Abstract
That cerebral blood flow remains unchanged at an increased blood viscosity, as long as the vascular supply is not compromised, was tested. To induce a reduced blood supply of some parts of the brain and to keep the supply unchanged in others both carotid arteries were occluded in anesthetized, ventilated rats. By this procedure, blood supply to the rostral brain, but not to the brainstem and cerebellum, was compromised. Blood viscosity was increased by intravenous infusion of 20% polyvinylpyrrolidone (high viscosity group) or decreased by infusion of 5% albumin (low viscosity group). Cerebral blood flow was measured by the [14C]iodoantipyrine method in 50 complete coronal sections of the rostral brain and 22 complete coronal sections of the brainstem and cerebellum in each rat. In the high viscosity group, mean cerebral blood flow of the rostral brain was significantly lower (46 ± 7 mL/100 g−1 · min−1) than in the low viscosity group (82 ± 18 mL/100 g−1 · min−1). No differences could be observed in brainstem and cerebellum between both groups (162 ± 29 mL/100 g−1 · min−1 vs. 156 ± 18 mL/100 g−1 · min−1). Local analysis of cerebral blood flow in different brain structures of the coronal sections showed the same identical results; i.e., in 29 of the 31 brain structures analyzed in rostral brain, local cerebral blood flow was lower in the high viscosity group, whereas no differences could be observed in the 11 brain structures analyzed in the brainstem and cerebellum. It is concluded that under normal conditions cerebral blood flow can be maintained at an increased blood viscosity by a compensatory vasodilation. When the capacity for vasodilation is exhausted by occlusion of supplying arteries, an increased blood viscosity results in a decrease of cerebral blood flow.
Keywords
Conflicting results have been reported that concern the effects of blood viscosity on cerebral blood flow (CBF). Some investigators have stated that changes in blood viscosity result in changes of CBF (Cole et al., 1994; Hurn et al., 1993; Massik et al., 1987; Muizelaar et al., 1986, 1992). Others have stated that changes in blood viscosity are counteracted by compensatory vascular responses in the cerebral microcirculation, resulting in a maintained CBF (Brown et al., 1985; Chen et al., 1989; Fan et al., 1980; Hudak et al., 1989; Jones et al, 1981; Krieter et al., 1995; Tomiyama et al., 1999). The latter studies are supported by findings from our group in which a more than threefold difference in blood viscosity of two groups of rats did not result in any difference of CBF when blood oxygen content was kept constant (Waschke et al., 1994). Whether these discrepant results and conclusions, which concern the effects of viscosity on CBF, may be based on differences in the experimental approach remains unclarified. It seems possible that under normal conditions an increased blood viscosity may be compensated for by a vasodilation, which results in an unchanged CBF. Conversely, CBF may be decreased by an increased viscosity in such cases in which the capacity of the vessels to dilate is exhausted. Until now, in most investigations, each of these possibilities has been tested in a separate experimental approach. Because of the different experimental conditions of each testing, it is not possible to compare the results directly with each other. We therefore have designed an approach in which both conditions (normal and compromised blood supply) can be found in the same brain. This experimental setting allows one to exclude any difference in the state of the experimental animals (blood pressure, acid–base status, and others) except for the difference in vascular supply. To this end, bilateral carotid artery occlusion was performed in rats (Eklof and Siesjö, 1972). In this model, blood supply is compromised in those brain areas that are supplied by the carotid arteries (Eklof and Siesjö, 1973). It can be expected that blood vessels in the forebrain are dilated, thus providing collateral blood flow from distant brain regions. If these vessels are maximally dilated for collateral blood flow, they should not be able to react to further factors that reduce blood flow, such as hyperviscosity of the blood. Conversely, brain regions that are supplied by unobstructed blood vessels should maintain their capability to react to an increased blood viscosity by vasodilation. Such a state should exist in the more caudal parts of the brain, which are supplied by the vertebral arteries. Because the induction of hyperviscosity by 20% polyvinylpyrrolidone induced some reduction of hematocrit, the control group of rats obtained an infusion of 5% albumin to match the control group and experimental group also with respect to hematocrit as closely as possible. The results show a clear difference in blood flow between the low viscosity and the high viscosity group in the forebrain but not in the medulla.
MATERIALS AND METHODS
After approval by the institutional animal care committee (Regierungspräsidium Karlsruhe, Germany), the experiments were performed in 16 male Sprague-Dawley rats (Charles River Deutschland, Sulzfeld, Germany), weighing 340 ± 17 g (mean ± SD). The animals were kept under temperature-controlled environmental conditions on a 14:10 light:dark cycle and fed a standard diet (Altromin 1324; Altromin, Lage, Germany) with free access to food and potable water until the start of the experiments.
The rats were randomly assigned to two experimental groups, a low viscosity group and a high viscosity group. They were placed in a small box and anesthetized by inhalation of isoflurane (Forene; Abbott, Wiesbaden, Germany) and 30% O2/air with a precalibrated vapor (Dräger, Lübeck, Germany). Anesthesia for surgery was maintained by 1.5% to 2.5% isoflurane via a nose cone. Body temperature was held at 37°C with a temperature-controlled heating pad. Polyethylene catheters (PE-50; Labokion, Sinsheim, Germany) were inserted into the right femoral artery and vein. Sutures were tied loosely around both common carotid arteries. A tracheostomy tube was inserted and connected to a small rodent ventilator (KTR-4; Hugo Sachs Elektronik, Freiburg, Germany). The arterial catheter was connected to a pressure transducer to monitor blood pressure continuously, and the venous catheter was used for drug and fluid administration.
After surgery, the concentration of isoflurane was set to 1 MAC (minimum alveolar concentration). For isoflurane, a MAC value of 1.4% (White et al., 1974) was used in this study. Pancuronium bromide (pancuronium; Organon, Eppelheim, Germany) was administered for muscle relaxation. To achieve a steady state, anesthesia was maintained for an equilibration period of additional 60 minutes. Thereafter, both common carotid arteries were occluded by tying both sutures. Ten minutes after vessel occlusion, 20% polyvinylpyrrolidone (molecular weight, 1,100,000; polyvinylpyrrolidone 90 Serva; Boehringer Ingelheim Bioproducts Partnership, Heidelberg, Germany), dissolved in isotonic saline, was infused at 6 mL/kg of body weight in the high viscosity group during 5 minutes. The solution of 20% polyvinylpyrrolidone, used in the present study, was prepared by dissolving an adequate amount of dry polyvinylpyrrolidone in saline during gentle stirring.
Polyvinylpyrrolidone is chemically defined as a variable molecular weight polymer of the monomer N-vinylpyrrolidone. Because of its properties as binder, dispersant, solubilizer, and emulsion stabilizer, it has had many industrial, cosmetic, and medical uses in past decades (Gosselin et al., 1984). Solutions of polyvinylpyrrolidone in low concentrations have been used as plasma expanders since 1942 (Hecht and Weese, 1943). The only pharmacodynamic actions of polyvinylpyrrolidone on blood vessels and body tissues are its uptake by the reticuloendothelial system and subsequent granuloma formation after prolonged administration (Kuo et al., 1997). Therefore, the use of polyvinylpyrrolidone as plasma expander has been abandoned in medicine.
Because former investigations and preliminary experiments had shown that this treatment led to a decrease of hematocrit from 40% to approximately 30%, probably because of the high colloid osmotic pressure of the infused polyvinylpyrrolidone, animals in the low viscosity group were subjected to hypervolemic hemodilution by 5% albumin in isotonic saline (human albumin 5%; Immuno, Heidelberg, Germany) 28 mL/kg of body weight in the same time period, which resulted also in a reduction of hematocrit to approximately 30%.
Blood gases were measured by using an automated blood gas analyzer (AVL Gas Check 939; AVL, Graz, Austria). Hemoglobin content, oxygen saturation, and oxygen content of blood were measured by a hemoximeter (OSM 3; Radiometer, Kopenhagen, Denmark). Expiratory CO2 was monitored continuously by a capnometer (EMG I; Heyer, Bad Ems, Germany). The hematocrit was determined by capillary tube centrifugation (Hettich, Tuttlingen, Germany). Plasma glucose concentration was measured by a polarographic method (Glucose Analyzer 2; Beckmann, München, Germany). The ventilator was adjusted to maintain an arterial po2 slightly greater than 100 mm Hg and normocarbia. Body temperature was monitored via a rectal probe and maintained within the range of 36.5°C to 37.5°C by a heating lamp.
Blood viscosity was measured by a rotating cone-plate viscometer (model DV-2; Wells-Brookfield, MA, U.S.A.) with an 0.8° cone spindle (CP 40). Before vessel occlusion and immediately before measurement of CBF, 500 μL of blood was drawn from each animal into heparinized Eppendorf tubes. Immediately thereafter, viscosity of this blood was measured at five different shear rates, starting with the lowest shear rate. The value of blood viscosity was read when it had stabilized on the display. After the highest shear rate had been reached, the measurements were repeated in each sample in the reverse order. The mean of the two measurements at each shear rate was used for further calculations.
Thirty minutes after the end of either infusion of polyvinylpyrrolidone or albumin, for the measurement of local CBF, 100 μCi/kg of body weight of 4-iodo-N-methyl-[14C]antipyrine (specific activity, 55 mCi/mmol; American Radiolabeled Chemicals, St. Louis, MO, U.S.A.) dissolved in 1 mL of saline was infused continuously at a progressively increasing infusion rate for 1 minute via the femoral venous catheter. The progressively increasing infusion rate, a modification of the method described earlier (Sakurada et al., 1978), was chosen to minimize equilibration of rapidly perfused tissues with arterial blood during the period of measurement. During the 1-minute infusion period, 14 to 20 timed blood samples were collected in drops from the free-flowing arterial catheter directly onto filter paper disks (1.3 cm in diameter) that previously had been placed in small plastic beakers and weighed. The samples were weighed and radioactivity estimated with a liquid scintillation counter (Tri-Carb 4000 series; Canberra Packard, Frankfurt, Germany) after extraction of the radioactive compound with ethanol. After the 1-minute infusion and sampling period, the animal was decapitated, the brain was removed as quickly as possible and frozen in 2-methylbutane chilled to −40°C to −50°C.
The frozen brains were coated with chilled embedding medium (M1 embedding matrix; Lipshaw, Detroit, MI, U.S.A.), stored at −80°C in plastic bags, and sectioned into 20-μm sections at −20°C in a cryostat. At each 200 μm, the first three serial sections were selected for autoradiography and the following seven sections were discarded. By this procedure, approximately 216 to 288 sections were obtained for each brain for autoradiography. These sections were autoradiographed along with precalibrated [14C]methyl methacrylate standards. Autoradiographic images were converted to digitized optical density images by an image-processing system (MCID; Imaging Research, St. Catharines, Ontario, Canada). Tissue optical densities were converted to [14C] concentration by comparison with the precalibrated standards. Local CBF was calculated from the local concentrations of [14C] and the time course of the blood 4-iodo-N-methyl-[14C]antipyrine, including corrections for the lag and washout in the arterial catheter (Sakurada et al., 1978). The washout correction rate constant was 100/min. A brain–blood partition coefficient for iodo[14C]antipyrine of 0.9 was used, as previously determined in our strain of rats (Schröck and Kuschinsky, 1988).
Individual brain structures were analyzed by using an ellipsoid cursor, which was adjusted to the size of the individual brain region. One value of local CBF was obtained from the mean of one measurement in six adjacent brain sections.
Mean CBF was measured in complete coronal sections at distances of 200 μm. For this, the first section at each 200 μm interval was taken (approximately 72 to 96 sections per brain). As a reference point dividing rostral brain from brainstem and cerebellum, the appearance of the lateral lemniscus was taken in the rostrocaudal direction of the coronal sections. Starting from this reference point, complete results could be obtained from 50 brain sections rostral to the lateral lemniscus and 22 brain sections in the caudal direction, the latter including the sections in which the lateral lemniscus was found. These 72 brain sections included all major parts of the brain and were taken for the measurement of mean CBF.
For the measurement of mean CBF in either the rostral brain or the brainstem and cerebellum, the values obtained from all coronal brain sections of either the rostral brain or the brainstem and cerebellum were summarized to obtain the area-weighted means.
Statistical analysis
Statistical differences between the experimental groups were evaluated by analysis of variance and paired and unpaired t-tests. Significance was assumed for P < 0.05. Data are presented as mean ± SD values.
RESULTS
Physiologic parameters
Physiologic parameters of the two groups are summarized in Table 1. At the time of CBF measurements, mean arterial blood pressure and plasma glucose concentration were significantly higher in the high viscosity group in comparison with the low viscosity group. In addition, a small but significant difference in blood pH between both groups was observed. All other physiologic parameters were not significantly different.
Physiologic parameters before carotid occlusion and after blood viscosity changes, immediately before blood flow measurements
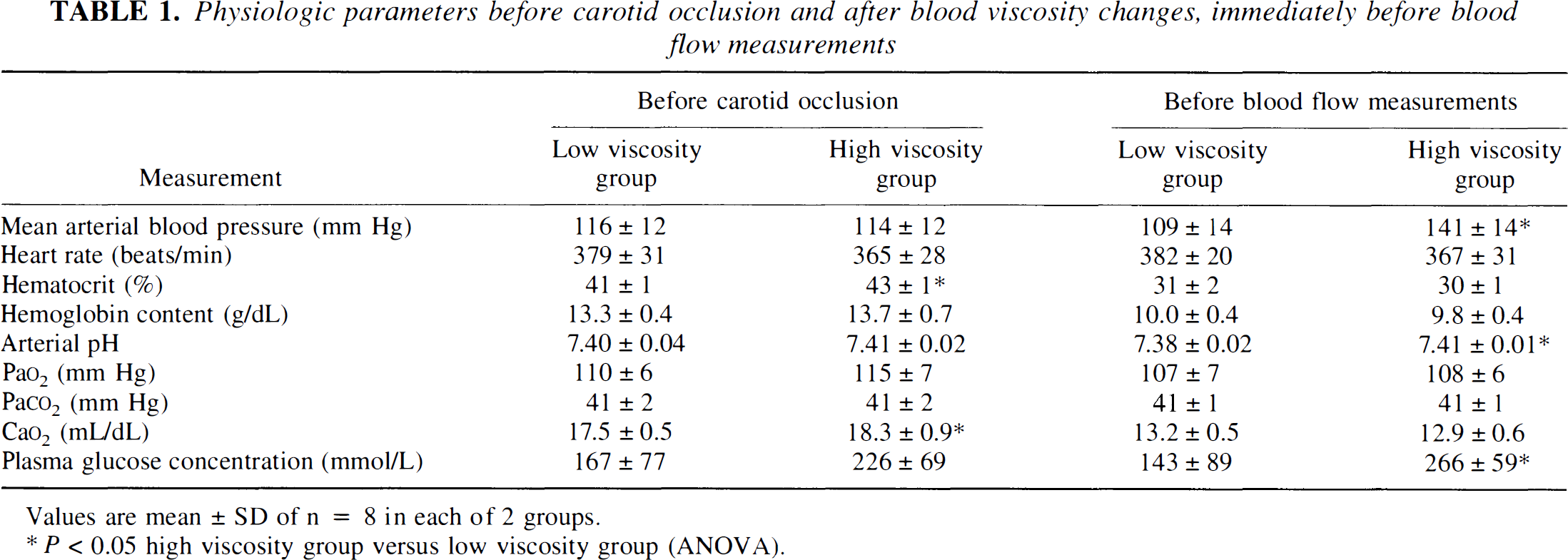
Values are mean ± SD of n = 8 in each of 2 groups.
P < 0.05 high viscosity group versus low viscosity group (ANOVA).
Viscosimetric data
The viscosimetric measurements of whole blood samples taken before and after infusion of either 20% polyvinylpyrrolidone or 5% albumin are summarized in Fig. 1. Before infusion, whole blood viscosity did not differ significantly between the two groups at the shear rates analyzed. Infusion of polyvinylpyrrolidone resulted in a more than threefold higher viscosity immediately before infusion of iodo[14C]antipyrine in the high viscosity group compared with infusion of albumin in the low viscosity group.
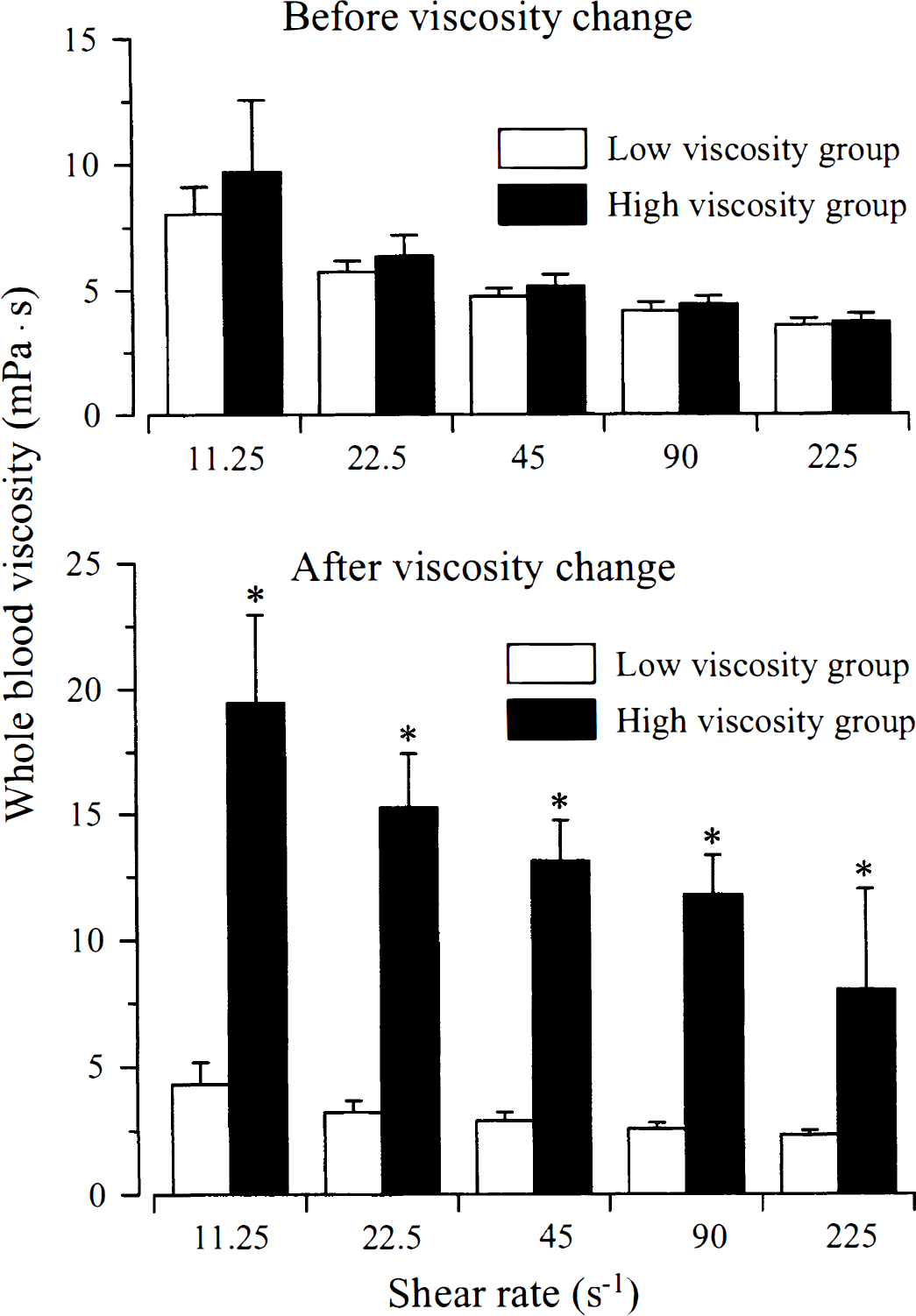
Whole blood viscosity in both experimental groups before and after changes of blood viscosity. *P < 0.05, high viscosity group versus low viscosity group.
Blood flow studies
Mean cerebral blood flow
Figure 2 shows mean CBF in the rostral brain and the cerebellum-brainstem of the two experimental groups. Compared with the low viscosity group, the mean CBF was lower in the rostral brain but not in the brainstem-cerebellum of the high viscosity group.
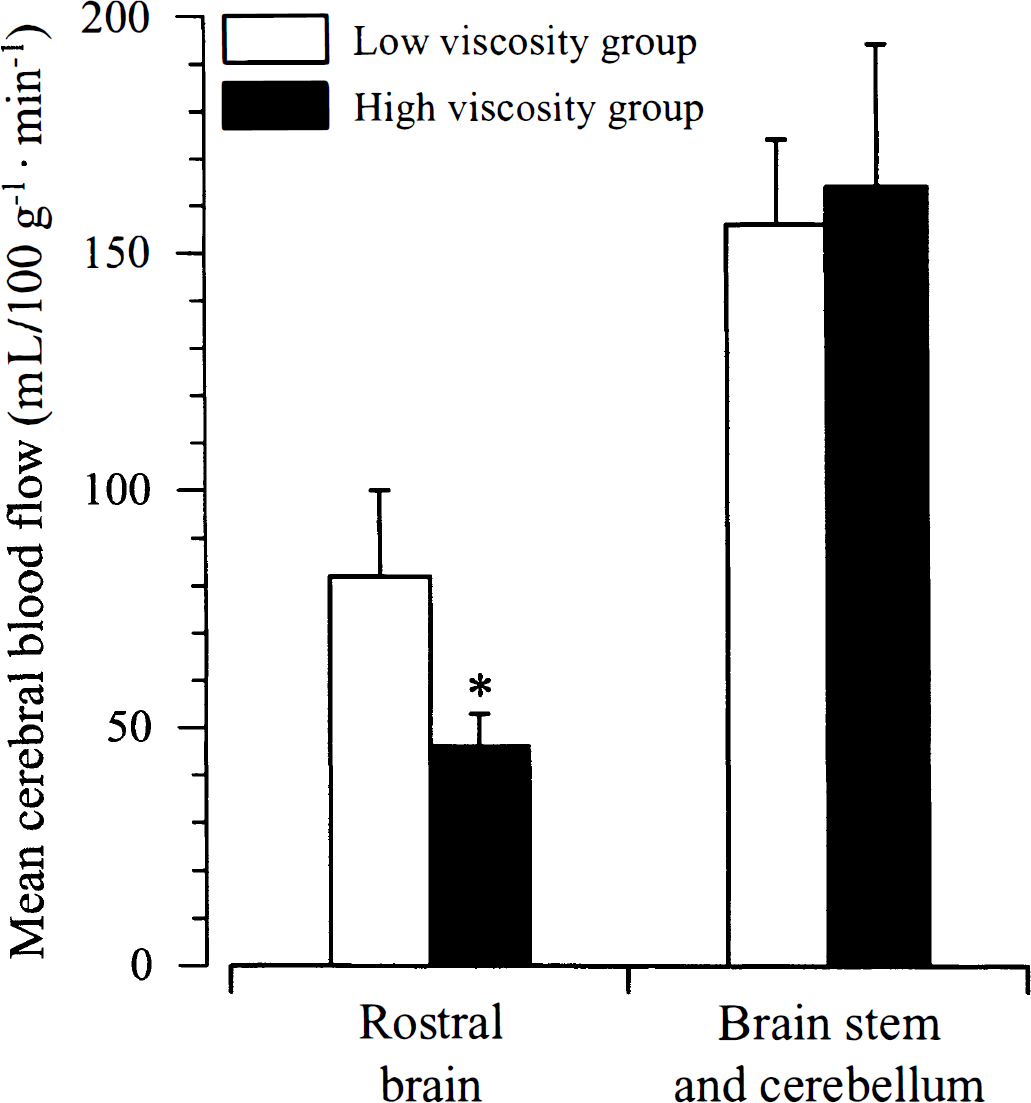
Mean cerebral blood flow (CBF) in rostral brain versus brain stem and cerebellum during bilateral carotid artery occlusion. *P < 0.05, high viscosity group versus low viscosity group.
Figure 3 shows the mean CBF in individual brain sections rostral and caudal to the reference point between the rostral brain and the brainstem-cerebellum (the appearance of the lateral lemniscus in the rostrocaudal direction of the coronal sections). In all corresponding brain sections rostral to the brainstem-cerebellum, the mean CBF was significantly lower in the high viscosity group than in the low viscosity group, whereas no difference could be found in the brainstem-cerebellum.
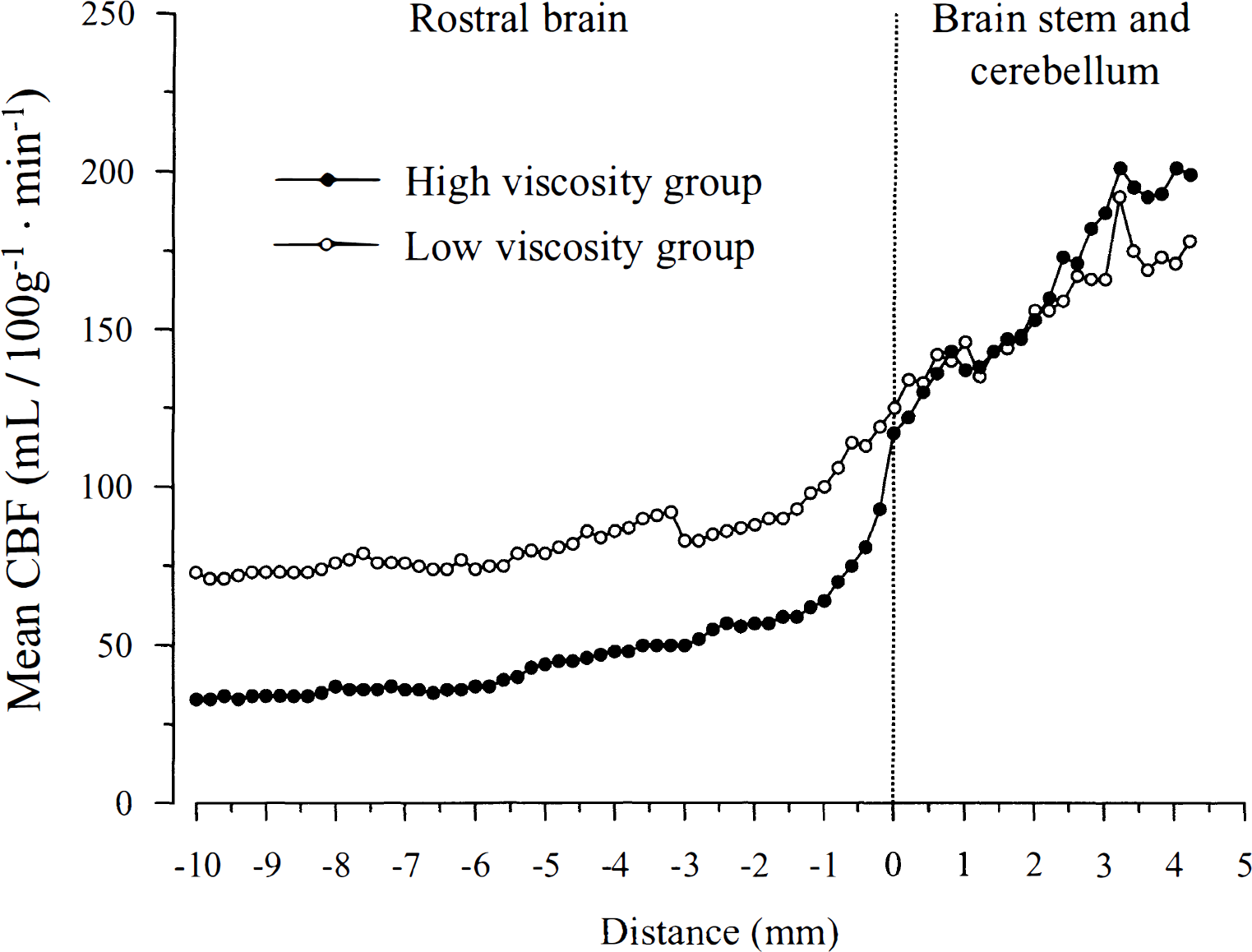
Mean cerebral blood flow (CBF) in coronal brain sections during bilateral carotid artery occlusion. Reference point dividing rostral brain from brain stem and cerebellum (0 mm) is the appearance of the lateral lemniscus in the rostrocaudal direction of the coronal sections.
Local cerebral blood flow
The differences in CBF of the rostral brain could also be verified by a comparison of local blood flow values in different brain structures. The results are shown in Table 2. Comparison of the regional values of CBF between the low and the high viscosity group showed significant differences in 29 of 31 brain structures investigated in the rostral brain, whereas no such differences could be measured in the 11 brain structures of the brainstem-cerebellum.
Local cerebral blood flow after bilateral carotid arteries occlusion and blood viscosity changes (mL/100 g−1 · min−1)
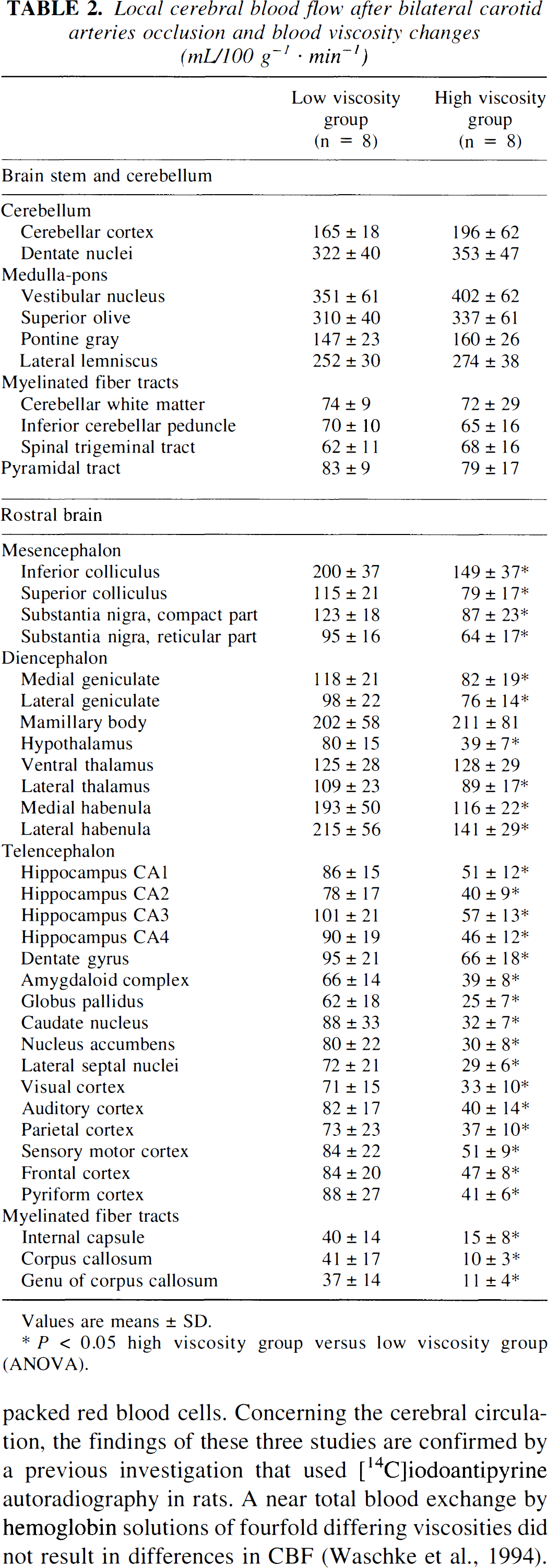
Values are means ± SD.
P < 0.05 high viscosity group versus low viscosity group (ANOVA).
DISCUSSION
The results of the present study support that changes in blood viscosity do not result in changes in CBF as long as the blood vessels retain their capacity to compensate for the changes in viscosity by vasodilation at an increased viscosity and a vasoconstriction at a decreased viscosity. When the capacity of the blood vessels to dilate is already exhausted to compensate for a reduced blood supply, an increased blood viscosity results in a decreased CBF. Such a situation exists in the rostral brain regions during bilateral carotid artery occlusion. Under these conditions, the supply of the rostral brain regions is dependent on collateral blood flow, which results in a reduced perfusion pressure in these regions (Fujishima and Omae, 1976). Although compensatory mechanisms such as metabolic (Golanov and Reis, 1997) or neurogenic (Golanov and Reis, 1996, 1999; Sun and Reis, 1994) vasodilation may become effective in rostral brain regions and diameter and blood flow in the basilar artery are increased (Fujii et al., 1991), these mechanisms are not sufficient to restitute blood flow to the control state observed before vessel occlusion (Choki et al., 1977; Eklof and Siesjö, 1973; Fujishima et al., 1981; He et al., 1997; Shiokawa et al., 1986). When the vascular compensatory mechanisms are outstripped in rostral brain regions during occlusion of both carotid arteries, blood flow cannot be maintained during hypotension (Eklof and Siesjö, 1973) or during an increase in blood viscosity. In contrast, regulatory mechanisms such as basilar artery dilation (Fujii et al., 1991) and pial arterial dilation are effective in preserving blood flow to caudal brain regions during hypotension (He et al., 1997; Shiokawa et al, 1986) or during an increased blood viscosity (present investigation). In contrast to the rostral brain, these mechanisms are not exhausted by a vascular occlusion of supplying carotid arteries.
The mean arterial blood pressure was higher in the high viscosity group than in the low viscosity group. This difference did not influence CBF in the caudal brain areas; the intact autoregulation in the brainstem and cerebellum is evident from the identical values of blood flow in both groups (Fig. 3). In the rostral brain areas, autoregulation may be disturbed. An abolished autoregulation in the rostral brain should result in a higher blood flow in these areas in the high viscosity group than in the low viscosity group, because blood pressure was higher in the high viscosity group. However, CBF in these regions was lower than in the low viscosity group, which indicates that any effect of arterial pressure, if it exists, is overridden by the effects of high viscosity on rostral brain blood flow.
The mean value of plasma glucose concentration was considerably higher in the high viscosity group already before infusion of polyvinylpyrrolidone. This difference was more apparent after infusion of polyvinylpyrrolidone. Whether this difference is just by chance or has any relation to the infusion of a high viscosity solution is not evident. Whatever the reason for this difference, it is unlikely that such a moderate increase in plasma glucose concentration has influenced CBF. The identity of CBF in the brainstem and cerebellum in both groups challenges any relevant effect of hyperglycemia on the CBF. This does not exclude the well-known damaging effect for hyperglycemia on ischemic brain tissue.
It has already been shown that CBF in the normal brain of rats, measured by the [3H]nicotine method, is kept constant during variations of intravascular volume expansion, cardiac output, and central venous pressure (Todd et al., 1993). The postulated existence of compensatory mechanisms for variances of blood viscosity in the cerebral circulation is supported by three previous studies in which radioactive microspheres were used. These studies showed that an increase in blood viscosity does not induce a corresponding decrease of CBF. Chen et al. (1989) exchanged the blood of anesthetized dogs for donor blood in which the viscosity had been increased by the addition of dextran, thus maintaining the oxygen content of the blood constant. They reported a decrease of blood flow in the spleen and the small intestine, whereas no change of regional blood flow was observed in the heart and in the brain. Krieter et al. (1995) infused small amounts of ultra high molecular weight dextran (4% of total blood volume) into beagle dogs. They obtained an increase in plasma viscosity from 1.06 to 2.99 mPa · s and a reduction of the hematocrit from 36% to 20%. Despite the increase in blood viscosity, they observed an increase in blood flow to all organs investigated, including the brain (Krieter et al., 1995). These observations are in accordance with those of Fan et al. (1980) who observed that oxygen transport to the brain, heart, kidney, liver, and small intestine is maintained at a constant level in anesthetized dogs when blood viscosity is either decreased by hemodilution with the use of blood plasma or is increased by hemoconcentration with the use of packed red blood cells. Concerning the cerebral circulation, the findings of these three studies are confirmed by a previous investigation that used [14C]iodoantipyrine autoradiography in rats. A near total blood exchange by hemoglobin solutions of fourfold differing viscosities did not result in differences in CBF (Waschke et al., 1994).
These findings are confirmed by further investigations that show that the maintenance of oxygen supply to the brain is a major driving force for CBF during different pathophysiologic conditions. Tomyiama et al. (1999) recently measured CBF by [3H]nicotine in hemodiluted and not-hemodiluted rats at atmospheric pressures. They compared the results with the values of CBF observed in hemodiluted animals whose blood oxygen content during hemodilution had been maintained by hyperbaric oxygenation in the same range as found in the not-hemodiluted control group at atmospheric pressure. Compared with the not-hemodiluted group, the increase in CBF in the hyperbaric hemodiluted group was approximately half the value that had been observed in the hemodiluted group at atmospheric pressure. Therefore, it was concluded that at least 40% to 60% of the increase in CBF at atmospheric pressure during hemodilution could be attributed to the reduction in arterial oxygen content (Tomiyama et al., 1999). The same group has shown previously in rabbits that, during hemodilution, maintenance of oxygen supply to the brain, beyond the increase in CBF as measured by radioactive microspheres, can be attributed to an increase in the oxygen extraction ratio from hemoglobin during normoxic conditions (Todd et al., 1994a), whereas oxygen supply to the brain during hypoxemic conditions is maintained by an exaggerated increase in CBF only (Todd et al., 1994a).
The effects of viscosity on the CBF have been investigated in brain tissue that was made ischemic by the occlusion of the middle cerebral artery in cats (Muizelaar et al., 1986) and in spontaneously hypertensive rats (Cole et al., 1994). Muizelaar et al. (1986) have lowered blood viscosity by infusion of 25% mannitol (1 g/kg of body weight). This treatment induced a decrease of hematocrit, an increase in mean arterial pressure, and an increase in CBF in the ischemic brain areas. Although the increased blood flow observed in the ischemic brain areas after the mannitol treatment was ascribed to the decreased blood viscosity, it cannot be excluded that the increase in cerebral perfusion pressure was the cause of the increased blood flow observed. Cole et al. (1994) induced a decrease in blood viscosity by exchange transfusion of blood against either albumin or hemoglobin solutions. These authors found an increase in CBF in the territory of the middle cerebral artery both in normal and in ischemic brain tissue. These observations suggest a lack of compensatory mechanisms for changes in blood viscosity in normal brain, which is at variance with the above-mentioned investigations (Chen et al., 1989; Fan et al, 1980; Krieter et al., 1995; Waschke et al., 1994). This study can be subjected to some points of criticism. One point is that the two different methods for reduction of blood viscosity resulted in inconsistent blood flow responses. At an identical blood viscosity, CBF in the ischemic area was approximately twice as high after exchange by the hemoglobin solution as it was after exchange by albumin. It seems possible that the exchange by hemoglobin per se induced an increased CBF in this study. Another point of criticism may be that the experimental groups in which blood had been exchanged against albumin showed a lower blood oxygen content than the control group. This lower blood oxygen content may have induced an increase in blood flow in those brain areas that had retained their reactivity to a decreased blood oxygen content (Cole et al., 1994).
In contrast, in the present investigation, blood oxygen content was identical in the high and the low viscosity groups, whereas the mean arterial pressure was lower in the low viscosity group. Thus, neither differences in blood oxygen content nor blood pressure can explain the higher CBF observed in the low viscosity group. In addition, because the brainstem and the cerebellum of the same animal were used as their own controls, a pharmacodynamic effect of polyvinylpyrrolidone, responsible for the differences in blood flow observed between the two experimental groups, can also be excluded. In addition to the points of criticism suggested, the main conclusions of Muizelaar et al. (1986) and Cole et al. (1994), that changes in blood viscosity may be responsible for the changes in CBF observed in brain regions exhibiting a compromised blood flow, are therefore confirmed by the present investigation. However, compared with the findings of Cole et al. (1994), compensatory mechanisms for differences in blood viscosity could be clearly demonstrated in the present investigation in those parts of the brain that were not affected by vessel occlusion.
The crucial role of the blood vessels in the maintenance of blood flow during changes in blood viscosity has been demonstrated under in vitro and in vivo conditions. Koller et al. (1993) studied the effect of an increase in viscosity of the perfusion solution on the diameters of isolated arterioles of rat cremaster muscle. At a constant perfusion rate, increases in viscosity of the perfusate by 2%, 4%, and 6% dextran caused a gradual vasodilation up to 22%. This vasodilation was explained by endothelial mechanisms, which maintained wall shear stress close to control conditions at a higher viscosity of the perfusate.
In vivo, the responses of pial arteries and of the basilar artery to decreases in blood viscosity induced by infusion of mannitol (Muizelaar et al., 1983) and hemodilution (Hudak et al., 1989; Muizelaar et al, 1992) have been studied. Under both conditions, a decrease of the oxygen-carrying capacity and of the blood viscosity, as a consequence of a declining hematocrit, was observed. An increase in CBF was verified in two of these studies by the use of a thermal diffusion probe (Muizelaar et al., 1992) or radioactive microspheres (Hudak et al., 1989). Despite the increase in CBF, the vessels observed were found to constrict when blood viscosity was decreased by hemodilution (Hudak et al., 1989; Muizelaar et al., 1992). A similar vasoconstriction was observed when blood viscosity was reduced by infusion of mannitol (Muizelaar et al., 1983). The vasoconstriction of the cerebral resistance vessels indicates a compensatory reaction that counteracts an increase in CBF that would occur as a reaction to the decrease in blood viscosity, which is induced by a reduced hematocrit from hemodilution or mannitol infusion. Such reactions seem to represent an adaptive mechanism of the cerebral vessels to changes in blood viscosity. Although the observations of these previous investigations were limited to superficial blood vessels, e.g., pial vessels and basilar artery, it seems likely that deep intracerebral vessels show a comparable reaction. In the present study, blood viscosity was increased. This should result in a compensatory vasodilation, which results in a constant CBF. The results of the present study are in accord with this hypothesis, because the CBF was maintained in brain regions with a normal blood supply, probably because of a compensatory vasodilation. In other regions of the same brain, the blood supply was compromised because of an exhausted capacity of the vessels to further dilate after carotid occlusion had been added to the blood hyperviscosity.
Mechanisms that may mediate vasomotor responses after changes in blood viscosity have been studied in vitro in endothelial cells. An increased blood viscosity induces an increased shear stress at the endothelial cells of the blood vessels. As a consequence, a K+-selective, shear stress–activated ionic channel is opened, which hyperpolarizes the endothelial cell (Olesen et al., 1988), possibly mediated by an inward rectifying potassium channel (Jacobs et al., 1995). The hyperpolarization is then followed by a relaxation of the vascular smooth muscle cells. In addition to the activation of K+ channels, shear stress can induce the release of nitric oxide from endothelial cells (Luscher, 1991). Nitric oxide would then cause vasodilation by the activation of a soluble guanylate-cyclase (Marietta, 1994; Nathan and Xie, 1994) and subsequent Ca2+-dependent mechanisms (Chen and Rembold, 1992; Sperelakis et al., 1994) in vascular smooth muscle cells.
Changes of blood viscosity have been discussed as a therapy of brain ischemia. An increase in regional CBF has been observed after a decrease of blood viscosity in the ischemic brain areas by Cole et al. (1994) and Muizelaar et al. (1986). However, clinical studies showed no benefit of a decrease in blood viscosity for the neurologic outcome in individuals suffering from focal cerebral ischemia (Berrouschot et al., 1999; Hemodilution in Stroke Study Group, 1989; Italian Acute Stroke Study Group, 1988; Scandinavian Stroke Study Group, 1987). The lacking clinical amelioration of stroke patients after hemodilution therapy has been explained in different ways. First, the time between the ischemic event and the start of the hemodilution therapy might have been too long to save ischemic brain tissue (Harrison, 1989). Second, the response to hemodilution therapy may be altered in the damaged brain tissue. Todd et al. (1994b) have observed such an alteration in the ipsilateral brain hemisphere of rabbits damaged by a cryogenic brain lesion. Whereas hemodilution increased CBF in the contralateral hemisphere and in the hindbrain, the response of CBF to hemodilution was markedly attenuated throughout the brain hemisphere ipsilateral to the lesion. Third, hemodilution results in two different effects, i.e., a decrease in blood viscosity and a decrease in the oxygen-carrying capacity of the blood. Thus, the benefit for the ischemic brain, exerted by an increase in CBF, may be offset by a lower blood oxygen content. This failure to enhance oxygen delivery to hypoxic brain regions seems to be the main problem of hemodilution in the treatment of clinical stroke. The present results show that the oxygen delivery to ischemic, but not to nonischemic, brain tissue can be increased by a decrease in blood viscosity when the oxygen content of the blood is not changed simultaneously. Such an effect may be achieved in the future by the addition of low viscosity oxygen carriers to the hemodilution solutions during the treatment of brain ischemia.
In conclusion, the findings of the present investigation are in accord with the hypothesis that a moderate increase in blood viscosity does not result in changes in CBF under normal conditions, because vascular compensatory reactions take place. In addition, the present study shows that such compensatory mechanisms are exhausted in brain areas compromised by occlusion of the supplying arteries.
Footnotes
Acknowledgments
The authors thank T. Lorenz, T. Fuchs, P. Strauβ, and M. Harlacher for excellent technical assistance.
