CH performed the research and analyzed results. JG and XX wrote the paper. JG, XX, and WC edited the manuscript and provided critical comments. All authors read and approved the final manuscript.
Availability of data and materials
The data that support the findings of this study are available on request from the corresponding author, [W.C.] who received permission from the Second Affiliated Hospital of Soochow University ethics committee. The dataset from the SEER database generated and/or analyzed during the current study is available in the SEER dataset repository (![]() ) in the Availability of Data and Materials.
) in the Availability of Data and Materials.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors .
