Abstract
Pulmonary actinomycosis is a rare pulmonary infectious disease that is often challenging to diagnose early and has a high misdiagnosis rate. In some cases, it can be particularly difficult to distinguish pulmonary actinomycosis from lung cancer. We herein report a rare case of pulmonary actinomycosis in which the preoperative examinations strongly suggested lung cancer, leading to the patient undergoing right upper lung resection and bronchoplasty. The patient had a good postoperative recovery; however, the postoperative pathology report indicated pulmonary actinomycosis. In this report, we summarize the key aspects of the diagnosis and treatment of pulmonary actinomycosis to aid clinicians in reducing the likelihood of misdiagnosis.
Keywords
Introduction
Actinobacteria are a type of prokaryotic organism that grow in a filamentous form and mainly reproduce through spores. They are microorganisms that lie between bacteria and filamentous fungi. Actinobacteria are commonly found in the oral cavity, dental caries, and tonsillar crypts of normal individuals. 1 Pulmonary actinomycosis is a chronic suppurative granulomatous disease caused by infection of the lungs with actinomycetes, particularly anaerobic Actinobacteria. The incidence of pulmonary actinomycosis is very low, with an annual incidence rate of approximately 1 in 300,000. 2 The disease can occur in all age groups, with the highest incidence rate among young adults. The male:female prevalence ratio is approximately 3:1.3–5 The risk of pulmonary actinomycosis is significantly increased in patients with underlying respiratory diseases such as emphysema, chronic bronchitis, and bronchiectasis. In addition, factors such as alcohol abuse, poor oral hygiene, and pre-existing oral diseases further elevate the risk. 6
It is sometimes difficult to differentiate pulmonary actinomycosis from lung cancer. This article focuses on the key points of differentiation between the two, presents some diagnostic considerations, and points out when surgical treatment is needed.
Case presentation
A 67-year-old woman was admitted to our hospital because of a 1-month history of coughing and expectoration. One month previously, the patient had developed a cough and expectoration without obvious inducement. The cough was non-irritating and produced a small amount of white, sticky sputum that was difficult to expel. The patient had no bloody sputum or fever. A chest computed tomography (CT) scan performed at a local hospital indicated a possible tumor in the right upper lung, prompting the patient to seek further treatment at our hospital. She had a history of hypertension but was not consistently taking antihypertensive drugs. Physical examination revealed no clinically significant findings.
Upon admission, an enhanced chest CT scan revealed a well-vascularized lesion in the right upper lobe of the lung, raising strong suspicion of lung cancer (Figure 1(a) and (b)). Positron emission tomography–CT (PET-CT) examination further supported the suspicion of pulmonary malignancy. Bronchoscopy revealed no obvious mass in the lumen of the right upper lobe of the lung (Figure 1(c)), and lavage and brushing of the right upper lobe showed no obvious abnormalities. Furthermore, other preoperative tests showed no obvious abnormalities.
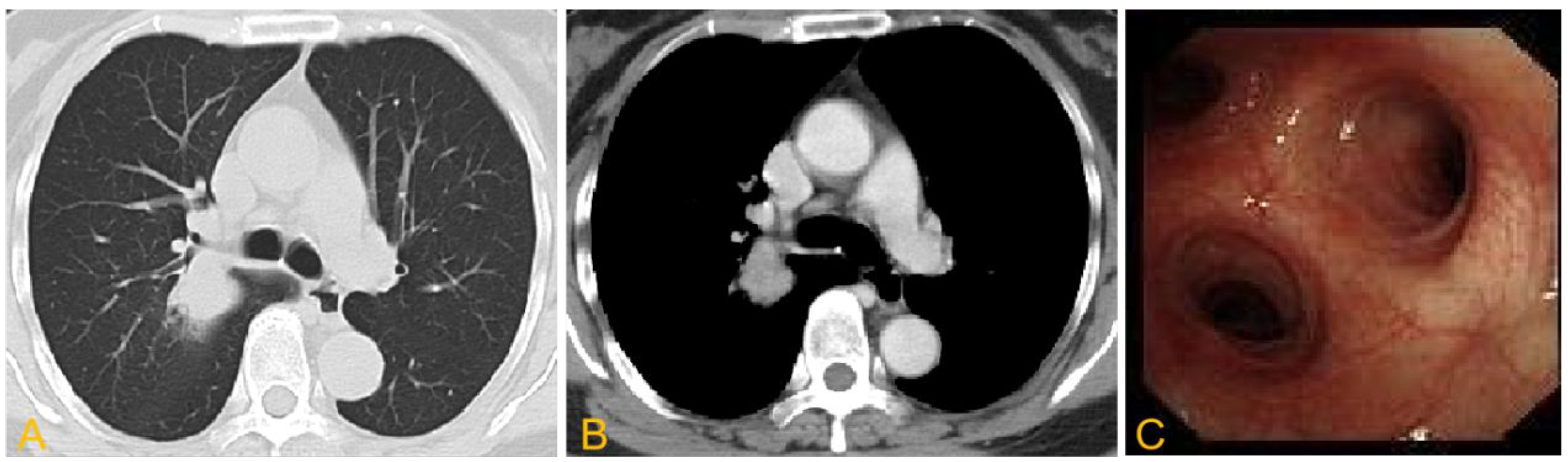
Imaging findings of suspected lung mass. (a, b) A mass-like soft tissue shadow was seen in the upper lobe of the right lung, measuring approximately 2.1 × 2.5 cm and accompanied by surrounding patchy, floccular shadows and moderate enhancement and (c) no obvious mass was found in the lumen of the upper lobe of the right lung during bronchoscopy.
After ruling out any surgical contraindications, the patient underwent right upper lobe resection and bronchoplasty under general anesthesia. During the operation, no pleural effusion was observed in the right thoracic cavity. A large, hard mass approximately 3.0 × 2.5 cm in size was palpable in the posterior segment of the right upper lobe near the hilum. The mass had invaded the dorsal segment of the right lower lobe, and the hilar, inferior tracheal, and upper mediastinal lymph nodes were swollen.
The anterior mediastinal pleura was incised, and the hilar lymph nodes were separated and resected. The right upper lobe pulmonary vein was isolated, clamped, and transected; the distal end was ligated, while the proximal end was ligated and sutured. Similarly, the anterior segmental artery was isolated, clamped, and transected; the distal end was ligated, while the proximal end was ligated and sutured. After incising the posterior mediastinal pleura and freeing the interlobar fissure, a tunnel was established along the interlobar artery. A linear cutting occluder was used to transect the posterior part of the horizontal and oblique fissures, and the invaded lower lung tissue was resected. The interlobar lymph nodes were separated and resected. The posterior ascending artery was separated and clamped, and the proximal ends were doubly ligated. The middle bronchus and right upper lobe bronchus lymph nodes were resected. The tumor in the right upper lung had invaded the outer wall of the bronchial opening of the right upper lung. To completely remove the tumor, we decided to perform right bronchoplasty. The right main bronchus and intermediate bronchus were incised at the upper and lower sides of the right upper lobe bronchus. A V-shaped incision was made to section the bronchus; the right upper lung tissue was then removed, and the pulmonary ligament was cut off. The right main bronchus and right intermediate bronchus were anastomosed end-to-end using 3-0 Prolene in a continuous pattern. The resected tissue was sent for rapid examination, which revealed inflammatory changes in the right upper lung. No abnormalities were found in the bronchial stump.
The patient recovered well after surgery and was discharged smoothly. After discharge, she was prescribed oral amoxicillin for 2 months and had an uneventful follow-up. Microscopic examination of the surgical specimen revealed a mass in the lung parenchyma near the hilum of the right upper lung. The mass contained organizing pneumonia, small abscesses, actinomyces-like clusters, fibrous tissue proliferation, and chronic inflammatory cell infiltration (Figure 2). The routine pathological diagnosis was an inflammatory pseudotumor with actinomyces infection.

Histopathological examination of lung tissue. (a) Actinomycetes colony with surrounding small abscesses (hematoxylin–eosin, ×400) and (b) evidence of organizing pneumonia in the lung tissue (hematoxylin–eosin, ×100).
Written informed consent for publication of this report was obtained from the patient. The reporting of this study conforms to the CARE guidelines. 7
Discussion
The early symptoms of pulmonary actinomycosis mainly include cough, expectoration of viscous purulent or bloody sputum, fatigue, night sweats, and fever. As the disease progresses, patients may exhibit signs of chronic debilitating infection such as emaciation, clubbing of fingers, anemia, and hypoproteinemia. Symptoms may worsen when multiple abscesses form in the lungs, manifesting as high fever, production of copious mucus and purulent sputum, blood-tinged sputum or massive hemoptysis, fatigue, night sweats, anemia, and weight loss.8,9 When the disease involves the pleura, patients may experience severe chest pain. Invasion of the chest wall can lead to the formation of subcutaneous abscesses and fistulas, often discharging pus mixed with bacterial masses. Involvement of the mediastinum can cause dyspnea or dysphagia, and in severe cases, it can lead to death. However, the clinical manifestations of pulmonary actinomycosis are nonspecific, and further examination is needed to confirm the diagnosis.
Laboratory tests can be performed to detect the presence of actinomyces in the sputum, bronchoalveolar lavage fluid, or biopsy specimens (obtained through bronchoscopy or aspiration). Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry and 16S ribosomal RNA sequencing are reportedly beneficial for the isolation of actinomyces,10–13 thereby facilitating the diagnosis of pulmonary actinomycosis. After actinomyces infection has become established in the lungs, local abscesses accompanied by sulfur granules may form. These sulfur granules are a characteristic manifestation of actinomyces infection and an important basis for the diagnosis of pulmonary actinomycosis. 14 Notably, the presence of sulfur granules does not necessarily indicate pulmonary actinomycosis; these granules may also be related to other lung diseases, such as nocardiosis. 15 When it is difficult to distinguish between pulmonary actinomycosis and pulmonary cryptococcosis, serological cryptococcal capsular polysaccharide antigen detection can be used for rapid clinical diagnosis of pulmonary cryptococcosis, especially for patients with normal immune function. Positive results are clinically significant for the diagnosis of pulmonary cryptococcosis. Metagenomic next-generation sequencing (mNGS) is also an effective differential method. With the development of molecular biological detection methods, mNGS has become widely used in clinical microbiological detection. It is especially suitable for detecting complex, rare, and atypical infectious diseases.16,17
Imaging examination is also an important means of diagnosing pulmonary actinomycosis. The CT features of pulmonary actinomycosis include segmental consolidation, peripheral enhancement of low-density areas, and pleural thickening. With chronic progression of the disease, pulmonary nodules may evolve into masses or airspace consolidation. Airspace consolidation with a central low-density area and multiple cavitations is a typical feature of pulmonary actinomycosis.18,19 These CT features can assist in the diagnosis and differentiation of pulmonary actinomycosis. PET-CT does not have an advantage in distinguishing lung cancer from pulmonary actinomycosis, and in many cases, actinomyces has mimicked the presentation of lung cancer on PET-CT. The proportion of pulmonary actinomycosis cases misdiagnosed as lung cancer by PET-CT can reportedly reach two-thirds, 20 making it difficult to use PET-CT for differential diagnosis between the two. In the present case, the preoperative diagnosis was considered lung cancer, and the purpose of the PET-CT examination was to stage the lung cancer before surgery.
Although the above-mentioned examinations can assist in diagnosis, it is almost impossible to correctly diagnose pulmonary actinomycosis during the first visit. 21 The four main reasons for misdiagnosis of pulmonary actinomycosis are as follows. First, pulmonary actinomycosis is a rare infectious disease, and clinicians often lack sufficient knowledge of it. This can lead to lack of awareness and a tendency to overlook the diagnosis. Second, the clinical and imaging manifestations of pulmonary actinomycosis are nonspecific and similar to those of common diseases such as tuberculosis and lung cancer, making it difficult to distinguish between these conditions.22,23 Third, it is difficult to isolate and cultivate actinomycetes. Because of the harsh cultivation conditions and high technical requirements for anaerobic bacteria, it is difficult to directly cultivate pathogenic bacteria, and positive results can only be obtained in a few cases through cultivation. Fourth, pulmonary actinomycosis is rarely seen in clinical practice, and many doctors are not familiar with its diagnostic criteria. Thus, it can easily be confused with other lung diseases.
The treatment of pulmonary actinomycosis involves three main aspects: general therapy, drug therapy, and surgery. General therapy includes ensuring adequate rest, nutrition, and physical fitness. Heavy physical activity and strenuous exercise should be avoided. With respect to drug therapy, penicillin is the first choice for the treatment of actinomycosis. This drug can destroy the protein of the bacteria, thus playing a role in sterilization. Antibiotics such as doxycycline, ceftriaxone, clindamycin, and erythromycin, to which pulmonary actinomycosis is sensitive, are also commonly used for sterilization therapy. These drugs also have antibacterial activity, thus controlling infection. Finally, if the drug therapy is ineffective or the condition progresses, surgical treatment should be considered. Notably, some patients are considered to have lung cancer before surgery, and only after surgical treatment are they found to have pulmonary actinomycosis.
Our literature review has revealed that bronchoalveolar lavage, lung biopsy, and surgical resection are the primary approaches for diagnosing pulmonary actinomycosis. Accurate preoperative diagnosis can avoid unnecessary surgical resection. However, culturing actinomyces from alveolar lavage and biopsy specimens is a challenging task because antibiotic use, improper specimen preservation, and microbial contamination can inhibit its growth, leading to diagnostic difficulties. In addition, bronchoalveolar lavage submitted for mNGS can serve as another diagnostic method. The mNGS technique does not rely on microbial culture; this can improve the identification of difficult-to-culture microorganisms, and the results are unlikely to be affected by prior exposure to antimicrobial drugs. 24 When the above tests fail to provide a definitive diagnosis, surgery may also be considered.
Conclusion
The incidence rate of pulmonary actinomycosis is low, and the diagnosis is difficult. Therefore, when diagnosing pulmonary actinomycosis, doctors must comprehensively consider the patient’s clinical manifestations, laboratory examinations, imaging manifestations, and other information to avoid misdiagnosis and missed diagnosis.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241275375 - Supplemental material for Pulmonary actinomycosis misdiagnosed as lung cancer: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605241275375 for Pulmonary actinomycosis misdiagnosed as lung cancer: a case report by Song Wu and Zhonghua Qin in Journal of International Medical Research
Footnotes
Authors’ contributions
Concept and design: ZHQ. Data collection and manuscript drafting: ZHQ. Administrative support: SW. Both authors discussed the results and revised the manuscript.
Availability of data and materials
The corresponding author can be contacted for further details.
Consent for publication
Written informed consent for publication was obtained from the patient. A copy of the written consent is available for review by the editor of this journal.
Declaration of conflicting interests
None of the authors has any conflict of interest to disclose.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Jiangyin People’s Hospital. Written informed consent was obtained from the patient.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
