Abstract
Polymyositis is a subgroup of idiopathic inflammatory myopathies characterized by symmetric proximal limb weakness and chronic skeletal muscle inflammation. We herein report the first case of bilateral leg pain and unilateral calf atrophy caused by polymyositis accompanying lumbar spinal stenosis and disc herniation. A 52-year-old man presented with intermittent claudication and calf pain that had become gradually aggravated during the last 3 months. Magnetic resonance imaging showed spinal stenosis at the L3/4 and L4/5 levels and lumbar disc herniation at the L4/5 level. Preoperative laboratory investigations revealed elevated muscle enzyme concentrations. Magnetic resonance imaging also showed atrophy, fatty degeneration, and edema in both calf muscles. Histological examination showed inflammatory myositis and fibrosis in the perifascicular connective tissues. The patient was diagnosed with polymyositis. We performed decompressive laminectomy at the L3/4 and L4/5 levels and discectomy at the L4/5 level. After administration of prednisolone for 6 months and methotrexate for 3 months, the patient’s bilateral calf pain and abnormal laboratory findings improved. The combination of surgical decompression and adequate medical treatment resulted in a successful recovery. Polymyositis should be suspected in patients with lumbar spinal stenosis or lumbar disc herniation who exhibit increased muscle enzyme concentrations or lower extremity muscle atrophy.
Keywords
Introduction
Idiopathic inflammatory myopathies are characterized by symmetric proximal limb weakness and chronic inflammation of skeletal muscles. 1 Polymyositis, a subgroup of idiopathic inflammatory myopathies, occurs over several weeks to months and causes pain in involved muscles.2,3 Lumbar spinal stenosis is defined as a narrowing of the spinal canal or neural foramen that causes intermittent claudication, pain, tingling, cramping, and muscle weakness of the lower extremities. 4 Symptoms of lumbar spinal stenosis gradually worsen over months to years. Lumbar disc herniation (LDH) is a common disease that causes low back pain and radiating pain in the lower extremities. 5 We herein present the first case of bilateral calf pain and claudication due to polymyositis accompanying simultaneous lumbar spinal stenosis and LDH. Polymyositis usually occurs in the proximal muscles of the lower extremities. In this patient, however, polymyositis occurred in the distal muscles of the lower extremities, making it difficult to discriminate between polymyositis and spinal stenosis.
Case report
A 52-year-old man visited our hospital for evaluation of intermittent claudication and calf pain in both legs that had begun 5 years previously. His symptoms had become gradually aggravated during the last 3 months. He was unable to walk for more than 5 minutes because of the calf pain. He had no personal or familial medical history. Physical examination revealed atrophy of the left calf muscle compared with the right side. His left calf diameter was 32.0 cm; however, his right calf diameter was 34.0 cm. Neurologic examination revealed normal findings with the exception of mild hypesthesia over the posterior and lateral aspects of the left calf. Plain radiography of the lumbar spine showed degenerative changes. Lumbar spinal magnetic resonance imaging (MRI) showed spinal stenosis at the L3/4 and L4/5 levels combined with LDH at the L4/5 level (Figure 1). We decided to perform decompressive laminectomy and discectomy for the lumbar spinal stenosis and LDH. However, preoperative laboratory investigations revealed elevated levels of creatine kinase (CK) (5506 IU/L; reference range, 55–215 IU/L), myoglobin (338.90 ng/mL; reference range, 16.00–116.00 ng/mL), aldolase (16.9 IU/L; reference range, <7.7 IU/L), lactate dehydrogenase (LDH) (1160 IU/L; reference range, 218–472 IU/L), CK-myocardial band (62.5 ng/mL; reference range, 0.0–5.0 ng/mL), aspartate aminotransferase (AST) (113 IU/L; reference range, 0–50 IU/L), and alanine aminotransferase (ALT) (127 IU/L; reference range, 1–50 IU/L). Other blood test results, including the white blood cell count, hemoglobin concentration, platelet count, serum erythrocyte sedimentation rate, blood urea nitrogen concentration, creatinine concentration, total protein concentration, albumin concentration, calcium concentration, phosphorus concentration, gamma-glutamyl transpeptidase concentration, total bilirubin concentration, and troponin I concentration, were within normal limits. The abnormal laboratory findings were sustained at follow-up. We considered it necessary to evaluate the patient for other diseases such as rhabdomyolysis and idiopathic inflammatory myopathies. However, the results of liver ultrasonography, abdominal pelvic computed tomography, a bone scan, and urinalysis were normal. We performed other laboratory investigations related to thyroid function to evaluate the patient for thyroid-related myopathies. However, the thyroid function tests revealed no major abnormalities in the concentrations of free thyroxine (0.93 ng/dL; reference range, 0.61–1.20 ng/dL), triiodothyronine (0.57 ng/mL; reference range, 0.60–1.55 ng/mL), and thyroid-stimulating hormone (4.021 μIU/mL; reference range, 0.380–5.330 μIU/mL). After consultation with a rheumatologist, pelvic MRI was performed to evaluate the patient for idiopathic inflammatory myopathies usually involving the skeletal muscles of the proximal lower limb. 6 However, there was no evidence of idiopathic inflammatory myopathies on pelvic MRI. We then decided to perform MRI of both lower extremities to identify the cause of the pain and left calf atrophy. MRI of both lower extremities showed atrophy, fatty degeneration, and edema in both distal thighs and calf muscles (Figure 2). We suspected that the patient had a rare case of idiopathic inflammatory myopathy invading the distal lower limb. Electromyography demonstrated diffuse bilateral radiculopathies mainly involving the bilateral L5 and S1 nerve root. There were no specific myopathic patterns of increased insertional and spontaneous activity with fibrillation potential, complex repetitive discharge, or short-duration/small-amplitude motor unit action potentials.

Preoperative magnetic resonance imaging of the lumbar spine. (a) Sagittal T2-weighted magnetic resonance image showing lumbar disc herniation at the L4/5 level with upward migration (arrow). (b) Axial T2-weighted magnetic resonance image showing spinal stenosis at the L3/4 level. (c) Axial T2-weighted magnetic resonance image showing lumbar disc herniation (arrow) at the L4/5 level.
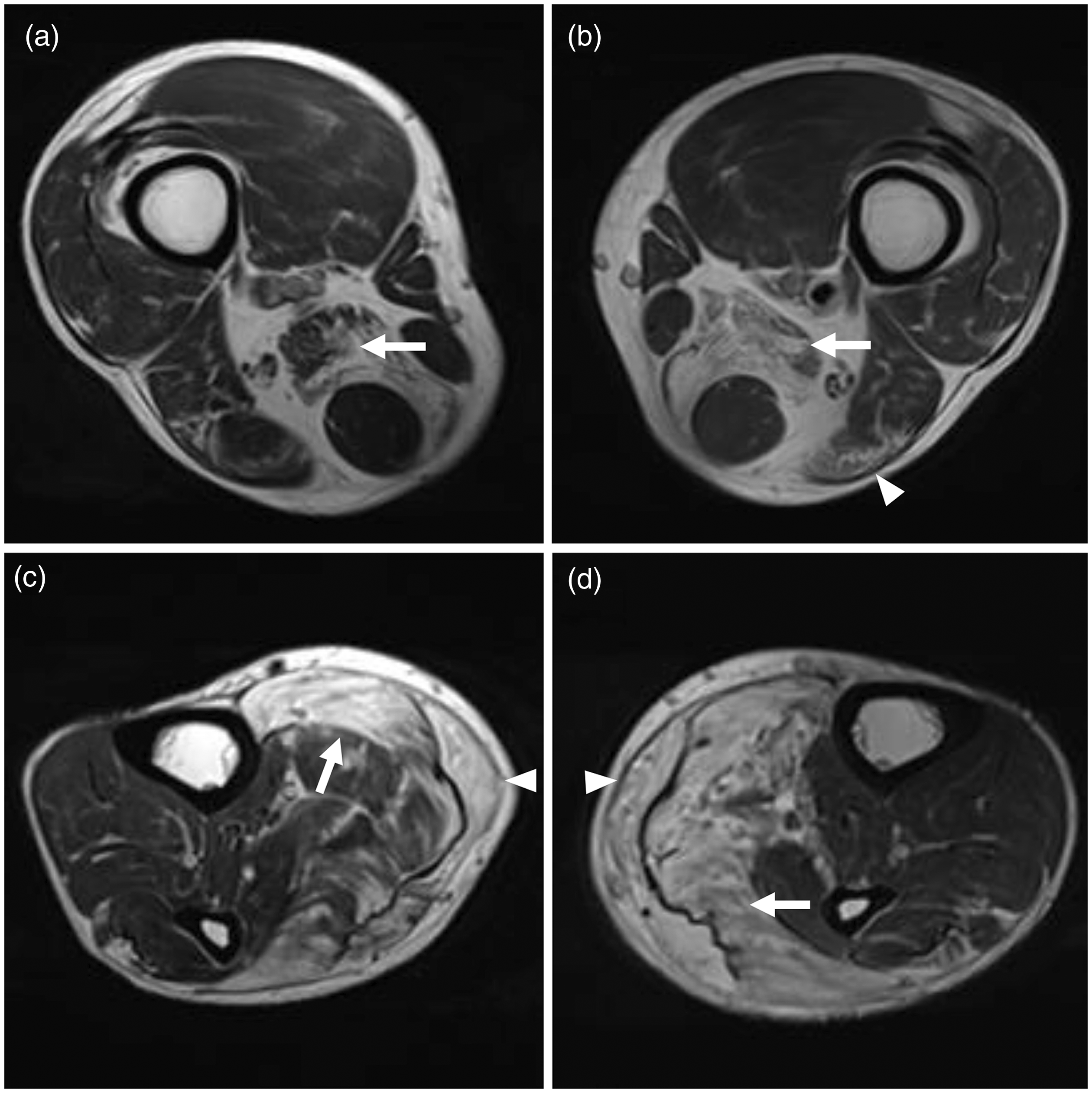
Magnetic resonance imaging of both legs. (a, b) Axial T2-weighted magnetic resonance images of the distal thighs showing diffuse muscle atrophy with fatty infiltration and muscle edema of both semimembranosus muscles (arrows) and the left biceps femoris muscle (arrowhead). (c, d) Axial T2-weighted magnetic resonance images of both calves showing diffuse muscle atrophy with fatty infiltration and muscle edema of both soleus muscles (arrows) and gastrocnemius muscles (arrowheads). (a, c) Right side. (b, d) Left side.
The patient had the following three problems: lumbar spinal stenosis at the L3/4 and L4/5 levels, LDH at the L4/5 level, and idiopathic inflammatory myopathy involving the distal muscles of both lower extremities. We explained to the patient that surgical decompression was necessary to treat his neurogenic claudication and bilateral calf pain. We also explained that a muscle biopsy and medical treatment such as corticosteroids or immunosuppressants were needed for the idiopathic inflammatory myopathy. Decompressive laminectomy at the L3/4 and L4/5 levels and discectomy at the L4/5 level were performed. The patient’s intermittent claudication improved after surgery. However, his bilateral calf pain persisted. On the 14th postoperative day, a bilateral calf muscle biopsy based on MRI findings was performed. Histological examination showed inflammatory myositis with inflammatory cell infiltration and fibrosis in the perifascicular connective tissue (Figure 3). No rimmed vacuoles were found in the biopsy specimens. We performed another physical examination and found no heliotrope rash, Gottron’s papules, or Gottron’s sign. Moreover, we found no weakness of the neck extensor or long finger flexor muscles and no dysphagia. These findings were consistent with the diagnosis of polymyositis according to the European League Against Rheumatism/American College of Rheumatology (EULAR/ACR) classification criteria. 7 Beginning on the 35th postoperative day, prednisolone (60 mg/day) was administered for treatment of the polymyositis in consultation with a rheumatologist. We administrated methotrexate (7.5 mg/week) with folic acid (2 mg/day) beginning 3 months after the administration of prednisolone in consultation with the rheumatologist. At the last follow-up, after treatment with prednisolone for 6 months and methotrexate for 3 months, the patient’s bilateral calf pain and laboratory results were improved. His CK concentration improved from 5506 to 1073 IU/L, his AST concentration improved from 113 to 37 IU/L, and his ALT concentration improved from 127 to 68 IU/L.
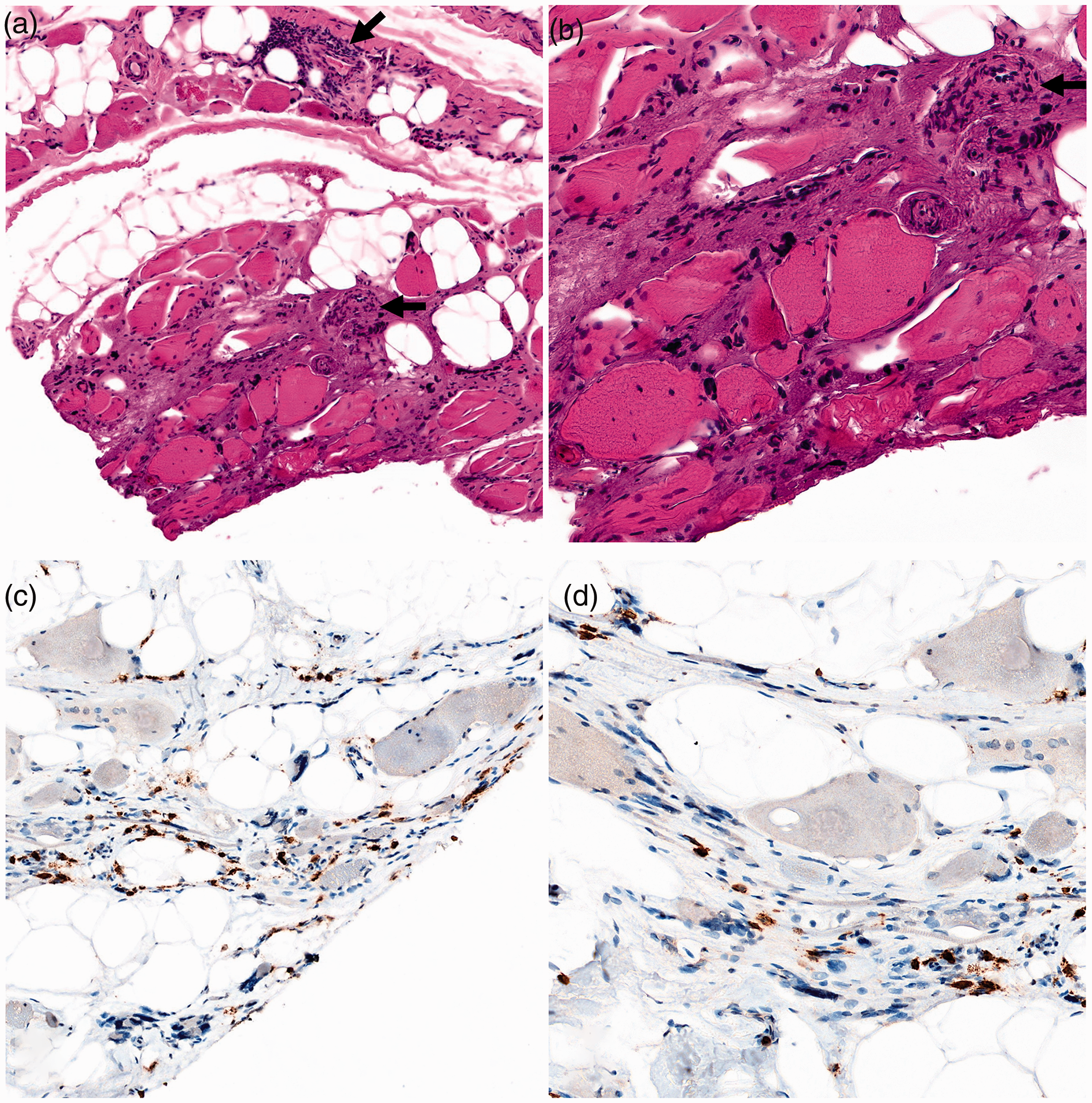
Biopsy of the left gastrocnemius muscle showing (a, b) endomysial infiltration of mononuclear cells surrounding myofibers and perivascular infiltration of mononuclear cells (arrows) with myofiber size variability. (c, d) Immunohistochemical staining of CD3 showing T cells invading the endomysium. No rimmed vacuoles were found in the biopsy specimens. (a, c) ×10 magnification. (b, d) ×30 magnification.
Written informed consent was obtained from the patient for the publication of this report and any accompanying images. All consent procedures and details of this study were approved by the Institutional Review Board of the Institutional Review Board of the Catholic University of Korea (approval no. PC20ZASI0170). The reporting of this study conforms to the CARE guidelines. 8
Discussion
Polymyositis is a subgroup of idiopathic inflammatory myopathies that initially involves the proximal lower limb muscles, particularly the vastus lateralis and intermedius muscles, before ascending to involve the buttocks, proximal upper limbs, neck flexors, and pharyngeal muscles.2,3,9,10 Early in polymyositis, typical MRI findings are bilateral and symmetric edema in the pelvic and thigh musculature.3,9,10 Huang et al. 11 reported that 80% of patients with polymyositis had definite thigh muscle involvement. Kim et al. 12 reported that about 75% and 50% of patients with polymyositis had proximal muscle weakness and muscle pain, respectively. However, our patient had a rare case of polymyositis with dominant involvement of the distal instead of proximal muscles of both lower extremities. Furthermore, the polymyositis was accompanied by simultaneous lumbar spinal stenosis and LDH. Although lumbar spinal stenosis and LDH can cause calf pain, polymyositis can also cause similar pain, making it difficult to discriminate between polymyositis and spinal stenosis based on symptoms alone.
CK has been traditionally considered the most useful serum enzyme for diagnosing and assessing idiopathic inflammatory myopathies in adult patients. 13 CK has been confirmed to be positive in more than 90% of patients with polymyositis. It usually increases to more than 10 times the upper limit of the reference range. 13 If the weakness of the extremities improves, the CK concentration also improves. 13 The myoglobin concentration is elevated at a similar frequency in patients with active myositis, 14 although it varies with disease activity. 15 Aldolase is less muscle-specific than CK. 13 However, when the CK concentration is normal, evaluation of aldolase is clinically useful. 13 Volochayev et al. 16 reported that the aldolase concentration was increased in 60.4% of patients with idiopathic inflammatory myopathies. In our patient, the myoglobin, CK, and aldolase concentrations were all elevated. Among various myositis-specific autoantibodies, anti-Jo-1 is the most common autoantibody identified in patients clinically classified as having primary polymyositis. 13 Identification of this autoantibody helps to diagnose and classify idiopathic inflammatory myopathies. In a previous study, the sensitivity of anti-Jo-1 was 22% and the specificity was 99%. 13 The presence of anti-Jo-1 is one of the EULAR/ACR classification criteria. 7 In our case, however, anti-Jo-1 was negative.
Focal myositis secondary to chronic nerve denervation can also cause calf pain and hypertrophy or amyotrophy. In patients with focal myositis, amyotrophy is one of the cardinal signs and is quite frequent.17,18 However, muscular hypertrophy due to nerve denervation may rarely occur. 17 According to the literature, most cases of neurogenic hypertrophy occur predominantly in the calf, and S1 radiculopathy is thought to be the main cause of this phenomenon.17,18 Costa et al. 19 reported that 86% of the patients in their study had S1 radiculopathy. The mechanism and progression remain unclear. 17 Gross et al. 17 reviewed the literature and reported that the mean CPK concentration in patients with focal myositis of the calf associated with S1 radiculopathy was 5.5 times higher than the normal concentration. It was necessary to rule out this condition in our patient because of the difference in calf diameter, elevated CK concentration, pain in the affected area, and spinal stenosis at the L3/4 and L4/5 levels combined with LDH at the L4/5 level. However, our patient had no S1 nerve root radiculopathy. The initial CK concentration was 5506 IU/L, which was much higher than the increase observed in patients with neurogenic hypertrophy. Moreover, the AST, ALT, aldolase, and LDH concentrations were simultaneously elevated. Furthermore, both distal lower limbs were involved. Based on these clinical features, we were able to rule out neurogenic muscular hypertrophy.
According to several studies, most patients with polymyositis respond well to corticosteroid treatment.1,20 As a result, corticosteroids are considered first-line therapy to treat polymyositis.1,20 Prednisolone is usually given orally at a dose of 0.5 to 1.0 mg/kg per day. 20 If there is no improvement in symptoms after 3 to 6 months of treatment with prednisolone, a second-line immunosuppressive agent such as methotrexate, azathioprine, or intravenous immunoglobulin G should be added. 1 However, no universal consensus has been reached regarding the treatment of refractory idiopathic inflammatory myopathies, and second-line agents are used according to empirical data. 1
We have herein described a unique case of polymyositis accompanying lumbar spinal stenosis and LDH. To the best of our knowledge, this is the first report of such a case. The combination of surgical decompression and adequate medical treatment resulted in a successful recovery. Polymyositis should be suspected in patients with lumbar spinal stenosis or LDH who have increased muscle enzyme concentrations or muscle atrophy of the lower extremities.
Footnotes
Acknowledgements
We thank Dr. Ki-Ok Min for her suggestions regarding preparation of the pathologic slides. We also thank the patient for providing consent for publication of this case report.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
