Abstract
Objectives
This study retrospectively evaluated the prognostic significance of downregulated protocadherin-10 (PCDH10) gene expression in bladder cancer.
Methods
To evaluate the prognostic significance of downregulated PCDH10 protein levels, immunohistochemistry was used to assess the level of PCDH10 protein in surgically-resected formalin-fixed, paraffin wax-embedded transitional cell carcinoma specimens. Relationships between PCDH10 protein levels, clinicopathological characteristics and overall survival were also evaluated.
Results
A total of 105 bladder transitional cell carcinoma specimens and 33 normal bladder epithelial samples were investigated using immunohistochemical staining. PCDH10 protein levels were downregulated in 63.8% (67/105) of bladder cancer specimens compared with control samples. Downregulated levels of PCDH10 were significantly associated with advanced stage, higher grade, larger tumour size, nonpapillary shape, tumour recurrence and decreased overall survival rates. Multivariate analysis indicated that downregulated PCDH10 levels were independently associated with decreased overall survival and had a relative risk of death of 4.571.
Conclusions
Downregulated PCDH10 levels correlated with malignant behaviour and poor overall survival in patients with bladder cancer. Downregulated PCDH10 levels might be useful as a prognostic biomarker for bladder cancer.
Keywords
Introduction
Bladder cancer is a common international public health problem, with an estimated global incidence of > 356,000 new cases per year and a prevalence rate of 2.7 million cases. 1 In the USA, bladder cancer is the second most common genitourinary malignancy, with an estimated 73,510 new cases and 14,880 deaths in 2012. 2 However, the incidence of bladder cancer is increasing in developing areas of the world, attributed to global changes in exposure to risk factors for bladder cancer, and growth and ageing of the population. 1 Risk factors correlated with bladder cancer include cigarette smoking, chronic bladder inflammation, exposure to carcinogenic chemicals, genetic predisposition and age. 3 Of newly diagnosed bladder cancer cases, over 90% are transitional cell carcinomas; in terms of invasiveness, ∼70–80% are nonmuscle-invasive tumours (tumour stages Ta–T1) and the remainder are muscle-invasive tumours (tumour stages T2–T4). 3 The outcome of bladder cancer patients is variable because tumours that are morphologically similar can behave differently. 3 Therefore, specific biomarkers that could serve as prognostic indicators are needed to identify patients who need more aggressive systemic therapy.
Protocadherin-10 (PCDH10) is a member of the protocadherin subfamily, which in turn belongs to the cadherin superfamily. 4 The cadherin superfamily of transmembrane glycoproteins includes the protocadherins, atypical cadherins, desmosomal cadherins, cadherin-related neuronal receptors and classic cadherins. 4 Cadherins play important roles in calcium-dependent homophilic cell–cell adhesion, and are involved in the establishment of cell polarity, cell-sorting, cell differentiation, proliferation and migration. 4 Studies have demonstrated that PCDH10 is a putative tumour-suppressor gene; the human PCDH10 gene is located at 4q28.3, and promoter methylation and downregulation of PCDH10 gene expression has been demonstrated in many human cancers including hepatocellular carcinoma, multiple myeloma, gastric cancer, medulloblastoma, prostate cancer, colorectal cancer, pancreatic cancer and cervical cancer.4–10 In addition, ectopic expression of the PCDH10 gene suppressed tumour cell growth, migration, invasion and colony formation. 11 However, the expression of the PCDH10 gene in bladder cancer and its clinical significance remains unclear.
The present study investigated levels of PCDH10 protein in bladder cancer tissues and control samples using immunohistochemical staining, in order to determine if there is any relationship between PCDH10 protein levels and clinicopathological features and the prognosis of patients with bladder cancer.
Patients and methods
Participants and Sample Collection
This retrospective study collected tumour samples obtained during surgery at the Department of Urology, Second Hospital of Tianjin Medical University, Tianjin, China, from patients with bladder transitional cell carcinoma. The patients had been diagnosed pathologically between January 2003 and June 2006. Tumour diagnosis, staging, treatment and follow-up were completed according to international standards. 12 The inclusion criteria for patients with bladder cancer were: histopathological diagnosis of bladder transitional cell carcinoma; no history of other tumours; availability of follow-up data.
Normal bladder epithelial tissues were collected as controls from inpatients with bladder calculi, treated at the same hospital, during the same time period; these tissue samples were examined pathologically to exclude the possibility of incidental tumours. Inclusion criteria for control subjects were no history of any malignant tumours and no prior treatment with anticancer therapy. All of the tissue samples were fixed with 10% formalin and embedded in paraffin wax.
All patients with bladder cancer were followed-up after primary treatment at the Department of Urology, Second Hospital of Tianjin Medical University, at intervals increasing from 3 months to 12 months. Follow-up continued until the death of the patient or to 60 months if the patient remained alive. Follow-up data were recorded using both hospital files and information from the urologist or family physician responsible for the patient. Overall survival data for the patients with bladder cancer were collected during follow-up; overall survival was defined as the time from the date of diagnosis to the date of death from any cause, or the date of last contact if the patient was still alive, as previously described.13,14 For data analysis, patients with bladder cancer were divided into two groups according to the level of PCDH10 protein immunoreactivity determined by 3,3′-diaminobenzidine (DAB) immunohistochemistry: the normal PCDH10 protein level group and the downregulated PCDH10 protein level group.
Written informed consent was obtained from each study participant. This study was conducted in accordance with the Declaration of Helsiniki (2008) 15 and approved by the Ethics Committee of the Second Hospital of Tianjin Medical University.
Immunohistochemical Analysis for PCDH10
Immunohistochemical staining was used to detect the level of PCDH10 protein in tissue samples, as reported previously.16,17 Briefly, 4-µm formalin-fixed and paraffin wax-embedded sections were deparaffinized in xylene, and xylene was removed through a graded series of alcohol. Endogenous peroxidase activity was blocked with 3% hydrogen peroxide for 10 min. Antigen retrieval was performed by boiling the sections in 10 mM sodium citrate buffer (pH 6.0) for 15 min. Sections were then incubated with mouse antihuman PCDH10 monoclonal antibody (1 : 100 dilution; Santa Cruz Biotechnology, Santa Cruz, CA, USA) at 4°C overnight. After washing three times with 10 mM phosphate-buffered saline (PBS; pH 7.4), sections were incubated with horseradish peroxidase goat antimouse secondary antibody (1 : 100 dilution; Santa Cruz Biotechnology) at 37 °C for 20 min and washed three times with 10 mM PBS (pH 7.4). Finally, the immunolabelling was visualized with 0.05% DAB (Sigma-Aldrich, St Louis, MO, USA) diluted with 0.05 M Tris-buffered saline (pH 7.6), and all sections were counterstained with haematoxylin and mounted for light microscopy.
Scoring of PCDH10 Immunoreactivity
Assessment of the immunohistochemical staining results was undertaken by two pathologists who were blinded to the patients’ clinicopathological data. Normal bladder epithelial tissues were used as internal positive controls. The level of PCDH10 protein immunoreactivity in cancer cells was compared with that of normal epithelial cells. In accordance with previously published criteria,16,18 cancer cells that were stained in similar ways to normal epithelial cells were defined as having normal PCDH10 protein levels. The level of PCDH10 protein immunoreactivity was graded according to the proportion of positive cells. The percentage of positive cells was calculated by dividing the total number of positive cells with the total number of all cells, in at least ten randomly chosen nonoverlapping high-power (×400) fields for each case, using a light microscope (Olympus Optical, Tokyo, Japan). When >90% of the cancer cells were positively stained for the PCDH10 protein, the tumours were considered to be uniformly positive and regarded as having normal PCDH10 protein levels. When 10–90% of the cancer cells were positively stained for the PCDH10 protein, the tumours were considered to be heterogeneous; when <10% of the cancer cells were positively stained for the PCDH10 protein, the tumours were considered to be negative. Heterogeneous and negative staining were counted together as a downregulated PCDH10 protein level.16,18
Statistical Analyses
All statistical analyses were performed using the SAS® statistical software package, version 8.0 (SAS Institute, Cary, NC, USA) for Windows®. The χ2-test was used to assess the associations between the level of PCDH10 protein immunoreactivity and clinicopathological features. Kaplan–Meier survival analysis and log-rank test were used to assess the difference of overall survival between patients with normal and downregulated levels of PCDH10 protein immunoreactivity. Multivariate Cox proportional hazard model analysis was used to estimate the independent prognostic effect of the level of PCDH10 protein immunoreactivity after controlling for classic risk factors of stage, grade, tumour size and tumour recurrence. A P-value <0.05 was considered to be statistically significant.
Results
Relationship between protocadherin-10 (PCDH10) immunoreactivity and clinicopathological characteristics in patients with bladder cancer (n = 105).
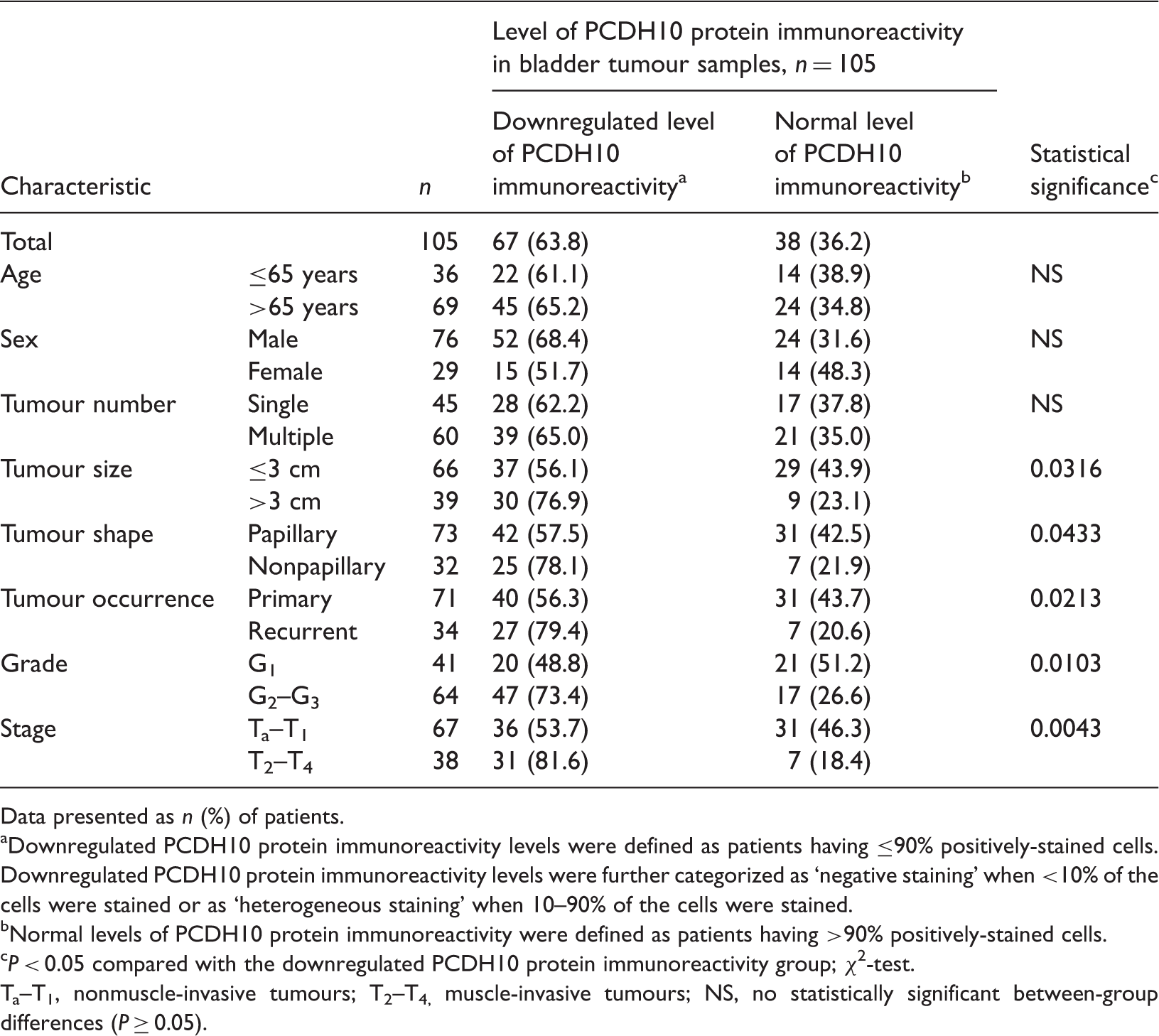
Data presented as n (%) of patients.
Downregulated PCDH10 protein immunoreactivity levels were defined as patients having ≤90% positively-stained cells. Downregulated PCDH10 protein immunoreactivity levels were further categorized as ‘negative staining’ when <10% of the cells were stained or as ‘heterogeneous staining’ when 10–90% of the cells were stained.
Normal levels of PCDH10 protein immunoreactivity were defined as patients having >90% positively-stained cells.
P < 0.05 compared with the downregulated PCDH10 protein immunoreactivity group; χ2-test.
Ta–T1, nonmuscle-invasive tumours; T2–T4, muscle-invasive tumours; NS, no statistically significant between-group differences (P ≥ 0.05).
Immunohistochemical analysis of the level of PCDH10 protein immunoreactivity in tissue specimens from 105 patients with bladder cancer and 33 control subjects found that all of the normal bladder epithelia showed equally strong PCDH10 protein immunoreactivity (>90% of the cells were positively stained) as shown in Figure 1A, which served as an internal positive control. Downregulated PCDH10 protein immunoreactivity was detected in 67 out of 105 (63.8%) tumour specimens, including 46 out of 105 (43.8%) heterogeneously stained tumours (Figure 1B) and 21 out of 105 (20.0%) negatively stained tumours (Figure 1C), which were all considered as having downregulated PCDH10 protein levels. Normal PCDH10 protein levels were detected in 38 of 105 (36.2%) patients with bladder cancer.
Representative light photomicrographs showing immunohistochemical staining for protocadherin-10 (PCDH10) protein in tumour samples from patients with bladder cancer and normal bladder epithelium from healthy control subjects. (A) PCDH10 immunoreactivity in normal bladder epithelium; (B) heterogeneous PCDH10 immunoreactivity in bladder cancer tissue; (C) negative PCDH10 immunoreactivity in bladder cancer tissue. Scale bars are indicated by the space between the square brackets, which represents 100 µm.
Downregulated levels of PCDH10 protein immunoreactivity were significantly associated with advanced disease stage (P = 0.0043), larger tumour size (P = 0.0316), poor differentiation (P = 0.0103), tumour recurrence (P = 0.0213) and a nonpapillary tumour shape (P = 0.0433) (Table 1). However, there was no association between downregulated levels of PCDH10 protein immunoreactivity and age, sex or tumour number.
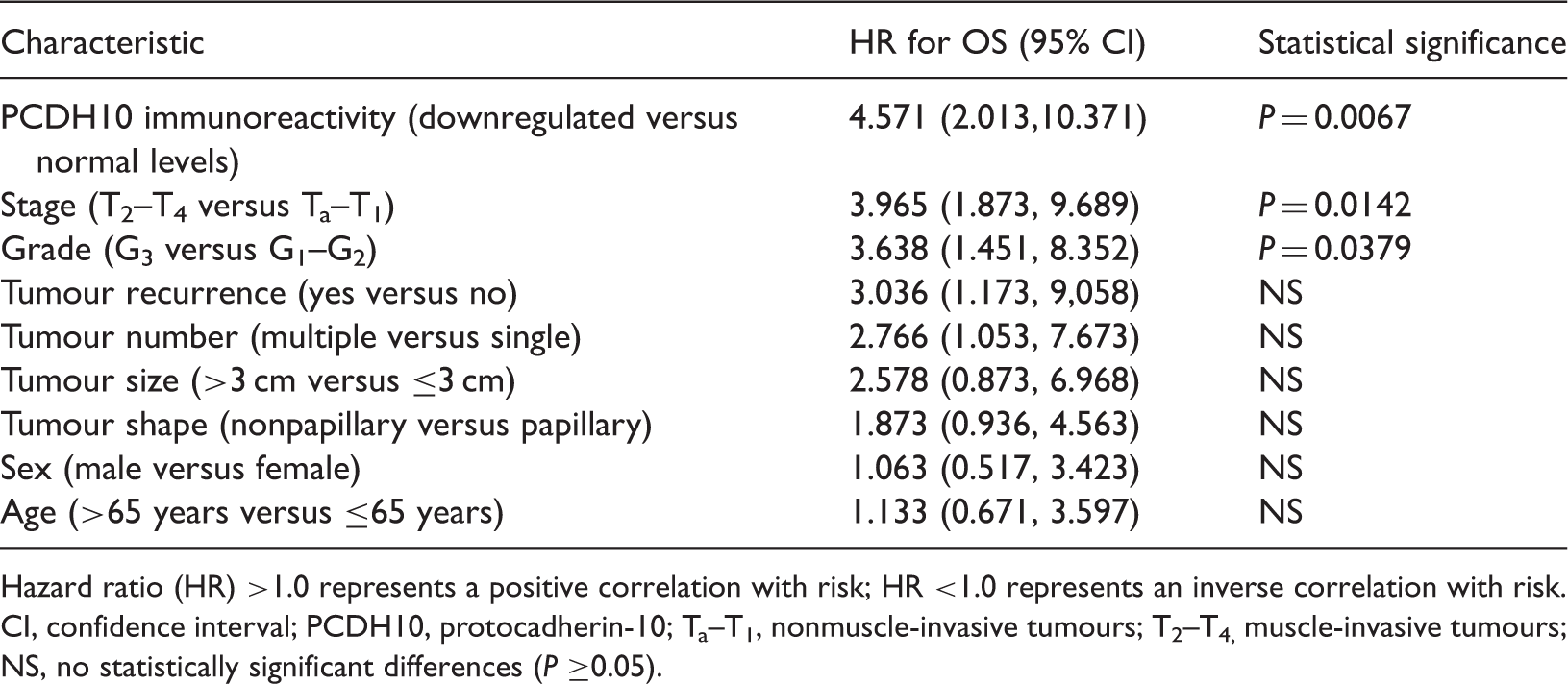
The mean ± SD follow-up duration among the 105 patients with bladder cancer was 51.9 ± 15.5 months (range 5–60 months). Kaplan–Meier survival analysis found that patients with downregulated levels of PCDH10 protein immunoreactivity had a significantly reduced survival time, compared with patients who had normal levels of PCDH10 protein immunoreactivity (P = 0.0055) (Figure 2). In the multivariate Cox proportional hazard model analysis (which controlled for the classic risk factors of stage, grade, tumour size and tumour recurrence), downregulated PCDH10 protein immunoreactivity was independently associated with poor overall survival and had a relative risk of death of 4.571 (95% confidence interval 2.013, 10.371; P = 0.0067) (Table 2).
Kaplan–Meier survival analysis for patients with bladder cancer (n = 105), according to the level of protocadherin-10 (PCDH10) protein immunoreactivity in tumour samples. Patients with normal levels of PCDH10 protein immunoreactivity (>90% of the cells were positively stained; n = 38) versus downregulated levels of PCDH10 protein immunoreactivity (≤90% of the cells were positively stained; n = 67). P = 0.0055 (log-rank test). Multivariate Cox proportional hazard model analysis of potential prognostic indicators for overall survival (OS) in patients with bladder cancer (n = 105). Hazard ratio (HR) >1.0 represents a positive correlation with risk; HR <1.0 represents an inverse correlation with risk. CI, confidence interval; PCDH10, protocadherin-10; Ta–T1, nonmuscle-invasive tumours; T2–T4, muscle-invasive tumours; NS, no statistically significant differences (P ≥0.05).
Discussion
Bladder cancer is a common malignancy, reported worldwide, that is associated with high levels of morbidity and mortality. 1 Unfortunately, the mortality rate for patients with bladder cancer increases with the extent of tumour invasion, despite the advances that have been made in tumour detection, treatment and surveillance strategies. For example, the approximate 5-year overall survival rates are ∼90% for nonmuscle-invasive disease, ∼60% for T2 disease and ∼35% for T3 disease, decreasing to ∼25% for T4 disease.19–21 Therefore, it is important to identify those patients who might benefit most from adjuvant therapy after surgery, so as to obtain better rates of prognosis. Currently, tumour grade and stage have been used as the most important prognostic characteristics, but both measures fail to predict patient prognosis accurately because tumours with similar morphology can behave differently. 3 Therefore, more reliable and objective biomarkers of prognosis are required.
The molecular aetiology of bladder cancer remains largely unknown, but many oncogenes and tumour suppressor genes that may play a role in the pathogenesis of bladder cancer have been identified, and could be useful when determining patient prognosis.3,17–21 The PCDH10 gene is a novel tumour suppressor gene; in many human cancer types, the PCDH10 gene has been shown to be downregulated by promoter methylation, and this downregulation correlates with a poor prognosis.4–11,22 In the present study, the level of PCDH10 protein immunoreactivity in bladder cancer specimens was downregulated compared with normal bladder epithelial tissues, suggesting that downregulation of the PCDH10 protein level might play an important role in the pathogenesis and progression of bladder cancer.
To elucidate the clinical significance of the levels of PCDH10 protein in bladder cancer, the present study evaluated the associations between PCDH10 protein immunoreactivity and clinicopathological characteristics. Invasion is the most important biological characteristic of human tumours, and is the major cause of treatment failure and death in patients with bladder cancer. 23 In the current study, downregulated levels of PCDH10 immunoreactivity were detected in 36 out of 67 (53.7%) nonmuscle-invasive tumours (Ta–T1), compared with 31 out of 38 (81.6 %) muscle-invasive tumours (T2–T4). This finding indicated that decreased levels of PCDH10 immunoreactivity were closely associated with bladder-cancer invasiveness. This result is in agreement with other studies, which showed that decreased levels of PCDH10 protein were also found in gastric cancer cells and tissues, and were associated with tumour invasiveness, 4 and that restoration of normal levels of PCDH10 protein in gastric cancer cells suppressed cell migration and invasion in vitro; 4 similar findings were demonstrated in medulloblastoma and prostate cancer studies.6,7 Taken together, these findings suggest that decreased PCDH10 protein levels may facilitate tumour invasion.
Differentiation is an important factor that contributes to tumour progression and determines patient outcomes. 23 In the current study, downregulated levels of PCDH10 immunoreactivity were detected in 20 of 41 (48.8%) well-differentiated tumours (G1), compared with 47 of 64 (73.4%) moderate or poorly differentiated tumours (G2–G3). This finding indicated that downregulated PCDH10 protein levels correlated with poor differentiation of bladder cancer. In addition, downregulated PCDH10 protein levels occurred in 37 out of 66 (56.1%) patients with tumours ≤3 cm in diameter and in 30 out of 39 (76.9%) patients with tumours >3 cm, which suggested that downregulated levels of PCDH10 protein might promote bladder cancer growth. This finding is similar to studies undertaken in gastric cancer, prostate cancer and medulloblastoma.4,6,7
Tumour recurrence is an important characteristic of bladder cancer that influences patient outcomes.24–26 In the present study, among the 105 patients with bladder cancer, the proportion of patients with downregulated levels of PCDH10 protein immunoreactivity in the recurrent group (27/34, 79.4%) was significantly higher than in the primary group (40/71, 56.3%) (P = 0.0213), suggesting that downregulated PCDH10 protein might contribute to the recurrence of bladder cancer. In addition, a downregulated level of PCDH10 protein immunoreactivity was significantly correlated with a nonpapillary tumour shape (P = 0.0433).
Taken together, current data suggest that downregulated PCDH10 protein levels, as identified by immunohistochemistry of tumour samples, are closely associated with the malignant behaviour of bladder cancer and might be used as a prognostic biomarker for bladder cancer patients. In order to confirm this hypothesis, overall survival was examined in this current study and patients with downregulated levels of PCDH10 protein immunoreactivity had a worse prognosis than those with normal PCDH10 protein levels. Moreover, multivariate Cox regression analysis indicated that a downregulated PCDH10 protein level was an independent predictor of poor overall survival after controlling for classic risk factors. This finding supports that of a previous report in gastric cancer. 22 These current findings suggest that a downregulated PCDH10 protein level might be a potential prognostic biomarker of overall survival in human bladder cancer.
In conclusion, the present study demonstrated that downregulated PCDH10 protein immunoreactivity was significantly correlated with the malignant behaviour of bladder cancer, and may be used as a potential prognostic biomarker of overall survival, for patients with bladder cancer. As the current study was limited by a small sample size, additional studies are needed to confirm these preliminary findings. In our opinion, if these current findings can be confirmed by larger independent studies, we would recommend using downregulated PCDH10 protein levels in tumour samples as a routine prognostic biomarker. This strategy would enable patients with bladder cancer to be offered the most appropriate therapeutic interventions following surgery, including radiotherapy, chemotherapy or biological therapy, with the goal of achieving the best attainable prognosis for each patient.
Footnotes
Declaration of Conflicting Interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
