Abstract
Objective
To investigate the clinical significance of protocadherin 17 (PCDH17) promoter methylation in bladder cancer.
Methods
Methylation-specific polymerase chain reaction was used to examine the promoter methylation status of PCDH17 in tumour tissue specimens obtained from patients with bladder cancer, and in normal bladder epithelial tissue specimens obtained from age- and sex-matched controls. The correlations between methylation status and demographic and clinicopathological parameters, and disease outcome, were assessed.
Results
Methylation of the PCDH17 promoter was detected in 77/115 (67.0%) patients with bladder cancer and 0/43 (0%) of the controls. Methylation was significantly associated with high cancer grade (G3), advanced cancer stage (T2–T4), large tumour diameter (> 3 cm) and tumour recurrence. Methylation was also associated with significantly shorter survival time compared with unmethylated PCDH17 in patients with bladder cancer, and was an independent predictor of overall survival.
Conclusions
PCDH17 promoter methylation is closely associated with bladder cancer malignancy and may be used as an independent predictor of clinical outcomes in patients with bladder cancer.
Introduction
Bladder cancer is one of the most common genitourinary cancers worldwide and is a significant cause of cancer-related mortality, with an estimated 72 570 new cases and 15 210 cancer-associated deaths expected in the USA in 2013. 1 The burden of bladder cancer-related morbidity is expected to increase due to changes in exposure to risk factors for bladder cancer development, and to the growth and increasing age of the global population.2,3 The initiation and progression of bladder cancer is characterized by the gradual accumulation of multiple genetic and epigenetic changes that lead to activation of proto-oncogenes or inactivation of tumour suppressor genes.4,5 Epigenetic alterations in bladder cancer include DNA methylation, histone modification and RNA interference. 6 Epigenetic modifications, particularly DNA methylation, play an important role in the progression of bladder cancer, since DNA methylation often results in the hypermethylation of selected gene promoters and the inactivation of tumour suppressor genes.7,8 Identification of novel methylated tumour suppressor genes may be used in the diagnosis, prognosis and therapy of bladder cancer.
Protocadherins (PCDHs) belong to a subfamily of the cadherin superfamily of calcium-dependent intercellular adhesion molecules that play a key role in cell adhesion and are also involved in the inhibition of tumour cell growth, migration and invasion.9,10 Several PCDH genes, including PCDH8, PCDH10 and PCDH20, are frequently silenced in human neoplasms,9–12 suggesting that they function as tumour suppressor genes. Since PCDH17 is in the same non-clustered subgroup, the present study focused on this gene as a candidate tumour suppressor.11,12 The PCDH17 promoter is a TATA-less promoter containing CG-rich sequences that may be susceptible to DNA methylation. PCDH17 methylation is found in several human carcinomas including urological cancers.7,12,13
In the present study, the promoter methylation status of PCDH17 in bladder cancer tissue was examined and compared with normal tissues. In addition, the correlation between PCDH17 promoter methylation status and clinicopathological parameters was examined, together with patient outcome, in order to elucidate the clinical significance of PCDH17 promoter methylation in patients with bladder cancer.
Patients and methods
Study population and tissue specimens
This case–control study included tumour samples from patients who had been diagnosed with bladder cancer between March 2004 and March 2008 at the Third Hospital of Hebei Medical University, Shijiazhuang, China. Criteria for sequential enrolment of patients with bladder cancer were: (I) histopathological diagnosis of bladder transitional cell carcinoma for the first time; (II) no history of other tumour; (III) no anti-tumour therapy prior to surgery; (IV) availability of sufficient tumour tissue and follow-up data. Tumour diagnosis, staging (Union for International Cancer Control system) and grading (World Health Organization histological system), treatment and follow-up were completed according to international standards. 14 Control bladder epithelial tissues were obtained from inpatients with nonmalignant urinary diseases, including benign prostatic hyperplasia and bladder calculus. Control tissues were examined pathologically to exclude the possibility of incidental tumours. Criteria for sequential enrolment of controls were: (I) no histopathological diagnosis of bladder cancer; (II) no history of tumour; (III) no anti-tumour therapy prior to surgery; (IV) availability of sufficient bladder epithelial tissues and follow-up data. Following surgery, patients with bladder cancer were followed-up for 5 years at 3–12 month intervals. Patients were excluded if they left the study half way or less through the follow-up period. Tumour samples were collected during surgery and all tissue specimens were promptly frozen in liquid nitrogen and stored at −80℃ prior to DNA extraction and methylation-specific polymerase chain reaction (PCR).
This study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of the Third Hospital of Hebei Medical University, Shijiazhuang, China. Written informed consent was obtained from each participant.
DNA extraction, bisulfite modification and methylation-specific PCR
Genomic DNA was obtained from tissue samples using the DNeasy® Tissue Kit (Qiagen, Valencia, CA, USA) following the manufacturer’s instructions. Prior to methylation-specific PCR, the extracted DNA was treated with bisulfite to convert unmethylated cytosines to uracils using the EpiTect® Bisulfite Kit (Qiagen) according to the manufacturer’s instructions. The methylation status of the PCDH17 gene was examined by methylation-specific PCR as reported previously. 7 Briefly, the primer sequences for the unmethylated PCDH17 reaction were as follows: PCDH17 unmethylated sense (UMS) (sense), 5′-AGATTATTGGGTGTTGTAGTTT-3′; and PCDH17 unmethylated antisense (UMAS) (antisense), 5′-AACCCTAACACAACATACACA-3′, which amplified a 90-base pair product. The primers for the methylated PCDH17 reaction were as follows: PCDH17 methylated sense (MS) (sense), 5′-GATTATCGGGTGTCGTAGTTC-3′; and PCDH17 methylated antisense (MAS) (anti-sense), 5′-CCCTAACGCAACGTACGCG-3′, which amplified an 87-base pair product. Each PCR reaction was carried out in a total volume of 25 µl, including: 100 ng DNA template and 0.2 µl (5 U/µl) TaKaRa Taq™ DNA polymerase (TaKaRa Biotechnology, Dalian, China), 0.2 mM of each primer, 2.5 mM magnesium chloride, 0.2 mM of each deoxynucleotide triphosphate, 2.5 µl 10 × PCR buffer (TaKaRa Biotechnology). PCR amplification of the modified DNA samples consisted of denaturation at 95℃ for 10 min, followed by 40 cycles at 94℃ for 30 s, 60℃ for 30 s, and 72℃ for 30 s for the methylated reaction or 42 cycles of 94℃ for 30 s, 60℃ for 30 s, and 72℃ for 30 s for the unmethylated reaction, and a final extension for all reactions at 72℃ for 5 min, using a BD Biosciences thermal cycler (Hercules, CA, USA). Water blanks were included with each assay. PCR products were separated on 2% agarose gels, stained with ethidium bromide, and visualized under ultraviolet illumination.
Samples were scored as follows: methylation positive, bands visible in the methylated reaction lane or in both the methylated and unmethylated lanes; methylation negative, bands visible only in the unmethylated reaction lane. Each methylation-specific PCR was performed in triplicate, and results were confirmed by repeating methylation-specific PCR assays for all samples.
Statistical analyses
Fisher’s exact test or χ2-test was used to evaluate the difference in PCDH17 methylation status between patients with bladder cancer and controls, and to assess the relationships between PCDH17 methylation and clinicopathological parameters. The difference in overall survival between patients with bladder cancer and methylated PCDH17 and patients with bladder cancer and unmethylated PCDH17 was assessed using Kaplan–Meier survival analysis and log-rank test. A multivariate Cox proportional hazards model was used to estimate the independent prognostic effect of PCDH17 methylation, controlling for risk factors, including grade, stage and tumour size.15–17 A two sided P-value < 0.05 was considered to be statistically significant. Statistical analyses were performed using SAS software, version 8.0 (SAS Institute, Cary, NC, USA) for Windows®.
Results
Protocadherin 17 (PCDH17) promoter methylation status and demographic, clinical and pathological features in patients with bladder transitional cell carcinoma (n = 115).

Data presented as n (%) of patients.
NS, no statistically significant difference (P ≥ 0.05; χ2-test; methylated versus unmethylated PCDH17 group).
Methylation of the PCDH17 promoter was present in 77/115 (67.0%) tumour samples from patients with bladder cancer (Figure 1) and none of the control samples (P < 0.0001). In patients with bladder cancer, PCDH17 methylation was found to be significantly associated with advanced stage (P = 0.0171), high grade (P = 0.0082), large tumour diameter (P = 0.0004) and tumour recurrence (P = 0.0063) compared with the unmethylated gene. No association was found between PCDH17 methylation and age, sex, smoking, tumour number or tumour morphology (Table 1).
Representative results showing methylation-specific polymerase chain reaction products for protocadherin 17 (PCDH17) promoter methylation in tumour-derived DNA from six patients with bladder transitional cell carcinoma. bp, base pairs; M, methylated gene promoter; U, unmethylated gene promoter. Cases 61, 63, 64 and 66 exhibited PCDH17 promoter methylation.
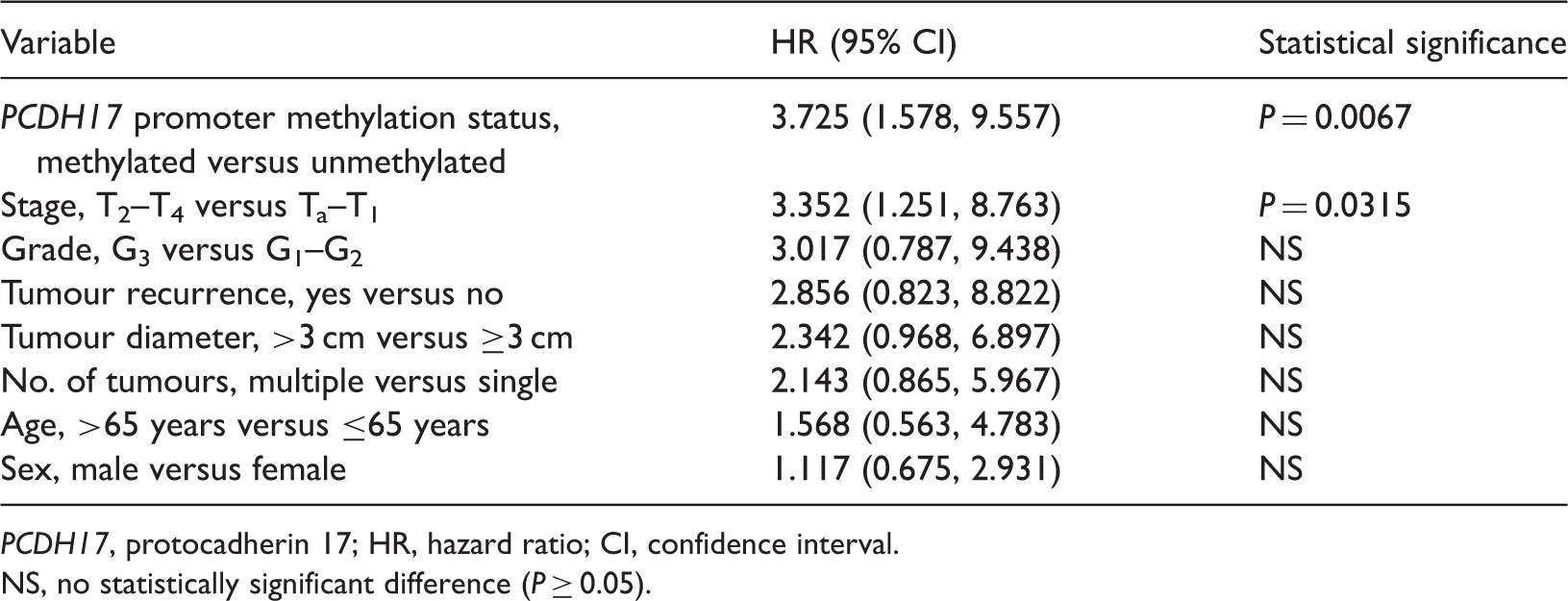
Patients with PCDH17 promoter methylation had significantly shorter survival times than those without methylation (P = 0.0036, Figure 2). Multivariate regression analysis revealed that PCDH17 promoter methylation (hazard ratio [HR] 3.725; 95% confidence intervals [CI] 1.578, 9.557) and advanced stage (HR 3.352; 95% CI 1.251, 8.763) were independent predictors of overall survival in bladder cancer (Table 2).
Kaplan–Meier survival curves for patients with bladder transitional cell carcinoma (n = 115), stratified according to protocadherin 17 (PCDH17) promoter methylation status assessed in tumour-derived DNA (P = 0.0036; log-rank test). Multivariate Cox proportional hazards analysis of independent predictors of overall survival in patients with bladder transitional cell carcinoma (n = 115), controlled for classic risk factors. PCDH17, protocadherin 17; HR, hazard ratio; CI, confidence interval. NS, no statistically significant difference (P ≥ 0.05).
Discussion
Bladder cancer is a common worldwide disease, with high morbidity and mortality rates. 18 More than 90% of bladder cancers are transitional cell carcinomas. 18 Thus, patients with bladder transitional cell carcinoma were selected as subjects in the present study. Bladder cancer is a disease driven by multistep accumulation of genetic and epigenetic changes. 6 Epigenetic alterations, including RNA interference, histone modifications and DNA methylation, may result in the silencing of cancer related genes, including tumour suppressor genes. 19 Moreover, DNA methylation is the most common epigenetic aberration in human malignancies. 20 Therefore, detecting promoter methylation changes in tumour suppressor genes could aid in the early diagnosis, monitoring and prognosis of bladder cancer. DNA methylation is a major mechanism of inactivation of tumour suppressor genes, besides genetic changes. In addition, DNA methylation can be used as a biomarker for tumour diagnosis, surveillance and prognosis, which will contribute to clinical decision-making and individualized treatment.20,21 Promoter methylation of PCDH17 has been reported in several human cancers, including bladder cancer, but the clinical significance of PCDH17 methylation in patients with bladder cancer remains unclear.7,13
The human PCDH17 gene is located on chromosome 13q21.2 and functions as a tumour suppressor in human cancers.7,13 The PCDH17 protein is a member of the cadherin superfamily. The protocadherins differ from classic cadherins in that their heterophilic interactions with other molecules may be more important for their various physiological functions than simple cell adhesion involving homophilic interactions. 22 PCDH17 may have multiple tumour-suppressive functions potentially contributing to cell–cell adhesion, signal transduction and growth control. In one study, methylation of the PCDH17 promoter was associated with the silencing of gene expression in oesophageal squamous cell carcinoma, which was associated with tumour malignancy, while restoration of PCDH17 expression was found to reduce cell proliferation, migration and invasion. 22 Similar results were also found in gastric and colorectal cancers. 7 Moreover, PCDH17 promoter methylation may frequently occur in urological cancers. 13 Thus, the current study aimed to investigate the clinical significance of PCDH17 promoter methylation in bladder cancer.
In the current study, promoter methylation of PCDH17 was found in 67.0% of bladder cancer tissues and was not detected in normal bladder epithelial tissues, suggesting that PCDH17 promoter methylation is tumour-specific and may be a potential biomarker in bladder cancer. Subsequently, the relationship between PCDH17 promoter methylation and clinicopathological parameters was analysed, revealing that methylation was significantly associated with advanced stage and high grade tumours, larger tumour diameter and tumour recurrence, which are all known risk factors for progression and poor prognosis in bladder cancer.23–25 The current results suggest that PCDH17 promoter methylation may be a useful biomarker for bladder cancer malignancy, similar to studies investigating gastric and colorectal cancers.7,12 In terms of prognosis prediction, patients in the current study with PCDH17 promoter methylation had significantly shorter survival times than those without methylation. In addition, multivariate Cox regression analysis indicated that PCDH17 promoter methylation was an independent predictor for prognosis in bladder cancer. The findings of the current study suggest that for patients with PCDH17 methylation, aggressive postoperative intervention should be performed, in order to achieve better prognosis.
Previous studies have demonstrated that other members of the cadherin superfamily, including CDH13, PCDH8 and PCDH10, were frequently found to be methylated in bladder cancer tissues, and this methylation was found to be associated with malignant behaviour and poor prognosis.10,15,26 These findings promoted the current investigation into the clinical significance of PCDH17 in bladder cancer, and suggest an important role for the cadherins in terms of disease progression and clinical outcomes.
This present study is limited by the small sample size. Larger scale prospective studies are needed to confirm our findings.
In conclusion, the current study suggests that PCDH17 promoter methylation is closely associated with malignant bladder cancer and may be used as an independent predictor of clinical outcomes in patients with bladder cancer. Detection of PCDH17 promoter methylation in tumour samples following surgery may be used to predict which patients need more aggressive postoperative intervention. In addition, DNA methylation is reversible. PCDH17 promoter methylation may therefore be a good therapeutic target in bladder cancer.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflict of interests.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
