Abstract
Carotid intima–media thickness (cIMT), a noninvasive marker of subclinical atherosclerosis beginning in childhood, has been linked to cognitive decline in adulthood. However, the nature and shape of this association remain unclear. We investigated linear and nonlinear associations between cIMT and cognitive function using observational and Mendelian randomization (MR) analyses. Observational and one-sample MR analyses were conducted in UK Biobank participants (n = 19,588). Two-sample MR used summary data from the CHARGE/UCLEB Consortia (n = 71,128) for cIMT and from a meta-analysis of UK Biobank and COGENT (n = 257,828) for cognitive function. Mediation MR evaluated the role of cerebrovascular disease. Observational analysis showed higher cIMT was associated with lower cognitive function (β = −0.17, 95% CI = −0.27 to −0.08, p = 3.24 × 10−4), with no nonlinearity (pnonlinearity = 0.49). One-sample MR supported a causal effect of higher genetically predicted cIMT on lower cognitive function (β = −1.18, 95% CI = −2.33 to −0.03, p = 4.51 × 10−2), with no evidence of nonlinearity (Pquadratic test = 0.88; PCochran Q test = 0.90). This was confirmed in two-sample MR (β = −0.83, 95% CI = −1.39 to −0.26, p = 4.03 × 10−3). Mediation analysis indicated that 9.9% of this association was mediated by small vessel stroke. Reverse MR showed no causal effect of cognitive function on cIMT. Our findings demonstrate a linear-no-threshold relationship between cIMT and cognitive function, underscoring the importance of early-life vascular health.
Keywords
Introduction
Dementia, the fourth leading cause of global neurological health loss, is characterized by a progressive decline in cognitive function. 1 One underlying explanation of such decline has been ascribed to atherosclerosis in carotid arteries, the vessels that deliver blood to the brain. 2 Indeed, carotid intima–media thickness (cIMT), a noninvasive surrogate marker of subclinical atherosclerosis and cardiovascular risk, has long been linked to adult cognitive decline.3,4 For example, the hitherto largest meta-analysis identified a significant association of higher cIMT with lower cognitive function (β = −0.15, 95% CI = −0.24 to −0.07; p < 0.001) in 15 cross-sectional studies (n = 15,118). Such a relationship, albeit not supported by synthesizing four cohort studies (β = −0.09, 95% CI = −0.20 to 0.02; p = 0.063) perhaps due to limited sample sizes (n = 3611) and insufficient follow-up periods (⩽4 years), 3 was confirmed by the Brazilian Longitudinal Study of Adult Health (β = −0.018, 95% CI = −0.024 to −0.013; p < 0.001) with an enlarged sample size (n = 9263) and a prolonged follow-up period (8 years). These findings collectively strengthen the link between cIMT and cognitive function.5,6
While well-designed observational studies have advanced our understanding of the association between cIMT and cognitive function, the causal nature and dose–response relationship of this association remain incompletely understood. First, observational associations are susceptible to residual confounding and reverse causality—inherent limitations preventing causal inference. While randomized controlled trials (RCTs) remain the state-of-the-art approach to establishing causal associations in clinical research, Mendelian randomization (MR), which employs genetic variants as a natural experiment analogous to a randomized trial, can offer more robust inferences about causal relationships between modifiable risk factors and health outcomes compared to conventional observational approaches. 7 To our knowledge, no prior study has yet investigated the causal associations between cIMT and cognitive function. Second, existing studies have not investigated the nonlinear associations,5,6 thereby hindering accurate characterization of the dose–response relationship. Given that nonlinear associations of cIMT with cardiovascular diseases have been reported,8,9 a similar pattern may exist for cognitive function. Third, while cIMT is strongly linked to stroke 10 and cerebral small vessel disease, 11 whether and the extent to which these cerebrovascular conditions mediate the causal pathway from cIMT to cognitive function remains unexplored. The potential mediating role of these vascular pathologies—critical for elucidating underlying mechanisms—has not been systematically addressed in previous research.
Therefore, we aimed to comprehensively characterize the nature and the shape of the cIMT-cognitive function association, leveraging the most comprehensive observational and genetic data from 19,588 UK Biobank participants. We performed the largest cross-sectional analysis to date to investigate both linear and nonlinear associations between cIMT and cognitive function. We then performed the first linear and nonlinear MR analyses to investigate causal associations between cIMT and cognitive function. We further performed two-step MR analyses to investigate whether cerebrovascular conditions mediate the pathway from cIMT to cognitive function. The overarching goal of our study was to provide genetic insights into the observed associations underlying cIMT and cognitive function, and eventually to advance precision prevention and medicine for cognitive decline and dementia.
Materials and methods
Data sources
UK Biobank
UK Biobank (UKB) is a prospective cohort study recruiting ~500,000 participants (aged 40–69 years at recruitment) from 22 assessment centers across the UK between 2006 and 2010. 12 All participants provided written informed consent, and the National Health Service North West Multi-Centre Research Ethics Committee provided ethical approval (11/NW/0382). We identified a subset of 37,919 unrelated participants of European ancestry with complete cIMT measurements at the imaging visit and genetic data that passed the quality control.13,14 cIMT was defined as the mean of the maximum of the four carotid artery measurements, 120° and 150° for the right carotid artery, 210° and 240° for the left carotid artery. We excluded participants without complete cognitive function measurements at the imaging visit. Cognitive function was assessed using a fluid intelligence score, which measures the ability to solve problems through logic and reasoning. Participants completed 14 five-option questions within a 2-min time limit. Any unanswered items due to time expiration were scored as zero. A simple unweighted sum of correct responses was calculated as the raw fluid intelligence score. To enhance the comparability of results, scores were standardized to z-scores with a mean of 0 and a standard deviation (SD) of 1. In total, 19,588 participants were included in both observational and genetic analyses (Supplementary Figure 1).
GWAS of cIMT
The largest genome-wide association study (GWAS) of cIMT meta-analyzed data from the UKB and the CHARGE/UCLEB consortia (Cohorts for Heart and Aging Research in Genomic Epidemiology/UCL–LSHTM–Edinburgh–Bristol), comprising 100,253 individuals of predominantly European ancestry (>98%). 14 cIMT was defined as the mean maximum value from multiple common carotid artery measurements. A total of 15 independent SNPs (p < 5 × 10−8) were identified. To mitigate potential horizontal pleiotropy bias, we excluded one SNP within the APOE region (Chr19: 45,116,911–46,318,605), retaining 14 SNPs as IVs (Supplementary Table 1). Since UKB contributed to ~32% of this meta-GWAS cohort, we utilized effect size estimates for these IVs derived from the CHARGE/UCLEB GWAS (n = 71,128) 15 to prevent overfitting.
GWAS of cognitive function
The largest GWAS of cognitive function meta-analyzed data from the UKB and the Cognitive Genomics Consortium (COGENT), comprising 257,841 individuals of European ancestry. 16 Cognitive function was defined as a fluid intelligence score or a statistically derived broadband index reflecting individual performance on a neuropsychological test battery. A total of 226 independent SNPs (p < 5 × 10−8) were identified and used as IVs (Supplementary Table 2).
GWAS of cerebrovascular diseases
To mitigate potential sample overlap bias, we utilized European-ancestry GWAS summary statistics for stroke phenotypes from the MEGASTROKE consortium: stroke (40,585 cases and 406,111 controls), ischemic stroke (34,127 cases and 404,630 controls) with its subtypes including large-artery atherosclerotic stroke (4373 cases and 200,868 controls), small vessel stroke (5386 cases and 248,959 controls), and cardioembolic stroke (7193 cases and 340,825 controls). 17 For intracerebral hemorrhage, we utilized summary statistics from the largest European-ancestry GWAS, meta-analyzing data from FinnGen, UKB, and the International Stroke Genetics Consortium (7605 cases and 711,818 controls). Cerebral small vessel disease was represented by European-ancestry GWAS data for lacunar stroke (6030 cases and 219,389 controls) 18 and four magnetic resonance imaging markers including white matter hyperintensity volume (n = 18,381), 19 fractional anisotropy (n = 17,663), 19 mean diffusivity (n = 17,467), 19 brain microbleeds (3556 cases and 22,306 controls; 97% European ancestry; Supplementary Table 3). 20 Independent SNPs (p < 5 × 10−8, r2 < 0.1 within a 1-Mb window) were selected as IVs. Given the limited number of genome-wide significant variants for small vessel stroke and brain microbleed, we relaxed the conventional IV selection threshold to p < 1 × 10−6.
Covariates
Age and sex were obtained from registries. Educational attainment (years of schooling completed) was calculated by mapping the highest level of education that a respondent achieved to an International Standard Classification of Education (ISCED) 1997 category and then imputing a years-of-education equivalent for each ISCED 1997 category. 21 The first 10 genetic principal components were calculated by UKB to account for population genetic structures. Sleep duration was categorized as short (⩽6 h/day), normal (7–8 h/day), and long (⩾9 h/day). Smoking status was categorized as never-smoker and ever-smoker. Alcohol intake frequency was categorized as daily or almost daily, three or four times a week, once or twice a week, one to three times a month, special occasions only, and never. Physical activity was categorized as low, moderate, and high according to the International Physical Activity Questionnaire. Body mass index (BMI) was calculated as the measured weight (kg) divided by the square of standing height (m2). Hypertension was defined as self-reported medical conditions code 1065; antihypertensive medication usage (Anatomical Therapeutic Chemical classification code C02); the International Classification of Diseases, Ninth Revision (ICD-9) code 401 and ICD-10 code I10; systolic blood pressure ⩾140 mmHg or diastolic blood pressure ⩾90 mmHg measured at the imaging visit. Type 2 diabetes was defined as self-reported medical conditions code 1223; antidiabetic medication usage (A10); ICD-10 code E11; fasting blood glucose ⩾7.0 mmol/L or glycated hemoglobin ⩾48 mmol/mol measured at baseline. Dyslipidemia was defined as lipid-lowering medication usage (C10AA); triglycerides >1.7 mmol/L, or total cholesterol >5.0 mmol/L, or low-density lipoprotein cholesterol >3.0 mmol/L, or high-density lipoprotein cholesterol <1.0 mmol/L (male) and 1.2 mmol/L (female) at baseline. Stroke was defined as self-reported medical conditions codes 1081, 1086, 1491, and 1583; ICD-9 codes 430, 431, 433, 434, and 436; ICD-10 codes I60, I61, I63, and I64. The detailed definitions are shown in Supplementary Table 4.
Statistical analysis
Observational analysis
We first fitted univariable linear regression models to investigate the observational associations of demographic (age, sex, educational attainment, and the first 10 genetic principal components), lifestyle (sleep duration, smoking status, alcohol intake frequency, and physical activity), and cardiometabolic (BMI, hypertension, type 2 diabetes, and dyslipidemia) variables with cognitive function. We next fitted multivariable linear regression models to estimate the beta coefficient (β) and 95% confidence interval (95% CI) for the observational association of cIMT with cognitive function. We implemented a series of models with incremental adjustments as follows: model 1 (the basic model) adjusted for demographic variables except age as a potential moderator 5 ; model 2 adjusted for model 1 covariates plus lifestyle variables; model 3 (the full model) adjusted for model 2 covariates plus cardiometabolic variables. We further added an interaction term between cIMT and age (continuous) to the full model to investigate the impact of age on the observational association between cIMT and cognitive function. Since the interaction term was significant, we performed a subgroup analysis stratified by age (<60 vs ⩾60 years). We finally performed a restricted cubic spline (RCS) with four knots (fifth, 35th, 65th, and 95th) to investigate the nonlinear observational association.
Mendelian randomization analysis
We first constructed a weighted genetic risk score (GRS) to instrument cIMT. We next evaluated the plausibility of the three core MR assumptions by assessing the associations of GRS with cIMT, cognitive function, age, sex, and cardiometabolic risk factors. 22 We performed a one-sample linear MR analysis using the two-stage least squares method to investigate the linear causal effect of cIMT on cognitive function. We further performed a nonlinear MR analysis using the fractional polynomial method to investigate the nonlinear association. Specifically, we performed stratified analyses in strata of the population constructed using the doubly ranked stratification method to estimate the localized average causal effect (LACE). 23 We performed meta-regression of LACE against the mean of the cIMT in each stratum using the derivative of fractional polynomial models of degrees 1 and 2. 24 We performed the quadratic test and Cochran Q test to examine whether a nonlinear model fits the LACE estimates better than a linear model. All analyses were adjusted for age at the imaging visit, sex, and the first 10 genetic principal components. We additionally performed a bidirectional two-sample MR analysis using the inverse-variance weighted method (IVW) as our primary approach. 25 We also performed several sensitivity analyses using the weighted-median 26 and MR-Pleiotropy Residual Sum and Outlier (MR-PRESSO) 27 method to assess the robustness of results. We finally performed a two-step MR analysis to evaluate the causal effects of cIMT on cerebrovascular disease and the causal effects of cerebrovascular disease on cognitive function. For significant mediators, we estimated the indirect effect by multiplying the effect of cIMT on the mediator by the effect of the mediator on cognitive function adjusting for cIMT. 28 The proportion mediated was calculated as the indirect effect divided by the total effect. Confidence intervals were derived from 1000 bootstrapping iterations. 29
All statistical analyses were performed using the R software version 4.5.1. A two-sided p < 0.05 was considered statistically significant. We followed the Strengthening the Reporting of Observational Studies in Epidemiology Using Mendelian Randomization (STROBE-MR) statement. 30 The study was performed in accordance with the principles of the Declaration of Helsinki.
Results
Participants characteristics
Characteristics of the study participants are shown in Table 1. Among 19,588 participants, the mean (SD) age was 64.6 (7.7) years and 53.2% were women; the mean years of schooling completed was 16.2 (4.4) years; the mean cIMT was 0.8 (0.1) mm. The distributions of cIMT and cognitive function are shown in Supplementary Figure 2.
Participant characteristics and their associations with cognitive function.
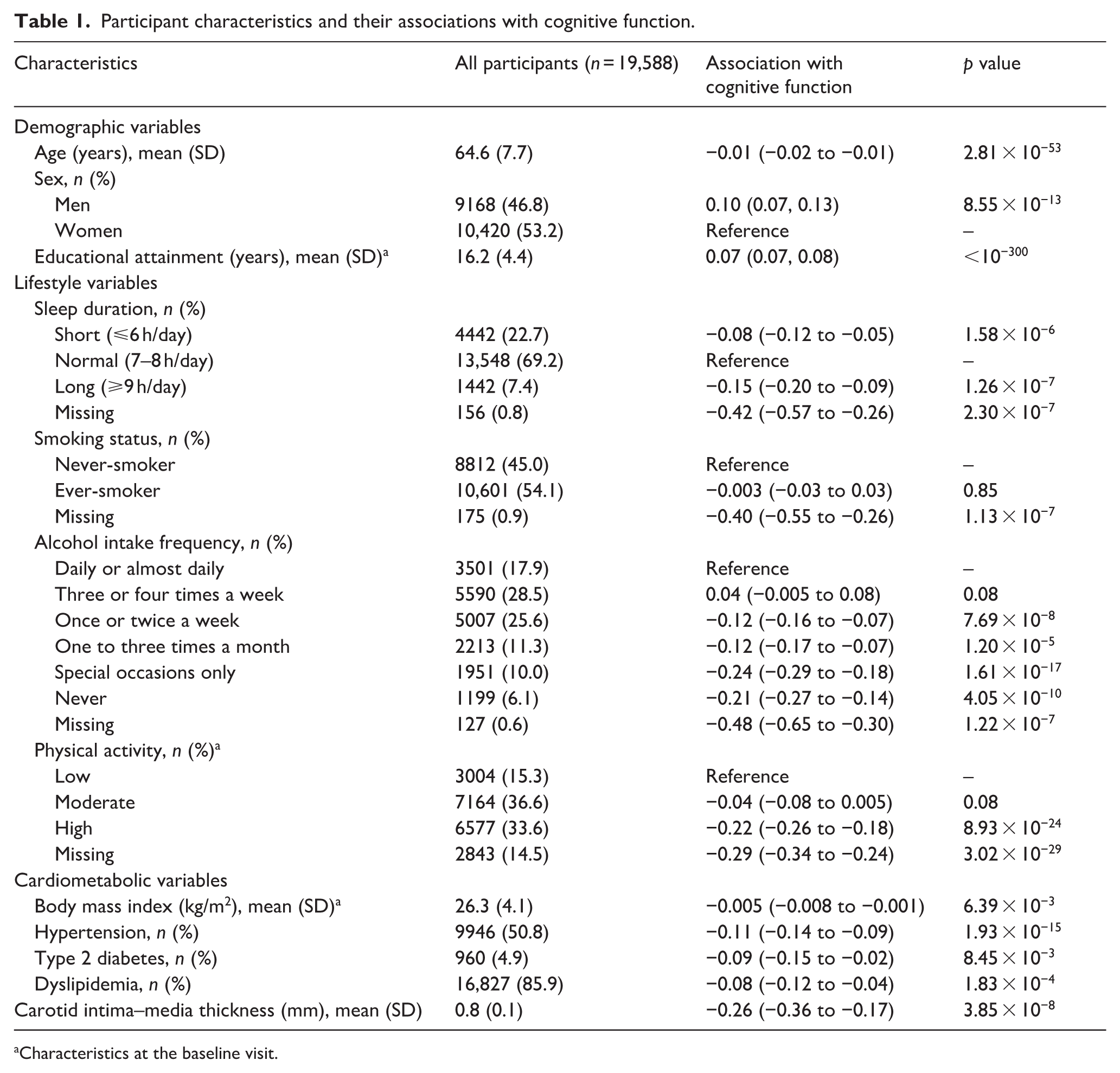
Characteristics at the baseline visit.
Older adults (β = −0.01, 95% CI = −0.02 to −0.01; p = 2.81 × 10−53), participants with short (β = −0.08, 95% CI = −0.12 to −0.05; p = 1.58 × 10−6) or long sleep duration (β = −0.15, 95% CI = −0.20 to −0.09; p = 1.26 × 10−7), infrequent alcohol consumers (once or twice a week: β = −0.12, 95% CI = −0.16 to −0.07; p = 7.69 × 10−8; one to three times a month: β = −0.12, 95% CI = −0.17 to −0.07; p = 1.20 × 10−5; special occasions only: β = −0.24, 95% CI = −0.29 to −0.18; p = 1.61 × 10−17; never: β = −0.21, 95% CI = −0.27 to −0.14; p = 4.05 × 10−10), participants with high-level physical activity (β = −0.22, 95% CI = −0.26 to −0.18; p = 8.93 × 10−24), higher BMI (β = −0.005, 95% CI = −0.008 to −0.001; p = 6.39 × 10−3), hypertension (β = −0.11, 95% CI = −0.14 to −0.09; p = 1.93 × 10−15), type 2 diabetes (β = −0.09, 95% CI = −0.15 to −0.02; p = 8.45 × 10−3), and dyslipidemia (β = −0.08, 95% CI = −0.12 to −0.04; p = 1.83 × 10−4) showed a lower cognitive function, while men (β = 0.10, 95% CI = 0.07–0.13; p = 8.55 × 10−13) and participants with higher educational attainment (β = 0.07, 95% CI = 0.07–0.08; p < 10−300) showed a higher cognitive function.
Observational associations
After adjusting for sex, educational attainment, and genetic principal components, higher cIMT was associated with lower cognitive function (β = −0.23, 95% CI = −0.32 to −0.14; p = 6.74 × 10−7; Table 2). Additional adjustment for sleep duration, smoking status, alcohol intake frequency, and physical activity had minimal impact on the association (β = −0.21, 95% CI = −0.30 to −0.12; p = 4.55 × 10−6). This association remained significant in the full model (β = −0.17, 95% CI = −0.27 to −0.08; p = 3.24 × 10−4). The interaction between cIMT and age on cognitive function was significant (p = 1.25 × 10−3). Stratified analysis showed a significant association of higher cIMT with lower cognitive function in participants aged 60 years or older (β = −0.12, 95% CI = −0.23 to −0.02; p = 2.42 × 10−2), but not in participants younger than 60 years (β = −0.03, 95% CI = −0.25 to 0.20; p = 0.83) despite a consistent direction of effect.
Linear observational associations of carotid intima–media thickness with cognitive function.
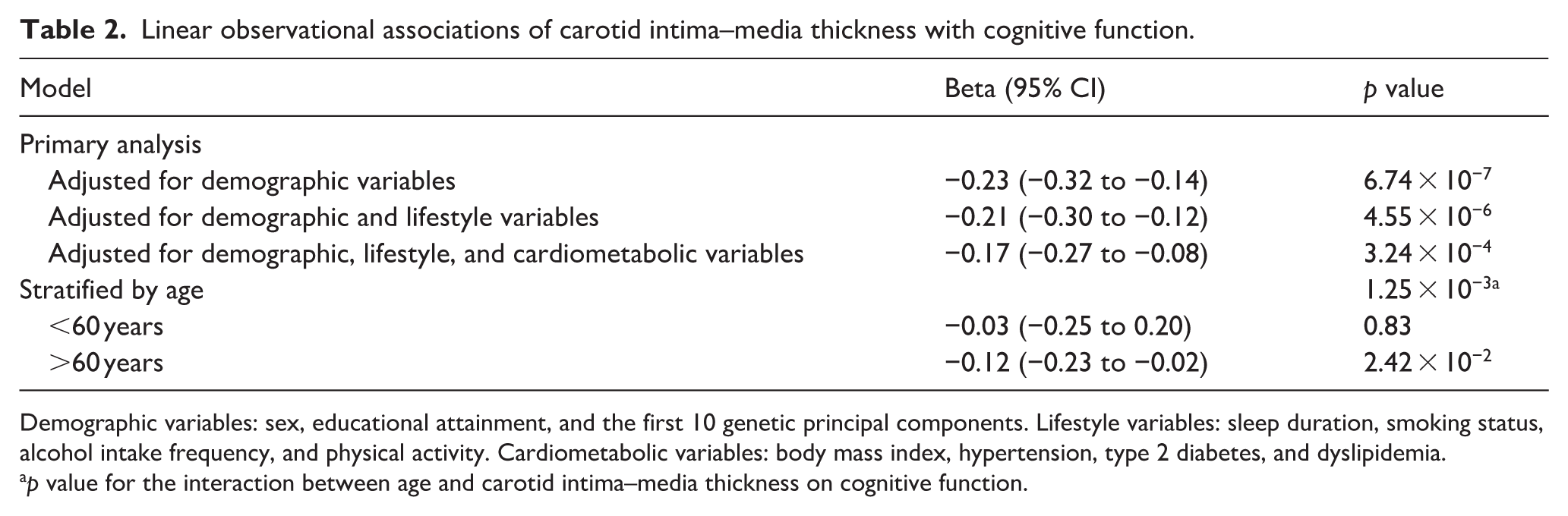
Demographic variables: sex, educational attainment, and the first 10 genetic principal components. Lifestyle variables: sleep duration, smoking status, alcohol intake frequency, and physical activity. Cardiometabolic variables: body mass index, hypertension, type 2 diabetes, and dyslipidemia.
p value for the interaction between age and carotid intima–media thickness on cognitive function.
Restricted cubic splines (Figure 1) supported a linear association between cIMT and cognitive function (pnonlinearity = 0.49). A similar result was observed in participants aged 60 years or older (pnonlinearity = 0.75).
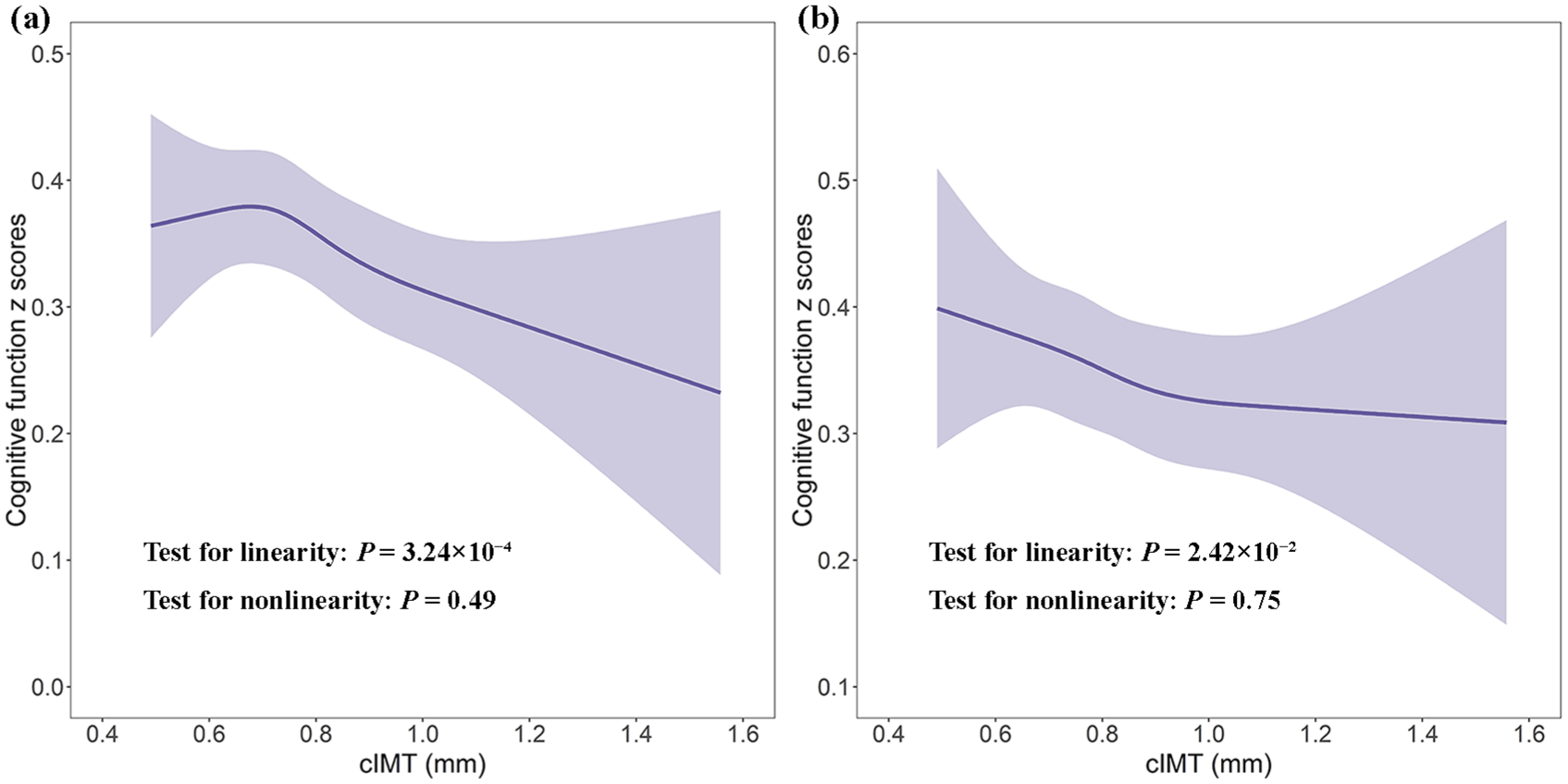
Nonlinear observational associations of carotid intima–media thickness with cognitive function. Panel (a) was performed in all participants, whereas panel (b) was performed in participants aged over 60 years. Estimates were adjusted for sex, educational attainment, the first 10 genetic principal components, sleep duration, smoking status, alcohol intake frequency, physical activity, body mass index, hypertension, type 2 diabetes, and dyslipidemia. The shaded areas represent 95% confidence intervals.
Genetic associations
The GRS showed a significant association with cIMT (p = 2.18 × 10−30), explaining 0.67% of the cIMT variance. The GRS was associated with cognitive function (p = 2.46 × 10−2), while this association attenuated to non-significant after adjusting for cIMT (p = 0.07). The GRS was not associated with age (p = 0.93), sex (p = 0.59), BMI (p = 0.87), and type 2 diabetes (p = 0.99), except for hypertension (p = 0.03) and dyslipidemia (p = 0.04), but the magnitudes of these associations were small. This negative association between GRS and cognitive function remained essentially unchanged after adjustment for hypertension and dyslipidemia (p = 3.62 × 10−2).
One-sample linear MR analysis identified a significant negative causal effect of genetically predicted cIMT on cognitive function (β = −1.18, 95% CI = −2.33 to −0.03; p = 4.51 × 10−2; Figure 2(a)). Nonlinear MR analysis supported a linear causal association between cIMT and cognitive function (pquadratic test = 0.88; pCochran Q test = 0.90; Figure 2(b)).
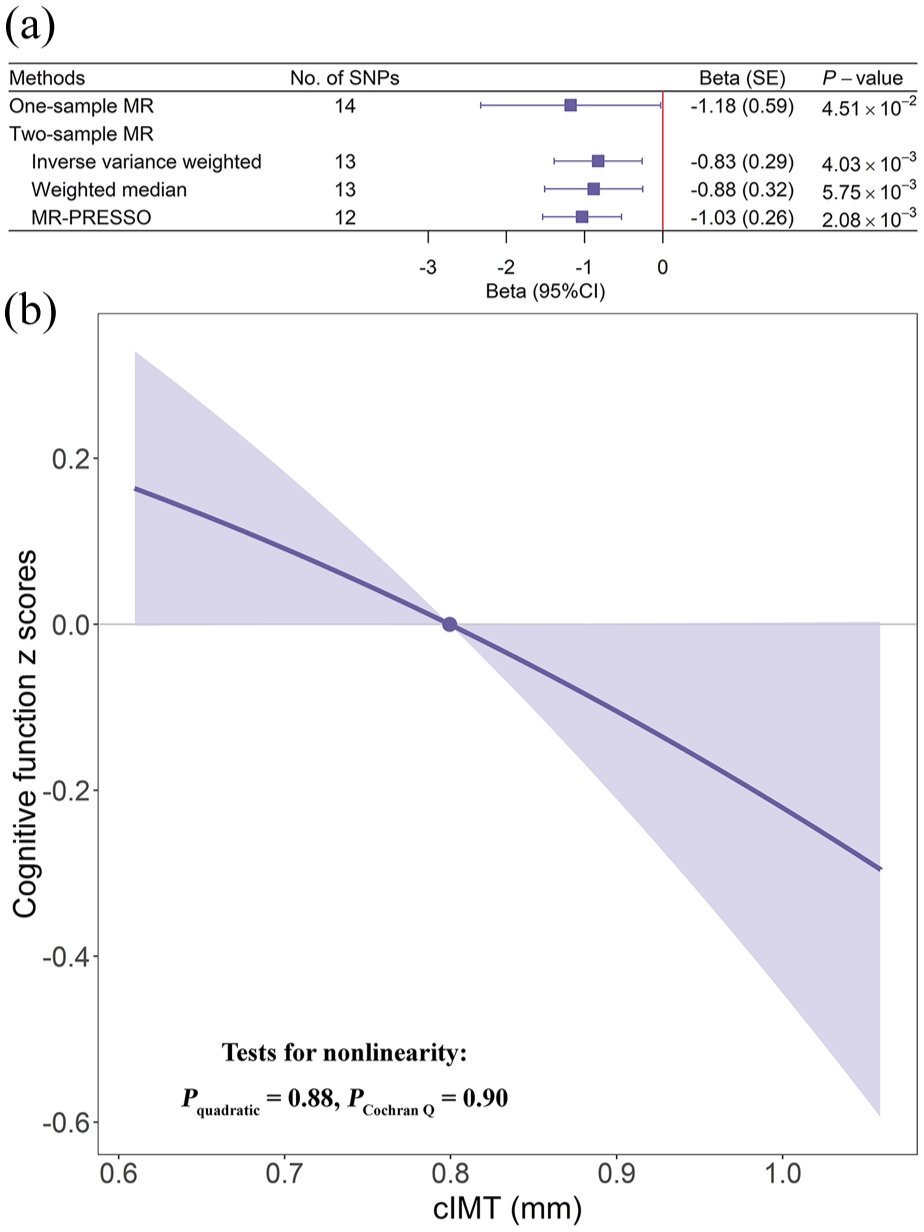
Linear and nonlinear Mendelian randomization estimates between carotid intima–media thickness and cognitive function. The boxes (a) denote point estimate of the causal effects (beta), and the error bars denote 95% CIs. The gradient (b) at each point of the curve represents the localized average causal effect, and the shaded areas represent 95% CIs. Estimates in one-sample linear and nonlinear MR were adjusted for age at the time of the imaging visit, sex, and the first 10 genetic principal components.
Two-sample MR analysis showed a consistent negative causal effect of genetically predicted cIMT on cognitive function (β = −0.83, 95% CI = −1.39 to −0.26; p = 4.03 × 10−3). This causal relationship was further supported by sensitivity analyses and was not affected by horizontal pleiotropy. In the reverse-direction MR, genetically predicted cognitive function was not associated with cIMT (β = −0.005, 95% CI = −0.011 to 0.001; p = 0.08; Supplementary Figure 3).
Two-step MR analysis identified small vessel stroke as a significant mediator in the cIMT-cognitive function association (Supplementary Figures 4 and 5). The proportion of this association mediated by small vessel stroke was estimated at 9.9% (0.3%–39.6%; Figure 3).
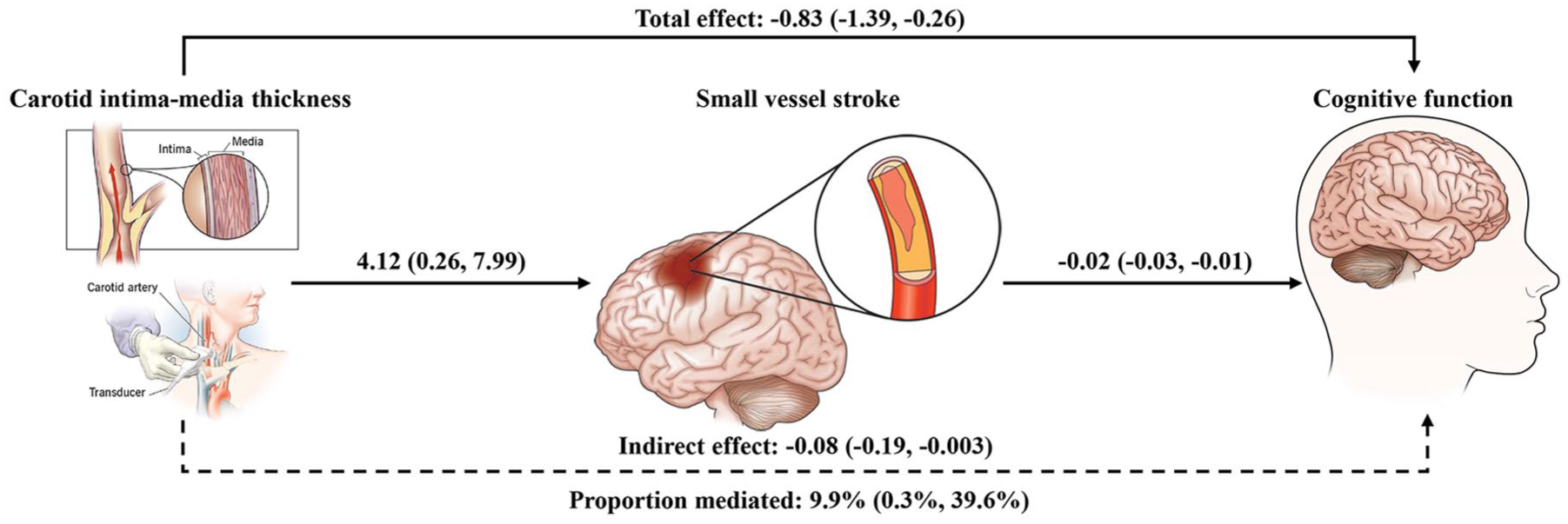
Mendelian randomization mediation analysis estimates between carotid intima–media thickness and cognitive function. The effect estimates (beta coefficients) are provided with standard errors.
Discussion
To our knowledge, this is the most comprehensive observational and genetic analyses that systematically investigate the nature and shape of the association between cIMT and cognitive function. Through evidence triangulation capitalizing on observational and genetic design, our results demonstrate that higher cIMT is putatively causally associated with cognitive decline, with no evidence of nonlinearity. Mediation analysis revealed that small vessel stroke accounts for a significant proportion of this causal relationship. The consistent findings across both observational and genetic analysis further suggest a potential benefit in slowing cIMT progression across the lifespan to mitigate cognitive decline.
While previous observational studies suggest a negative association between cIMT and cognitive function, such a relationship has not been supported by solid evidence from causal inference methods. Our study thus used a stepwise analytical framework: first investigating linear causal associations using conventional MR, followed by a state-of-the-art nonlinear MR to further characterize the dose–response relationship and a two-step MR to elucidate causal pathway. First, our linear MR provided convincing evidence supporting a potentially causal, detrimental effect of higher cIMT on cognitive function, consolidating current knowledge,3,5,31 and revealing novel insights into the role of cIMT in dementia pathogenesis. 32 This finding carries significant public health implications given that 27.6% of individuals aged 30–79 years in the general population in 2020 globally exhibit an abnormal cIMT of 1.0 mm and above (increased cIMT). 33 Crucially, cIMT progression is modifiable as an international collaborative meta-analysis aggregating 119 RCTs (n = 100,667) demonstrated a 9% reduced cardiovascular risk per 10 μm/year decrease in cIMT progression. 34 Since various forms of dementia may deliver a continuum spectrum rather than a strict dichotomy as in Alzheimer’s disease or vascular dementia, 35 the fluid intelligence score used in the current study may be a best fit to investigate the complexity of this pathological condition. 36 Second, our nonlinear MR revealed no threshold effects, corroborating a linear causal association observed in our linear MR. This linear-no-threshold relationship implies proportionality between cIMT and cognitive decline, supporting the concept of “the lower the better,” and further promoting our understanding of the dose–response relationship. Third, our MR mediation analysis uncovered the role of small vessel stroke mediating the causal effect of cIMT on cognitive function, elucidating causal pathways underlying this association. Collectively, these findings underscore the significance of establishing preventive intervention targeting cIMT progression in early life, raise the possibility of preventing atherosclerosis of brain-supplying arteries to maintain lifelong cognitive health. These results further support cIMT as a candidate screening marker for the early detection of cognitive decline, including mild cognitive impairment and dementia.
Our study features several key strengths. First, the MR design mitigates residual confounding and reverse causality limitations inherent in conventional observational studies. We implemented complementary one-sample and two-sample MR approaches with extensive sensitivity analyses leveraging the largest available genomic data, ensuring sufficient statistical power. Triangulation across these MR methods and observational analyses yielded consistent results, strengthening the robustness and validity of our findings. Second, we used a state-of-the-art nonlinear MR to test for nonlinearity in the cIMT-cognitive function association, providing novel insights into the dose–response relationship. Third, MR mediation analysis elucidated cerebrovascular disease’s role in the causal pathway, advancing mechanistic understanding of how higher cIMT impairs cognitive function.
We also acknowledge potential limitations. First, we restricted all analyses to participants of European ancestry to minimize the bias from population stratification. Therefore, our findings might not be generalizable to other ancestral populations. In particular, further analyses are warranted to investigate the potential effect of cIMT on cognitive function for individuals living in the Western Pacific region, as there is the largest share of global cases of increased cIMT and dementia.33,37 Second, cIMT might be important only at critical times during the decline of cognitive function. Our observational analysis identified age as a moderator of this relationship, and age-stratified analysis supported a significant association only in participants older than 60 years but not in those younger than 60 years. Despite a lifelong causal effect confirmed by our MR analyses, this critical time effect is consistent with previous prospective studies,5,38 indicating the intervention to cIMT progression commences at a different period of the life course might not translate into equivalent benefit. 39 Third, the UKB cohort is not fully representative of the general population. Participants are generally healthier, wealthier, and more health-conscious than nonparticipants, which may introduce selection and collider bias—particularly because both socio-behavioral variables and cognitive function are related to study enrollment and retention. Such biases can attenuate true associations and may lead to underestimation of relationships involving lifestyle behaviors. Therefore, findings related to lifestyle covariates (such as alcohol consumption and physical activity) in this study should be interpreted with appropriate caution.40,41
Conclusions
In conclusion, leveraging large-scale observational and genomic data of European ancestry, our study establishes a causal effect of cIMT on cognitive function acting partially through small vessel stroke. Our work finds no evidence of nonlinearity in this relationship, indicating that there is no safe level of cIMT for cognitive decline. These findings position cIMT as an actionable target for preserving cognitive health and preventing dementia.
Supplemental Material
sj-docx-2-jcb-10.1177_0271678X261415785 – Supplemental material for A linear-no-threshold association between carotid intima–media thickness and cognitive function: The mediating role of cerebrovascular disease
Supplemental material, sj-docx-2-jcb-10.1177_0271678X261415785 for A linear-no-threshold association between carotid intima–media thickness and cognitive function: The mediating role of cerebrovascular disease by Wenqiang Zhang, Lingli Qiu, Yunjie Liu, Yutong Wang, Mingshuang Tang, Lin Chen and Xia Jiang in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X261415785 – Supplemental material for A linear-no-threshold association between carotid intima–media thickness and cognitive function: The mediating role of cerebrovascular disease
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X261415785 for A linear-no-threshold association between carotid intima–media thickness and cognitive function: The mediating role of cerebrovascular disease by Wenqiang Zhang, Lingli Qiu, Yunjie Liu, Yutong Wang, Mingshuang Tang, Lin Chen and Xia Jiang in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgements
The UK Biobank analysis was conducted within the application 99713. We would like to thank all investigators who shared genome-wide summary statistics.
Author contributions
Conceptualization: WZ. Methodology: WZ and XJ. Investigation: WZ, LQ, YL, YW, MT, LC, and XJ. Visualization: WZ. Supervision: XJ. Writing—original draft: WZ. Writing—review and editing: WZ and XJ. WZ is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Science Fund for Creative Research Groups of Science and Technology Bureau of Sichuan Province (2024NSFTD0030), Jiangxi Province Key Laboratory of Molecular Medicine (2024SSY06231), the National Natural Science Foundation Incubation Project of the Second Affiliated Hospital of Nanchang University (2024YNFY12012), the Recruitment Program for Young Professionals of China, the Experimental Discipline Revitalization Plan of West China School of Public Health and West China Fourth Hospital (2023SY-03), the Promotion Plan for Basic Medical Sciences and the Development Plan for Cutting-Edge Disciplines, Sichuan University and other Projects from West China School of Public Health and West China Fourth Hospital, Sichuan University. The sponsors of this study had no role in study design, data collection, analysis, interpretation, writing of the report, or the decision for submission.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
The UK Biobank was granted by the National Health Service North West Multi-Centre Research Ethics Committee (11/NW/0382) and was conducted in accordance with the Declaration of Helsinki. All UK Biobank participants provided written informed consent. All GWAS summary statistics are publicly available, and the corresponding studies have obtained proper institutional review board approval. Participants of the GWAS summary statistics also provided informed consent.
Data availability statement
Supplemental material
Supplemental material for this article is available online.
