Abstract
A 48-year-old female with long-standing type 2 diabetes mellitus presented with acute onset of bilateral lower limb weakness. She had been previously well and denied any constitutional symptoms. Physical examinations revealed generalized lower limb weakness with bilateral lower limb hypotonia, power of 0 over 5, reduced deep tendon reflexes, and loss of peripheral sensations up to the level of T10. Upper limb functions were normal. Rectal examination showed a lax anal tone and reduced anal grip. Blood investigations showed elevated serum alkaline phosphatase, corrected serum calcium, and parathyroid hormone. Magnetic resonance imaging of the spine revealed an expansile mass at the posterior element of the ninth thoracic vertebrae, causing spinal compression with possible impingement of the right T9 exiting spinal nerve. An urgent surgical decompression and tissue biopsy were performed for stabilization of the spine. Intraoperative findings included spinal cord compression secondary to an epidural tumour mass extending from T9 to T10 disc levels. Histopathological analysis showed a giant cell tumour of the spine. A 99mTc Sestamibi-SPECT parathyroid scintigraphy showed an ectopic parathyroid adenoma at the left suprasternal region. A diagnosis of Brown tumour secondary to ectopic parathyroid adenoma was made. She underwent an exploratory parathyroidectomy procedure with removal of the ectopic parathyroid gland, which resulted in a normalization of the serum calcium and parathyroid hormone. Unfortunately, her lower limb functions did not return to normal, and she remained paraplegic at 6 months postoperatively.
Keywords
Introduction
A Brown tumour is a non-neoplastic giant cell lesion characterized by an increased circulating level of parathyroid hormone. It is the localized form of osteitis fibrosa cystica and most commonly affects the pelvis, ribs, long-bone shafts, clavicle, and mandible. 1 Hyperparathyroidism associated with brown tumours is generally rare, the incidence of which is less than 5%. 2 We describe a case of a 48-year-old lady who presented with sudden onset of bilateral lower limb weakness and it was associated with an expansile spinal mass which was surgically removed with the diagnosis of primary bone tumour. Histopathological examination subsequently revealed the mass as a manifestation of primary hyperparathyroidism caused by a parathyroid adenoma.
Brown tumour is more commonly found in ribs, clavicles, pelvis, femur, and facial bones.3,4 Spine involvement with a brown tumour in patients with primary hyperparathyroidism is rare and even rare to be reported as the first presentation of hyperparathyroidism. 5 Out of 22 cases of brown tumours of the spine that have been reported, the thoracic spine ( 59%) was observed to be the most commonly involved part of the spine, followed by lumbar ( 27%), cervical (9%), and sacral (4.5%) regions. 6 Clinical manifestations include pain, pathologic fracture, and spinal cord compression with related neurologic deficits. 6 Gradual paralysis, pathological fracture, and decrease in muscle strength can be observed in more severe cases. 6
Ectopic parathyroid glands are not common. It is mainly arising from abnormal migration of parathyroid tissue during embryogenesis. 7 In about 6%–16% of primary hyperparathyroidism, one or more hyperfunctioning parathyroid glands are located ectopically, which may often remain undetected and are a cause for failed parathyroid exploration.8,9 This case highlights that spinal Brown tumour should be considered as the differential diagnosis of lytic, expansile spinal tumours, especially with the histopathological diagnosis of giant cell tumours. Urgent interventions that include both treating the underlying primary cause and surgical decompression of the neural structures could help preserve neural function and stabilize the spine.
Case summary
A 48-year-old female with a background history of Type 2 diabetes mellitus, presented with acute onset of bilateral lower limb weakness. The symptoms initially started with right-sided weakness, numbness, and paraesthesia, which gradually progressed to total loss of power of both lower limbs 24 h later. There was no history of falls, trauma, lifting heavy objects, or preceding back pain. There was no loss of consciousness, headache, altered speech, or abnormal vision. The patient denied any urinary retention or loss of anal tone. On further history, there was no exposure to tuberculososis contact nor any symptoms to suggest tuberculosis. There was no family history of malignancy. She was on metformin and denied taking any alternative medicine.
Her physical examination revealed bilateral lower limbs muscle wasting and hypotonia, with the power of 0 out of 5 and reduced deep tendon reflexes. There was loss of sharp and blunt discrimination up to the level of T10. The plantar reflexes were equivocal bilaterally Rectal examination showed a lax anal tone and reduced anal grip, with the absence of urinary and bowel symptoms. Upper limb examinations were normal.
Her baseline investigations showed a normal full blood count, renal profile, and liver profile. However, her serum albumin was low 25 mg/L (normal value: 35–55 mg/L) with low magnesium; 0.55 mg/dL (normal level: 1.7–2.2 mg/dL). Erythrocyte sedimentation rate (ESR) was not elevated.
Her magnetic resonance imaging (MRI) of the spine showed an expansile mass at T9 posterior element causing spinal compression with possible impingement of the right T9 exiting spinal nerve (Figures 1 and 2). There was no focal spinal cord lesion to suggest transverse myelitis. Computed tomography sonography (CT scan) was performed to detect any underlying neoplasm, which showed no evidence of a primary tumour in the abdomen, thorax, and neck.

Sagittal T2W MRI whole spine revealed an expansile mass at T9 posterior element causing spinal compression with possible impingement of right T9 exiting spinal nerve. There was no focal spinal cord lesion to suggest transverse myelitis.
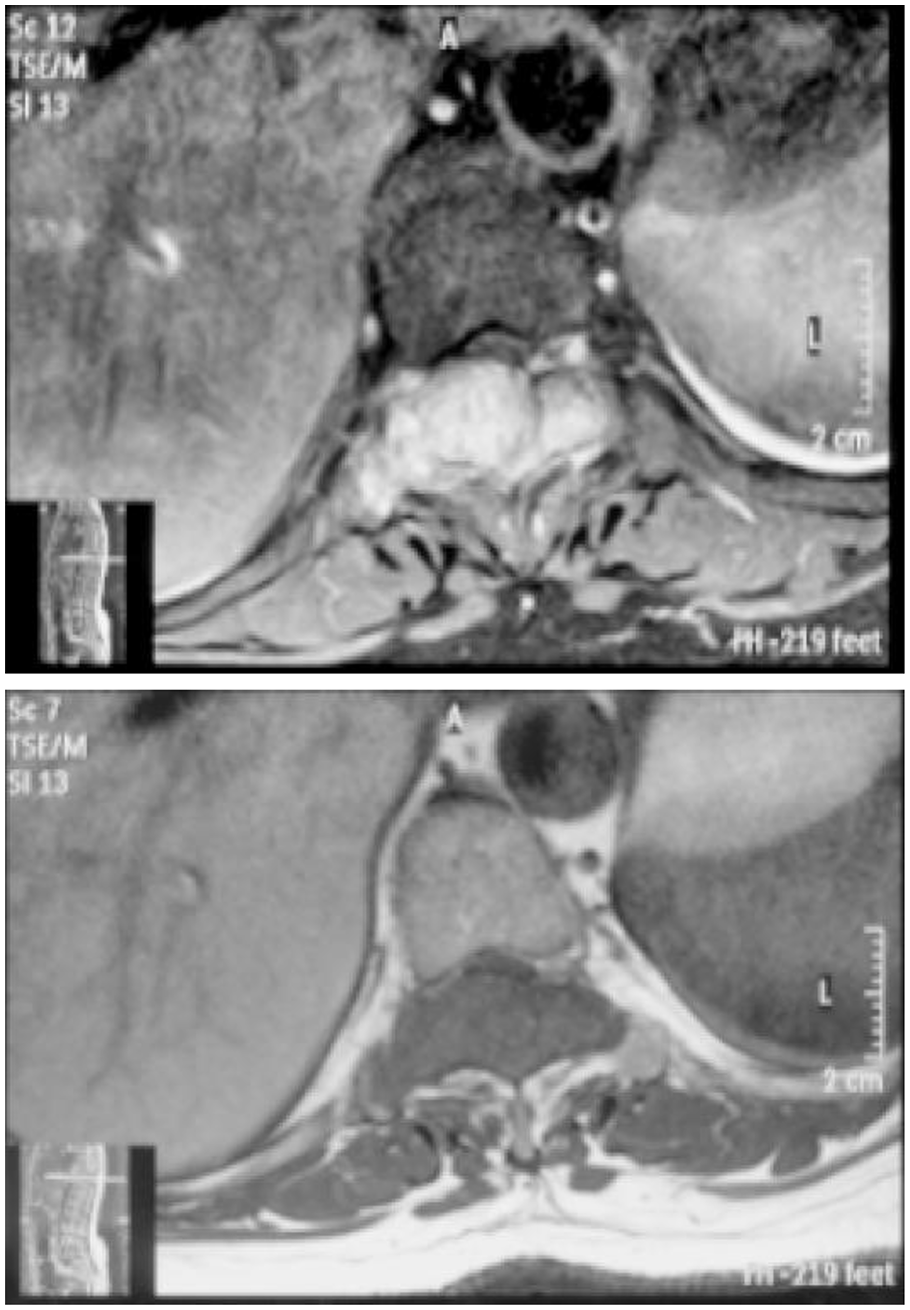
Axial T2 MRI showed the expansile mass at level T9 causing spinal canal stenosis, cord compression, and nerve root impingement.
The initial differentials diagnosis made were spinal stenosis secondary to primary bone malignancy or bone metastases. An urgent surgical decompression was performed for stabilization of the spine. Intraoperative findings include spinal cord compression secondary to an epidural tumour mass seen from T9 to T10 disc levels. Decompression was achieved through a posterior spinal instrumented fusion of T7–T11 with laminectomy accompanied by a T9 posterior element resection via a standard posterior midline approach (Figure 3). Post-surgical CT images showed no residual mass with stable instrumentation. Subsequent histopathological analysis from the intraoperative bone biopsy showed osteoclast-like giant cells that appear uniformly round shape mononuclear cells (Figure 4). A giant cell tumour of the spine or ‘brown tumor’ was confirmed as the diagnosis.

Plain CXR showed posterior spinal instrumented fusion T7–T11. Pedicle screw inserted bilateral T7, T8, T11, T12.

HPE at low and high resolution showed large numbers of evenly distributed multinucleated osteoclast-like giant cells in a background of sheets of round to oval-shaped mononuclear cells. This confirmed the diagnosis of giant cell tumour of parathyroid adenoma or Brown tumour.
Further biochemical analyses showed increased values of alkaline phosphatase of 220 U/L (normal range: 100–172 U/L), corrected calcium of 3.9 mmol/L (normal range: 2.25–2.5 mmol/L), and serum parathyroid hormone level of 62.93 pmol/L (normal range: 1.05–6.83 pmol/L). An ultrasound scan of the neck failed to reveal any parathyroid tumour. Subsequently, a 99mTc Sestamibi-SPECT parathyroid scintigraphy was performed which localized an ectopic parathyroid adenoma at the left suprasternal region.
She subsequently underwent an explorative procedure with removal of the ectopic parathyroid gland with prompt normalization of the serum calcium. A histopathological examination of the mass confirmed the diagnosis of parathyroid adenoma. Postoperatively, her serum corrected calcium, phosphate, and alkaline phosphatase levels were within normal range. There was no other clinical manifestation of hungry bone syndrome. Her serum parathyroid hormone level also within normal range throughout the follow up. Unfortunately, her lower limb function remained impaired, and she remained paraplegic at 6 months postoperatively. She is currently undergoing a spinal rehabilitation programme.
Discussion
Here, we highlighted an unusual presentation of Brown tumour secondary to ectopic parathyroid adenoma. Brown tumour is a relatively rare non-neoplastic lesion, which occurs in patients with hyperparathyroidism, most commonly affects large bones, such as ribs, clavicles, femur, jaw, and pelvis. 10 – 12 It affected mainly patients with primary (44.3%) and secondary (51.5%) hyperparathyroidism compare with tertiary hyperparathyroidism (4.2%). 13 Brown tumours often overlooked in patient with underlying end-stage renal failure disease as calcimemtic and vitamin D analogues were used as treatment for secondary hyperparathyroidism. 14
Brown tumour is described as the localized form of osteitis fibrosa cystica and characterized by abnormal osteoclastic and osteoblastic activities via prolonged excessive parathyroid hormone production causing bone resorption and bone marrow fibrosis. 15 – 17 Histologically, it is characterized by multinucleated giant cells in a background of spindle cell proliferation along with a large amount of hemosiderin deposition, within the vascularized bone marrow fibrosis giving a brown appearance to the lesion.2,15,18
Brown tumours tend to be misdiagnosed as malignancies due to their unspecific radiographic appearances. Other differential diagnoses including non-ossifying fibroma, aneurysmal bone cyst, fibrous dysplasia, chondroma, osteosarcoma and myloplax tumour, and metastatic disease must be considered.5,19,20 Early clinical suspicion, biochemical markers, parathyroid ultrasonography, parathyroid scintigraphy (99mTc-MIBI), and bone scintigraphy (99mTc-MDP) are important to reach the correct diagnosis. 21
Our patient presented with manifestation of Brown tumour affecting her spine, later found to be secondary to primary hyperparathyroidism from ectopic parathyroid adenoma. Unfortunately, symptoms of hypercalcaemia did not manifest earlier, causing her to become paraplegic as consequences of brown tumour.
Ectopic parathyroid adenoma constitutes a common etiology of persistent or recurrent hyperparathyroidism. Ectopic parathyroids are most frequently found in the anterior mediastinum, in association with the thymus or the thyroid gland, and tracheoesophageal groove and retroesophageal region. 22 It has been reported that brown tumour at left mandibular bones had occurred in patient with ectopic parathyroid adenoma that was associated with acute thyroxine ingestion, though the exact mechanism was not clear. 16 Another case reported of a brown tumour situated at maxilla due to ectopic parathyroid adenoma situated in the anterior mediastinum. 23 In both cases, patients were initially presented with mass and swelling of the jaw.
The mainstay of treatment for brown tumours focuses on treating the hyperparathyroidism.2,16 The resection of parathyroid adenomas promptly corrected the hypercalcaemia as shown in our patient. 23 Brown tumours may subsequently regress and almost all radiographic changes return to normal with correction of serum parathyroid and calcium levels.2,16 However, there were cases where brown tumour resection was the treatment of choice when there was persistent tumour growth following parathyroidectomy despite normalization of parathyroid hormone levels. 24 In other cases of the brown tumour without any severe disfigurement, the use of systemic or intralesional corticosteroid has been reported to reduce the size of the lesion. 16 As for almost all neurological manifestations, surgical intervention should be done early and promptly to increase the probability of full neurological recovery. Although the patient reported here described an acute presentation of the lower limb weakness, it was more likely that she had been manifesting the symptoms over a longer period which would explain the failure of full recovery after surgery.
After 3 days of surgery, her parathyroid hormone and serum corrected calcium returned normal. In patient with severe serum PTH level, there is higher chance for patient to develop hungry bone syndrome after removal of parathyroid adenoma which did not occur in our patient in view of pre-existing normal parathyroid gland. Follow-up radiological findings showed no residual development of the tumour. Unfortunately, her neurological function did not return to normal. Despite early detection and surgical intervention, her mobility had been compromised. This differs from previous reports that showed neurological function was improved in almost all patients with spine brown tumours after the operation. 6
Conclusion
Although uncommon, brown tumours constitute a real challenge and should be considered as a plausible diagnosis in a patient with an isolated bony lesion. Correlations between clinical, histological aspects, and imaging findings were essential in establishing an accurate diagnosis. Promptly performed decompression surgery may improve neurological deficits and delay in the management may result in a permanent disability that unfortunately was demonstrated in this patient. The intervention of brown tumour should include early diagnosis, complete biochemical assays, and complete skeletal survey. In the absence of any obvious parathyroid abnormalities, ectopic adenomas should be identified with more advanced imaging techniques including radioisotope scans.
Footnotes
Acknowledgements
The authors thank their patient and all individuals who contributed to the management of this case.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
