Abstract
Objectives
To investigate the effect of recombinant human luteinizing hormone supplementation (rLH priming) during the early follicular phase on in vitro fertilization (IVF) and intracytoplasmic sperm injection (ICSI) outcomes.
Methods
In order to evaluate available evidence regarding the efficacy of rLH priming in IVF/ICSI procedures, a systematic review and meta-analysis was preformed. Searches were conducted on MEDLINE®, EMBASE and the Cochrane Database of Clinical Trials without language limitation, but were restricted to randomized controlled trials (RCTs).
Results
Three RCTs including 346 patients were included in this meta-analysis, which demonstrated that rLH priming did not increase ongoing pregnancy rate. Although less recombinant follicle-stimulating hormone (rFSH) was required and the oestradiol level was higher on the day of human chorionic gonadotropin administration in the rLH priming group, the numbers of oocytes retrieved and embryos produced were comparable between patients treated with rLH priming and those treated with rFSH alone.
Conclusions
This systematic review and meta-analysis has demonstrated that at present there is insufficient evidence that patients undergoing IVF/ICSI may benefit from rLH priming during the early follicular phase.
Introduction
According to the two-cell, two-gonadotrophin theory, both follicle-stimulating hormone (FSH) and luteinizing hormone (LH) are important for follicle development in humans. FSH plays a crucial role in recruitment, selection and dominance; LH acts through the middle-to-late follicular phase by contributing to dominance maturation and ovulation. Meanwhile, LH promotes the biosynthesis of androgens, which are the substrate for oestradiol (E2).1,2
The effect of LH supplementation on in vitro fertilization (IVF) outcomes has been evaluated in several areas. For example, in the gonadotropin-releasing hormone (GnRH) agonist protocol, because of pituitary desensitization, endogenous levels of LH are very low during the late stimulation phase. 3 What is more, around 50% of patients undergoing IVF/intracytoplasmic sperm injection (ICSI) using GnRH agonist are LH deficient. 3 Thus, it would seem logical that LH supplementation would be beneficial to IVF/ICSI results. However, whether LH supplementation during the late follicular phase can increase the pregnancy and implantation rates or decrease the spontaneous abortion rate is still a subject for debate.4–7
In contrast, the effect of LH supplementation on IVF outcomes for patients who show a suboptimal response to FSH has also been evaluated by many investigators.8,9 Even though it is still unclear exactly how LH supplementation affects follicular development, a growing amount of evidence demonstrating the beneficial effect of LH supplementation in patients who show suboptimal response to FSH has been published.8,9 Studies have shown that LH supplementation not only improves embryo quality but also increases the pregnancy rate in women with poor ovarian reserve.8–10
Several studies have been conducted to assess the role of LH supplementation in the early phase of follicular growth.11–13 It has been shown that LH might increase granulosa cell FSH sensitivity by increasing androgen synthesis during the early stage of folliculogenesis. However, because of relatively small sample sizes, these studies were underpowered for pregnancy rate as a primary outcome and were inconclusive as to the benefit of recombinant LH (rLH) priming in patients undergoing IVF/ICSI.11–13
The aim of this systematic review and meta-analysis was to summarize the existing evidence on the effect of rLH priming during the early follicular phase on IVF/ICSI outcomes.
Materials and methods
Literature search
Literature searches were conducted to retrieve randomized controlled trials (RCTs) on the effect of early follicular phase rLH supplementation in patients undergoing IVF with normal ovary reservation on IVF/ICSI outcome. MEDLINE®, EMBASE and the Cochrane Database of Clinical Trials were searched up to May 2013 using the following search strategy: (‘recombinant luteinizing hormone’ or ‘luveris’ or ‘lutropin alfa’ or ‘recombinant rLH’ or ‘rLH’) AND (‘recombinant follicle stimulation hormone’ or ‘rFSH’ or ‘recombinant FSH’ or ‘rhFSH’) AND (‘assisted reproductive techniques’ or ‘ART’ or ‘IVF’ or ‘ICSI’ or ‘in vitro fertilization’ or ‘intracytoplasmic sperm injections’). The searches were not limited by language, but restricted to RCTs. A combination of text words and specific database indexing terminology were used to search available RCTs.
Inclusion and exclusion criteria
Inclusion criteria were RCTs that compared the effect of rLH priming in the early follicular phase on IVF/ICSI outcomes and ovarian stimulation parameters. Exclusion criteria included failure to report appropriate randomization procedures, participants as poor responders, or early follicular phase LH priming with other agents (e.g. human chorionic gonadotropin [hCG], dehydroepiandrosterone, letrozole or human menopausal gonadotropin [hMG]).
Data collection
Studies were screened by two reviewers (Z.B. and L.H.) independently and there were no disagreements in the studies identified for inclusion. First, all titles and abstracts from the databases were examined, but only those with the possibility of meeting the predefined criteria were kept for further evaluation. Secondly, final inclusion decisions were made on examination of the full manuscripts. If the published study was judged to contain insufficient information, study authors were contacted. The primary outcome was ongoing pregnancy rate. Secondary outcomes included the number of oocytes retrieved, recombinant FSH (rFSH) dosage, E2 level on the day of hCG administration and number of embryos produced.
Data synthesis and analysis
All data extracted were combined for meta-analysis using RevMan version 5.0 software (Cochrane Collaboration, Oxford, UK). Dichotomous data for each unit of analysis were expressed as an odds ratio (OR) with 95% confidence intervals (CIs). Continuous variables were expressed as weighted mean difference (WMD) with 95% CIs. Heterogeneity was evaluated using the Q-test and I2-index values, and reported for each outcome as a P-value and percentage, respectively. Data from the studies were combined using a fixed-effects model when heterogeneity was negligible, or a random-effects model when significant heterogeneity existed. A P-value < 0.05 was considered statistically significant.
Results
The process of literature identification and selection is summarized in Figure 1. A total of 367 citations were identified; 36 full text articles were reviewed in detail, and of these, five RCTs were considered eligible for inclusion.11–15 Of these five studies, one study examined whether exogenous LH administration during the early follicular phase had a beneficial effect on the IVF outcomes.
15
However, the ovarian stimulation protocol in the study group started with 1 ampule of hMG (75 IU FSH + 75 IU LH activity) daily for 4 days, with simultaneous administration of rFSH.
15
Another study assessed the effect of short-term pre-rFSH administration of LH in women entering an ICSI cycle on follicular development, oocyte quality and early embryo development.
14
LH priming was achieved by 7 days’ administration of 200 IU/day hCG when ovarian suppression had occurred.
14
Therefore, these two trials were excluded because the aim of this study was to explore the effect of rLH priming on IVF/ICSI outcomes.
Flow chart of the study selection process used for a systematic review and meta-analysis, undertaken to investigate the effect of recombinant human luteinizing hormone supplementation (rLH priming) during the early follicular phase on in vitro fertilization and intracytoplasmic sperm injection outcomes.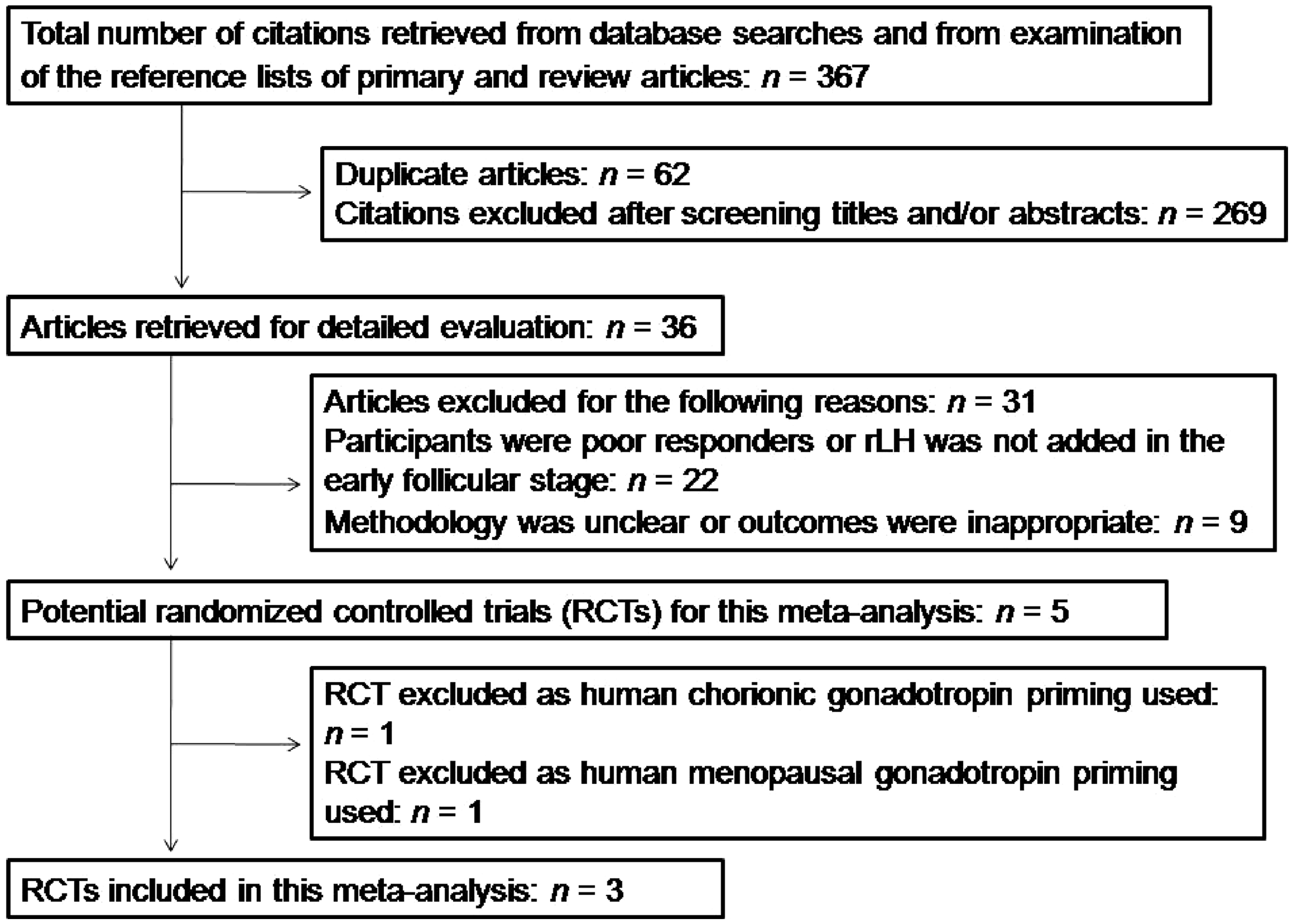
Three studies were considered eligible for inclusion in this meta-analysis, which included 346 patients undergoing IVF/ICSI with or without rLH priming.11–13 In these three studies, all patients were treated with the standard long protocol. Pituitary desensitization was achieved by using GnRH-a (depot tryptorelin, buserelin or short-acting triptorelin) to inhibit a premature LH surge.
All three trials with a total of 325 women provided data on the ongoing pregnancy rates (note that not all 346 women included in the studies were able to undergo embryo transfer). The ongoing pregnancy rate varied between 25.3% and 44.0% in the rLH priming group and between 18.7% and 37.1% in the control group (Figure 2).11–13 All three RCTs showed a higher ongoing pregnancy rate in women treated with rLH priming compared with those treated with rFSH alone. However, the pooled analysis with these three trials did not show differences between rLH priming and the control groups (OR 0.75; 95% CI 0.46, 1.21; I2 = 0%).
Forest plot of ongoing pregnancy rates in patients undergoing in vitro fertilization or intracytoplasmic sperm injection, treated with or without recombinant human luteinizing hormone (rLH) priming during the early follicular phase (n = 325). rFSH, recombinant follicle-stimulating hormone; M-H, Mantel–Haenszel; CI, confidence interval.
Meta-analysis of the three trials that reported the number of oocytes retrieved as an outcome showed no significant difference between women treated with rLH priming compared with the control group (WMD −0.33; 95% CI −2.43, 1.77; Figure 3).11–13 Durnerin et al.
11
reported a higher oocyte yield in the rLH priming group (10.8 versus 9.2) and Kovacs et al.
12
also reported a higher oocyte yield in the rLH priming group (10.2 versus 9.0), but the differences were not significant. However, the remaining study showed a significantly lower oocyte yield in the rLH priming group (6.2 versus 7.4; P < 0.05).
13
There was heterogeneity in this comparison (I2 = 75%, Pheterogeneity < 0.05). Data were combined using a random-effects model.
Forest plot of the number of oocytes retrieved in patients undergoing in vitro fertilization or intracytoplasmic sperm injection, treated with or without recombinant human luteinizing hormone (rLH) priming during the early follicular phase (n = 346). rFSH, recombinant follicle-stimulating hormone; SD, standard deviation; IV, inverse variance; CI, confidence interval.
Only two RCTs with a total of 200 patients that reported on the dosage of rFSH used in ovarian stimulation.12,13 Meta-analysis of these two RCTs showed that the total dosage of rFSH used was significantly lower in those with rLH priming, compared with those treated with rFSH alone (WMD 169.45; 95% CI 9.10, 329.80; Figure 4). No significant heterogeneity was suggested by the results of the Q-test and the I2-index.
Forest plot of the recombinant follicle-stimulating hormone (rFSH) dosage in patients undergoing in vitro fertilization or intracytoplasmic sperm injection, treated with or without recombinant human luteinizing hormone (rLH) priming during the early follicular phase (n = 200). SD, standard deviation; IV, inverse variance; CI, confidence interval.
Two RCTs with a total of 200 patients reported data for E2 level on the day of hCG administration.12,13 The pooled analysis of these two RCTs showed a significantly higher E2 level in the rLH priming group compared with the control group (WMD −381.4; 95% CI −612.58, –150.22; Figure 5). Heterogeneity tests were nonsignificant (I2 = 0%), indicating that there was no statistical inconsistency between these two studies.
Forest plot of oestradiol level on the day of human chorionic gonadotropin administration in patients undergoing in vitro fertilization or intracytoplasmic sperm injection, treated with or without recombinant human luteinizing hormone (rLH) priming during the early follicular phase (n = 200). rFSH, recombinant follicle-stimulating hormone; SD, standard deviation; IV, inverse variance; CI, confidence interval.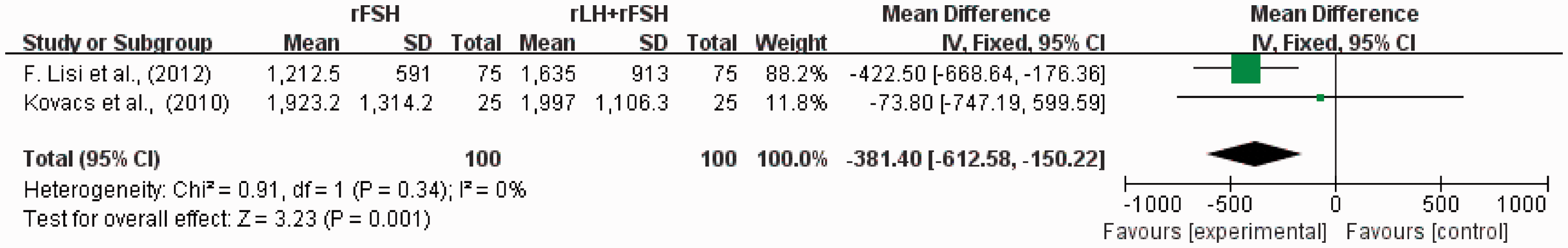
All three RCTs reported on the number of embryos produced as an outcome.11–13 The random-effects model showed no significant difference between those treated with rLH priming, compared with those treated with rFSH alone (WMD −0.42; 95% CI −1.71, 0.87; Figure 6). There was heterogeneity in this comparison (I2 = 66%, Pheterogeneity = 0.05).
Forest plot of the number of embryos produced in patients undergoing in vitro fertilization or intracytoplasmic sperm injection, treated with or without recombinant human luteinizing hormone (rLH) priming during the early follicular phase (n = 346). rFSH, recombinant follicle-stimulating hormone; SD, standard deviation; IV, inverse variance; CI, confidence interval.
Discussion
To the best of our knowledge, this is the first meta-analysis evaluating the efficacy of rLH priming during the early follicular phase for IVF/ICSI outcomes. According to the results of the present meta-analysis, women undergoing IVF/ICSI using a GnRH agonist protocol with rLH priming appear to have similar outcomes compared those treated with rFSH alone.
Both FSH and LH play an important role in folliculogenesis. FSH stimulates growth and recruitment of immature follicles in the ovary. In the early follicular phase, FSH is the major survival factor that rescues the small antral follicles (2–5 mm in diameter for humans) from apoptosis. LH, which is well known for its importance in the late follicular phase, supports theca cells in the ovaries that provide androgens and hormonal precursors for oestradiol production.16,17 Thus, in the process of IVF, to imitate the changes of FSH and LH in natural folliculogenesis, rLH or hMG are usually added in the late follicular phase during ovarian stimulation. Although controversy remains about the efficacy of this procedure, LH supplementation during the late follicular phase is employed worldwide to help follicles reach maturation.
Most of the studies concerning the effect of LH supplementation on IVF outcomes were designed to administer LH in the late follicular phase or in patients who showed a suboptimal response to FSH.9,10 However, little is known about the role of LH in the early follicular stage. The complex roles played by FSH, LH and other hormones during folliculogenesis have become better understood. High LH and androgen concentrations, which used to be considered as indicators of less healthy follicles in the early follicular phase, are now found to have a positive effect on granulosa cell activity of smaller follicles. 18 LH not only promotes the growth of larger follicles, but also increases granulosa cell FSH activity by increasing androgen synthesis. 19 In addition, it is known that the cytoplasmic maturational process is controlled by E2. 19 Since LH is also responsible for the production of androgen precursors by theca cells, which are required for E2 biosynthesis, LH is crucial for the process of oocyte maturation. 19 Therefore, it is reasonable to hypothesize that if rLH is administered in the early follicular phase before ovarian stimulation, in women undergoing IVF/ICSI with the GnRH agonist protocol, (i) more follicles might be recruited, less rFSH may be required and more oocytes may be retrieved, since LH has a synergistic effect with FSH in follicular recruitment and granulosa cell proliferation; (ii) oocyte quality might be increased, which leads to an increased yield of viable embryos and even a higher pregnancy rate.
Indeed, according to the results of the present meta-analysis, patients treated with rLH priming needed less rFSH to achieve oocyte maturation and tended to yield more oocytes, compared with those treated with rFSH alone. However, it must also be borne in mind that patients treated with rLH priming needed more LH compared with control patients. Meanwhile, the number of embryos was comparable between these two groups, even though the peak E2 level was significantly higher in the rLH priming group. All of these results were consistent with our hypothesis. However, all three RCTs observed a higher pregnancy rate in the rLH priming group, but the differences were not statistically significant.
The validity and interpretation of a meta-analysis mainly depends on the quality and homogeneity of the included studies. Therefore, only RCTs with LH priming using rLH were included into this study. However, it is acknowledged that this systematic review and meta-analysis has several potential limitations. First, there was clinical and methodological heterogeneity between the three included RCTs. Durnerin et al. 11 used depot GnRH agonist for pituitary downregulation and a dose of 300 IU/day of rLH for 7 days of priming, whereas the other two studies used daily GnRH agonist downregulation and 75 IU/day of rLH for 4 days of priming.12,13 It cannot be excluded that these clinical variations might be associated with the effectiveness or not of rLH priming. Another problem encountered in this meta-analysis was that the total number of RCTs included was relatively small. Although no heterogeneity was observed in the analysis of ongoing pregnancy rate, rFSH dosage and E2 level on the day of hCG administration, there was significant heterogeneity in the analysis of the number of oocytes retrieved and the number of embryos produced. In addition, given the small sample sizes, the CIs for some of the outcomes were extremely wide. Meanwhile, due to the limited number of studies included in this meta-analysis, it was not possible to perform funnel plots to examine publication bias, undertake subgroup analyses and meta-regression analyses.
In conclusion, based on the best available evidence, this systematic review and meta-analysis found that there is insufficient evidence that patients undergoing IVF/ICSI may benefit from rLH priming during the early follicular phase. However, because of the limited number of RCTs included in this study, larger well-designed RCTs are needed to determine whether rLH priming is beneficial to IVF/ICSI outcomes.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This work was supported by the National Natural Science Foundation of China (Grant numbers 31271605 and U1304315) and the Youth Foundation of The First Affiliated Hospital of Zhengzhou University, Zhengzhou, Henan Province, China.
