Abstract
To what extent sildenafil, a selective inhibitor of the type-5 phosphodiesterase modulates systemic redox status and cerebrovascular function during acute exposure to hypoxia remains unknown. To address this, 12 healthy males (aged 24 ± 3 y) participated in a randomized, placebo-controlled crossover study involving exposure to both normoxia and acute (60 min) hypoxia (Fi
Keywords
Introduction
Sildenafil was primarily developed for the treatment of hypertension and angina pectoris 1 owing to its highly selective ability to inhibit type-5 phosphodiesterase (PDE-5) and modulate the nitric oxide (NO)/cyclic guanosine monophosphate (cGMP) pathway. 2 Sildenafil has since been of interest for providing neuroprotection,3 –5 in light of its ability to cross the blood-brain barrier (BBB) 6 and increase the expression of PDE-5 mRNA and protein in neurons, glial cells and cerebrovascular endothelial cells7 –9 while also activating angiogenesis and neurogenesis.10 –13 While limited data exists, sildenafil has been shown to increase basal cerebral blood flow (CBF) in healthy humans14,15 and more recently, lower the risk of Alzheimer’s Disease particularly in those most frequently issued prescriptions for erectile dysfunction. 16
The vascular protective benefits of sildenafil are likely attributed to its direct and indirect impact on redox status. The selective inhibition of PDE-5 has been shown to enhance endogenous antioxidant defenses given observed increases in the expression of erythrocyte superoxide dismutase and catalase activity17,18 and inhibition of nicotinamide adenine dinucleotide phosphate (NADPH) activity,2,19 collectively effecting a reduction in free radical and associated reactive oxygen species (ROS) formation preceding liberation of vascular NO. 17 Sildenafil acts to increase the biological effect of NO by attenuating cGMP hydrolysis, 20 subsequently increasing intracellular cGMP concentration that mediates the NO-induced activation of cGMP-dependent protein kinases and ion channels. 21
Exposure to the hypoxia of high-altitude promotes local elevations in oxidative-nitrosative stress (OXNOS), reflected by a free radical-mediated reduction in vascular NO bioavailability. 22 This has been further associated with cerebrovascular dysfunction, namely attenuated vasoreactivity to hypercapnia 23 and select metrics of dynamic cerebral autoregulation (dCA).23,24 While sildenafil has been used prophylactically to attenuate the hypoxia-induced elevation in pulmonary artery systolic pressure, the primary risk factor for high-altitude pulmonary oedema,25,26 there is only one study that has explored its impact on the cerebrovasculature during inspiratory hypoxia. Accordingly, Chan, et al. 18 reported a single dose (50 mg) of sildenafil increased cerebral oxygenation on days 1 and 3 following rapid ascent to 3,480 m. However, to what extent sildenafil could recover the aforementioned hypoxia-induced cerebrovascular dysfunction23,24 remains to be established.
In light of these findings, we conducted a randomized, double-blind, placebo-controlled crossover trial utilizing a functionally integrative translational approach to examine the impact of PDE-5 inhibition on hypoxia-induced systemic OXNOS and corresponding implications for cerebrovascular function. We hypothesized that sildenafil would attenuate hypoxia-induced systemic OXNOS and promote improvements in select indices of cerebral hemodynamic function.
Methods
Ethics
The experimental protocol was approved by the Research Ethics Committees of the University of South Wales (#17070LSE) and Cardiff Metropolitan University (#16/8/01 R). All experimental procedures were carried out in accordance with the most recent (7th) amendment of the Declaration of Helsinki of the World Medical Association, 27 with the exception that it was not registered in a publicly accessible database, with verbal and written informed consent obtained from all participants.
Participants
Twelve healthy, physically active young (26 ± 12 y) males with a body mass index (BMI) of 26 ± 3 kg·m−2 were recruited into the study (see power calculations in statistical analysis). All participants were free of medication, non-smokers and abstained from taking nutritional supplements, including oral antioxidants and anti-inflammatories. Participants were specifically asked to refrain from physical activity, caffeine and alcohol for a period of 48 h prior to formal experimentation, to minimize biological variation.
24
They were also encouraged to follow a low-nitrate/nitrite (
Design
The study adopted a randomized, double-blind, placebo-controlled crossover design (Figure 1). All participants completed two trials (oral administration of sildenafil and placebo) and were exposed to both normoxia
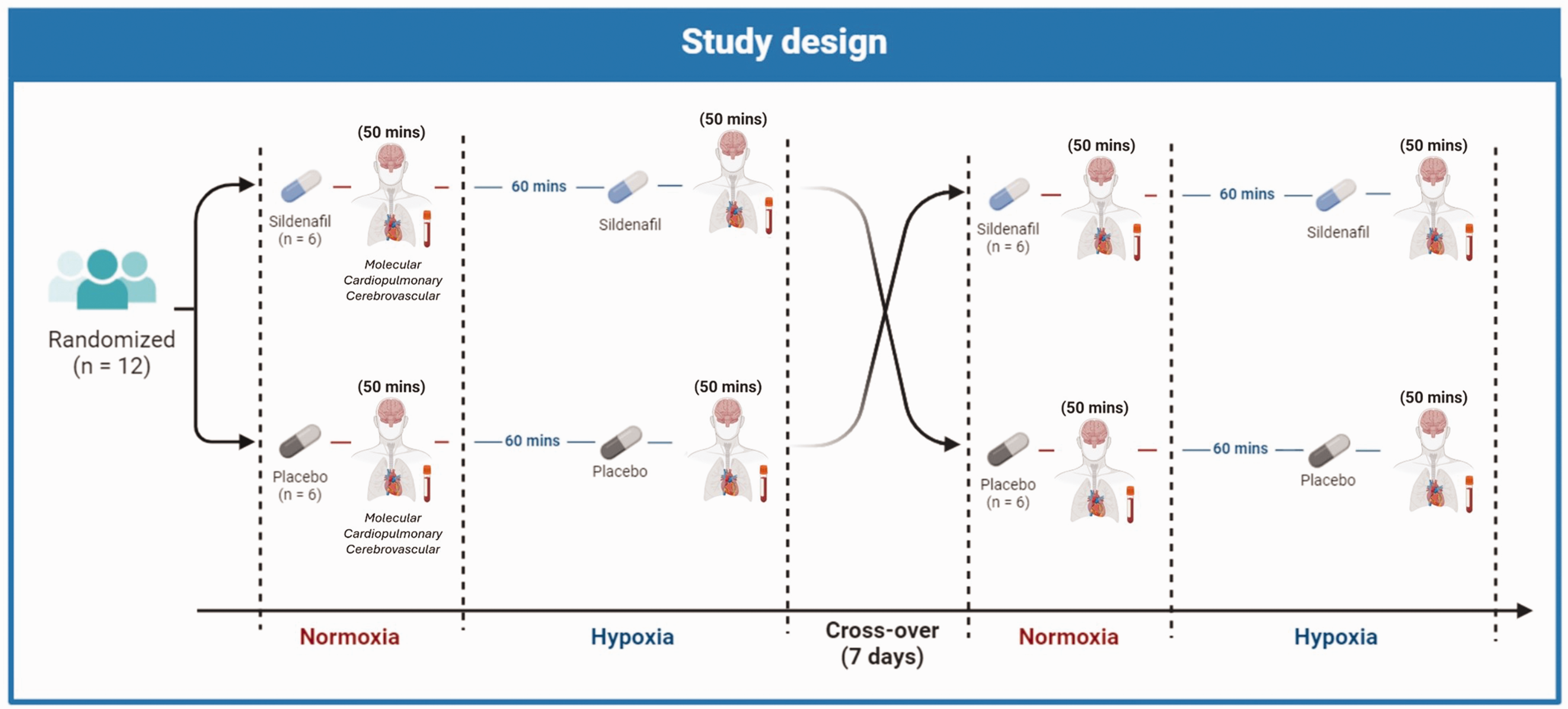
Study design. The study adopted a randomized, double-blind, placebo-controlled crossover design. All participants completed two trials (sildenafil and placebo) and were exposed to both normoxia
Measurements
Molecular function
Blood was collected from an indwelling cannula into Vacutainers® (Becton, Dickinson and Company, Oxford, UK) before centrifugation at 600 g (4°C) for 10 min. Plasma (K-EDTA) and red blood cell (RBC) supernatant were decanted into cryogenic vials (Nalgene® Labware, Thermo Fisher Scientific Inc, Waltham, MA, USA), immediately snap-frozen in liquid nitrogen and subsequently stored at −80°C. Prior to batch analysis, each sample were thawed at 37°C in the dark for 3 min. Whole blood was also assayed for hemoglobin (Hb) and hematocrit (Hct). Hb was measured photometrically (HemoCue 201+, Radiometer, UK) and Hct was prepared via ultracentrifugation (Hawksley and Sons Ltd, Sussex, UK) and measured using a Hawksley Micro Hematocrit Reader (Hawksley and Sons Ltd, Sussex, UK).
Free radicals
The ascorbate free radical (A•−) was employed as a direct measure of global free radical formation.30,31 Plasma (1 mL) was injected directly into a high-sensitivity multiple-bore sample cell (AquaX, Bruker Daltonics Inc., Billerica, MA, USA) housed within a TM110 cavity of an EPR spectrometer operating at X-band (9.87 GHz). Samples were recorded by cumulative signal averaging of 10 scans using the following instrument parameters: resolution, 1024 points: microwave power, 20 mW; modulation amplitude, 0.65 G; receiver gain, 2 × 105; time constant, 40.96 ms; sweep rate, 0.14 G/s; sweep width, 6 G; centre field, 3486 G. Spectra were filtered identically (moving average, 15 conversion points) using WINEPR software (Version 2.11, Bruker, Karlsruhe, Germany) and the double integral of each doublet was determined using commercial software (OriginLab Corps, MA, USA). The intra- and inter-assay CVs were both <5%. 31
NO metabolites
Ozone-based chemiluminescence (Sievers NOA 280i, Analytix Ltd, Durham, UK) was employed to detect NO liberated from plasma and RBC samples via chemical reagent cleavage as previously described in detail.32,33 This facilitated detection of total plasma (combined concentration of nitrite [
Cardiopulmonary function
Beat-by-beat arterial blood pressure was assessed via finger photoplethysmography and arterial volume clamping (Finometer PRO, Finapres Medical Systems, Amsterdam, Netherlands) and used to calculate mean arterial blood pressure (MAP) after calibrating values to the average of three automated brachial blood pressure measurements (Life Source, A&D Medical, model: UA767FAM), taken over a 5 min resting baseline period. Heart rate (HR) was assessed using a 3-lead electrocardiogram (ECG, ADI BioAmp ML132, ADInstruments, Colorado Springs, CO, USA). Respiratory flow was measured with a pneumotachometer (model HR 800 L, Hans Rudolph, Shawnee, KS) and expired gases (end-tidal partial pressures of
Cerebrovascular function
Cerebral haemodynamics
The proximal segment of the right middle cerebral artery (MCA) was insonated using a 2 MHz-pulsed transcranial Doppler (TCD) ultrasound system (Multi-Dop X4; DWL Electroniche Systeme, Singen, Germany). Following standardized search techniques,
36
the Doppler probe was secured over the middle trans-temporal window using a custom fit headband device (Spencer Technologies) to securely insonate the MCA and measure blood velocity (MCAv). Indices of cerebrovascular conductance (CVCi) and cerebrovascular resistance (CVRi) were calculated as mean MCAv/MAP and MAP/mean MCAv, respectively. Pulsatility index (PI) was calculated as: systolic MCAv−diastolic MCAv/mean MCAv and subsequently normalized to the prevailing MAP (PINorm = PI/MAP). Cerebral
1.34
Cerebral oxygenation
Pulsed continuous-wave near-infrared spectroscopy (NIRS, Oxymon Mk III; Artinis Medical Systems BV, Zetten, The Netherlands) was used to monitor changes in cerebral oxygenation at optical densities of 780 and 850 nm. 37 One set of NIRS optodes (5.0 cm source detector spacing, consistent with recent recommendations) was placed on the skin over the left frontal prefrontal cortical region of the forehead between Fp1 and F3, consistent with the anatomical landmarks of the International 10–20 system for EEG placement. 38 Optodes were secured in place using double-sided adhesive tape and positioned underneath the custom fit headband device. Concentration changes (Δ) in oxyhemoglobin (O2Hb) and deoxyhemoglobin (HHb) were calculated using the modified Beer–Lambert Law 39 incorporating a differential pathlength factor (DPF) of 5.93% for cerebral tissue. 40 We also calculated the difference between Δ[O2Hb] and Δ[HHb] as a surrogate ‘oxygenation index’ (OI), consistent with NIRS studies that have also employed similar continuous-wave devices (Grassi et al. 2003). Finally, Δ[THb] was calculated as the sum of Δ[O2Hb] and Δ[HHb] signals and used as a surrogate for changes in regional blood volume (Van Beekvelt et al. 2001). All NIRS signals were normalized to reflect changes relative to a 5 min resting baseline control in normoxia following administration of the placebo (arbitrarily defined as 0 µmol·L−1). Signals were recorded at 50 Hz, displayed in real time and stored on a computer for offline analysis. A 60 s average at the end of a five min resting baseline was calculated in each condition.
Dynamic cerebral autoregulation (dCA)
Following 10 min of seated rest, 5-min segments of MAP and MCAv data were obtained for spectral analysis of spontaneous oscillations to assess dCA via transfer function analysis (TFA). 41 Beat‐to‐beat MAP and MCAv mean signals were calculated across each cardiac cycle, linearly interpolated, and resampled at 4 Hz in accordance with formal recommendations of the Cerebrovascular Research Network.42,43 Spontaneous MAP and MCAv mean power spectrum density and the mean value of TFA coherence, gain, and phase were band averaged across the very low frequency (VLF: 0.02–0.07 Hz, 50 to 14.3-s cycles) and low frequency (LF: 0.07–0.2 Hz, 14.3 to 5-s cycles) ranges where CA is considered to be most operant. 41 The squared coherence function reflects the fraction of output power (i.e., MCAv mean) that can be linearly related to the input power (i.e., MAP) at each frequency. Similar to a correlation coefficient, this value varies between 0 and 1 with 1 indicating that all the variability in MCAV is linearly explained by the variability in MAP. To ensure that robust phase and gain estimates were entered for subsequent analysis, we averaged only those gain and phase (positive to eliminate wrap-around) values where the corresponding coherence was ≥0.34, applied to single harmonics only. An increase in gain and reduction in phase were taken to reflect reduced dCA, indicative of a more pressure-passive relationship between MAP and MCAv. 24
Cerebrovascular reactivity to carbon dioxide (CVRCO2)
Following the 5 min resting baseline for TFA, the inspired air was rapidly changed to 5% C
Statistical analysis
Power calculations
Prospective power calculations were determined using G*Power (V.3.1.9.4). Assuming comparable differences (15%) and corresponding effect size (0.60) previously observed in basal plasma A•− (η2 = 0.54) 44 between normoxia and hypoxia, our primary end-outcome variable for OXNOS, the present study required a (minimum) sample size of 20 participants (10 per group) in order to achieve adequate power (1-β = 0.80 at P < 0.05). We chose to further inflate this by 20% during recruitment (n = 24 or 12 per group) given the potential for loss to follow-up or incomplete data collection.
Inferential analyses
Data were analyzed using the Statistics Package for Social Scientists (IBM SPSS Statistics Version 29.0). Shapiro-Wilk W tests (all P > 0.05) confirmed that all data sets were normally distributed. A 2-way (Inspirate: normoxia vs. hypoxia × Drug: placebo vs. sildenafil) repeated measures analysis of variance (ANOVA) was used to detect differences between conditions and presented alongside effect sizes (partial eta squared: ηp2) and estimates of statistical power (β). Where interaction effects were detected, post-hoc comparisons were made using paired samples t-tests with Bonferroni correction. Significance was established at P < 0.05 for all two-tailed tests and data expressed as mean ± standard deviation (SD).
Results
Compliance and randomization
A total of 12 participants completed both placebo and sildenafil trials with no loss to follow-up. Randomization led to 6 participants completing the sildenafil trial first with 6 participants completing the placebo trial first (Figure 1).
Molecular function
During the placebo trial, hypoxia did not impact
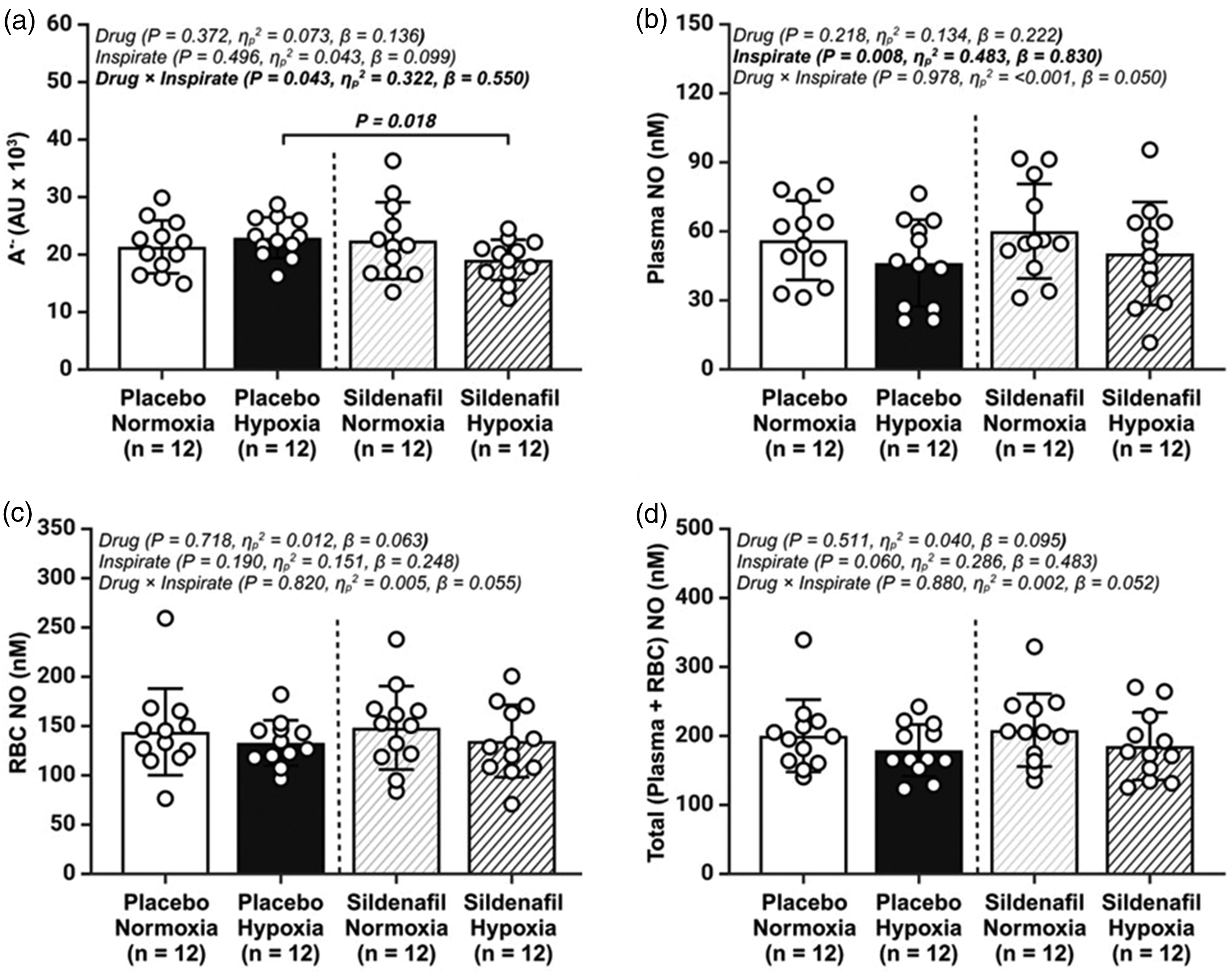
Basal metrics of systemic oxidative-nitrosative stress (OXNOS). (a) plasma ascorbate free radical (A•−); (b) total plasma nitric oxide (NO) concentration (nitrite + S-Nitrosothiols); (c) total red blood cell (RBC) NO (nitrite + S-Nitrosohemoglobin +hemoglobin-bound NO); and (d) total (plasma + total RBC) NO concentrations; AU, arbitrary units; Values are mean ± SD. n = 12 for all parameters.
Cardiopulmonary function
Hypoxia increased Hb (P = <0.001), Hct (P = 0.004),
Cardiopulmonary function.
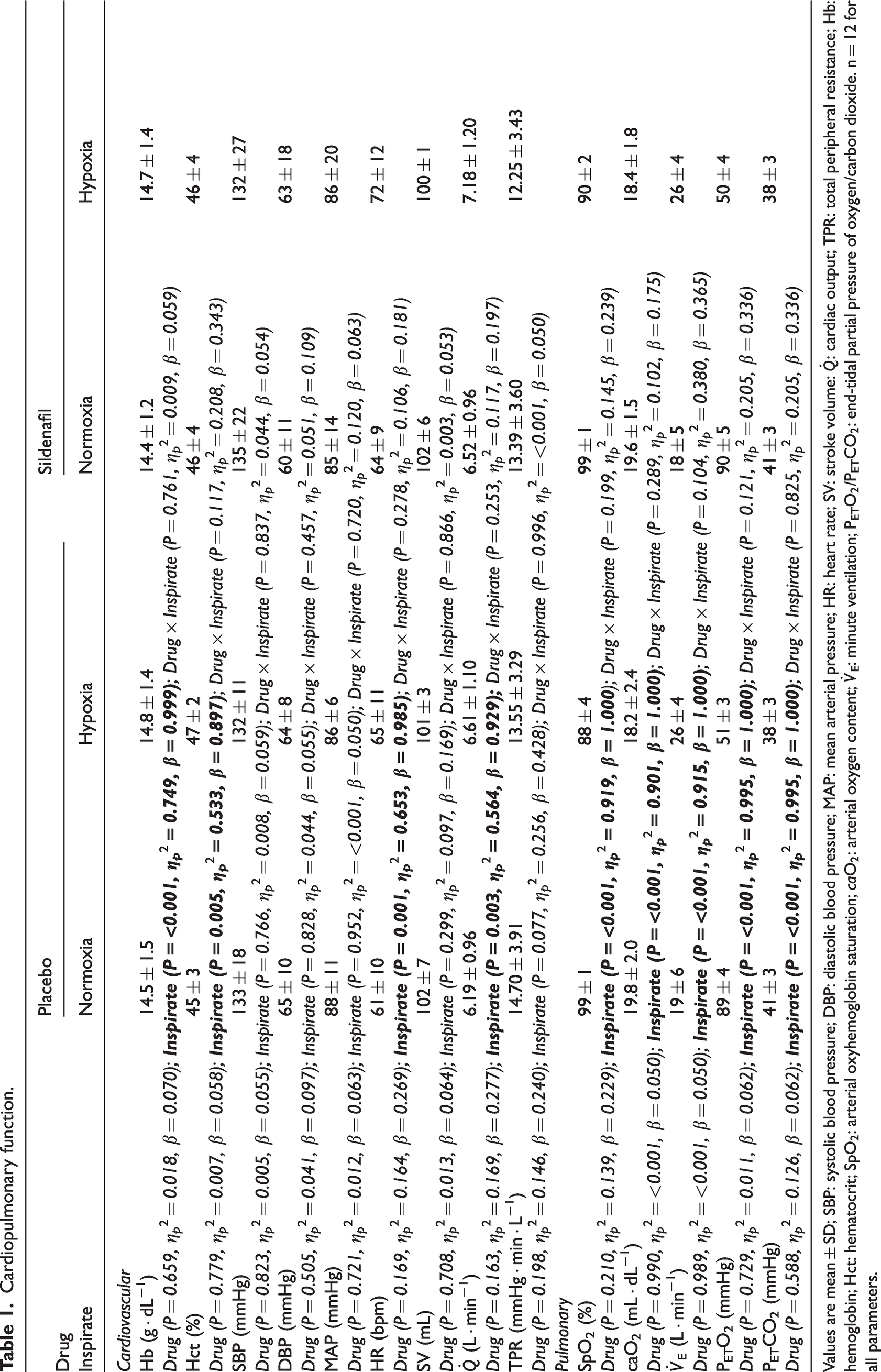
Values are mean ± SD; SBP: systolic blood pressure; DBP: diastolic blood pressure; MAP: mean arterial pressure; HR: heart rate; SV: stroke volume:
Cerebrovascular function
Cerebrovascular function.
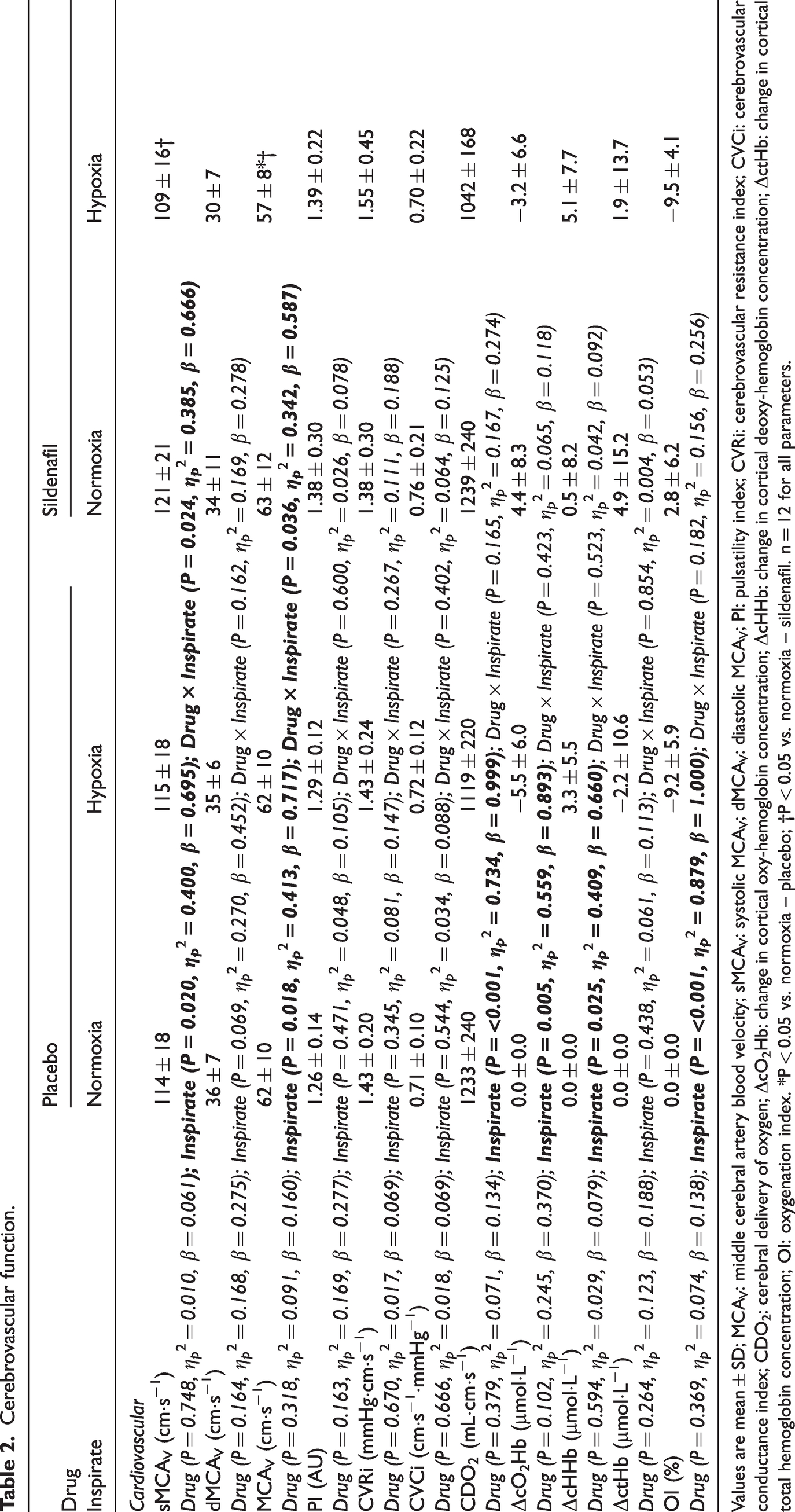
Values are mean ± SD; MCAV: middle cerebral artery blood velocity; sMCAV: systolic MCAV; dMCAV: diastolic MCAV; PI: pulsatility index; CVRi: cerebrovascular resistance index; CVCi: cerebrovascular conductance index; CDO2: cerebral delivery of oxygen; ΔcO2Hb: change in cortical oxy-hemoglobin concentration; ΔcHHb: change in cortical deoxy-hemoglobin concentration; ΔctHb: change in cortical total hemoglobin concentration; OI: oxygenation index. *P < 0.05 vs. normoxia – placebo; †P < 0.05 vs. normoxia – sildenafil. n = 12 for all parameters.
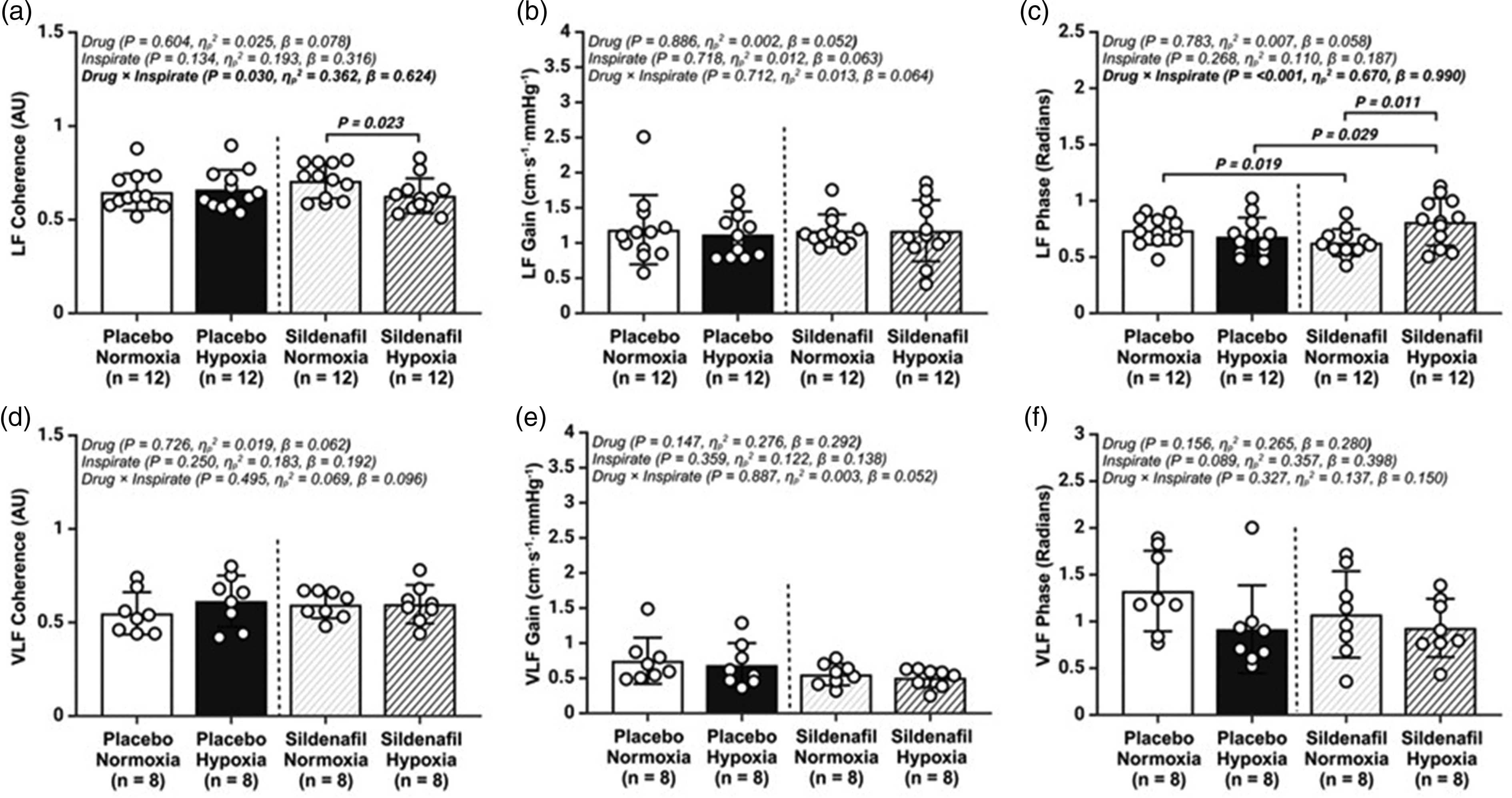
Transfer function analysis of the cerebral pressure–flow relationship during spontaneous oscillations in mean arterial blood pressure (MAP) and middle cerebral artery velocity (MCAv). (a) low frequency (LF) Coherence; (b) LF Gain; (c) LF Phase; (d) very low frequency (VLF) Coherence; (e) VLF Gain and (f) VLF Phase; AU, arbitrary units; Values are mean ± SD. n = 12 for LF parameters, whereas n = 8 for VLF data.
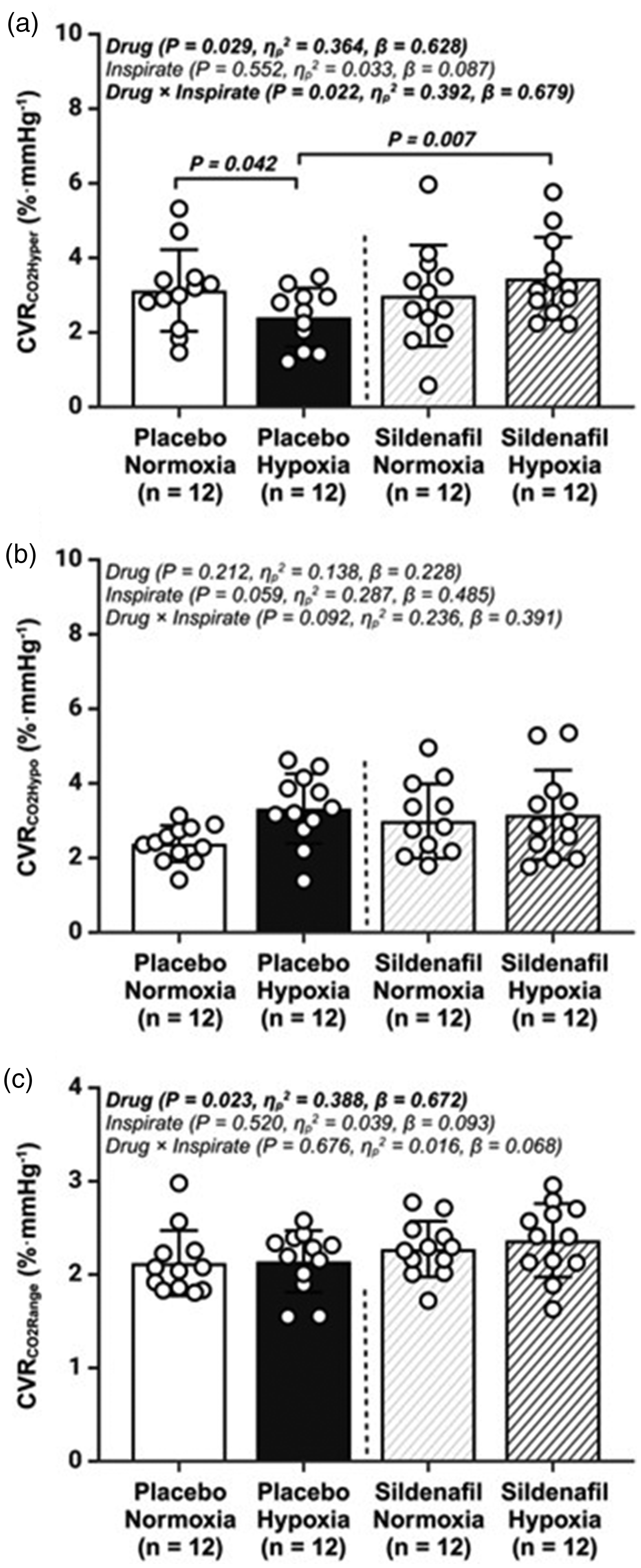
Cerebrovascular reactivity to carbon dioxide (CVRCO2). (a) cerebrovascular reactivity to hypercapnia (CVRCO2Hyper); (b) cerebrovascular reactivity to hypocapnia (CVRCO2Hypo) and (c) cerebrovascular reactivity to carbon dioxide range (CVRCO2Range); Values are mean ± SD. n = 12 for all parameters.
Discussion
Taking a functionally integrative translational approach, the present study has identified two important findings. First, sildenafil selectively reduced systemic free-radical formation in hypoxia yet, contrary to original expectations, this was not accompanied by a reciprocal elevation in vascular NO bioavailability consistent with (attenuated) systemic OXNOS. Second, despite a selective reduction in MCAv that likely reflected enhanced (hypoxic) vasodilation given the attenuated hypoxia-induced cerebral deoxygenation and clearly independent of hyperventilation-induced hypocapnia-mediated cerebral vasoconstriction (i.e. PETCO2 invariant), sildenafil increased LF phase and restored the hypoxia-induced attenuation in CVRCO2Hyper. Collectively, these findings provide evidence for a PDE-5 inhibitory pathway that enhances select aspects of cerebrovascular function in hypoxia, subsequent to a systemic improvement in redox homeostasis that is independent of systemic NO bioavailability.
Molecular function
Owing to the low reduction potential for the
Cerebrovascular function
The functional recovery of hypoxia-induced cerebrovascular impairments following sildenafil administration, taking the form of elevated CVRCO2HYPER and a less pressure-passive dCA response (increased LF phase) are equally novel findings. Not only does sildenafil have the capacity to cross the blood-brain-barrier 6 and act directly upon the central nervous system where PDE-5 has been localized to neurons and glial cells, 6 sildenafil can also target PDE-5 within cerebrovascular endothelial cells. 9 Accordingly, the NO/cGMP signal pathway has been shown to inhibit neuroinflammation, increase CBF and promote neurogenesis and synaptic plasticity.48,49
Additionally, these findings may be attributed to sympathetic hyperactivity following sildenafil administration in hypoxia,50,51 given that ganglion blockade with trimethaphan diminishes dCA 52 and cerebral vasomotor reactivity 53 . Although sympathetic nervous activity (SNA) was not assessed directly in the present study, we did observe an elevation in heart rate with sildenafil, indicative of increased sympathetic tone. 54 Equally, whilst we did not observe any changes in TFA Gain estimates in either the low or very-low frequency bands, the use of Phase estimates alone has been shown to be a more reliable measure of dCA than Gain in clinical studies.43,55 –57
It should be noted that while our findings under conditions of normoxia are supported by previous research,
58
there does exists a discrepancy with that of Jahshan, et al.
59
who demonstrated sildenafil to increase CVRCO2HYPER without affecting basal cerebrovascular tone or static/dynamic indices of CA. These discrepancies may be related to differences in drug dosage/timing notwithstanding technical nuances associated with the analytical assessment of CVRCO2HYPER and dCA. In our study, we administered 50 mg of sildenafil in normoxia and employed a 3-min inhalation of 5% C
Clinical importance
The findings from this study are clinically relevant given that free radical-mediated impairments in cerebrovascular function caused by arterial hypoxaemia precede myriad infectious and degenerative neurological disorders.
62
While this is the first investigation to explore the cerebrovascular implications of sildenafil under controlled conditions of acute inspiratory hypoxia utilizing a placebo-controlled cross-over design, these findings are commensurate with research in those with existing cerebrovascular impairments. In patients with pulmonary hypertension that are characterized by lower CBF and cerebrovascular reactivity, sildenafil enhanced visually-evoked flow responses indicating improved neurovascular coupling.
15
More recently, a single dose (50 mg) of sildenafil has been shown to increase global CBF and CMR
Limitations
The present study has several limitations that warrant consideration. First, MCAv represents an indirect surrogate measure of CBF and given that MCA diameter appears to increase in hypoxia 64 , we cannot rule out the possibility that we underestimated the volumetric changes incurred by hypoxia and sildenafil. In support, the selective reduction observed in MCAv during the sildenafil hypoxic trial may reflect an increase in CBF subsequent to enhanced vasodilation, highlighting the importance of duplex imaging of global CBF (flow and velocity metrics of internal carotid and vertebral arteries) in future studies. Second, there has been an on-going debate regarding the best method to quantity dCA and in particular, the use of spontaneous versus forced oscillations on blood pressure as the TFA input. The present study utilized spontaneous oscillations in blood pressure which is arguably more reflective of basal activity, safer to reproduce in a clinical population and importantly, does not challenge either the autonomic or cardiorespiratory systems. 65 However, this approach assesses the minimal level of blood pressure variations leading to lower TFA coherence and diminished linearity within the cerebral pressure-flow relationship. 66 Alternatively, it has been recommended that the TFA input should utilize forced oscillations in blood pressure (i.e. repeated squat-stands) to increase the signal-to-noise ratio, 43 arguing that this reflects everyday challenges associated with large swings of blood pressure (i.e. bending down, standing up, walking upstairs) and that owing to the high coherence (e.g. close to 1.0), the changes in phase and gain are interpretable. 66 However, we have consistently identified that >50% of young, healthy participants experience vasovagal syncope and associated cephalalgia during forced oscillations at an FIO2 of 0.12 (DM Bailey et al, unpublished observations), hence why we constrained our analyses to spontaneous oscillations only. Lastly, these findings are constrained to that of normobaric hypoxia with comparison to chronic high-altitude (hypobaric hypoxic) accordingly difficult owing to different physiological responses.67,68 However, we, among others, have demonstrated that acclimatization to high-altitude hypoxia increases basal systemic nitric oxide bioavailability33,69 which is responsible, at least in in part for, increasing cerebral substrate delivery and preserving bioenergetic function consistent with the conservation of mass principle. 70 An improvement in cerebral redox homeostasis may represent the unifying mechanism that explains the functional improvements in cerebral oxygenation documented by Chan, et al. 18 following sildenafil administration after chronic exposure to high-altitude.
Conclusion
The present study has demonstrated that PDE-5 inhibition has the potential to restore redox homeostasis, namely attenuate systemic free radical formation without altering vascular NO bioavailability and recover select impairments in cerebral hemodynamic function following acute exposure to poikilocapnic hypoxia. These findings are clinically relevant given that free radical-mediated impairments in cerebrovascular function caused by arterial hypoxaemia precede myriad infectious and degenerative neurological disorders.
Footnotes
Data availability statement
Original data arising from this research are available directly from Professor Damian Miles Bailey upon reasonable request.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Royal Society Wolfson Research Fellowship (no. WM170007 to D.M.B) and the Higher Education Funding Council for Wales (postdoctoral fellowships for B.S.S, T.S.O, T.A.C and A.I to D.M.B).
Acknowledgements
We appreciate the commitment of all participants and technical assistance provided by Mr Trevor Harris.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.M.B. is Editor-in-Chief of Experimental Physiology, Chair of the Life Sciences Working Group and member of the Human Spaceflight and Exploration Science Advisory Committee to the European Space Agency and is a member of the Space Exploration Advisory Committee to the UK Space Agency. D.M.B. is affiliated to the companies FloTBI and Bexorg, Inc., focused on the technological development of novel biomarkers of brain injury/bioenergetics in humans.
Authors’ contributions
All experiments were conducted in the Neurovascular Research Laboratory at the University of South Wales. B.S.S and D.M.B conceived and designed the research. B.S.S, C.J.M, H.T, T.G.D, T.S.O, T.A.C, L.F, A.I, I.L, J.M.C, M.S and D.M.B contributed to the acquisition, analysis and interpretation of data. B.S.S and D.M.B drafted the manuscript, and all authors revised it critically and contributed intellectual content. B.S.S, C.J.M, H.T, T.G.D, T.S.O, T.A.C, L.F, A.I, I.L, J.M.C, M.S and D.M.B approved the final version of the manuscript, agree to be accountable for all aspects of the work, and will ensure that any questions concerning the accuracy or integrity of any part of this work are appropriately investigated and resolved. All persons designated as authors qualify for authorship, and all those who qualify for authorship are listed.
