Abstract
Segmental zoster paresis is a rare complication of herpes zoster, characterized by focal motor weakness that does not always present simultaneously with skin lesions. Zoster paresis can be easily confused with other neuromuscular or spinal diseases. This case report describes the case of a 72-year-old woman with herpes zoster and cervical spinal stenosis at the same spinal level, where it was difficult to distinguish segmental zoster paresis from cervical radiculopathy combined with motor neuropathy. Although segmental zoster paresis in the upper extremity is rare, it should be included in the differential diagnosis of segmental pain and weakness in the extremities, especially in older or immunocompromised patients. Correct diagnosis is required, to avoid unnecessary surgery and allow timely antiviral treatment.
Introduction
Herpes zoster is a very common infectious disease, characterized by painful vesicular skin eruptions distributed along unilateral sensory dermatomes. Extensive long-term general population studies indicate the incidence of herpes zoster to be 1.2–4.8 cases/1000 people per year in all ages, and 7.2–11.8 cases/1000 per year in those aged > 60 years.1,2 Reactivation of latent varicella zoster virus occurs decades after initial infection in older subjects or immunocompromised individuals, therefore the incidence of herpes zoster increases with age and in patients with immunodeficiency.2,3 Segmental zoster paresis is a rare complication (observed in 1–5 % of patients with herpes zoster) which involves focal motor weakness in the same segment as the cutaneous eruption.4–6 Making a diagnosis of zoster paresis is straightforward when characteristic herpetic eruptions present alongside motor symptoms, but it is difficult to determine the cause of weakness when it presents before cutaneous lesions, the myotomal and dermatomal distributions are dissociated, or any abnormal magnetic resonance imaging (MRI) or electromyography (EMG) findings are consistent with the symptoms.7–9 An erroneous diagnosis in post-zoster paresis may result in inappropriately aggressive surgical intervention and could prevent timely antiviral treatment. We report a case of limb paresis that occurred concurrently with cutaneous herpes zoster and existing degenerative cervical spinal stenosis.
Case report
A 72-year-old woman was referred to the pain clinic at The Catholic University of Korea, College of Medicine, Incheon St Mary’s Hospital, Republic of Korea on 29 August 2011. The patient presented with a pin-pricking, burning pain in the left shoulder that had started approximately 2 weeks previously, with the typical herpes zoster rash appearing 3–4 days later. The patient had been prescribed 750 mg/day famciclovir by mouth 2 days before presenting at the hospital. At the time of referral, the hyperalgesic skin lesion was distributed throughout the left C4–T2 dermatomes, with lancinating pain (visual analogue scale [VAS] score 9–10/10, where 10 represented the worst possible pain) being most severe in the left T1 segment (Figure 1). The patient did not complain of motor weakness during the initial visit and the results of routine laboratory analyses were normal.
A 72-year-old woman with hyperalgesic skin lesions characteristic of herpes zoster, distributed in the left C4–T2 dermatomes.
The patient underwent stellate ganglion and cervical epidural blocks with 20 mg triamcinolone, and received anticonvulsant (450 mg pregabalin) and antidepressant (10 mg nortriptyline) for treatment of neuralgia. After 1 week the pain and allodynia had improved, but the patient noted progressive weakness of the left shoulder. Neurological examination revealed left shoulder abduction weakness (grade 1–2/5).
10
This weakness began 1 week before the painful eruptions occurred, but was not reported by the patient because the pain was so severe at her initial visit. MRI of the cervical spine revealed degenerative changes with multilevel neural foraminal stenosis at C3–T1, and combined cervical cord compression caused by a bulging disc at C3–4 (Figure 2A). The foraminal stenosis was most severe at the left C4–5 foramen (Figure 2B). Deep tendon reflexes were equal and active bilaterally, and the patient had no other physical signs of cervical myelopathy. Needle EMG showed denervation potentials (positive sharp waves) in the left biceps, deltoid, brachioradialis and C5 – 6 paraspinal muscles. Sensory and motor nerve-conduction studies were normal and there were no abnormalities in somatosensory evoked potentials. These findings were consistent with a left C5–6 radiculopathy. The patient was informed of the possibility of combined zoster paresis and weakness from the foraminal stenosis and a cerebrospinal fluid (CSF) study was recommended, which she refused.
(A) Sagittal T2-weighted magnetic resonance image of a 72-year-old woman with left shoulder abduction weakness, showing cord compression (arrow) at the C3–4 vertebral level. (B) Axial T2-weighted magnetic resonance image showing left neural foramen narrowing at the C4 – 5 vertebral level (arrows).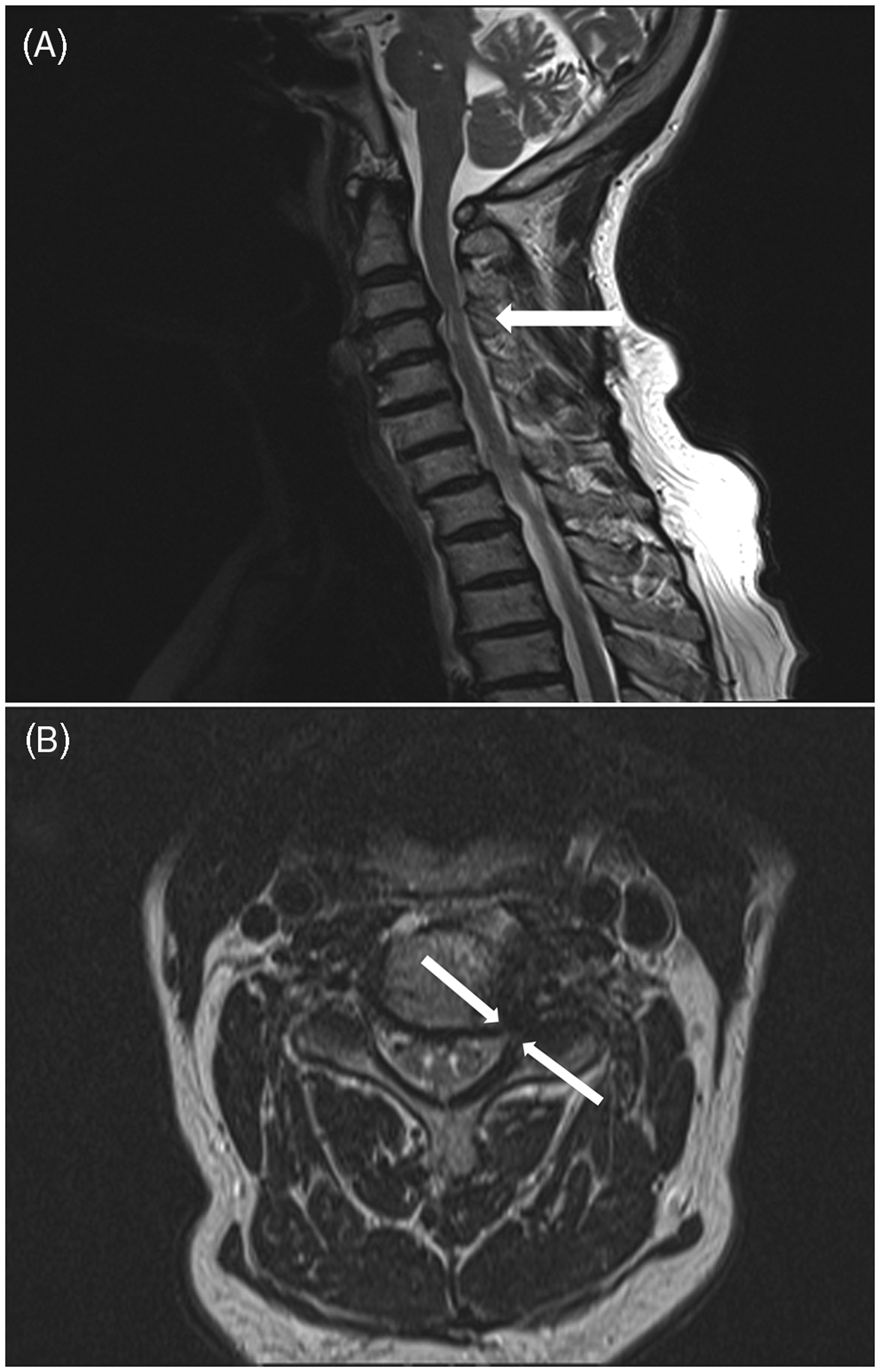
Over the next 4 weeks the patient received two further triamcinolone cervical epidural blocks and a stellate ganglion block, and continued pregabalin and tricyclic antidepressant treatment. She underwent physiotherapy, which focused on rehabilitation and strengthening of the left shoulder. After 2 months, the pin-pricking pain had disappeared and burning pain was reduced (VAS score 2/10), but the weakness remained. Left shoulder abductor power had improved to grade 3/5 at 6 months after initial presentation.
Discussion
Although acute herpes zoster and postherpetic neuralgia are familiar to physicians, zoster-associated motor symptoms are rare and are often misdiagnosed as another type of motor neuropathy.4,7–9 The current case presented with shoulder weakness in the presence of zoster skin lesions, leading us to suspect segmental zoster paresis even though cervical spine MRI and EMG findings were consistent with radiculopathy caused by foraminal stenosis at the C4–6 levels.
The reported incidence of segmental limb paresis with cutaneous zoster is 1–5%,5,6,11,12 but the true incidence of this complication is unknown because of the difficulty in detecting symptoms of thoracic and upper cervical motor weakness.5,7,13 EMG studies enable the detection of additional motor involvement that is not associated with clinical symptoms.14,15 The exact mechanism of this unusual spread of herpes zoster virus has not been established, but pathological and neuroradiological studies indicate that the virus spreads proximally as well as distally, causing local neuritis in the spinal nerve, anterior horn cells and the anterior root (as well as in the posterior horn cells and dorsal roots).14,15 The basic pathological neural reaction to herpes zoster is axonal degeneration with a degree of secondary segmental demyelination.16,17 Facial muscles are most susceptible to zoster-induced paresis. 4 The cervical and lumbosacral segments show a low incidence of zoster paresis, and it is commonly reported in a lower cervical (C5–7) or lumbosacral distribution. 7 Motor manifestations in the upper limbs involve various neurological syndromes including cervical radiculopathy or brachial plexopathy, and multiple mononeuropathy associated with painful eruptions on the upper limbs. 17
Segmental zoster paresis does not always involve the same distribution as the sensory dermatomal distribution, which may cause diagnostic confusion.7,8 Weakness usually occurs within 2–3 weeks of rash onset, but can precede the skin eruption or occur long after the skin eruption in rare cases.9,18 Zoster paresis peaks within hours or days,7,14,17,19 with weakness varying from mild to very profound. Prognosis is favourable, with complete functional recovery in 55% of cases and an additional 30% of cases showing substantial improvement. 4 Appropriate antiviral therapy may reduce the incidence and severity of segmental zoster paresis. 7
Given its variable clinical manifestations, segmental zoster paresis can be confused with spinal radiculopathy, rotator cuff injury, impingement syndrome or spinal myelopathy; misdiagnosis may lead to incorrect treatment such as unnecessary surgery.8,9,11,15 It is difficult to distinguish zoster paresis from complications of spinal surgery when the weakness occurs postoperatively, either without or before a skin eruption. 9 In cases with unclear clinical symptoms, spinal MRI may help to exclude degenerative disorders of the cervical spine, such as stenosis or cord malignancy.9,20 In addition, EMG or CSF studies could be useful for the differential diagnosis of zoster paresis. Tests for zoster virus-specific antibodies or polymerase chain reaction (PCR) of varicella zoster virus DNA in the CSF can be helpful, especially in suspected cases of zoster myelitis, encephalitis or zoster sine herpete (herpes zoster infection without skin lesions).21,22 However, PCR results are time dependent, with diagnostic sensitivity decreasing steeply 1–2 weeks after disease onset.8,21,23 Typical EMG findings are similar in segmental zoster paresis and radiculopathy due to root compression: they include reduced recruitment, fibrillations, and positive sharp waves or polyphasic motor unit potentials in muscles supplied by the affected root. 14 Sensory conduction studies can be used to distinguish between patients with zoster paresis and those with spinal compressive radiculopathy, as the majority of patients with zoster paresis present with reduced-amplitude sensory nerve action potentials and, less frequently, absent or reduced compound motor action potentials.14,15,19 Sensory and motor nerve-conduction test results were normal in the current case, and there were no abnormalities in somatosensory evoked potentials. Taken together with cervical-spine MRI findings, this suggested a C5–6 radiculopathy due to spinal stenosis rather than zoster radiculopathy. Electrodiagnostic studies of muscles and nerves are affected by the lesion location, the degree of axonal damage and the disease stage. 24 In addition, MRI examinations to investigate spinal disorders are associated with a high false-positive rate in asymptomatic individuals, which increases with age. 25 The incidence of asymptomatic spinal cord compression also increases with age and could be considered a normal aspect of the degenerative changes of the spine. 26 The findings of electrodiagnostic studies and MRI concur in only 60% of patients with symptoms suggestive of radiculopathy, and in 76% of cases when weakness is present.26,27
The patient described in this report clearly presented with herpes zoster and spinal stenosis in the same segment, but it was not possible to determine whether the weakness was due to zoster paresis or cervical foraminal stenosis. Judging from the clinical course, it is probable that the ongoing zoster-induced neural inflammation caused swelling of the spinal root in the presence of foraminal stenosis. Proposed indications for surgery in patients with cervical radiculopathy are unremitting radicular pain despite 6–8 weeks’ conservative treatment, progressive motor weakness or presence of myelopathy. 28 The neurosurgeon treating the current case cautiously recommended decompression surgery for severe foraminal stenosis, based on MRI evidence of cervical nerve root compression which was consistent with clinical signs and symptoms. However, the weakness did not progress, and the patient refused spinal surgery and invasive diagnostic procedures. Epidural steroid injection (including transforaminal epidural injection) is known to be effective in zoster paresis, cervical spondylotic radiculopathy and postherpetic neuralgia.29,30 Cervical epidural steroid injection in the current case resulted in a decrease in neuralgic pain (to a VAS score of 2/10) within 2 months, and a partial improvement in segmental paresis at 6 months, after presentation.
In conclusion, we describe a case in which it was difficult to distinguish between segmental zoster paresis and cervical radiculopathy combined with motor neuropathy. Although segmental zoster paresis in the upper extremity is rare, it should be included in the differential diagnosis of segmental pain and weakness in the extremities, especially in older or immunocompromised patients. Correct diagnosis is required, to avoid unnecessary surgery and allow timely antiviral treatment.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
