Abstract
It has been estimated that approximately two million neurons, sixteen billion synapses and twelve kilometers of axons are lost each minute following anterior large-vessel stroke. The level of collateral blood flow has become recognized as a primary determinant of the pace of this loss and an important factor in clinical decision-making. Many of the topics in this review cover recent developments that have not been reviewed elsewhere. These include that: the number and diameter of collaterals and collateral blood flow vary greatly in the brain and other tissues of healthy individuals; a large percentage of individuals are deficient in collaterals; the underlying mechanism arises primarily from naturally occurring polymorphisms in genes/genetic loci within the pathway that drives collateral formation during development; evidence indicates collateral abundance does not exhibit sexual dimorphism; and that collaterals—besides their function as endogenous bypass vessels—may have a physiological role in optimizing oxygen delivery. Animal and human studies in brain and other tissues, where available, are reviewed. Details of many of the studies are provided so that the strength of the findings and conclusions can be assessed without consulting the original literature. Key questions that remain unanswered and strategies to address them are also discussed.
Introduction
It has been estimated that, on average, approximately two million neurons, sixteen billion synapses and twelve kilometers of axons are lost each minute following anterior large-vessel stroke. 1 However, the pace of this loss is highly variable among patients for reasons that are beginning to come into focus. The major determinants of the severity of tissue injury after acute arterial obstruction are the sensitivity of the tissue type to hypoxia/ischemia, location and duration of the occluding thrombus, and the amount of blood flow provided by the native pre-existing collateral circulation supplying the territory at risk.1–17 Collaterals also provide alternative routes for perfusion in slowly developing steno-occlusive diseases. 18 They are present in brain, eg, the pial or leptomeningeal collaterals of the neocortex, and in most other tissues. By cross-connecting a fraction of the distal-most arterioles in the crowns of adjacent arterial trees (0.01–0.001%10,11,19,20), watershed collaterals provide, for example in brain, perfusion to sustain the penumbra after acute ischemic stroke (AIS) and thus lessen infarct volume in proportion to their “extent” (number and average anatomic lumen diameter) when one of the trees becomes obstructed.11,19,21,22 In the absence of spontaneous or therapeutic recanalization, they become the sole source of perfusion. They also aid delivery of lytic agents to the occluding thrombus and neuroprotectant treatments to the ischemic tissue. 23
Unfortunately, pial collateral flow as assessed by collateral score on neuroimaging varies widely in patients following AIS, correlating inversely with early core size, infarct progression, risk of hemorrhagic transformation, risk of early neurological deterioration after thrombectomy, and directly with efficacy of thrombolysis and thrombectomy and functional outcome.1–6,10,12–17 For example, flow supplied by the pial collateral network in patients presenting with proximal middle cerebral artery occlusion (MCAO)—the most common cause of ischemic stroke—partitions into approximately 30% with poor, 45% with intermediate, and 25% with good collateral flow.1–6,10,12–17
The primary cause of this wide variation and collateral insufficiency in such a large fraction of individuals has been suggested, assuming mice predict humans, to arise primarily from genetic differences in the abundance of collateral blood vessels.19,22,24–27 For example, pial collateral number was recently found to vary by 47-fold (and diameter by 3-fold) among a panel of 60 inbred mice created to capture the naturally-occurring genetic diversity in the mouse species as a model of the outbred human population. 22 Collateral extent in other tissues of the same individual mirrors that present in brain, due to genetic coherence of collateral abundance among tissues.22,24–27 Hence, collateral flow also varies widely in the human heart and lower extremities,28,29 with major contribution to the severity of coronary and peripheral artery ischemic diseases. Variation of this magnitude in an anatomic phenotype is remarkable. It has been shown to arise from polymorphisms in genes within the pathway that drives collateral formation during development.22,26,30–32 This process, denoted “collaterogenesis”, involves a unique endothelial cell (EC)-led angiogenic mechanism that, in mouse, occurs late during gestation after the arterial and venous trees have formed.33–35 Maturation of the nascent collaterals then follows during the first few weeks after birth, wherein a fraction are pruned away and those that remain acquire their smooth muscle cell (SMC) medial layer and undergo lumen enlargement and wall thickening to yield the number and anatomic diameter that are present in the adult.33–39
Genetic factors are not the only determinant of collateral insufficiency. Studies in animals have found that aging or the presence of certain cardiovascular risk factors cause a pruning away of a fraction of collaterals and a decline in diameter of those that remain.20,35,39 This process, termed collateral rarefaction, is accompanied by more severe tissue injury in models of stroke and other ischemic diseases. Interestingly, aging-induced collateral rarefaction can be prevented by regular aerobic exercise or genetic or pharmacologic increase in eNOS/nitric oxide (NO) activity/bioavailability. 40 As well, acute arterial obstruction causes additional new collaterals to form, a process termed neo-collateral formation (NCF) to distinguish it from collateral formation during development.41,42 Occlusion also causes the native, pre-existing collaterals that are present to undergo remodeling, ie, to increase their lumen diameter and wall thickness over the subsequent days-to-weeks.11,19,24,35–38,42–46 Prolonged exposure to reduced inspired oxygen also induces NCF and remodeling.42,47
While the above “environmental” factors contribute to variation in collateral extent, they have a much smaller effect than differences in genetic background. 35 The “collateral genes” responsible for the variation in the vigor of collaterogenesis are only beginning to be distinguished. Identifying them is important for the reasons mentioned above and also because knowledge of a patient’s collateral status has recently become recognized as an important factor in determining whether endovascular treatment will be of benefit.1–6,8–10,12–17,48 And because collateral insufficiency, itself, is a risk factor for stroke, myocardial infarction, and other obstructive conditions.
A number of recent reviews are available regarding collateral blood vessels, the collateral circulation,1–11,35,49–51 and non-genetic factors affecting collateral extent, ie, collateral rarefaction, remodeling, and new collateral formation in adulthood. 35 This review draws primarily on work done in the last fifteen years aimed at identifying the genetic factors responsible for differences in collateral formation and maturation during development and thus collateral extent in the adult. Understanding in this area is in its infancy and has not been reviewed previously. Particular attention is given to what is known regarding collaterals in brain, and where reports are available, other tissues since such comparisons strengthen conclusions. Recent evidence that collaterals may serve a physiological function in the absence of arterial obstruction is also reviewed. Findings from both animal and human studies are provided where extant, as are questions that remain outstanding. Details of many of the studies are included to aid in assessing the strength of the findings and conclusions. Additional areas for future investigation are also discussed.
Collateral blood flow and collateral extent vary widely in brain and other tissues
Early studies in humans observed a surprisingly large amount of variation in collateral blood flow. Bang et al 52 and Christoforidis and coworkers 53 found that pial collateral score varied widely in patients with MCA-AIS. Wustmann et al54,55 reported that coronary collateral flow index (CFI = Paorta-Pwedge pressure/Paorta-Pcentral venous pressure) differed by 10-fold in healthy individuals with no evidence of coronary artery or other cardiac disease. They subsequently found that CFI in the lower extremities also evidenced significant variation. 29 The underlying basis for these findings was unclear. Data from earlier studies in mice suggested that genetic-dependent variation in collateral extent might be involved, but were not connected to those above in humans: Infarct volume after permanent unilateral distal M1-MCAO (pMCAO) differed substantially between two commonly studied strains of inbred mice, C57BL/6 (B6) and BALB/c,56–59 as did recovery of hindlimb perfusion after femoral artery ligation (FAL).60,61 Subsequent findings led the authors to suggest that BALB/c mice may have fewer native (pre-existing) collaterals in their lower extremities. 62 However, limitations in angiographic resolution, possible differences in collateral lumen diameter at baseline (not measured), collateral remodeling, and ischemic-induced capillary angiogenesis, left this possibility uncertain.
Chalothorn, Zhang et al19,43 subsequently developed methods to measure collateral number and lumen diameter with high-fidelity using casting of the arterial vasculature after pharmacologic-induced maximal SMC relaxation followed by fixation at 100 mmHg pressure—the latter because SMCs have low sensitivity to hypoxia and can undergo contraction during angiographic casting. (See Refs 22 and 42 for methodological details, including measurement of diameter based on pial collateral lumen area. 22 ) They then found19,24,25,30 that the number and diameter of native collaterals in B6 and BALB/c mice differed widely in brain and intestine, as did collateral blood flow in their hindlimbs (quantification of collaterals in the 3-dimensional arterial trees of hindlimb skeletal muscle was, and remains, a challenge; however supporting data was obtained for the 2-dimensional abdominal wall 20 and spinotrapezius 63 muscles). For example, the number of MCA-to-ACA collaterals and their diameters differed by ∼10-fold and ∼2-fold, respectively19,24,25,30 (cortex size and MCA and ACA tree territories were identical 19 ). Tissue injury in brain and hindlimb after arterial occlusion followed accordingly, as did penumbra volume in brain. 21 Collateral extent also differed widely among 13 other “classical” strains, with infarct volume after pMCAO closely following. 19 Interestingly, approximately two thirds of the 15 strains had abundant large-diameter collaterals, while the remaining had lower numbers of smaller-diameter collaterals. These findings showed that native collateral extent in mice varies substantially with genetic background and is a major contributor to the variation in tissue injury following arterial obstruction. The investigators suggested that the same may be true in humans. Recently, Faber et al 22 strengthened this conclusion by examining a panel of 60 mouse strains designed to capture greater than 90 percent of the genetic diversity in the mouse species, as compared to the above 15 classical strains which share a high degree, approximately 40–70 percent, of genetic relatedness.see 22 for Refs. The 60-set panel thus better models the “outbred” human species. Collateral number and diameter varied by 47- and 3-fold, respectively (Figure 1), and infarct volume followed accordingly. The distribution of strains with zero-to-poor (14%), poor-to-intermediate (25%), intermediate-to-good (47%), and good collaterals (13%) approximated CTA collateral scores obtained at admission in patients with AIS.1–6,13–17 For example, collaterals were absent in 7%, poor in 32%, moderate in 39% and good in 23% in 1988 patients with AIS-MCAO. 16 Given the many factors that affect resistance downstream from the collateral network (eg, SMC tone, edema, platelet/leukocyte adhesion, viscosity35,64,65) and thus collateral flow post-stroke, these findings suggest that genetic differences in collateral extent are the primary determinant in mice and, by inference, in humans. 35 The latter is consistent with the fact that the collateral network is the major series resistor that determines the pattern and time-course of contrast-filling of the downstream arterial tree on scoring collaterals in humans.
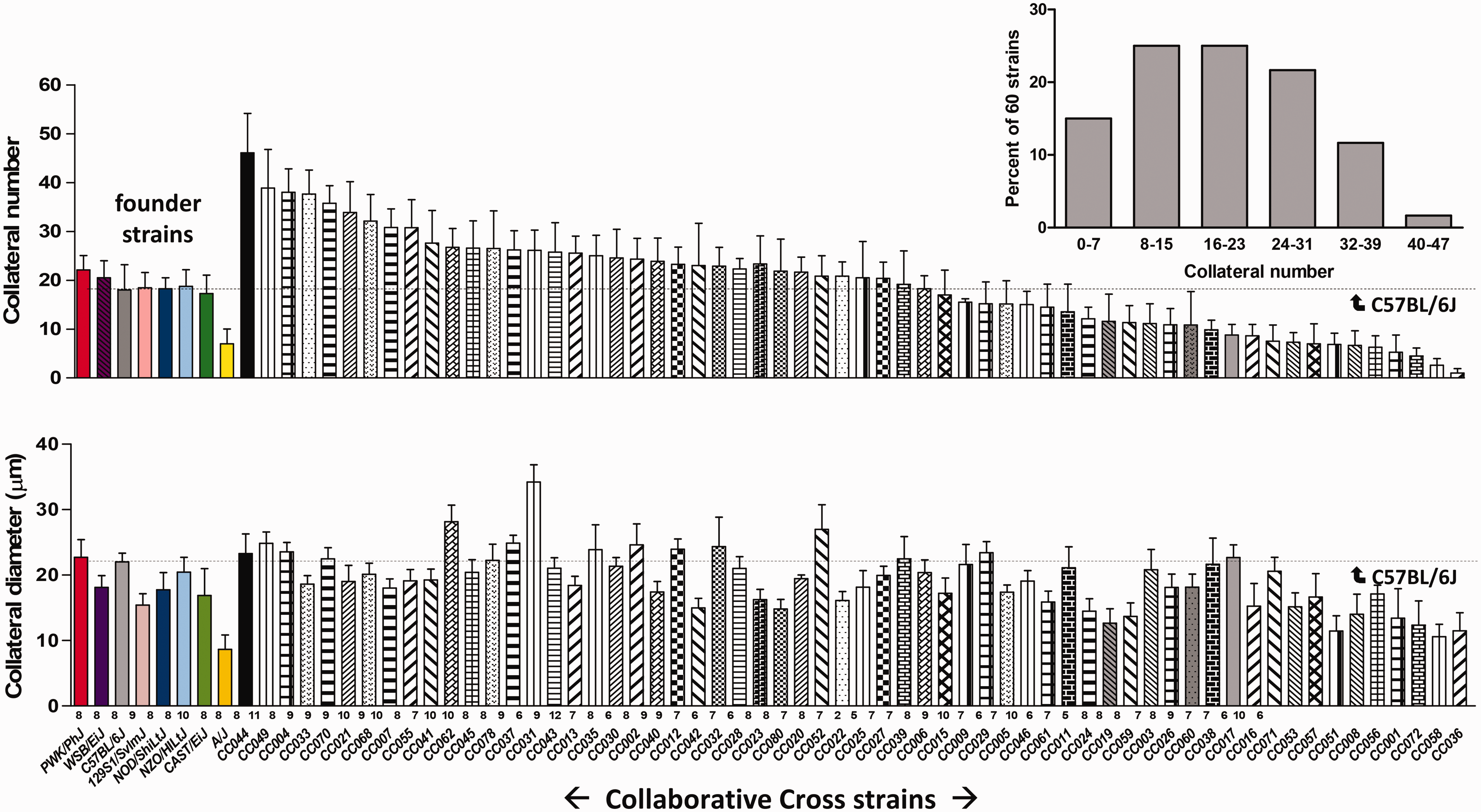
Number and diameter of MCA-to-ACA collaterals vary by 47.0- and 3.4-fold among 60 Collaborative Cross strains. Names of strains and number of animals per strain are given below each bar. Values are mean ± SD. Inset, Distribution of collateral number among the strains: 14% with poor (0–7), 25% poor-to-intermediate (8–16), 47% intermediate-to-good (17–31), and 13% with good collateral number (32–47). Modified from literature. 22
Identifying “collateral genes” that underlie collateral insufficiency
Current understanding of the signaling pathways that direct collaterogenesis and collateral maturation has come from studies that have used two different approaches. The first, which has been applied most often to explore biological processes, relies on methods to abolish, reduce, or increase, the activity of a potentially causal candidate molecule. Examples are administration of an agent that mimics or blocks its action, and use of transgenic, knockdown/knockout, or gene-editing methods. When the latter are employed to test a protein known to be involved in a similar or related process, the approach is referred to as a reverse genetic experimental design. The second, the forward genetic approach, entails conducting a genome-wide genotype-phenotype linkage analysis in an unbiased manner without a priori nomination of a candidate molecule. 22 Forward genetic studies thus often identify novel genes. The initial step is to identify a quantitative trait locus (QTL), ie, a genomic region harboring a naturally occurring sequence variation/mutation, most often a single nucleotide polymorphism (SNP, insertion/deletion, etc) within a gene or regulatory element, that causes loss-of-function (or much less often, gain-of-function) of a protein involved in determining the strength of the phenotype of interest. Such a “functional SNP/variant” is usually one that changes an amino acid, resulting in a change in the protein’s activity. A finding of statistical linkage of the SNP’s presence to the strength of the phenotype suggests the protein is the cause or a contributor to the variation in the trait. Performing such “QTL mapping” requires that a wide range of variation in the phenotype has been identified in a cohort of genetically distinct individuals. The latter has traditionally been obtained by creating an “F2 population” of several hundred individuals through brother-sister mating of the offspring (the “F1 population”) of breeder pairs of two parental strains that, themselves, display a large difference in the phenotype. Brother-sister mating shuffles the strains’ genomes, resulting in an allele-dose-dependent difference in the phenotype among the F2 individuals (ie, aa, ab, bb, where a,b equal the parents’ nucleotide type at the functional SNP), if the allele effect follows simple additive genetics for the causal gene’s protein activity or level of expression. Genotyping is done for each F2 individual at several hundred “marker” SNPs, ie, SNPs ∼equally spaced across each chromosome that mark the region (haplotype block) of DNA as coming from one or the other parent after crossover of their chromosomes during meiosis. The allele status of the marker SNPs is then tested in each F2 individual for statistical association, usually expressed as the log of the odds (LOD) ratio, with the strength of the phenotype. A significant association at the locus, which is then denoted a QTL, indicates that the causal gene/genetic element is contained within the DNA block that segregated during crossover. Scanning is subsequently done of the gene sequences present within a pre-defined interval (eg, ±5–10 megabases) surrounding the position along the chromosome having the highest statistical linkage (the QTL peak) to identify the presence of predicted loss-of-function SNPs in the genes of the parental strain with the low-value phenotype and establish a list of candidate genes. The latter are identified using in silico prediction algorithms that estimate the likelihood that the SNP will cause a damaging change in the protein’s structure or create a transcription stop codon. The candidate genes are then ranked according to strength of the loss-of-function predictions and other criteria (see Ref 22) followed by subjecting the top candidates genes to deletion, editing, or other methodology to identify the causal gene/genetic element(s) at the identified QTL.
Reverse genetics studies
Reverse-genetic studies have identified that several well-known signaling proteins in developmental, physiological, and pathological angiogenesis are also involved in collaterogenesis and/or collateral maturation (Table 1). Chalothorn and colleagues 24 conducted an in silico expression-quantitative trait locus analysis of B6 and BALB/c mice with abundant and sparse collaterals, respectively, that suggested involvement of the pathway: hypoxia→Hif1α [Shh, AP2] (factors in brackets denote interaction with the aforementioned factor/s)→VEGF-A→Flk1 (aka, VEGF-Receptor2)[Shp2]→Angpt1→EphrinB2→collateral formation, and that the activity of these signals may differ between the two strains (Figure 2); reduced Vegfa expression was confirmed in BALB/c (notation for a gene or its mRNA is given in italics: first letter capitalized for non-human, all-caps for human; non-italic notation denotes the protein). 24
Studies in mice demonstrating that genes known to be involved in vascular development are also determinants of collateral formation and maturation, and thus the extent/abundance of collaterals in the adult (Figure 4).
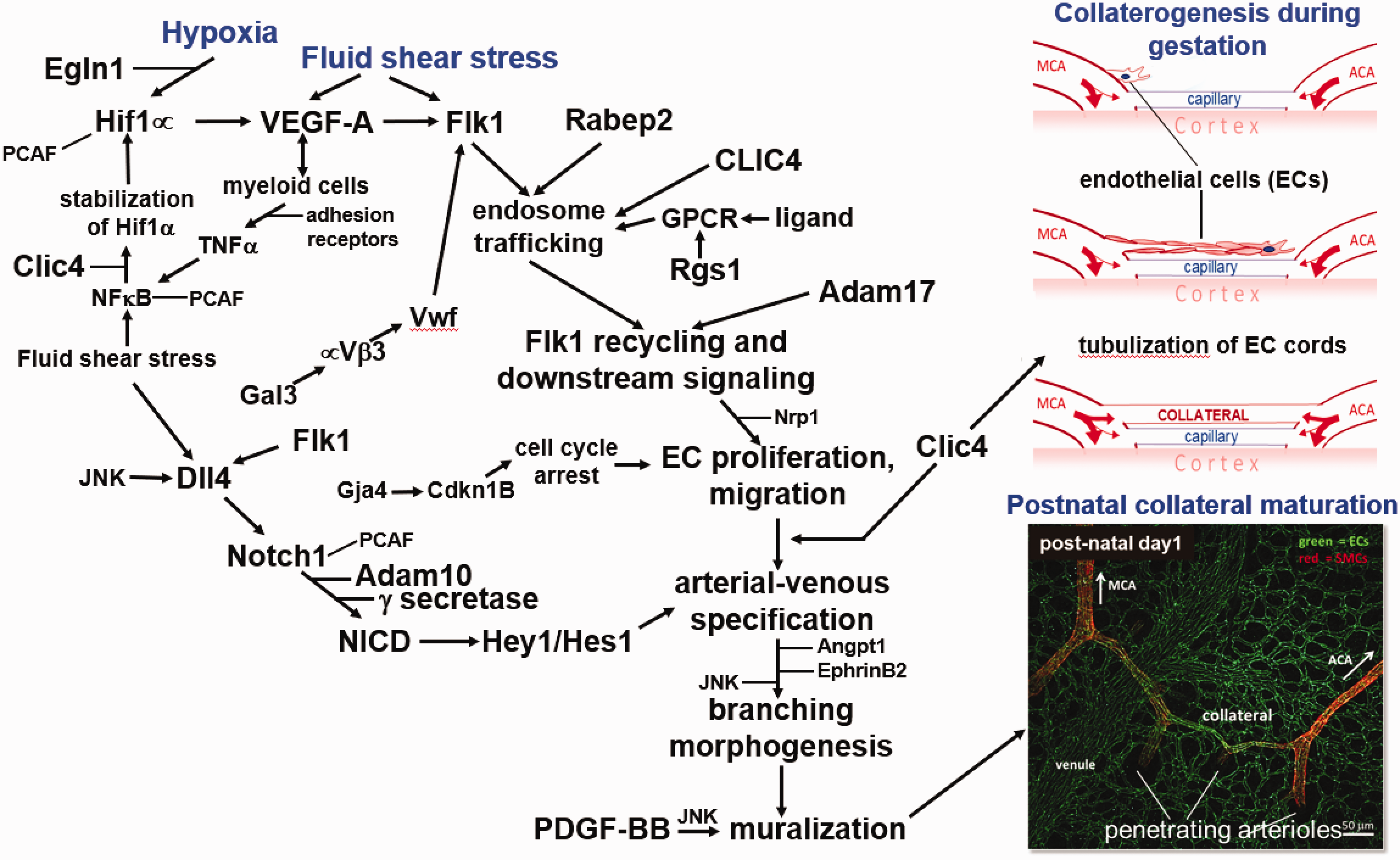
Proposed signaling mechanisms that direct collaterogenesis. See text for supporting studies. Inset, nascent collateral undergoing investment with smooth muscle cells (SMCs, red) is seen overlying a venule and capillaries in the pia and two penetrating arterioles (ECs, green).
Following on this work, Clayton et al 36 tested whether the prototypic angiogenic growth factor, VEGF-A, is required for collaterogenesis. Pial collaterals were quantified in postnatal day-1 (P1), P21 and 3–4 months-old (young-adult) gene-targeted mutant mice (CD1 strain) expressing reduced (Vegfalo/+), normal (Vegfa+/+; wildtype) and increased (Vegfahi/+) VEGF-A. Mice with homozygous alleles or with global knockout of Vegfa (ie, non-cell-type specific germline gene deletion) could not be examined because they die early in gestation from failure to form a functional vasculature. The above strains displayed allele-dose-dependent increases in collateral number at the time-points examined and an inverse relationship for collateral number versus 24 h-infarct volume post-pMCAO (r2 = 0.98; 50% decrease in infarct volume in Vegfahi/+ mice). Similar results were obtained for hindlimb collaterals, limb use, and tissue injury score after FAL. Postnatal maturation (ie, pruning away of a portion of the nascent collaterals) was accentuated in Vegfalo/+ and prevented in Vegfahi/+ mice. Additional pruning after P21 occurred in the Vegfalo/+ mice. Collateral remodeling (increase in anatomic lumen diameter and wall thickness), which reaches maximum 21–28 days after FAL (pial collateral remodeling after pMCAO was not assessed), was reduced by 40% in the Vegfalo/+ mice. These findings demonstrate that VEGF-A—which is a critical factor in developmental angiogenesis, tumor angiogenesis, and adaptive and pathological increases in capillary density in the adult—is required for collaterogenesis, collateral maturation, and the remodeling of collaterals that follows arterial obstruction. The results also show that the level of VEGF-A and several other factors in the collaterogenesis pathway (discussed below), which are determinants of the number and lumen diameter of pial collaterals in the adult, are also determinants of collaterals in other tissues of the same individual. Thus the collateral network of the mouse neocortex, which is readily accessible for quantification and tissue sampling because its entirety is arranged in two-dimensions on the surface of the brain, provides an excellent model system for studying collateral vascular biology, compared to other tissues such as the heart and lower extremities whose collaterals are arranged in three dimensions usually deep in the tissues.
Lucitti et al 34 extended these findings (Table 1). The Vegfa mutant mice described above evidenced allele-dose-dependent collateral formation during embryonic day-(E) 14.5–18.5—the time during which pial collaterals form in the mouse.33,34 The relationship correlated with the rate of EC proliferation in the watershed zone between the MCA and ACA trees and the level of expression by E16.5 brain ECs of Flk1, Flt1, Adam17, Adam10, Nrp1, Dll4, Hey1 and Hes1 (transcription factors, Hey1 and Hes1, are downstream of Notch1). The results suggested that the pathway: hypoxia→Egln1 (known to link hypoxia to Hif1α)→Hif1α→VEGF-A→Flk1 [Adam17,Nrp1]→Dll4→Notch1[ADAM10]→NICD (Notch Intracellular Cleavage Domain product whose concentration depends on ADAM10 and γ−secretase)→Hey1, Hes1—a pathway that is well-known to be involved in developmental angiogenesis—is also involved in collaterogenesis (Figure 2). Consistent with this, subsequent global knockdown of Vegfa or Flk1 during E13.5–16.5 in B6 mice decreased collateral formation. Endothelial-specific conditional heterozygous deletion of Adam17 and Adam10 during the same period decreased and increased collaterogenesis, respectively. Adam17 is known to positively regulate intracellular trafficking of Flk1, and Adam10 cleaves Notch1, liberating it for further cleavage by γ-secretase. Pharmacological inhibition of γ-secretase increased collateral formation, supporting that Notch1→γ-secretase→NICD→Hey1/Hes1 negatively regulates collaterogenesis. Deficient formation of collaterals by embryonic reduction of VEGF-A and Flk1 did not undergo spontaneous correction during growth to adulthood. Deficient formation also did not associate with a difference in vascular branch-patterning, confirming a previous report. 33 The above results suggested that collateral number in the adult is determined by the vigor of the above pathway (and additional factors, see below) during a narrow time window late in gestation after the arterial and venous trees and capillary beds have become established. Cristofaro and colleagues 66 (Table 1) subsequently confirmed the above-suggested role of Dll4→Notch1 in collaterogenesis in mouse pia using Dll4+/−, Dll4 overexpressing (both on CD1 background), and Notch1 conditional knockout (B6 background) mice examined at P9 and E13.5–14.5, respectively. Supportive results were also obtained in Dll4+/− adult hindlimb. The authors also observed postnatal pruning of pial collaterals in dll4+/− mice, indicating that Dll4 opposes this aspect of maturation of the collateral network.
Vegfa expression was lower in BALB/c versus B6 mice, in association with a SNP at or near the Vegfa locus,24,33,34 suggesting that a cis-acting polymorphism in BALB/c contributed to the above deficiencies. Fewer second-order branches of the MCA were observed, which is consistent with earlier evidence that VEGF-A level is a determinant of branching morphogenesis during development.67–69
Chloride intracellular channel 4 (Clic4) is involved in EC cell hollowing, proliferation, apoptosis, and capillary network formation, among other functions.see 37,38,70–73 Global deletion of Clic4 (CD1 mice) had fewer pial collaterals at P1 (ie, reduced collaterogenesis), greater pruning away of a portion of the nascent collaterals during the next 3 weeks, less growth in their diameter by P21, and thus fewer collaterals of smaller diameter in the adult; 37 as expected, CBF decreased more and infarct volume was greater following pMCAO. Similar results were obtained in hindlimb after FAL, including less increase in Hif1a, Vegfa, and Angpt2 in gastrocnemius muscle. Lucitti et al 38 confirmed these findings, with the exception that the reduction in collaterogenesis missed significance. However, different methods were used to identify perinatal collaterals in the two studies (ie, Ephrin-B2 expressing mutant mice 37 and Flk1-eNOS double staining 38 ). Overexpression of Vegfa reversed the excessive post-natal pruning and impaired maturation of diameter in Clic4−/− mice, restoring adult collateral number and diameter to that of wildtypes, and reversed the increase in infarct volume post-pMCAO. Clic4−/− mice had reduced expression of VEGF-C, Flk1, Flt1, N-cadherin, desmin and smooth muscle α-actin in pial watershed tissue. 38 The above findings suggest Clic4 exerts its action early in the collaterogenesis pathway by promoting VEGF-A signaling, EC proliferation, and Ephrin-B2-positive EC cords to undergo tubulation and vessel assembly (Figure 2). The authors also found that Clic4 positively regulates collateral maturation. Clic4 has been shown to interact with NFkB→Hif1α stabilization 72 and GPCR→endosome trafficking.73,74
c-Jun NH2-terminal kinase (JNK) is required for normal EC sprouting, branch-patterning, pruning, and vessel wall assembly during development.75,76 Ramo and coworkers 77 found that EC-specific deletion of all three isoforms in mouse led to reduced Dll4/Notch signaling, excessive EC sprouting, and chaotic branching (similar to Dll4+/− mice, see above) within the early vascular plexus in hindlimb and retina, and thereafter, abnormal arterial tree branching and failure to form collaterals in the hindlimb (retina lacks native collaterals 78 ). Thus, JNK is essential for normal vascular morphogenesis and collaterogenesis.
The gap junction protein, connexin-37 encoded by Gja4, is expressed in ECs and SMCs where it mediates intercellular communication during vascular development and remodeling.79–81 Fang and colleagues 82 found that mice with global knockout of Gja4 (B6 background) had increased collateral number. This suggests connexin-37 is involved in collaterogenesis and/or collateral pruning (evaluation in embryos and/or neonates is needed*, Table 1 ), since it is known that adult collateral number is determined by pre-natal collateral formation and early postnatal pruning.33,36–38 Also, fluid shear-stress activates a Notch→Gja4→Cdkn1B axis that promotes EC cycle arrest, leading to arterial-venous specification during embryonic vascular formation. 83 The above findings suggest Gja4 is downstream of Notch1 signaling in the collaterogenesis pathway.
Egl-9 family hypoxia-inducible factor 1 (prolyl hydroxylase 2, Phd2; encoded by Egln1) and its family members, Phd1 and Phd3, transduce reduced oxygen levels to increased Hif1α activity. 84 Phd2 is required for hypoxia-driven vascular network formation during development. 85 Takeda et al 86 concluded that Egln1−/+ mice (129/S6 background; homozygous knockout is embryonic lethal) have increased hindlimb collaterals. However, this was based on histology and micro-CT, neither of which can distinguish collaterals from arterial and venous vessels which were both increased in number and diameterr.** Table 1 Increased expression of Pdgfb, Sdf1, Tie2, Mmp2, and Nrp1 was observed in macrophages sorted from adductor muscle. Hindlimb perfusion and exercise performance were increased and tissue injury decreased 12 h after FAL. However, whether this reflects increased collateral number or diameter at baseline before ligation, as the authors concluded, or augmented remodeling of pre-existing collaterals and/or new collateral formation (NCF) afterwards (and/or expansion of the arterial and venous trees as noted above), was not determined. Findings supporting one or more of the above possibilities were reported by Lijkwan et al. 87 after shRNA knockdown of Phd2 in calf muscle of B6 mice (note, the primary collateral network supplying the hindlimb in mice resides in the thigh). And by Zhou and coworkers 88 in patients with congenital cyanotic heart disease, wherein those with a mutation in Phd2 had increased Rentrop-identified coronary collaterals and less incidence of cardiac ischemia. The mutant allele had no effect on baseline expression of Phd2 in a human cell line, but was associated with increased expression of Phd2, Hif1a, Vegfa and erythropoietin in response to hypoxia. Notwithstanding the above uncertainties** Table 1 , these findings, together with the known role of Phd2 in sensing hypoxia—and the fact that collateral status in hindlimb and other tissues parallels that in brain22,24–27,36,37,40—suggest that Phd2 is involved in collaterogenesis.
Tirziu and colleagues 89 linked the NFkB transcription factor family to the above findings of Takeda et al 86 vis-a-vis collaterogenesis. NFkB proteins are ubiquitous transcription factors involved in many processes including proliferation, differentiation, cell death, immune function and tissue development. 90 Knockdown of NFkB (knockouts are embryonic lethal) by EC-specific transgenic overexpression of IkBa, which targets NFkB proteins for degradation, increased pial collateral number in mice. 89 This was associated with reduced Hif1α, Dll4, NICD, VEGF-A and PDGF-BB in hindlimb muscle following FAL, and with formation of excessive arterial branching during vascular development as assessed in newborn retina. The latter was reversed after restoration of Notch1 signaling by treatment with its Dll4 co-ligand, Jagged-1. Genetically induced increase in NFkB activity in cultured ECs increased VEGF-A and PDGF-BB. The investigators thus proposed that NFkB activation of VEGF-A and PDGF-BB expression, combined with NFkB→Dll4→Notch1→induction of normal branch patterning (versus the excessive branching that occurs when Notch signaling is reduced, which leads to poorly perfused distal vessels) are within the collaterogenesis signaling pathway.
Von Willibrand Factor (VWF), which is synthesized and stored in Weibel-Palade bodies within the EC cytoplasm, is essential for primary hemostasis. VWF is also involved in blood vessel formation during development, possibly through interactions with αvβ3, Anpt2, and galectin-3 (release of Anpt2 and galectin-3 from Weibel-Palade bodies is regulated by VWF) that are known to be involved in VEGF-A signaling. 91 Consistent with this, global knockout of VWF decreased pial collateral number in B6-129S2 mice. 92 Supportive evidence was also obtained in hindlimb after FAL, including reduced remodeling of gracilis muscle collaterals and capillary angiogenesis in the ischemic gastrocnemius. These data show that while VWF’s modulatory role in vascular development is modest—since no severe developmental abnormalities were apparent with deficiency of VWF 92 —VWF is essential in the collaterogenesis pathway. This same reasoning applies to Clic4 and Gja4 as discussed above.
Vitamin D deficiency (VDD), which occurs with heritable mutations in the vitamin D receptor gene (Vdr) and other conditions, is associated with morphological and functional alterations of cerebral arteries and increased risk and severity of ischemic stroke. 93 The active form of vitamin D, 1,25-dihydroxyvitamin D, is involved in many processes including brain and cerebrovascular development. 94 Pial collateral number at baseline and CBF after unilateral carotid occlusion were reduced in B6 mice with Vdr rendered unable to bind DNA by targeted germ-line mutagenesis (VdrD/D). 93 This suggest Vdr is involved in collaterogenesis, with the following caveat:*** Table 1 Although VdrD/D breeders and offspring were fed supplemental calcium, phosphate, and lactose lifelong to normalize calcium homeostasis, MCA and ACA tree sizes were increased and decreased, respectively (brain weight was unaffected), suggesting alteration in developmental maturation of the cortex or its arterial trees, either of which could affect collaterogenesis. Also, collateral tortuosity, which increases with aging in association with a loss of collateral number and reduced diameter in those that remain (collateral rarefaction), 39 was increased at baseline in the 3 months-old young-adult VdrD/D mice. Given that rarefaction has been linked to age-associated reduction in NO bioavailability/EC dysfunction,20,39,95,96 and that the latter associates with VDD, 93 it is possible that the decreased collateral number reflects VDD-induced premature rarefaction rather than reduced collaterogenesis.
Bastiaansen and colleagues 97 found that B6 mice with deletion of the transcription coactivator, PCAF (p300/CBP-associated factor), had a modest 11% reduction in pial collateral number, with a similar trend (p = 0.07) in hindlimb. PCAF’s known actions to modulate Hif1α, Notch and NFkB-mediated expressionsee 97 suggests its involvement in the collaterogenesis pathway (Figure 2).
Hecht et al 98 found that intravenous injection of mouse embryonic progenitor cells (EPCs) into rats immediately, 7 and 14 days after ligation of the vertebral and left carotid arteries increased pial collateral number 24% (diameter was not increased), and increased cerebrovascular reserve capacity, diameters of the anterior circle of Willis arteries, and intracerebral capillary density. See literature 99 regarding potential involvement of several pathway elements in Figure 2 and supportive findings by others, including in the hindlimb ischemia model; and for a recent review of this area.
The above studies examining Gja4, Egln1, Nfkb, Vwf, Vdr, PCAF, and EPCs were conducted in adults. Since these proteins and EPCs have been shown in other studies to be involved in vascular formation during embryogenesis, the effects on collateral number most likely reflect their involvement in collaterogenesis. However, collateral status needs to be examined in embryos or from P1 through the first four weeks after birth when pruning and maturation occur to be certain the findings do not reflect alterations in the latter.
Forward genetics studies
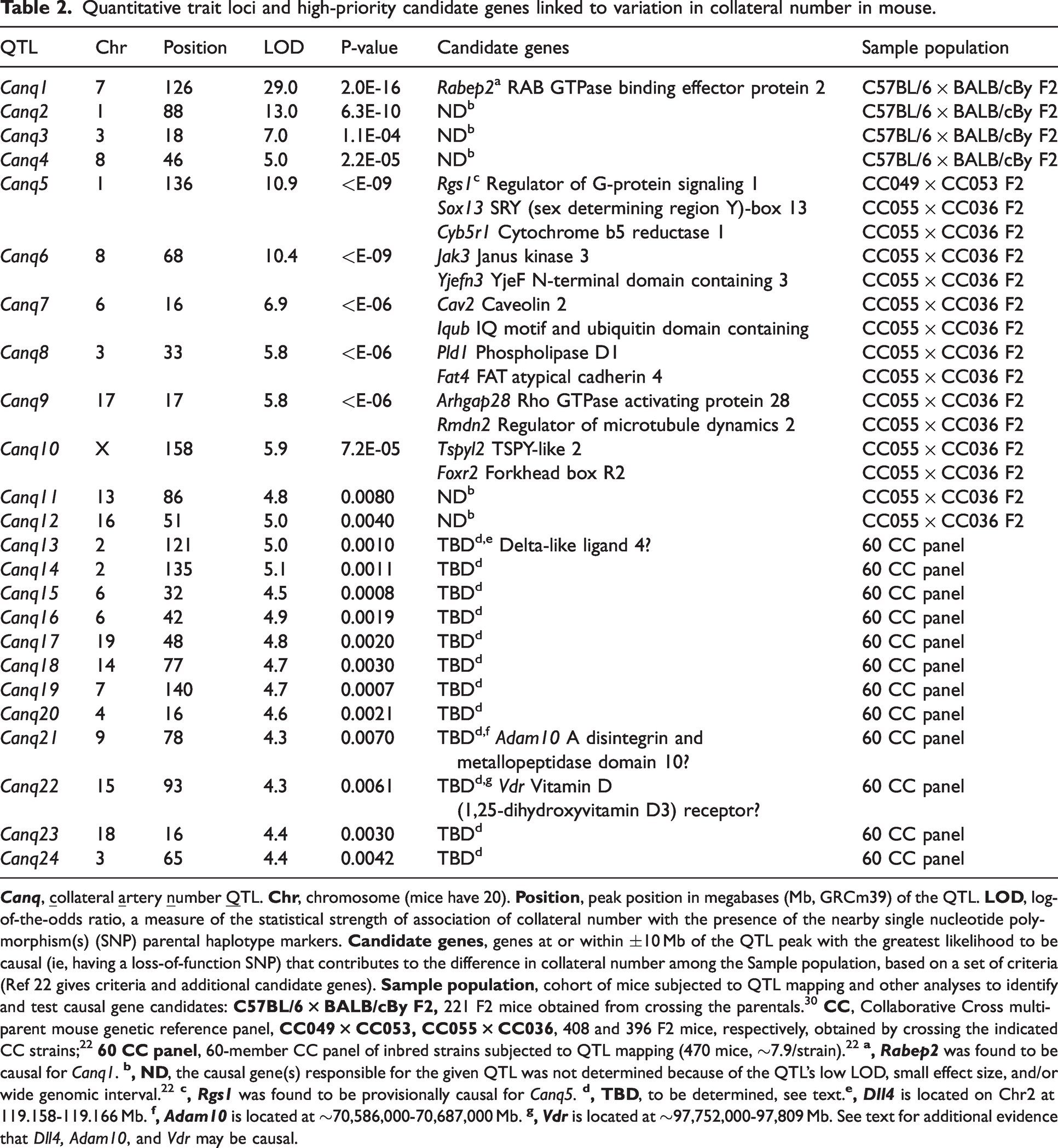
Reverse genetics studies (Table 1) indicate that proteins known to play primary roles in developmental, physiological, and pathological angiogenesis are also involved in collateral formation during development (barring the caveats mentioned for the last 6 proteins). The findings of Zhang and coworkers 19 permitted a second approach—unbiased forward genetic analysis—toward identifying signaling proteins within the collaterogenesis pathway. The number and lumen diameter of pial collaterals was found to vary widely, ∼30-fold and 3-fold, respectively, among 15 strains of mice that were selected from the ∼100 classic laboratory-inbred strains so as to capture the latters’ genetic diversity. This wide range in phenotype allowed Wang et al 30 to use QTL mapping and its accompanying downstream methods toward identifying responsible variant genes. They crossed the strain with the lowest number and smallest lumen diameter, BALB/cBy (averaging 0.5 MCA-ACA collaterals per hemisphere, 13 um diameter), with the B6 strain. B6 mice were chosen for their abundant (9 per hemisphere) large-diameter collaterals (23 um) and because B6 is the genetic reference strain for the Mus musculus mouse species. Importantly for comparing collaterals, both strains had the same body weight, areas of their neocortex and MCA, ACA and PCA tree territories, and average collateral length. 19 As well, small variation in these parameters among the 15 strains did not correlate with their differences in collateral number and diameter. 19 Wang and colleagues 30 reciprocally crossed BALB/cBy and B6 to create F1 breeders that yielded a cohort of 221 F2 mice whose collateral number and diameter ranged from 0–15 and 7–28 um. Four QTL were identified on chromosomes (Chr) 7, 1, 3 and 8, denoted Canq1-4 respectively, that linked to the variation in collateral number independent of sex or parent-of-origin (Table 2). The Canq1 locus, which contributed most to the difference in number, also associated with the variation in collateral diameter. Chromosome substitution analysis (ie, interchanging Chr7 between strains) and SNP association fine-mapping of an expanded number of strains (21) further confirmed the dominant contribution of Canq1.25,31
Quantitative trait loci and high-priority candidate genes linked to variation in collateral number in mouse.
Sealock and colleagues 26 used congenic fine-mapping to narrow the genomic interval underlying Canq1 to one (denoted Dce1) that accounted for ∼80% of the difference between the BALB/cBy and B6 strains in collateral number and diameter at baseline, and in blood flow and infarct volume after pMCAO. They also obtained similar findings in hindlimb at baseline and after FAL. Haplotype-SNP association analysis was then used to identify and prioritize a list of potentially causal genes. The top candidate, Rabep2 (Rab GTPase effector binding protein-2), was subsequently shown by Lucitti et al., 32 using additional congenic, knockout, and CRISPR methods, to be causal, conferring the same ∼80% difference in the above-identified endpoints. They also showed, as expected from previous work, 33 that the difference in collaterogenesis between the two strains during gestation was responsible for the differences present in the adult (ie, postnatal collateral maturation was comparable in the strains). The collateral-poor BALB/cBy mice were shown with CRISPR gene editing to harbor a loss-of-function SNP that changes an amino acid in Rabep2 to one that is predicted in silico to have a damaging effect on the protein’s putative binding domain. The genotypes at this same SNP (or at a different but nearby SNP in two of the six poor-collateral strains that was also predicted to be deleterious) were concordant with collateral number among the 21 strains previously phenotyped, except for one outlier for which several different predicted damaging SNPs were identified.31,32 These results strengthened the authors’ conclusion that naturally occurring variants of Rabep2 are a major determinant of collateral variation among B6, BALB/cBy and at least 19 other classical strains of mice. Lee et al 100 confirmed that conventional knockout of Rabep2 in B6 mice reduced pial collateral number by 66% and increased infarct volume 5-fold after pMCAO. They also showed that forced (vector-mediated) expression of either mouse (B6) or human Rabep2 in B6.Rabep2 knockout mice rescued their poor collateral and enlarged infarct volume phenotypes. Furthermore, forced expression of predicted damaging variants of RABEP2 present in the human population (albeit at low frequencies in certain genetic ancestries) led to reduced collateral number and increased infarct volume in B6 mice, demonstrating that deleterious variants of RABEP2 also exist in humans.
At the time of the above report by Lucitti et al 32 little was known about Rabep2 (which is still the case). It had been shown in vitro to interact, like its paralog, Rabep1 (but with much less effectiveness), with several other Rab family members involved in intracellular trafficking of early endosomes.see 32 Early endosomes transport membrane receptors such as Flk1/VEGF-Receptor2 to golgi vesicles, lysosomes, or back to the cell membrane, or couple them to downstream signaling proteins—such as for Flk1—to ERK1/2 and Akt that are known mediators of Flk1-induced angiogenesis. Accordingly, Lucitti and colleagues found in vivo that endosomal trafficking in endothelial cells within the pial watershed region was impaired in mice deficient in Rabep2 when examined late in embryogenesis when collaterogenesis was underway. These findings were confirmed and extended in cultured human ECs, wherein RABEP2 was found to regulate cell surface FLK1 abundance and downstream signaling, and when knocked down to reduce EC migration. 101 Rabep2 was also shown to be a substrate for glycogen synthase kinase-3 in heart, 102 and in an epithelial cell line to localize to endosomes as well as the microtubule organizing center and cilium basal body, wherein Rabep2 was found to be required for ciliogenesis. 103 The relevance of these findings to collaterogenesis awaits additional investigation.
Reduced collaterogenesis is not the only perturbation in Rabep2 knockout mice. Deletion altered branch patterning in the pial capillary plexus when examined late in gestation. 32 However, previous studies found that the difference in collaterogenesis in B6 versus BALB/cBy mice did not appear to be secondary to this difference. 33 Rabep2 status also had no effect on: development of the general arterial-venous vasculature, developmental angiogenesis in the neonatal retina; capillary and microvessel density in adult brain; tumor angiogenesis and growth; and in sprouting angiogenesis in vitro, leading to the conclusion that Rabep2 is required for collaterogenesis but not angiogenesis. 32 Rabep2 deletion did not affect collateral maturation on comparing collateral extent in late-gestation versus adulthood. 32 Lucitti et al 32 did not seek to identify the polymorphic genetic elements underlying Canq2-4 (Table 2) because of their small effect-sizes and wide genomic intervals identified in the B6xBALB/cBy F2 mice. 30
It has recently become apparent with completion of in-depth sequencing of the classical mouse strains that traditional QTL mapping of their F2 populations is limited by their high degree of genetic relatedness.see 22 That is, studies such as the above 30 have access to <40% of the naturally occurring polymorphisms predicted to be present in the mouse species, and thus do not optimally model the “outbred” human population. The Collaborative Cross mouse genetic reference panel (CC) was recently developed to address this limitation. 22 The CC was created by a decade-long mating effort undertaken by an international consortium that involved five carefully chosen genetically diverse classical strains and three strains derived from the three wild sub-species of Mus musculus: M.m.musculus, M.m.domesticus, and M.m.castaneous (the eight CC “founder” strains). It is well recognized that mice and humans share a high degree of genetic similarity and that fundamental biological pathways are conserved between them. Thus, it is likely that genes that harbor functional variants affecting collaterogenesis across the 60-strain panel of CC mice are also determinants of collateral variation in humans.
With this rationale, Faber and colleagues 22 conducted QTL mapping of the 60 CC strains and identified twelve peaks with LOD ≥4.3, denoted herein as Canq13-Canq24, that linked to differences in collateral number (Figure 3, Table 2). None of these “provisional QTL” reached the genome-wide p < 0.05 threshold (however each was individually significant at p < 0.008). This likely reflected that only 60 CC strains were available for study out of the at least 500 that the breeding scheme was designed to yield 22 (recall that four QTL at genome-wide p < 0.05 were identified on mapping the considerably larger 221 B6 × BALB/cBy F2 individuals in the above study 30 ). This arose from unexpected reproductive incompatibilities among the 8 founder strains. Several provisional QTL for diameter were also identified whose locations differed from those above for number. However, identifying their causal genes was not pursued because 1) of the small 3.4-fold trait range for diameter, 2) collateral number displays much greater genetic variation, and 3) collateral number has a greater influence than diameter on the distribution of collateral flow and infarct volume after MCAO. The finding that the above QTL peaks for collateral number and diameter do not co-localize is consistent with evidence that anatomic lumen diameter of arterial vessels is primarily determined by physical forces, ie, the prevailing levels of fluid shear stress and pressure-dependent circumferential wall stress.33,104,105
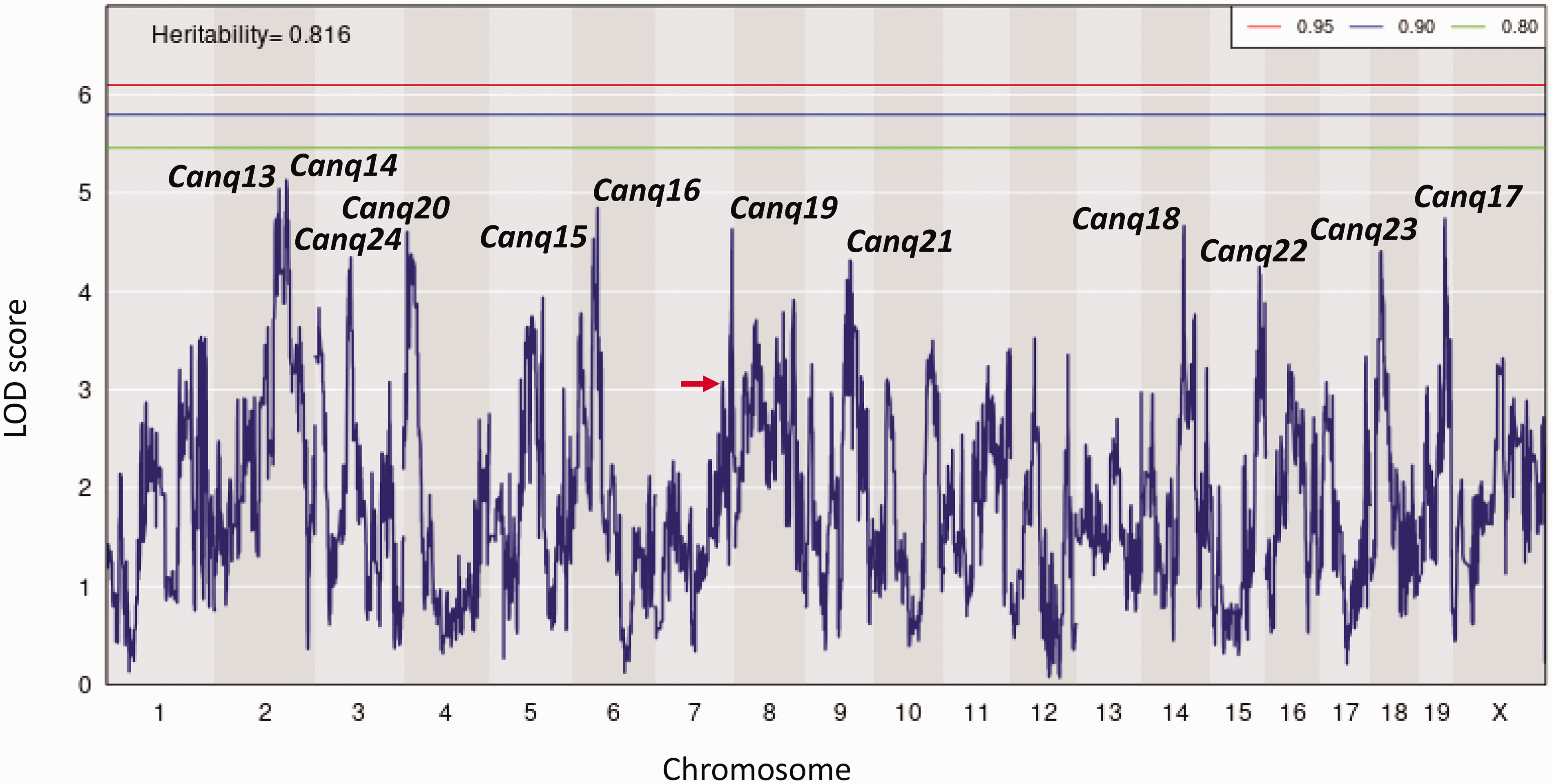
Collateral number is a highly polygenic trait. QTL mapping of collateral number in Collaborative Cross mice. Table 2 gives additional data for
None of the above provisional QTL for collateral number overlapped with the four QTL identified in the B6 × BALB/cBy F2 population (see QTL peak values in Table 2), or with the genes listed in Table 1 with the exception of the following: Dll4, which is located at 119 Mb on Chr2, is within the confidence interval of Canq13 whose peak is at 121 Mb; Adam10 (at 71 Mb on Chr9) and Vdr (at 98 Mb on Chr15) are within the confidence intervals of Canq21 (78 Mb peak) and Canq22 (93 Mb peak) respectively. The peak marking Canq1/Rabep2 was below the genome-wide significance levels (red arrow in Figure 3). This indicates that while variants of Rabep2 are major determinants of differences in collateral number and diameter among B6, BALB/cBy and at least 19 other closely related classical strains, 32 it is not a major determinant in the Mus musculus species per se, as represented by the genetically diverse 60 CC panel, and thus may also not be a major determinant in the general human population. This is not an unexpected finding, since Canq1/Rabep2 and Canq2-Canq4 described previously, 30 plus the twelve additional QTL identified above, make clear that collateral extent is a highly polygenic trait. In fact, no suspect SNPs within or near human RABEP2 have been identified in the extant GWAS of ischemic stroke. As well, several coding variants predicted to be damaging are present in RABEP2, albeit at low frequency in the general human population (https://gnomad.broadinstitute.org/). Interestingly, the four most common of these variants when placed in mouse Rabep2 in B6 mice each reduced collateral number and increased infarct volume. 100
Since Canq13-Canq24 did not reach genome-wide significance, the investigators 22 crossed twelve CC strains that have high- versus low-collateral number to simplify genetic structure and increase the sample size toward identifying additional QTL. The twelve strains were also chosen to have homologous loss-of-function alleles for Rabep2 in order to strengthen detection of additional QTL by preventing its re-identification. F1 progeny of the strains were phenotyped for collateral number to identify optimal parentals, which were then crossed to generate two F2 populations each of ∼400 individuals. Subsequent mapping identified eight QTL for collateral number, denoted Canq5-Canq12 (Table 2). A number of genes within ±10 Mb of Canq5-Canq10 were found to have in silico-predicted loss-of-function SNPs in the low-collateral strains. Table 2 lists their top-ranked (high-priority) candidate genes. The investigators also identified 335 predicted-deleterious SNPs present in their human orthologs. They then tested selected candidate genes underlying Canq5-10 with several assays, including an in vivo assay for collaterogenesis that they developed. However, supportive findings were only obtained for Rgs1 as possibly causal for Canq5. Canq11 and Canq12 were not studied because of their lower LOD values and poor differentiation from surrounding signal. The product of Rgs1, Regulator of G-protein signaling-1, is a GTPase-activating protein that limits Gα and Gβγ signaling from certain G protein-coupled receptors. Little is known about its possible role in vascular development. 22 Interestingly, Clic4 has recently been shown to interact with GPCR signaling in EC function. 106 Overall, the above study provided a set of candidate genes for future investigations in mice and humans aimed at identifying proteins within the collaterogenesis pathway whose variants potentially underlie genetic-dependent collateral insufficiency in brain and other tissues. The supplement to this review provides resources needed for future identification of the causal genes underlying Canq13-Canq24 (See Section ‘Genetics of collateral variation in humans’).
Among the genes listed in Table 1 (and the wider list of genes in Tables 1 and 2 in literature 22 ), the well-known “angiogenic” genes that are required for embryonic vascular development—Egln1, Vegfa, Flk1, Dll4, Notch1, Adam10, Adam17, RelA/NFkB—cause embryonic lethality if deleted, yet, with the possible exception of Dll4 and Adam10 (see above), are not within ±5–10 Mb of the QTL listed in Table 2. This indicates that the contributions of functional variants in these genes to the differences in collateral number between BALB/cBy and B6, the 60 CC, and the parental CC strains for which Canq5-Canq10 were identified, which vary by as much as 47-fold, are at most nominal, ie, variants in these angiogenic genes, if contributory, are below detection by QTL analysis. This may reflect compensations for, or redundancies in, the action of these proteins within the collaterogenesis pathway. In contrast, since they are required for development of the general arterial-venous vasculature during gestation, loss-of-function variants in them have predictably been selected against during evolution. None of the other 16 genes shown in Figure 2 are within ±10 Mb of Canq13-Canq24.
Other studies examining gene variants
Huang and colleagues 107 found that poor collateral score (determined within seven days post-stroke) and greater admission NIHSS associated with a putative damaging SNP in BIRC5 (OR 3.6, 1.7, respectively) among 428 patients with severe stenosis or occlusion of the MCA or common/internal carotid artery (ICA). BIRC5 encodes the anti-apoptosis and angiogenic factor, survivin. 108 These findings suggested that individuals with the low-collateral score SNP may be at risk for poor collateral flow and increased NIHSS post-stroke. Since the location of mouse Birc5 (Chr 11, 118 Mb) does not align with any of the QTL listed in Table 2, these findings suggest that survivin is a determinant of a factor(s) that affects collateral flow other than collateral extent (eg, resistance downstream of the collateral network, perfusion pressure, etc 35 ). The same uncertainty is inherent in all studies employing collateral scoring discussed below.
A genotype-phenotype relationship was also reported for VEGF-A and coronary artery disease. Individuals with the A allele of SNP rs699947 located within the promotor of VEGF-A expressed higher levels of VEGFA mRNA,109–111 and the variant associated with increased angiogenic responsiveness.112–115 Alidoosti et al 116 recently examined the allele in 195 patients having: stable/unstable angina or prior MI; ≥70% narrowing of at least 1 coronary artery; and no prior acute MI, revascularization surgery, anemia, or other conditions known to increase VEGF-A. Plasma VEGF-A did not differ between patients with poor (63% of the cohort) versus good (37%) Rentrop collaterals. However, the allele was present at higher frequency in those with good collaterals (P = 0.014; OR 1.96) when compared with the CC genotype. This finding may reflect VEGF-A’s involvement in collaterogenesis and collateral maturation (see above, Table 1, Figure 2), and/or collateral remodeling induced by occlusion,34,36 and/or hypoxia-induced NCF.42,47 Or in addition to/alternative to these possibilities, VEGF-A’s vasodilator and endothelial permeability-enhancing activities could affect determinants of collateral flow that are independent of anatomic collaterals. 35 The above reports107,116 are noteworthy because they associate a genetic variant with collateral score in humans.
Studies examining gene expression
Several studies have tested association of collateral score with gene expression in patients with AIS. Qin, Tian and colleagues117,118 found that IGF2, LYVE1 (both involved in angiogenesis) and FSAP (thrombosis) negatively, and THBS1 (angiogenesis) positively, associated with good collaterals and outcome at 90 days in blood collected from patients within seven days after M1 ± distal ICA occlusion who did not receive thrombolysis or thrombectomy. Since collateral score and blood were not obtained on admission, it is unclear whether the associations reflected: an inherent difference in expression in patients with poor versus good collaterals—ie, that the gene might be a modulator of collaterogenesis and collateral extent pre-stroke—versus that a signal(s) from the ischemic tissue affected expression of a factor(s) that determines collateral flow independent of collateral extent. 35 Similar uncertainties also arise regarding the finding that plasma levels of a long non-coding RNA (LncRNA) associated with Rentrop-scored collaterals in patients with coronary artery disease (CAD). 119 Likewise, patients with CAD, angina, and no MI in the previous 48 hours had higher serum VEGF-A and VEGFA mRNA in blood mononuclear cells whose levels associated with good Rentrop coronary collaterals and preserved ejection fraction. 120 However, it is not possible to know if these findings reflect an inherent genetic difference between the two groups or whether those with increased VEGF-A and good collaterals have more severe coronary obstruction—which itself increases circulating VEGF-A and promotes greater remodeling.24,36,47
In contrast to the above studies,117,118 Wu et al121,122 obtained plasma and collateral score on admission of patients with proximal M1-MCA ± distal ICA stroke. Expression profiling was conducted for levels of LncRNAs that were altered after AIS. 121 Among twenty-three patients examined, ENST00000422956, which was provisionally found to target Pax8 (identified, thus far, to be a thyroid-specific transcription factor), correlated positively with collateral status. They also examined RGMa, which had been reported to negatively affect angiogenesis post-stroke. 122 Levels of mRNA were higher in patients with poor versus good collaterals and healthy controls (which were comparable) and associated negatively with baseline admission NIHSS. Since plasma and collateral score were obtained on admission in both studies, the findings—if they reflect genetic differences in expression pre-stroke—raise the possibility that ENST00000422956 and RGMa may be modulators of an individual’s genetic background-dependent collateral extent or determinants of downstream resistance, perfusion pressure, or other determinants of collateral flow. 35
Collaterals and sex
Premenopausal women and ovary-intact young-adult female rodents sustain smaller infarctions than age-matched males, an effect partly attributed to the neuroprotective, anti-apoptotic, vasodilatory, and anti-inflammatory effects of estrogen. 123 Greater collateral extent in females rodents is not an additional contributor: Pial collateral extent was the same in females and males in 221 B6 × BALB/cBy F2 mice with 15- and 4-fold range in collateral number and diameter and equal numbers of both sexes. 30 The investigators 123 also found no sexual dimorphism for pial collateral extent, PCom number or diameter, or diameter of primary cerebral arteries in several strains of mice, nor for the decline in pial collateral number and/or diameter (collateral rarefaction, see Introduction and literature 35 ) caused by aging, obesity, or hypertension, nor for collateral remodeling 35 after pMCAO. However, rarefaction was greater in females with long-standing hypertension, which would favor greater infarct volumes. Li and coworkers 124 confirmed and extended these findings in rats. No sex-dependent differences were found in pial collateral extent, diameter of arterial segments of the circle of Willis (CoW), or CBF in the territory at risk during MCAO and reperfusion. Responsiveness of in vitro isolated pial collaterals to myogenic, vasodilator, and vasoconstrictor stimuli also did not differ with sex. As an aside to the above findings, the causative allele-dosage of the Canq10 QTL on Chr × (Table 2), whose estimated contribution (10%) to the difference in number of MCA-ACA collaterals (15-fold) between the parental strains was lowest among the seven QTL identified, 22 is expected to be the same in both sexes due to X-chromosome inactivation and non-recessive inheritance.
Recent studies have investigated whether collateral score varies with sex in patients with AIS; because scores were obtained on admission within at most 24 h since last known well, they would be affected little by remodeling of pre-existing collaterals and not at all by formation of new ones which requires more than 24 h after occlusion to occur ( 42 ; Zhang, Faber, unpublished): In 182 patients (median age 70 y) that presented with proximal M1 ± distal ICA-AIS within 16 h (52% female, 3 y older than males), Dula et al 125 found that women had better collateral scores (hypoperfusion intensity ratio, p = 0.006) and smaller core and penumbral volumes on admission, followed by slower core progression. Fifi et al 126 reported that women also had better collaterals (hypoperfusion intensity ratio, p < 0.001) in 285 patients (49% female; 69 y versus 65 y for males) that presented with anterior circulation LVO-AIS within 24 h. Wiegers and coworkers 16 examined 1988 patients (45% female; 69 y) that presented with anterior LVO-AIS within 6 h and found that women had better CTA collaterals (0–3 scoring scale; p < 0.001), as did Chalos et al 127 (p < 0.001) in 1762 patients (47% female, 70 y versus 66 y for males). In contrast, Rebchuk and coworkers 128 reported no difference in CTA collateral score with sex in 575 patients (49% female, 70 y) that presented with anterior LVO-AIS within 12 h, nor did Nannoni et al 129 in 857 patients (48% female, 72 y) presenting with M1-LVO within 24 h.
As discussed earlier, collateral score reflects collateral-dependent filling/“flow” within the branches and trunk of the occluded arterial tree downstream of the collaterals. The latters’ anatomic diameter at baseline average ∼25 ums in mouse and ∼60 um in human and are below the resolution of CTA or MRA. Collateral score is therefore unable to distinguish differences in collateral extent from other factors 35 that affect collateral flow. If the above findings in rodents that collateral extent is not greater in females are applicable to humans, then the aforementioned reports’ finding that women more frequently have better collateral flow during the subacute phase of stroke suggests that factors other than anatomic collaterals 35 are responsible.
Variation in the posterior communicating collateral arteries of the circle of Willis (PComs) and other collateral arteries: Genetic, stochastic, and environmental contributions
The above discussion addressed genetic-dependent variation in watershed collaterals. A second type of collateral exists, ie, collateral arteries, which are anastomoses found in specific locations, eg, the superior ulnar and genicular collateral arteries around the elbow and knee, palmar and plantar arch collaterals in the hand and foot, the anterior and posterior communicating/commissural collateral arteries (ACom and PCom “primary” collaterals) of the CoW, etc. 11 Unlike watershed collaterals, collateral arteries in healthy young adults generally exhibit minimal or no tortuosity at baseline and undergo much less remodeling on a percentage basis in response to a chronic increase in shear stress in obstructive disease.130,131 Different mechanisms underlie formation of collateral arteries and microvascular watershed collaterals: Based primarily on studies examining embryonic development of the CoW, which has been investigated in detail, the PCom and ACom collaterals arise by either retention of an artery(s) that is present in the early embryo or formation of a different collateral connection later in gestation that replaces the earlier one.see 130 Variation in their presence, diameter, and length (and “pattern” of the ACom) is evidenced in the adult by the wide range of embryonic (termed “remnant”) versus adult morphologies of the PComs and ACom on imaging of patients with cerebrovascular disorders and in cadavers free of such disorders. Since the variants are present at birth, they are attributed to “normal developmental variation”. However, the responsible mechanisms have not been examined. Understanding this is important not only to basic vascular biology but because deficiency in the CoW collaterals impacts treatment decisions and is a risk factor for stroke, stroke severity, recurrence, TIA, and subarachnoid hemorrhage.130,132 As well, Ospel and colleagues 133 reported that most fast-infarct progressors had lower pial collateral scores and higher frequencies of carotid-T or -L occlusions. This indicates, as predicted from hemodynamic considerations and suggested in recent reports,130,134,135 that PCom/ACom status can impact flow across the pial collateral network. Also, Westphal et al 136 found that “incomplete” PComs on MRA, which were present in 77% of 193 patients with M1-MCA AIS and 59% of 73 with TIA, tended to associate with higher mortality (p = 0.08).
The diameter and number of PComs (ie, whether present bilaterally, unilaterally, or absent) was reported in earlier studies to vary in several mouse strains, suggesting involvement of genetic factors.see 130 However, the findings were discordant. For example, PComs in B6 mice were described as “bilateral and well-developed”, “bilateral and hypoplastic”, or “highly variable in number”. Likewise, reports of PCom status in the two strains of rats that have been examined are also discordant. Such discrepancies may derive from the lack of perfusion fixation at maximal dilation, reliance on qualitative rather than quantitative assessment, and small sample sizes, given the below findings of high within-strain stochastic (random biological) variation.
A recent study 130 found that PCom anatomic lumen diameter (ie, fixation at pharmacologically induced maximal dilation and 100 mmHg pressure) varied widely among six strains, along with an unexpectedly high amount of stochastic variation in PCom number: cohorts of B6 and BALB/cBy mice each evidenced large percentages of individuals with absent, unilaterally, or bilaterally present PComs, whereas all individuals of the other four strains had bilateral PComs. Crossing 129S1/SvIm mice, which have large-diameter bilaterally present PComs, with B6 mice which have small diameter, variably present PComs, completely rescued the latter’s poor PCom status. This indicated that PCom extent varies widely in mice due to genetic and genetically-modulated stochastic factors. In contrast, extent of pial collaterals among the seven strains evidenced the expected small amount of within-strain variability typical of other anatomic vascular features. Deletion of Rabep2 had no effect on PCom extent, which indicated that at least one of the variant genes that contributes to variation in watershed collaterals differs from those that contribute to variation in PComs. This is consistent with evidence, mentioned above, that microvascular collaterals and collateral arteries form by different mechanisms.
The above investigators 130 found that variation in perinatal growth rate did not underly the stochastic variability in PCom extent. And that environmental factors—aging and hypertension—can also contribute to variation in PComs, causing a decrease in diameter, although with a smaller impact than genetic and stochastic factors. Obesity, hyperlipidemia, metabolic syndrome, diabetes mellitus, and intrauterine growth restriction had no effect. The investigators also showed that pMCAO-induced outward remodeling of PComs on both the ipsi- (more-so) and contralesional sides occurred and varied in magnitude with genetic background. This contrasts with the common assumption that flow in PComs, which is low or net-zero due to lack of a pressure difference in healthy individuals, is not recruited when the site of occlusion is distal to the CoW.see 130
The above findings additionally help to clarify sources of the large variation in infarct volume and other CBF-dependent endpoints that are seen in B6 and certain other strains subjected to models of ischemic stroke that induce flow across the PComs (eg, stenosis or ligation of the internal carotid artery 137 ), as well as hemorrhagic stroke. 132 They also suggest that genetic, stochastic, and environmental mechanisms may underlie the well-known variation in PComs seen in humans. The authors 130 proposed that differences in genetic and stochastic factors that modulate vascular patterning during development—and/or growth rate of the anterior versus posterior telencephalon—underlie whether the “fetal” versus “adult” PCom configuration is present in an individual. And that aging, hypertension, and possibly other cardiovascular risk factors can further affect PCom diameter. No studies have sought to identify SNPs associated with PCom variation in humans, although several have identified ethnicity-associated differences, and a single-case study found apparent familial linkage.see 130
Support for the above findings that collateral arteries (eg, PComs) display prominent genetic and stochastic variation, comes from several studies examining palmar arch and rete carpale palmare collateral arteries that inter-connect the radial and ulnar arteries in humans: 138 Bigler and colleagues 139 found that CFI, obtained on temporary occlusion of the radial and/or ulnar arteries in 250 patients undergoing coronary angiography, showed a bell-shaped distribution that ranged from no collateral conductance to such extensive conductance that occlusion caused little or no decline in pressure. Hollander et al 140 confirmed these findings in 50 individuals. Coronary CFI correlated with palmar CFI, supporting the coherence of collaterals among tissues that has been found in mice. 22,24–27,36,37,40 In a striking example of stochastic variation of collateral arteries, Cambron and coworkers reported robust palmar arch collateral circulation in the right but none in the left hand of a healthy young adult. 141
Genetic-dependent inner retinal artery tree-structure predicts collateral extent and stroke severity
Prabhakar and colleagues 78 recently reported the above finding. The authors’ rationale for the study derived from several considerations. First, knowledge of the status of watershed collaterals is clinically important. Second, since the average anatomic lumen diameter of native collaterals in humans (∼60 um) is below the resolution of imaging modalities, collateral score, digital subtraction angiography, and CFI are used to estimate relative blood flow across the collateral network in brain, heart, and other tissues. However, these are invasive procedures and have limitations: i) scoring pial collateral flow relies on advanced neuroimaging that is often not available (but see literature 10 ); ii) angiography has limited resolution (∼150 microns), and is complicated by the 3-dimension arrangement of arterial and venous trees in thick tissues; iii) determining CFI requires intra-arterial balloon occlusion; iv), all three assessments are affected by multiple downstream factors and perfusion pressure. 35 Thus, identification of a non-invasive biomarker for collateral extent would be of interest. Third, findings in mice suggested that genetic differences in pial collateral number may align with subtle qualitative differences in branch patterning of the MCA tree.33,34 Review of images of brain and other tissues from the investigators’ prior studies and those of others 63 supported this notion and the hypothesis that variant genes that cause differences in collateral extent may also cause differences in arterial tree patterning. And that branching geometrics obtained from a tissue amenable to non-invasive imaging might correlate with (predict) collateral extent in brain and other tissues (the latter since genetic variation in pial collaterals extends to variation in other tissues in the same individual, at least in mice21,24–27,36,37,40). Fourth, the inner retinal arterial circulation is the only vascular bed that is arrayed in two dimensions and amenable to non-invasive, high-fidelity imaging. Unfortunately, it lacks native collaterals because it is supplied by a single artery and is thus devoid of watershed regions. This precludes simply measuring collateral extent in the retina as a means to predict extent in other tissues. Fifth, heritability in several metrics of retinal artery tree structure has been identified in humans. 78 Sixth, formation and maturation of the retinal and cortical vasculatures share several anatomic features, as do topographic changes that occur with aging, cardiovascular diseases, and hereditary angiopathies.142,143 This suggests that certain metrics of retinal artery tree structure may align with anatomic features of the cerebral circulation, including pial collateral extent.
Given this rationale, Prabhakar and colleagues 78 measured collateral extent, infarct volume, and retinal artery tree patterning in ten strains of mice selected for their large differences in collateral extent. Twenty-one out of forty-six Euclidean and non-Euclidean metrics correlated with collateral number and diameter across the strains (p < 0.01). The most informative—fractal lacunarity, bifurcation angle, optimality, and six other metrics—predicted collateral number and diameter across seven multivariable regression models. The best model predicted (p < 0.0001) number (R2 = 0.91; R2 of 1.00 equals perfect correlation/prediction), diameter (0.81), and infarct volume post-pMCAO (0.86). These metrics, when extracted from images of the MCA tree, also predicted collateral number, diameter, and infarct volume (p < 0.0001). The authors advanced the hypothesis that certain variant genes important in the signaling pathways that control arterial tree formation/patterning and collaterogenesis are shared; however, they operate in parallel since genetic differences in tree patterning did not account for differences in collaterogenesis.33,34 Likewise, collateral-like connections between branches within a given arterial tree, denoted “intra-tree anastomoses” (ITAs), form at the same time during embryogenesis as watershed collaterals and exhibit the same genetic background dependence for their differences in abundance, yet the strain-dependent differences in the number that form also did not correlate with the internal branching structure of the trees.33,34
Khan et al 144 recently extended these findings. Retinal fundus images and CTA collateral scores were obtained on admission from 35 patients with acute MCA-AIS. Compared to those with good collaterals, poor-collateral patients had higher retinal vessel multifractals (0.01 < p < 0.04), lower NIHSS, and higher 90-day mRS. Derivation of a retinal vessel “classifier” metric that differentiated patients with poor versus good collaterals exhibited 0.74 sensitivity and 0.71 specificity. If these findings are confirmed in a larger cohort, geometric analysis of an individual’s retinal artery trees may provide a rapid, inexpensive, non-invasive biomarker to aid existing neuroimaging and hemodynamic methods for scoring collaterals toward patient stratification into clinical studies and choice of treatment options, and to aid analysis of retinal registries toward integrating stroke, coronary artery, and peripheral artery disease with twin, family-, and population-based genetic data (Figure 4). Determination of palmar collateral CFI (see above) could also provide a minimally invasive procedure for the above such efforts.

The genetic-dependent pattern of branching of an individual’s inner retinal arterial trees correlates with the individual’s pial collateral number, diameter, and infarct volume in mice 78 and with collateral score and NIHSS on admission in AIS patients 144 after middle cerebral artery (MCA) occlusion.
Collateral variation in other species
Maxwell and colleagues 145 found that collateral flow assessed using microsphere trapping within the dependent territory immediately after coronary artery ligation was, as a percentage of pre-occlusion orthograde flow: 100% in guinea pig, 16% in dog, 12% in cat, 6% in rat, 2% in ferret, baboon and rabbit, and 1% in pig, leading the authors to conclude that collaterals vary widely between species (a potential underlying mechanism (s) was not offered). There are uncertainties about this conclusion, including that findings for several of the species are discordant with other studies 27 and that individuals of each species were from closely-related outbred stock or interbred litters. Given the wide genetic variation in native collaterals among mouse strains19,22,31 and substantial variation in collateral flow among “outbred” humans (Sections ‘Variation in the posterior communicating collateral arteries of the circle of willis (PComs) and other collateral arteries: Genetic, stochastic, and environmental contributions’ to ‘Future questions’),1–8,10,12–16,28,29 the often-stated conclusion that the above findings reflect species-specific variation in native collaterals145—which is at variance with the implication of the aforementioned “between species” conclusion—requires comparing genetically diverse individuals or different isogenic strains of each species. Large within-species variation, if found, would refute the conclusion. In support, compared to Wistar rats, Long-Evans rats had 23% fewer pial collaterals (diameters were comparable), 2.7-fold larger infarct volume after pMCAO, and 38% lower hindlimb blood flow immediately after femoral artery ligation. 27 Future studies are needed to determine if large, genetic-dependent variation in collateral extent is a shared feature in all wild outbred mammalian species, with the exception of species that have evolved at high altitude (see next section).
High altitude species have abundant collaterals: physiological function of collaterals
Recent evidence 27 suggests species that have evolved at high altitude are an exception to the preceding paragraph’s concluding sentence. Based in part on findings suggesting that low tissue oxygen in watershed regions drives collateral formation during development33,34 and de novo formation of additional collaterals in the adult,41,42,47 together with the finding that guinea pigs—which evolved as a species at high altitude 27 —have exceptionally good collateral circulation in heart, 145 a recent study 27 compared collaterals in several strains of guinea pigs (Cavia) and high-altitude-evolved deer mice (Peromyscus) with lowlander Peromyscus conspecifics, Mus musculus mice, and rats (Rattus). Strains of guinea pigs and high-altitude Peromyscus were found to have greater pial collateral number than lowlanders, and evidenced, remarkably, complete and 80% protection against infarction after cerebral artery occlusion, respectively. They also sustained significantly less infarction in heart (including none in guinea pig) and hindlimb after arterial ligation, and had robust and complete CoW configurations. Interesting, the same Rabep2 variants that confer abundant collaterals among 21 mouse strains 32 are also present in guinea pig, respectively (positions of residues between the two species differ by 12): P200, P212; R298, R310; R426, R438. The authors proposed that species and specie-populations native to high-altitude have undergone selection for alleles within coding regions (eg, gain-of-function mutations) or regulatory regions of collaterogenesis pathway genes, which assures abundant collaterals as a fitness advantage for survival in their low-oxygen environment. The authors also hypothesized, based on their findings and other work (ie, neo-collateral formation induced by arterial occlusion or systemic hypoxemia41,42,47) that collaterals not only provide protection in obstructive disease but also serve to optimize oxygen delivery to meet oxygen, particularly when oxygen is limiting.27,35 In support, they cited evidence that polymorphisms in various human genes, including several in Figure 2—many whose expression is increased by reduced oxygen—associate with lower risk-severity of ischemic disease in high altitude human populations.27,42 Also, although not mentioned therein,27,42 the incidence of myocardial infarction and severe ischemic heart disease is unusually low in Bantu Black South Africans (BSAs) compared to White South Africans (WSAs) or Europeans,146–151 including autopsy findings that incident and occult infarction was 34-fold lower than in WSAs despite comparable CAD. Also, coronary collateral number in young and old healthy BSA cadavers was double that of WSAs and unrelated to cardiac mass or coronary artery tree size. These findings support the following hypothesis: The proto-Bantu originated ∼6,000 years ago in Nigeria and Cameroon 152 where low dietary iron and high maternal, newborn, and adult anemia from malarial and helminthic infections are prominent. These environmental conditions selected for hereditary hemaglobinapathies that, while providing a level of protection, are the major cause of the endemic anemia.153–156 While evidence that these conditions have selected for abundant collaterals has not been sought, including for example using single-gene association analyses (see below), Occam’s razor supports its investigation.
Future questions
Several areas for future investigation are highlighted below, in addition to those mentioned in the preceding sections.
Genetic mouse models of collateral variation
Analysis of Collaborative Cross (CC) mice demonstrated that collateral number and diameter are polygenic traits that vary ∼50 and ∼3-fold due to variant alleles in “collateral genes” that drive collaterogenesis during development (Section ‘Forward genetics studies’). 22 This striking variation is a primary determinant of the variation in blood flow to the at-risk territory post-stroke (rarefaction induced by aging and risk factor presence have a much smaller effect on collateral extent 35 ) and is likely shared by humans. This is supported by the fact that mechanisms that direct vascular development are highly conserved among vertebrates. The CC strain-set, plus the finding that a single gene, Rabep2, is responsible for ∼80% of the difference in collateral extent between the relatively closely related classical B6 and BALB/c (and BALB/cBy substrain) strains, 32 provide two experimental systems to model humans with good, intermediate, and poor collaterals: Comparing CC strains with abundant, intermediate, and sparse collaterals, for example, CC044 or CC049, CC027 or CC039, and CC036 or CC058 (Figure 1) has the advantage that genetic diversity is maximized, which better represents the “outbred” human population than classical mice. On the other hand, comparing B6, B6.Rabep2−/− and BALB/cBy, which are ∼70% genetically identical,see 22 has the advantage that genetic diversity is lessened (to even a single gene/SNP when comparing B6 and B6.Rabep2−/− mice), thus minimizing genetic variation in the other determinants of collateral flow 35 (see the Discussion in literature 22 for additional considerations in selecting between these two systems). Also, body weight, MCA, ACA and PCA tree size, collateral length, number of penetrating arterioles per collateral, and collateral remodeling after MCAO do not differ between B6 and BALB/cBy strains; 19 this is likely to be the case among many of the CC strains, although this would have to be confirmed among those selected for study. While use of these models is in its infancy in stroke research, new understanding has already begun to emerge.157–160
Measurement of collateral extent and collateral conductance in humans
Unlike in experimental animals, pial collateral extent cannot be quantified in humans because of limitations in imaging resolution. Instead, pial collaterals are inferred by scoring retrograde filling patterns and other indexes of collateral flow on obstruction.1–6,10,12–18,161–165 However, the ability of these measurements to infer collateral extent is confounded by differences in the many other factors that affect collateral flow.
35
It is possible that pial collateral number could be determined (for experimental purposes) using intrathecal/subarachnoid, contrast-enhanced, high-field strength MRI to identify regions of interest within the cortical grey matter watersheds, based on imaging their low oxygen signature,
166
and counting the number of perivascular (Virchow-Robins) spaces (PVSs) that surround the penetrating arterioles that branch from the collaterals (Figure 2; quantifying PVSs with higher versus lower oxygen would differentiate penetrating arterioles from penetrating venules). Assuming that each collateral has a finite number of penetrating arterioles that does not differ with genetics, as is the case in mouse,
19
an index of relative collateral number could be obtained, although the gyrencephalic human cortex presents an additional imaging challenge, compared to rodents. The known association of enlarged PVSs with aging and certain pathological states would also have to be factored in. If the necessary methods were found feasible, testing could be done in a large-animal model, with comparison to anatomic collaterals obtained by port-mortem angiography (and, for added value, to collateral score on conventional imaging). It is also possible that collateral extent could be quantified from prospectively collected human brains on necropsy and/or from extant cadaver brain repositories, wherein genetic material could also be obtained. There are a number of publications examining the extent of PCom and ACom collateral arteries in cadavers.see
130
Collateral score is generally confined to a several-point scale (dynamic rLMC employs a 20-point scale 163 ; see also literature 165 ). This limits testing for association between collaterals, infarct size, and other variables in humans, using SNP-association and other genetic approaches to identify putative collateral genes. It is possible that collateral flow index (CFI = Pabove occlusion-Pbelow occlusion/Pabove occlusion-Pjugular vein), which correlates closely with collateral flow in heart, lower extremity, and hand28,29,139,140 and, importantly, is a continuous variable, could be obtained in patients with MCA-AIS during evaluation for thrombectomy. Tahir and colleagues 167 recently compared CFI to four widely-accepted grading scales obtained on pretreatment CTA and DSA. CFI correlated best with CTA Maas score, and better than any of the grading scales with 30-day mRS. Next-generation pressure-reporting guide-wires are under development that could be employed 168 . Feasibility is further supported by the recent introduction of trans-clot micro-catheter blood sampling in LVO-AIS patients. 169
An intriguing question is why collaterals vary so greatly in lowlander rodents and humans. It has been proposed that this reflects the relaxation of stabilizing genetic selection for abundant collaterals in low-altitude populations, since variation in collateral extent may have little or no relevance to fitness under normoxemia and normal hemoglobin status (however, see literature 35 ); and that the opposite has occurred with prolonged reduction in oxygen availability.27,42,47 Interestingly, expression of many of the genes thus far identified in the collaterogenesis pathway (Figure 2) are increased by reduced oxygen.27,42,47
Regarding the proposed physiological role for collaterals (Section ‘High altitude species have abundant collaterals: physiological function of collaterals’ and literature 35 ), once candidate collateral genes identified in mice are confirmed in patients with stroke, myocardial infarction, or peripheral artery disease, their analysis could be extended to genetic association studies in humans with hereditary hemoglobinopathies, sickle cell disease, and populations that have resided at high-altitude for thousands of years and undergone genetic adaptation to reduced oxygen availability (eg, Tibetan, Andean and Ethiopian highlanders), employing collateral score, CFI, or retinal branch patterning. 35 Several studies have noted lower incidence of stroke and better outcomes in high-altitude populations.see 27,35,42
Genetics of collateral variation in humans
Future work is required to identify the candidate genes underlying the QTL, Canq12-24 (Table 2). Since these were derived from mapping the genetically diverse 60-strain CC panel, they mark the genes that contribute the most to variation in collateral number in the mouse species—and by inference in humans. Toward this end, the genetic mapping resources and dataset used to identify the above QTL are provided in the Supplement to this review that will be needed in future studies to identify the primary “collateral genes” underlying these QTL in mice, and in turn in humans, using the recently published in silico pipeline. 22 Doing so will greatly expand understanding of the collaterogenesis pathway elements (Figure 2), potential therapeutic targets, and may allow development of a blood- or saliva-based test to identify individuals with risk alleles for poor collaterals before stroke or other occlusive disease occurs. Additionally, such a biomarker could aid clinical decision-making and stratification of patients into studies. 170 Given that mice and humans share greater than 90% genetic identity,171,172 it is likely that human orthologs of the causal collateral genes underlying the QTL identified in CC mice (Table 2) will harbor polymorphisms responsible for the majority of the variation in collaterals in humans.
Candidate genes identified in mice can be tested for confirmation in humans using single-gene SNP association analysis, with its significant statistical advantage over GWAS, against collateral score, retinal branch patterning, or CFI in heart, lower extremities and possibly brain. For example, VEGAS gene-wise analysis of all SNPs (GRCh38) in RABEP232 and seven well known “angiogenic” genes as “controls” (VEGFA, KDR, EGLN1, CLIC4, ADAM10, ADAM17, Notch1) (Table 2) in 532 patients with ischemic stroke (GASROS cohort) found, after adjustment for age, sex, principal components and Bonferroni correction, that RABEP2 associated at nominal significance (p = 0.098) with DWI infarct volume (top SNP rs11645302, p = 0.004; rs35979293 p = 0.048 for differential effect in LVO versus non-LVO cases); control genes were 0.261<p < 0.823, respectively. 173 On replication analysis of 283 SNPs among 20 genes using a cohort of 1700 patients versus 7900 non-stroke controls from METASTROKE, two SNPs in RABEP2 had the lowest (p = 0.0004, rs35979293) and second lowest (p = 0.0012, rs4072401) nominal p-values for cardioembolic ischemic stroke versus non-stroke, with a RABEP2 gene-wise p = 0.0083. The top SNP in GASROS was in high-linkage disequilibrium with the two METASTROKE SNPs (D’ = 0.94EU, D’ = 1.0AF), and in GASROS, rs35979293 was associated with DWI volume (G/T, OR 0.2589, p = 0.0483). 173 Hence, RABEP2 and other genes that associate with ischemic stroke risk and infarct size, potentially by impacting collateral status, may be amenable to identification in humans. Mendelian randomization 174 and exome sequencing175 could also be employed.
Conclusions
Remarkable discoveries have been made in the last fifteen years regarding the development of thrombectomy; understanding the importance of collaterals in the efficacy of thrombectomy, thrombolysis, and patient outcomes; collateral variation as a clinical entity; and stroke genetics. A new area of investigation, collateral genetics, has also been introduced. Hopefully, this review will aid future researchers interested in continuing to advance understanding of this unique vascular network—the collateral circulation—in brain and other tissues.
Abbreviations
ACA: anterior cerebral artery; ACom: anterior communicating collateral artery; AIS: acute ischemic stroke; B6: C57BL/6 inbred mouse strain; CAD: coronary artery disease; CBF: cerebral blood flow; Chr: chromosome; CFI: collateral flow index; CoW: circle of Willis; E#: embryonic day-1, 2, etc; EC: endothelial cell; FAL: femoral artery ligation; FIO2: fraction of inspired oxygen; ICA: intracranial internal carotid artery; LOD: log of the odds ratio; LVO: large vessel occlusion (anterior cerebral circulation); MCA: middle cerebral artery; MCAO: middle cerebral artery occlusion; mRS: modified Rankin score; NCF: neo-collateral formation (formation of new additional collaterals in the adult); NIHSS: NIH stroke scale; OR: odds ratio; P#: Postnatal day-1, 2, etc; PCA: posterior cerebral artery; PCom: posterior communicating collateral artery; pMCAO: permanent middle cerebral artery occlusion; QTL: quantitative trait locus; SMC: smooth muscle cell; SNP: single nucleotide polymorphism
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X251317880 - Supplemental material for Genetic determinants of insufficiency of the collateral circulation
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X251317880 for Genetic determinants of insufficiency of the collateral circulation by James E Faber in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-xlsx-2-jcb-10.1177_0271678X251317880 - Supplemental material for Genetic determinants of insufficiency of the collateral circulation
Supplemental material, sj-xlsx-2-jcb-10.1177_0271678X251317880 for Genetic determinants of insufficiency of the collateral circulation by James E Faber in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NIH-NINDS R01 NS083633, NIH-NHLBI R01 HL111070.
Acknowledgements
The author gratefully acknowledges Hua Zhang, previous lab members, and collaborators.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
