Abstract
Objective
To establish a diagnostic model of idiopathic central precocious puberty on the basis of transrectal pelvic ultrasound and basal gonadotropin.
Methods
A total of 669 girls with Tanner breast development stage II were enrolled in this study from January 2015 to December 2018. The participants were divided into the ICPP group and the premature thelarche group. We analyzed various variables, including age at initial diagnosis, basal luteinizing hormone levels, the long diameter of the uterus, the transverse diameter of the uterus, the anterior–posterior diameter of the uterus, the volume of the uterus, maximum ovarian diameter, average ovarian volume, maximum ovarian volume, number of follicles (≥4 mm), maximum follicular diameter, endometrial thickness, and vaginal wall thickness.
Results
The following diagnostic model was established: Y=−14.123 + 0.630 × age at initial diagnosis + 1.119 × transverse diameter of the uterus + 1.278 × anterior–posterior diameter of the uterus + 0.637 × average ovarian volume + 1.316 × maximum ovarian diameter + 0.146 ×number of follicles ≥4 mm + 2.925 × endometrial thickness + 0.559 × basal luteinizing hormone value. The area under curve was 0.922, sensitivity was 84.9%, and specificity was 86.2%.
Conclusion
Basal LH levels and transrectal pelvic ultrasound should be applied together to improve the accuracy of diagnosis in ICPP.
Keywords
Introduction
Recently, the incidence of precocious puberty in children has shown a yearly increase. 1 The incidence of precocious puberty in girls is 5 to 10 times higher than that in boys and more than 90% of these cases are idiopathic. 2 Idiopathic central precocious puberty (ICPP) and premature thelarche (PT) are diseases with the primary manifestation of secondary sexual characteristics.3–5 Early diagnosis and timely treatment of these diseases are important and there is a need for a more convenient and efficient alternative method for diagnosis and treatment in clinical practice. Previous studies have shown that basal luteinizing hormone (LH) levels and pelvic ultrasound findings are important predictors of central precocious puberty.6–8
Therefore, we conducted this study to establish an ICPP diagnostic model with transrectal pelvic ultrasound and basal gonadotropin as variables, and to evaluate the diagnostic efficacy of this model.
Material and methods
Subjects
We performed an observational study from January 2015 to December 2018. We recruited girls with suspected precocious puberty in the Pediatric Endocrine Department of the Second Hospital of Hebei Medical University. This study was conducted in accordance with the declaration of Helsinki and approved by the Ethics Committee of the Affiliated Hospital of Hebei University on 20 January 2015. The parents or guardians of all subjects provided written informed consent. The study was registered at https://www.researchregistry.com/browse-the-registry#home/ (No: researchregistry5592).
Inclusion and exclusion criteria
Inclusion criteria for the study were as follows: (1) the age of initial breast development was <8 years old and (2) breast development was assessed as Tanner B stage II. Exclusion criteria were as follows: (1) patients with organic diseases affecting the hypothalamic–pituitary–gonadal axis; (2) patients with an abnormal sexual development sequence; and (3) patients with incomplete auxiliary examination data.
Instruments and methods
Gonadotropin-releasing hormone was injected subcutaneously at a dose of 2.5 µg/kg (maximum dose of 100 µg). An experienced ultrasound physician used the Philips IU22 ultrasound instrument (Philips Medical Systems Inc., Bothell, WA, US) for examinations. The frequency of the transvaginal and direct dual-purpose ultrasound probe was approximately 7.5 MHz. The probe entered through the anus. The internal genitals of the children were measured.
The normal values of sex hormones are as follows. Follicle-stimulating hormone (FSH) levels are 1.5 to 10 mIU/mL in the early stage of ovulation, 8 to 20 mIU/mL in the ovulation stage, and 2 to 10 mIU/mL in the late stage of ovulation. FSH levels of 5 to 40 mIU/mL are generally normal values. LH levels are 2 to 15 mIU/mL in early ovulation, 30 to 100 mIU/mL in ovulation, and 4 to 10 mIU/mL in late ovulation. LH levels of 5 to 25 mIU/mL are generally normal values. The sample size formula was as follows: N=Z2 × [P × (1−P)]/E2=1.962 ×[0.5 × (0.5)]/0.12=96 (N: sample size; Z: statistics, Z=1.96 when the confidence is 95%; E: error value; P: probability value).
Statistical analysis
Normally distributed data are expressed as mean ± standard deviation and were compared between the groups using the independent sample t-test. The receiver operating characteristic (ROC) curve was established and the parameters with differences between the ICPP and PT groups were determined. Statistical analysis was performed by SPSS version 23.0 (IBM Corp., Armonk, NY, USA) and Medcalc 12.7 software (MedCalc Software bvba, Ostend, Belgium). P<0.05 was considered statistically significant.
Results
General data
A total of 669 female subjects were included in the study. The subjects were divided into the ICPP group (LH levels ≥5 IU/L, n=350) with a median age of 8.0 years old and the PT group (LH levels <5 IU/L, n=319) with a median age of 7.5 years.
Age at initial diagnosis and basal gonadotropin levels
The age of initial diagnosis, basal LH levels, basal FSH levels, and basal LH/FSH were significantly higher in the ICPP group than in the PT group (all P<0.001). However, because gonadotropins are secreted in pulses, the basal LH and FSH values of the two groups often overlapped (Table 1).
Comparison of gonadotropins between the ICPP and PT groups.

Values are median and interquartile range. ICPP: idiopathic central precocious puberty; PT: premature thelarche; LH: luteinizing hormone; FSH: follicle-stimulating hormone.
Pelvic ultrasound-related parameters
The long diameter of the uterus, the transverse diameter of the uterus, the anterior–posterior diameter of the uterus, and the volume of the uterus were significantly greater in the ICPP group than in the PT group (all P<0.001). The long diameter of the cervix, the anterior–posterior diameter of the cervix, average ovarian volume, maximum ovarian volume, and maximum ovarian diameter were also significantly greater in the ICPP group than in the PT group (all P<0.001). Furthermore, the number of follicles ≥4 mm, maximum follicular diameter, endometrial thickness, and vaginal wall thickness were significantly greater in the ICPP group than in the PT group (all P<0.001) (Table 2).
Comparison of pelvic ultrasound-related parameters between the ICPP and CT groups.
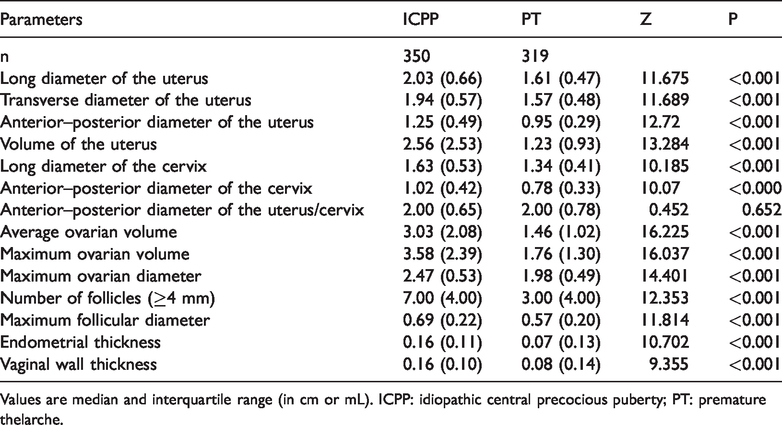
Values are median and interquartile range (in cm or mL). ICPP: idiopathic central precocious puberty; PT: premature thelarche.
The diagnostic accuracy of age at initial diagnosis, basal FSH levels, and basal LH/FSH for girls in the ICPP group was low (AUC >0.6 and <0.7). Other indices also improved the diagnostic accuracy of ICPP in patients to a certain extent (AUC >0.7 and <0.9), but they were still slightly low (Figure 1a–c).
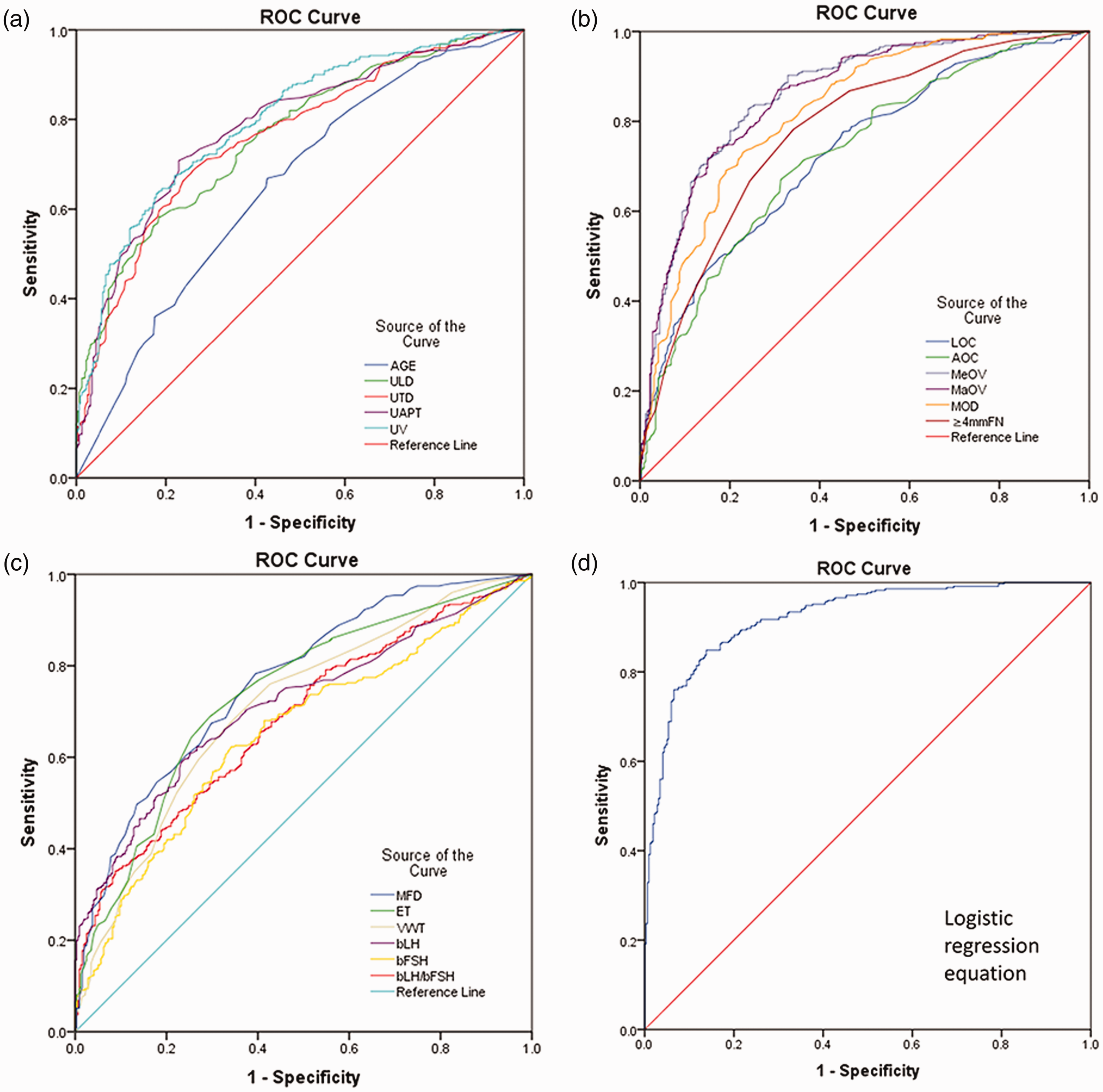
ROC curves of parameters in the idiopathic central precocious puberty and premature thelarche groups and the combined diagnostic model.
Establishment of the combined prediction model
The results of binary logistic multivariate regression analysis were as follows: Y=−14.123 + 0.630 × age at initial diagnosis +1.119 × transverse diameter of the uterus +1.278 × anterior–posterior diameter of the uterus + 0.637 × average ovarian volume +1.316 × maximum ovarian diameter + 0.146 × number of follicles (≥4 mm) + 2.925 × endometrial thickness + 0.559 ×basal LH value (Figure 1d). For the combined prediction model, the AUC was 0.922, the standard error was 0.0102, the 95% confidence interval was 0.899 to 0.941, the sensitivity was 84.9%, the specificity was 86.2%, Youden’s index was 0.7106, and the cut-off value was >0.5045. The AUC, sensitivity, and specificity of this model were significantly higher than those of single parameters alone (all P<0.05).
Verification of the combined prediction model
Another 69 girls aged between 6 and 9 years with suspected precocious puberty were recruited. The prediction results of the combined prediction model for 11 (15.94%) patients were inconsistent with the consensus criteria. Thirty-three girls were diagnosed with ICPP and 24 of them showed good agreement with the results of the combined prediction model.Thirty-six girls were diagnosed with PT, and 34 of them showed good agreement with the results of the combined prediction model. For the combined prediction model, the sensitivity was 72.73%, the specificity was 94.44%, the total coincidence rate was 84.09%, and Youden’s index was 0.67. Additionally, the positive likelihood ratio was 13.09%, the negative likelihood ratio was 0.29%, the positive predictive value was 92.31%, and the negative predictive value was 79.07%.
Discussion
Measurement of basal gonadotropin levels requires multiple venipuncture samples and is not readily accepted by parents or children. Pelvic ultrasound has the advantages of being noninvasive, simple, and economical, and is widely used in clinical practice. A transrectal ultrasonic examination can directly observe the morphology, volume, and follicular condition of the target organs of sex hormones, such as the uterus and ovaries, and is widely used in clinical practice in China.
Previous studies have shown that basal LH levels are significantly correlated with the peak value of LH in gonadotropin-releasing hormone provocative tests and basal LH has certain significance in the diagnosis of ICPP. 9 However, another study showed that basal gonadotropin levels usually overlapped between patients with ICPP and those with PT, and they were only suitable as a screening index for diagnosing ICPP. 10 A study on the relationship between basal gonadotropin and activation of the hypothalamic–pituitary–gonadal axis that was conducted by Ding et al. 8 also showed that basal LH levels had the highest predictive value for hypothalamic–pituitary–gonadal axis activation. This finding is consistent with the results of our study. However, diagnosing ICPP accurately based on basal LH levels alone is difficult.
In early adolescence, ovaries are the first organ to undergo changes. Early ICPP in children may simply show ovarian enlargement, but the uterus shows no significant change. The patients in the present study were girls aged between 6 and 9 years with clinically suspected precocious puberty. Their clinical breast development was confined to Tanner B stage II to ensure that the patients were in pre-adolescence or the early adolescence stage when pelvic ultrasound had a higher diagnostic value.
Paesano et al. 11 showed that the AUC of uterine length was 0.919 and its diagnostic efficiency was the best among the parameters of pelvic ultrasound. This finding may have been related to the large age range of their study subjects and the obvious signs of development of girls who were in adolescence. Yu et al. 12 further showed that the sensitivity was relatively high (91.6%) and the specificity was relatively low (68.7%) for the uterine body for diagnosing ICPP in girls, and this easily causes misdiagnosis and overtreatment. This finding may have been related to the fact that Yu et al. 12 only included girls with ICPP who needed gonadotropin-releasing hormone treatment and they were older and developed faster than those in our study. Our results are consistent with the results of their previous study.
This study has some limitations. First, this was an observational study and not a randomized, controlled trial. Second, this was a single-center clinical trial and the sample size was small. Third, only 58 of 669 cases were identical to the consensus diagnosis and follow-up, and a few patients were missed. Therefore, we need to emphasize the relatively minor specificity of the model.
Conclusion
A diagnostic model of ICPP was successfully established by transrectal pelvic ultrasound. Therefore, in the diagnosis of ICPP, basal LH levels and transrectal pelvic ultrasound should be applied together to improve the accuracy of diagnosis.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by Hebei Excellent Talents Project in 2016, Hebei Provincial Science and Technology Department Project (09276102D-1), Baoding Science and Technology Plan Project (18ZF058), and the Hospital Fund of the Affiliated Hospital of Hebei University (2019Q012).
