Abstract
Background
Penicilliosis marneffei is a rare disease caused by Talaromyces marneffei, which is endemic in Southeast Asian countries, and usually occurs in immunocompromised or immunodeficient hosts. We report an unusual misdiagnosed case of penicilliosis marneffei in an immunocompetent patient from a non-endemic area of China.
Conclusion
Timely fungal culture and pathological examination of clinical specimens are critical for the diagnosis of penicilliosis marneffei, especially in non-endemic regions and immunocompetent patients.
Introduction
Penicilliosis marneffi (PSM) is an opportunistic fungal disease that can prove fatal, especially in immunosuppressed and immunodeficient patients, such as those with human immunodeficiency virus (HIV), autoimmune diseases, and malignant tumors, and after organ transplantation. 1 Disalvo et al. 2 first described human PSM infection in 1973. Bamboo rats are the primary source of human PSM infection. 3 PSM infection is highly endemic in tropical Asia, especially Vietnam, Thailand, India, and South China. 4 However, previous studies have shown a low incidence of PSM in non-endemic areas or in immunocompetent patients. We describe here a rare case of Talaromyces marneffei infection in an immunocompetent patient from the non-endemic region of China. This infection was misdiagnosed as lung cancer and had distinct pathological features in the lungs.
Case report
A 59-year-old man from the central part of China presented to hospital with a 3-month history of cough and progressive hemoptysis. He had pre-existing hypertension for 10 years and no prior family history of malignancy or any other diseases. He is a farmer who was born and worked in Hubei Province, and had never visited other parts of China.
Chest computed tomography (CT) showed a space-occupying lesion in the lower lobe of the left lung. Positron emission/CT showed malignant lesions with mediastinal and bilateral hilar lymph node metastasis. Blood tests showed an increase in the following inflammatory biomarkers: white blood cell count, 11.85 × 109/L (range: 3.5–9.5 × 109/L); neutrophils, 76.2% (range: 40%–75%); and C-reactive protein level, 60.5 mg/L (range: 0–10 mg/L). The tumor markers neurone specific enolase (0.8 ng/mL, range: 0–16.3 ng/mL) and CA125 (69.9 U/mL, range: 0–30.2 U/ml) were also increased. However, conventional anti-infection therapy in his local hospital was ineffective. He was diagnosed with lung cancer and administered gefitinib anti-tumor treatment for 1 month. The patient had visited multiple hospitals where the above-mentioned findings were obtained before the first visit. The diagnostic process is shown in Table 1. At admission to our hospital, his symptom of hemoptysis was aggravated, especially the frequency of hemoptysis. He did not have a fever, and no gut, skin, or nervous system symptoms, and no obvious abnormal signs were observed in a general physical examination.
Hospitalization process of the patient.
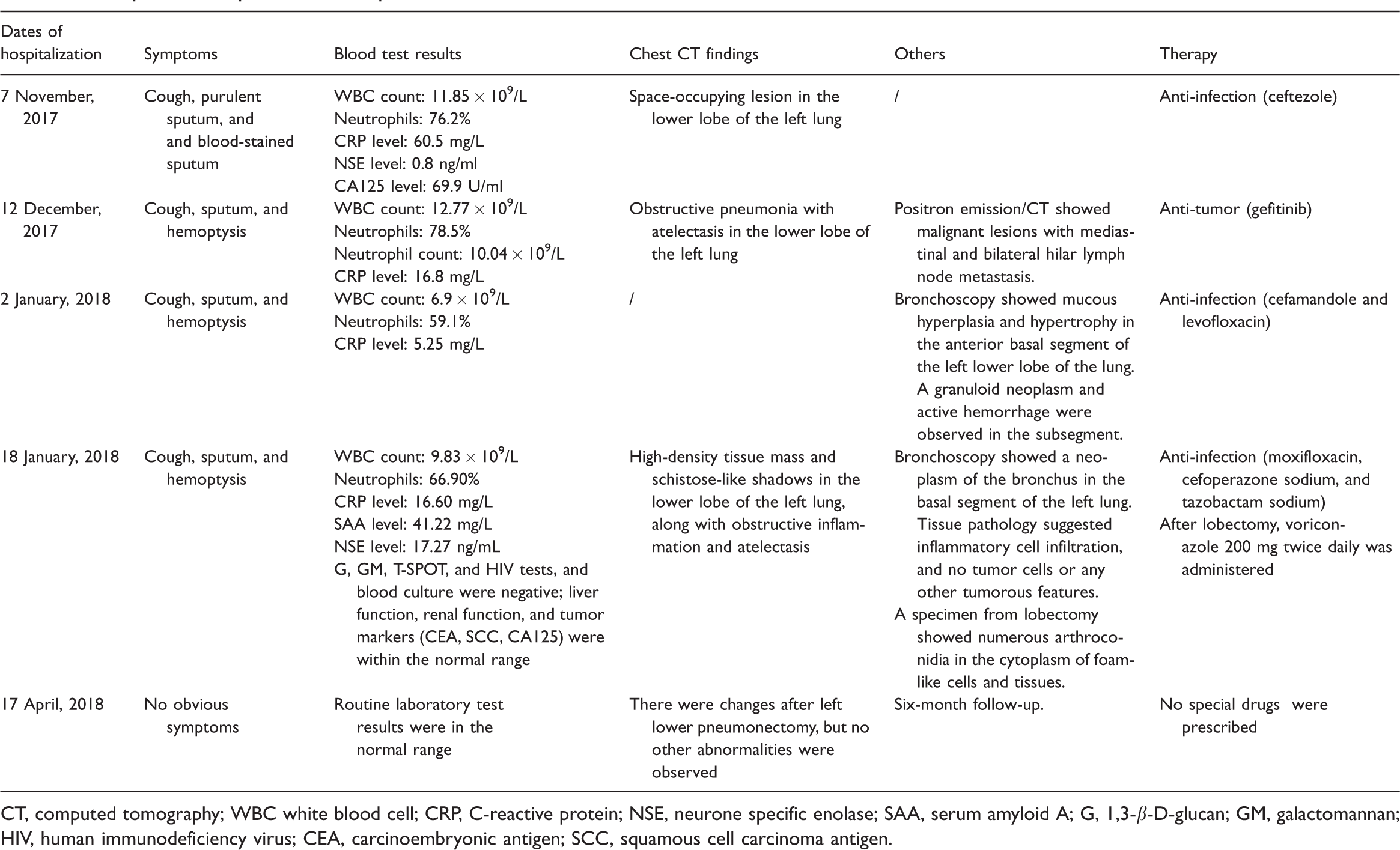
CT, computed tomography; WBC white blood cell; CRP, C-reactive protein; NSE, neurone specific enolase; SAA, serum amyloid A; G, 1,3-β-D-glucan; GM, galactomannan; HIV, human immunodeficiency virus; CEA, carcinoembryonic antigen; SCC, squamous cell carcinoma antigen.
After admission to our hospital on 18 January 2018, routine laboratory tests showed that inflammatory biomarker levels were high, including a white blood cell count of 9.83 × 109, neutrophil count of 6.57 × 109, C-reactive protein level of 16.6 mg/L, and serum amyloid A level of 41.22 mg/L (range: <10 mg/L). The erythrocyte sedimentation rate was elevated (65 mm/hour, range: 0–21 mm/hour), but the hemoglobin content was low (118 g/L, range: 130–175 g/L). A 1,3-β-D-glucan test, aspergillus galactomannan test, tuberculosis T-SPOT, purified protein derivative test, and HIV test were negative. No fungal growth was detected in blood culture. A chest CT scan showed a high-density tissue mass and schistose-like shadows in the lower lobe of the left lung, along with obstructive inflammation and atelectasis (Figure 1a & b). A bronchoscopic examination indicated bronchial neoplasm in the basal segment of the left lung (Figure 1c & d). A tuberculosis gene X-pert test (Cepheid Co., Sunnyvale, CA, USA) of bronchoalveolar lavage fluid from bronchoscopy to distinguish tuberculosis was negative, but tissue pathology did not contribute to a diagnosis. Because of the uncertain property of the mass and the patient’s symptoms, we performed pulmonary lobectomy followed by a morphological and pathological examination with the consent of the patient.

Chest contrast-enhanced computed tomography scan shows a high-density tissue mass (arrows) and schistose-like shadows in the lower lobe of the left lung, along with obstructive inflammation and atelectasis (a, b). A bronchoscopy examination shows a neoplasm of the bronchus in the basal segment of the left lung (arrow) (c, d).
Immunohistochemical and specific staining was performed. Routine hematoxylin and eosin staining showed yeast-like organisms aggregated in the alveolar cavity and interstitium, with hyperplasia of interstitial fibers, and scattered lymphocytic infiltration (Figure 2a). The tissue was positive for CD138, CD38, and immunoglobulin G, but negative for immunoglobulin G4 in immunohistochemical staining. Moreover, hexamine silver staining (Figure 2b) showed numerous yeast-like arthroconidia in the cytoplasm of histocytes, and periodic acid–Schiff staining (Figure 2c) showed specific characteristics of binary fission and arthroconidia with a central transverse septum. Finally, based on immunohistochemical and specific staining, the patient was diagnosed with penicilliosis marneffei. He was then prescribed specific anti-fungal treatment of voriconazole 200 mg twice daily for 12 weeks. At a 1-year follow-up, no obvious respiratory symptoms were observed, and he was not limited in his daily physical activities. The most recent chest X-ray showed changes in imaging findings after left lower pneumonectomy, but no other abnormalities were observed (Figure 3).

Hematoxylin and eosin staining shows yeast-like organisms aggregated in the alveolar cavity (arrows) and interstitium, with hyperplasia of interstitial fibers and scattered lymphocytic infiltration (a). Hexamine silver staining (b) shows numerous yeast-like arthroconidia in the cytoplasm of histocytes (arrows). Periodic acid–Schiff staining (c) shows penicilliosis marneffei-specific characteristics. The yellow arrow indicates binary fission, the red arrow indicates arthroconidia with a central transverse septum, and the blue arrow indicates a budding yeast cell. Magnification, ×400 for each image.

Chest X-ray shows normal changes after left lower pneumonectomy, but no other abnormalities can be seen.
Discussion
PSM is a fungal disease, which is extremely unusual in immunocompetent hosts and non-endemic regions. 5 PSM invades the mononuclear macrophage system and the severity of infection varies with the gradation of immunosuppression. 6 Clinical manifestations of PSM are nonspecific, and the most common symptoms are fever, skin and soft tissue lesions, swollen lymph nodes, cough, and dyspnea. A laboratory examination of PSM can show abnormalities of leukocytes, anemia, thrombocytosis, elevated inflammatory biomarkers, abscess formation, and osteolytic lesions. 7 Various presentations of PSM infection of the lungs are observed by radiographic imaging, including simple or multiple lobular consolidation, cavitation, diffuse honeycombing or interstitial infiltration, and mediastinal and hilar lymph node lesions. 8 PSM is a temperature-dependent dimorphic fungus, presenting as yeast at 37°C, but as mycelium at 25°C. Although microbiological fungal culture is currently the gold standard for diagnosis of PSM, diagnosis can also be made from histopathological and cytological examinations of clinical specimens. 9 Recently, diagnosis of PSM by high-throughput sequencing and dermatoscopy has been reported. 10
The present case was a patient from a non-epidemic area who was immunocompetent and showed the symptoms of cough and hemoptysis. Tumor marker levels were increased, while a fungal test and blood culture were negative. Imaging showed an irregular tissue mass associated with obstructive inflammation and atelectasis, and enlarged mediastinal and bilateral hilar lymph nodes. CT findings of lung cancer generally include a lobulated mass, burr-like mass, and pleural depression, which are different from the presentations of PSM with a CT scan. However, such a mass is difficult to distinguish from a tumor and easily misdiagnosed as lung cancer, but pathology plays a crucial role in diagnosis. Because the patient was admitted to several different hospitals, and the process of diagnosis and treatment was complicated, diagnosis of PSM was not considered in the early stage. Failure to carry out fungal culture was a critical limitation of the present case. Moreover, the previous long-term treatment of different drugs may also have affected the blood culture and fungal test results, which were negative in the later stage. As mentioned above, the diagnosis of PSM can also be made from histopathological and cytological examinations of clinical specimens. Based on findings in the present case, physicians should take into account some rare diseases with common symptoms. Finally, diagnosis of PSM in our patient was confirmed by a histopathological examination after lobectomy in our hospital.
Currently, the recommended treatment for patients with HIV and PSM infection is amphotericin B, 11 but there is no standard treatment of PSM in non-HIV patients. 12 Our patient was prescribed with specific anti-fungal treatment of voriconazole 200 mg twice daily for 12 weeks. His general condition was good, his activity tolerance recovered, and a chest CT scan showed no abnormalities at a 1-year follow-up.
In conclusion, our findings from this rare case report will hopefully improve the awareness of PSM among clinicians in non-endemic areas, especially in immunocompetent patients with atypical symptoms. Timely fungal culture of clinical specimens and a pathological examination should be performed to reduce misdiagnosis and improve the survival rate of PSM.
Footnotes
Acknowledgements
The authors are greatly appreciative of the patient for providing a detailed medical history and agreement for submission of the case report. We thank Dr. Luke R. Bonser from the University of California, San Francisco for carefully reading and revising the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This project was supported by grants from the National Natural Science Foundation of China (81600023) and the China Scholarship Council Fund (201806275100).
Ethics statement
The study protocol was approved by the ethics review committee of Renmin Hospital of Wuhan University. Informed written consent was obtained from the patient for publication of this case report and accompanying images.
