Abstract
Objective
As a new minimally invasive surgery developed in recent years, the procedure of endoscope-assisted transforaminal lumbar interbody fusion (TLIF) has not been well described. We herein describe a patient with lumbar disc herniation who underwent direct foraminoplasty and thorough decompression using a self-designed endoscopic trephine in endoscope-assisted TLIF at L4–5.
Methods
A 57-year-old man presented with a 3-month history of low back and right leg pain. Lumbar spine magnetic resonance imaging indicated protrusion of the L4–5 intervertebral disc in the right posterior direction. The patient underwent endoscopic surgery involving direct foraminoplasty, removal of the L4–5 intervertebral disc, and thorough decompression around the nerve roots.
Results
During the 1-year follow-up, the patient was generally in good condition and had no significant limitation of lumbar spine activity.
Conclusions
Endoscopic resection for the treatment of lumbar disc herniation is not a novel concept, but the improvement in the efficacy of endoscope-assisted TLIF and other endoscope-assisted spinal surgeries deserves to be reported. Through the innovation and improvement of surgical equipment for endoscope-assisted TLIF, direct foraminoplasty and thorough decompression can be effectively realized. These advances will help to promote the postoperative efficacy and enhance the prognosis.
Keywords
Introduction
Despite the steep learning curve, 1 spine surgeons have obtained good clinical outcomes after skillfully mastering the operating equipment and techniques of endoscope-assisted transforaminal lumbar interbody fusion (TLIF).2,3 However, because of the narrow space and relatively fixed position of the endoscopic channel, the conventional step-by-step trephine technique has a relatively unsatisfactory effect in endoscopic decompression, especially in the process of intervertebral foraminoplasty, resulting in postoperative residual pain in some patients. 4 We herein describe the efficiency of a self-designed endoscopic trephine for thorough decompression in endoscope-assisted TLIF as a novel technique for managing lumbar disc herniation.
Case presentation
A 57-year-old Chinese man presented with a 3-month history of continuous low back and right leg pain. Before presentation to our department, he was treated in a private clinic; however, conservative treatment was ineffective and the patient’s symptoms gradually worsened. Physical examination revealed percussion pain in the low back and limited lumbar spine activity, and the right straight leg raise test was positive. Lumbar spine magnetic resonance imaging and computed tomography indicated that the L4–5 intervertebral disc had protruded in the right posterior direction, the bilateral crypts were compressed and narrowed, the cauda equina was significantly compressed, and the spinal canal was narrowed (Figure 1).
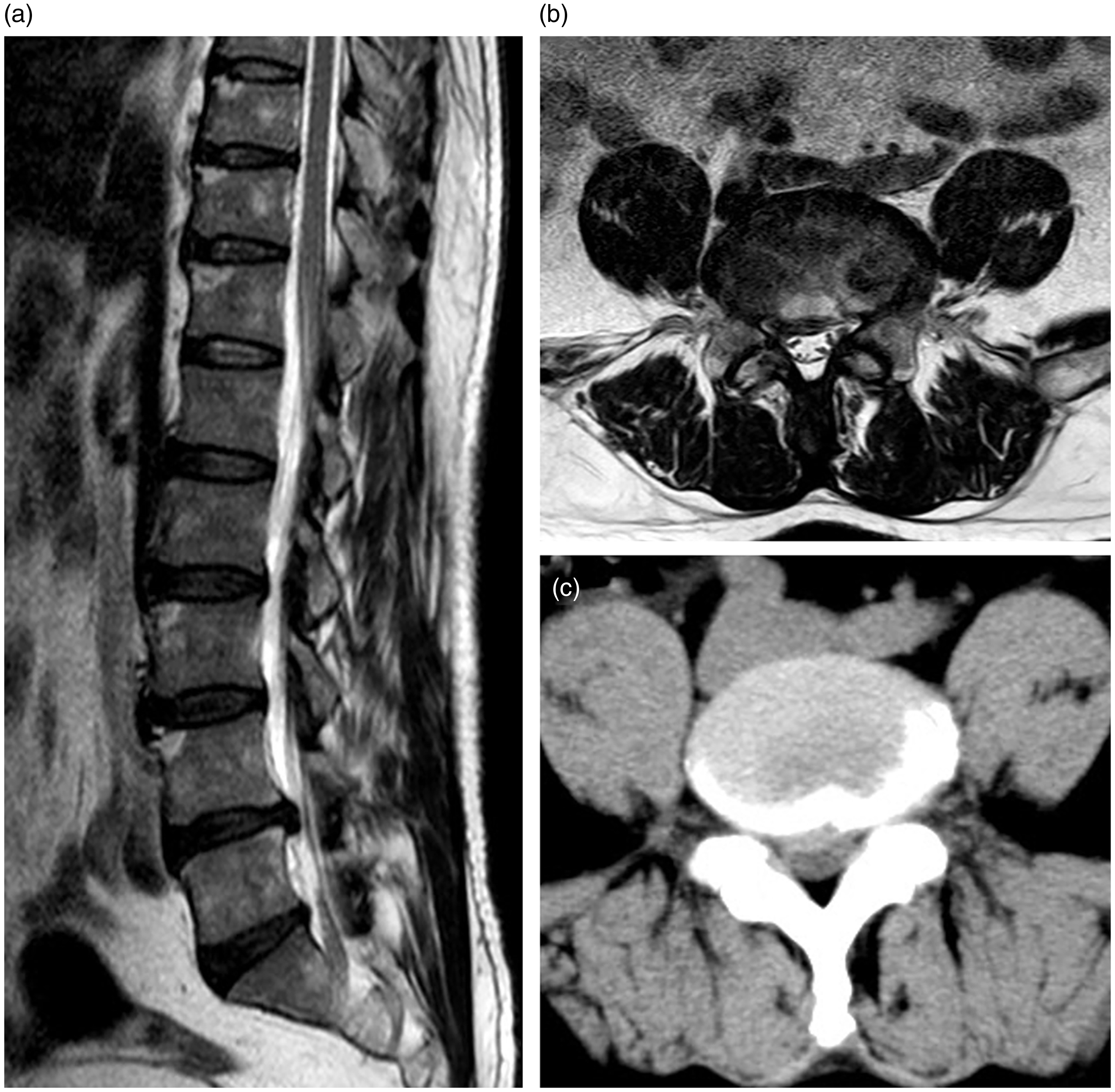
Lumbar spine magnetic resonance imaging (MRI) and computed tomography (CT) of a 57-year-old man at admission. (a) Sagittal MRI of the lumbar spine shows protrusion of the L4–5 intervertebral disc. (b) Axial MRI of the lumbar spine shows that the bilateral crypts and cauda equina are compressed and narrowed. (c) Axial CT of the lumbar spine shows severe stenosis of the spinal canal.
The patient was a middle-aged Chinese laborer engaged in a large amount of daily work, and he sternly refused to undergo open surgery. Additionally, in selecting either simple endoscopic decompression or endoscopic decompression with fusion, the patient considered his requirement to devote himself to daily work as soon as possible after the surgery. Thus, to enhance the stability of the spine after surgery, an additional fusion was recommended to avoid potential instability. The patient consented to the procedure and was scheduled for an endoscope-assisted TLIF at L4–5. He was placed into the prone position under conscious sedation, and routine fluoroscopy was performed to examine the L4–5 intervertebral space. After identification of the L4–5 intervertebral space, the posterolateral skin entry point was located 10 to 12 cm from the midline. The endoscope was introduced from the right flank through successive dilators and reamers. The trephine applied in our system was self-designed and characterized by a strong holding force, allowing it to be firmly fixed on the superior articular process, and a large diameter, which effectively shortened the entry time. Under endoscopic direct vision, the superior articular process was carefully cleared by the self-designed trephine, and the endoscopic channel could be smoothly inserted to effectively remove the L4–5 intervertebral disc and perform complete decompression around the nerve roots.
An interbody cage and bone blocks were subsequently implanted through the endoscopic channel and firmly fixed in the intervertebral space. Because the patient only had pain on the right side before surgery and no decompression was performed on the left side, the general stability was satisfactory. Thus, the unilateral cage was implanted into the intervertebral space. Intraoperative fluoroscopy suggested that the position of the interbody cage was satisfactory. In addition, to enhance the stability of the posterior column, a facet screw was inserted in the left articular process of L4–5 (Figure 2). According to previous studies by Jang and Lee 5 and Eskander et al., 6 the facet screw system can provide mechanical stability equal to that provided by a pedicle screw system, whether in single-segment or multi-segment fixation. Additionally, our patient was under conscious sedation, and the application of facet screw fixation effectively reduced the surgical time, relieved intraoperative pain, and was minimally invasive.

Surgical equipment and intraoperative images in endoscope-assisted transforaminal lumbar interbody fusion. (a) The self-designed endoscopic trephine with increased thickness, wide serrated teeth, and sharpness. (b) The self-designed trephine grinds the superior articular process under a protective sleeve during performance of direct foraminoplasty. (c) A work sleeve is placed after completion of foraminoplasty. (d) Thorough decompression around the nerve roots. (e) A lag screw is inserted in the left articular process of L4–5 to enhance the stability of the posterior column.
With the preoperative symptoms apparently alleviated, the patient was discharged the following day. During the 1-year postoperative follow-up, he was generally in good condition and had no significant limitation of lumbar spine activity. Postoperative imaging examination suggested that the interbody cage was fixed in place and that bone fusion had been achieved (Figure 3).

Postoperative imaging examination at the 1-year follow-up. (a, b) The interbody cage and lag screw are fixed in place (a, frontal view; b, lateral view). (c–e) Effective bone fusion has been achieved (c, sagittal image; d and e, axial images).
Discussion
Minimally invasive lumbar spine surgery has had a long history since the initial description of minimally invasive TLIF in 2002 by Foley et al.7,8 In recent years, with the rapid development of minimally invasive spinal surgery technology, endoscopy has also been ingeniously combined with TLIF, and the surgical indications for lumbar disc herniation have been expanding. 9 The surgical scope has also progressed from the common locations at L4–5 and L5–S1 to more challenging cases such as L3–4, L2–3, and even L1–2. Increasingly more clinical studies are confirming that endoscope-assisted TLIF is a new and effective method for the treatment of lumbar disc herniation.2,3,10 Additionally, the fusion rate and efficacy of endoscopic fusion have been affirmed and advocated by many clinical centers.11–13
Anatomically, the size of the intervertebral foramen is the main factor affecting the puncture and catheterization in the process of endoscope-assisted TLIF. The normal intervertebral foramens tend to be ear-shaped on the lateral radiograph of the lumbar spine, with a wide top and narrow bottom, 14 and the shape of the foramen from L1–2 to L5–S1 is an inverted triangle from large to small. Moreover, because the superior articular process of the lower vertebral body is directly opposite the upper intervertebral disc, and because the superior articular process is also closely adjacent to the upper vertebral body and intervertebral disc, it is difficult to accommodate the conventional 7.5-mm-diameter channel. Therefore, only by carefully clearing the superior articular process and enlarging the intervertebral foramen can the endoscopic channel be smoothly placed in the position of the herniated or prolapsed intervertebral disc and thus obtain more thorough and effective decompression.
In this case, we demonstrated how to directly shape the intervertebral foramen by improving the design of endoscopic trephine while avoiding damage to the surrounding tissue. By prolonging the length and changing the radian of the serrated teeth, the effective contact area between the serrated teeth and target tissues is increased, and more protruding tissue and the bone of the superior articular process can be removed smoothly. Endoscopic resection for the treatment of lumbar disc herniation is not a novel concept,15,16 but the improvement in the efficacy of endoscope-assisted TLIF and other endoscope-assisted spinal surgeries deserves to be reported. The 1-year postoperative follow-up of our patient indicated that he recovered well after endoscopic direct foraminoplasty and thorough decompression.
Conclusion
Through the innovation and improvement of surgical equipment for endoscope-assisted TLIF, direct foraminoplasty and thorough decompression can be effectively realized to promote the postoperative efficacy and enhance the prognosis.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics and consent statements
This study was conducted in accordance with the declaration of Helsinki and with approval from the Ethics Committee of Jiangxi Provincial People’s Hospital Affiliated to Nanchang University (Ethics Committee reference number: 8187302541). Written informed consent was obtained from the patient.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
