Abstract
Nitric oxide is a potent vasodilator. Infusion of its precursor, L-arginine, results in increased cerebral blood flow (CBF) in experimental animals. We examined the effects of L-arginine infusion on CBF in humans using positron emission tomography and the quantitative H215O method. Six subjects received 500 ml of 0.9% NaCl solution, and six subjects received an infusion of L-arginine (16.7 mg/kg/min; 500 mg/kg). Before and after the i.v. infusion, paired CBF measurements were performed at baseline and with vibrotactile stimulation of the right hand. In scans performed without vibrotactile stimulation, mean whole-brain CBF increased from 34.9 ± 3.7 ml 100 g–1 min–1 to 38.2 ± 4.4 ml 100 g–1 min–1 (9.5%; p < 0.005) after L-arginine infusion. The temporal pattern of CBF changes differed from that of plasma growth hormone and insulin levels and of arterial pH. In contrast, in the saline group, mean whole-brain CBF did not change significantly (35.8 ± 5.9 ml 100 g–1 min–1 to 35.9 ± 6.4 ml 100 g–1 min–1; 0.3%). Vibrotactile stimulation produced significant focal increases in CBF, which were unaffected by L-arginine infusion. L-arginine infusion was associated with an increase in plasma L-citrulline, a byproduct of nitric oxide synthesis.
L-Arginine is a precursor for nitric oxide (NO), a potent dilator of blood vessels. Infusion of L-arginine results in vasodilation and increased blood flow in several vascular beds (Hishikawa et al., 1991; Morikawa et al., 1992; Sun et al., 1992; Bode-Böger et al., 1994). Morikawa et al. (1994) reported that L-arginine dilates pial arterioles and increases blood flow in the cerebral circulation of rats. This finding suggests that NO is an important mediator in cerebral autoregulation.
It has also been postulated that NO mediates the increased CBF observed with functional activation of the brain. Administration of NO synthase inhibitors attenuates the CBF response to neuronal activation (Dirnagl et al., 1993; Akgören et al., 1994). In knockout mice lacking neuronal NO synthase, however, Ma et al. (1996) observed that this inhibitory effect did not occur. This finding suggests that NO derived from endothelial cells does not mediate coupling between flow and metabolism and that neuronal NO may not be the sole mediator of coupling.
L-Arginine infusion reduces the size of experimental brain infarction (Morikawa et al., 1994; He et al., 1995), raising the possibility that manipulation of the L-arginine-NO system may be a novel therapeutic strategy for human cerebral ischemia. This finding led us to examine the effects of L-arginine infusion on human basal cerebral blood flow (CBF) and the CBF response to neuronal activation using positron emission tomography (PET) and the quantitative H215O method of estimating CBF.
SUBJECTS AND METHODS
Twelve neurologically normal subjects were studied. Six subjects (four males; mean age, 22.8 years) received 500 ml of 0.9% NaCl solution and six subjects (six males; mean age 26.7 years) received an infusion of L-arginine (16.7 mg/kg/min; 500 mg/kg). This dose of L-arginine was similar to one that produced an increase in CBF in rats (300 mg/kg; Morikawa et al., 1992). In all subjects, paired CBF measurements were performed at baseline and with vibrotactile stimulation of the right hand. Pairs of measurements were performed before and after i.v. infusion of saline or L-arginine (Fig. 1). The duration of the infusion was 30 min. Baseline scans (without stimulation) were performed 20 and 45 min after the start of the infusion. Scans with stimulation were performed 30 and 55 min after the start of the infusion. All subjects provided informed consent, and the study protocol was approved by the ethics committee of the Montreal Neurological Hospital and Institute.
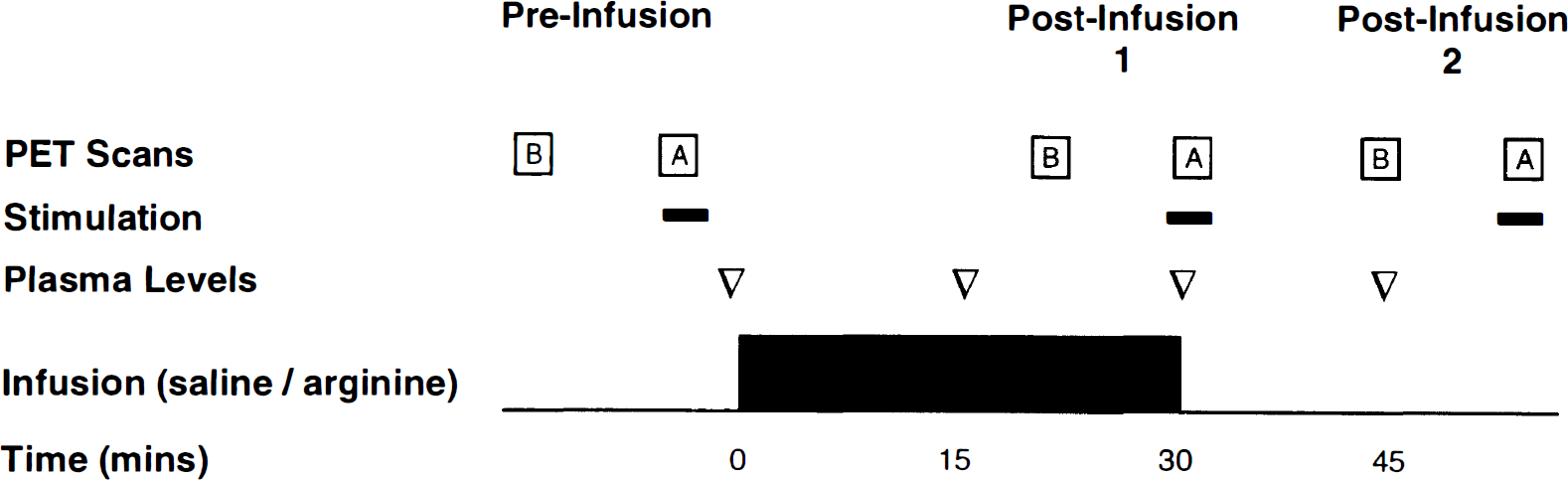
Design of study.
Vibrotactile activation protocol
Vibrotactile stimulation at a frequency of 110 Hz and an amplitude of 2 mm was provided by a mechanical vibrator (Model 91, Daito, Osaka, Japan) fixed to the fingertips of the subjects' right hand. The vibratory stimulus was intermittent (1 s on, 1 s off) to avoid adaptation (Applegate et al., 1988). Stimulation was commenced 30 s before the start of PET scanning and continued throughout the 3-min scanning period. All scans were performed with subjects' eyes closed in a darkened room.
PET scanning
PET studies were performed with a Scanditronix PC-2048 15B scanner, which produces 15 slices with an intrinsic resolution of 6 × 6 × 6 mm (Evans et al., 1992). A foam head mould was fitted to minimize head movement. Subjects were positioned in the scanner so that the uppermost slice was parallel to the orbitomeatal line at the level of the high cerebral convexity. A 68Ge orbiting-rod transmission source was used for attenuation correction. Cerebral blood flow was measured using a previously described technique (Raichle et al., 1983; Ohta et al., 1996). Briefly, an i.v. bolus of ∼1.5 GBq of H215O was administered at the start of a 3-min multiframe dynamic scan. At the same time, continuous monitoring of arterial radioactivity was commenced.
At times designated in Fig. 1, 10 ml of arterial blood was withdrawn for analysis of pH and PCO2 and L-arginine, L-citrulline, growth hormone, and insulin concentrations. Heart rate and blood pressure (sphygmomanometry with Korotkoff sounds) were also measured.
All subjects underwent magnetic resonance imaging (MRI; Philips Gyroscan 1.5T) with scans consisting of 160 contiguous slices of 1-mm thickness and 1 × 1-mm pixel dimension.
Image analysis
PET images were reconstructed using a 12-mm FWHM Hanning filter, and individual time–activity curves for each voxel were fitted with a two-compartment model to obtain regional maps of CBF (Ohta et al., 1996). The PET and MRI images were coregistered, and MRI-based whole-brain regions of interest were drawn. These regions were used to calculate mean global CBF.
Identification of the activation focus.
All images were stereotaxically normalized to correspond to the standard brain dimensions of Talairach and Tournoux (1988). For each subject, t statistic volumes were generated for the change in CBF between the averaged baseline and averaged activation scans (Worsley et al., 1992). The t statistic peak corresponding to the focus of activation in the left somatosensory cortex was identified, and mean CBF in a volume of interest containing all voxels within 8 mm of the peak was calculated for each PET scan.
CBF in representative gray and white matter regions.
Mean CBF was calculated in representative gray and white matter volumes of interest in the parasagittal frontal gray matter, the insular cortex, and the thalami and in the centrum semiovale, respectively. The sizes of the volumes of interest were frontal cortex, 2.1 cm3; insular cortex, 4.6 cm3; thalamus, 5.3 cm3; and centrum semiovale, 2.1 cm3.
To determine the uniformity of changes in CBF, images of the average change in CBF were generated. The stereotaxically normalized CBF images averaged across subjects for the scan without activation before infusion were subtracted from the first postinfusion images without activation.
For statistical analysis, we used the paired t test to compare CBF values under corresponding baseline or activation conditions for each of the postinfusion scans with CBF in the preinfusion scans. The paired t test was also used to compare the results of plasma assays, blood pressure, and pulse rate after infusion with the results before infusion.
RESULTS
Mean whole brain blood flow
Before infusion, baseline whole-brain CBF was 35.8 ± 5.9 ml 100 g–1 min–1 (mean ± SD) in the saline group (Table 1) and 34.9 ± 3.7 ml 100 g–1 min–1 in the L-arginine group (Table 2). In scans performed without vibrotactile stimulation, a 9.5% (p < 0.005) increase in mean whole-brain CBF occurred after infusion of L-arginine, in contrast to a 0.3% increase after saline infusion. The effect of L-arginine infusion was no longer apparent 45 min after the start of the infusion (whole-brain CBF without vibrotactile stimulation: 33.4 ± 4.5 ml 100 g–1 min–1).
Cerebral blood flow, plasma hormone and amino acid assays, blood pressure, and heart rate for the preinfusion and the two postinfusion measurements after 0.9% NaCl infusion (n = 6)
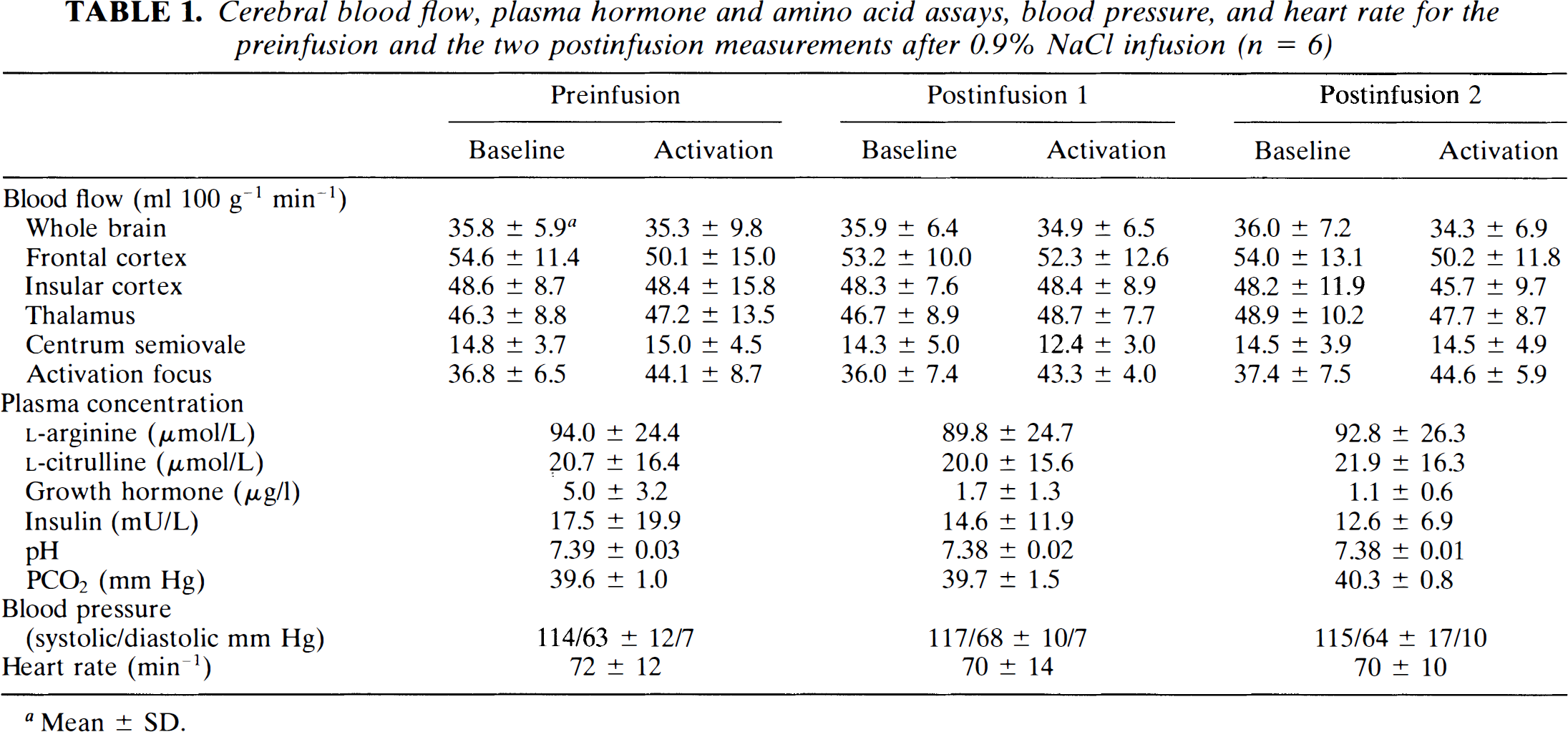
Mean ± SD.
Cerebral blow flow, plasma hormone and amino acid assays, blood pressure, and heart rate for the preinfusion and the two postinfusion measurements after L-arginine infusion (n = 6)
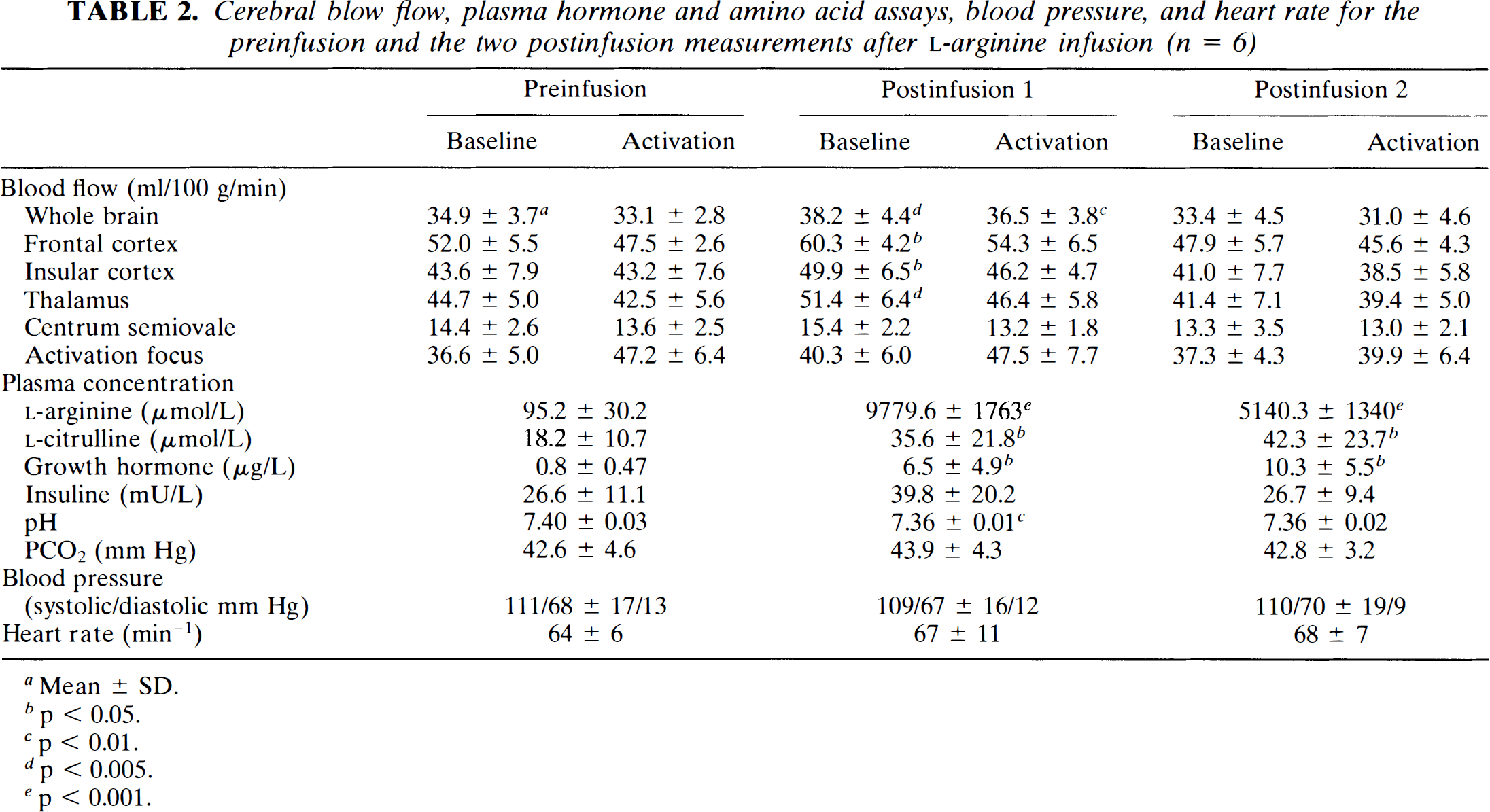
Mean ± SD.
p < 0.05.
p < 0.01.
p < 0.005.
p < 0.001.
Effects on gray and white matter blood flow
The effect of L-arginine infusion was observed predominantly in gray matter structures (Table 2; Fig. 2). In the cortical and subcortical gray matter regions, a 14 to 16% increase in CBF was observed (p < 0.05), whereas in the centrum semiovale, mean CBF increased by only 6.9% (p > 0.05).
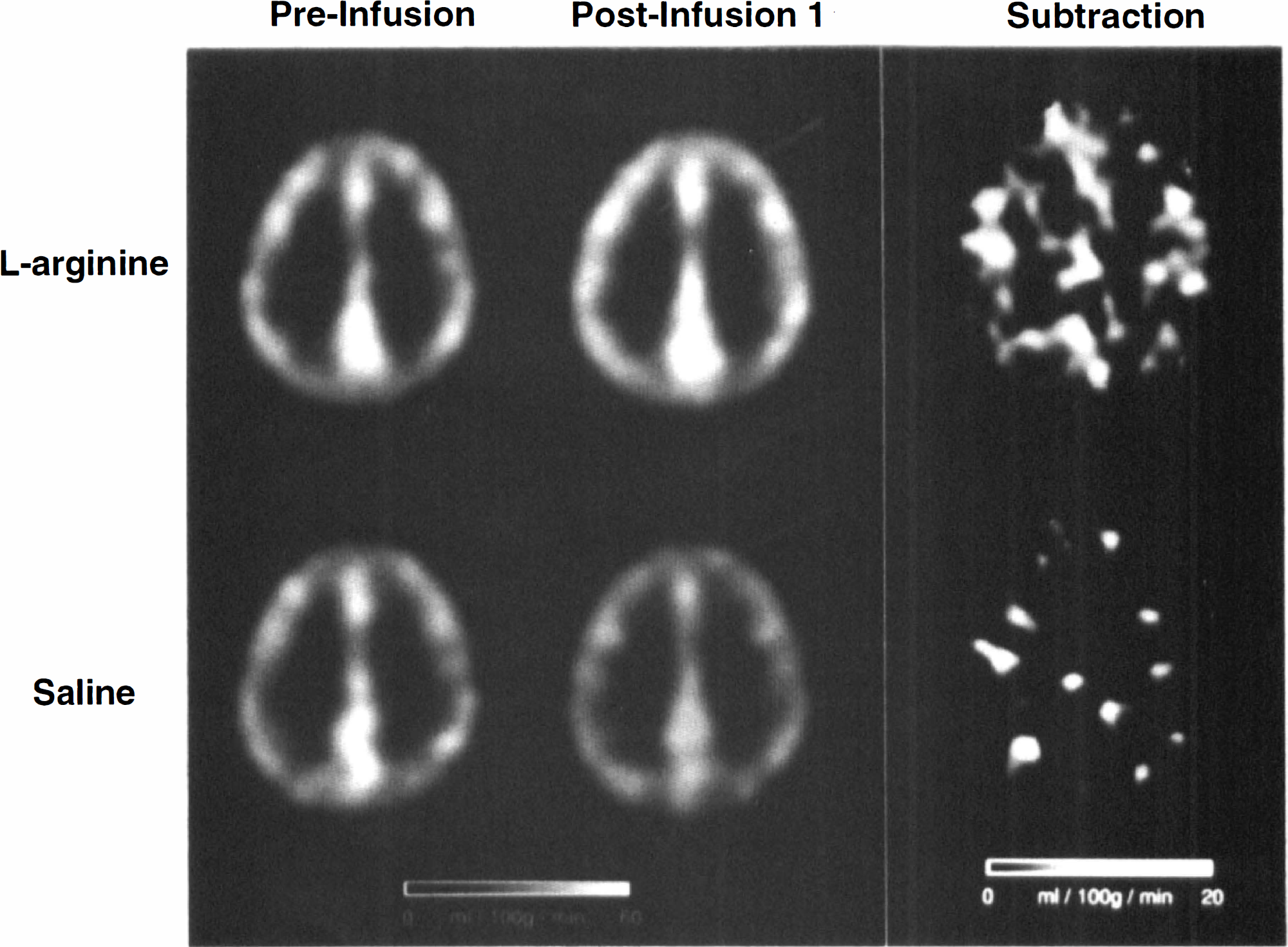
Preinfusion averaged CBF images, CBF images for the first post-infusion baseline scan, and images of the mean change in CBF for groups receiving
Effects on blood flow in the activation focus
For both the saline and arginine groups, CBF in the activation focus was higher with vibrotactile stimulation than in the baseline state (Fig. 3). This difference was statistically significant (p < 0.05) for the preinfusion and the first postinfusion scans in both groups. The increase in CBF in the activation focus was smaller in the second pair of scans after the infusion of L-arginine (37.3 ± 4.3 ml 100 g–1 min–1 to 39.9 ± 6.4 ml 100 g–1 min–1 and was not statistically significant (p = 0.18).
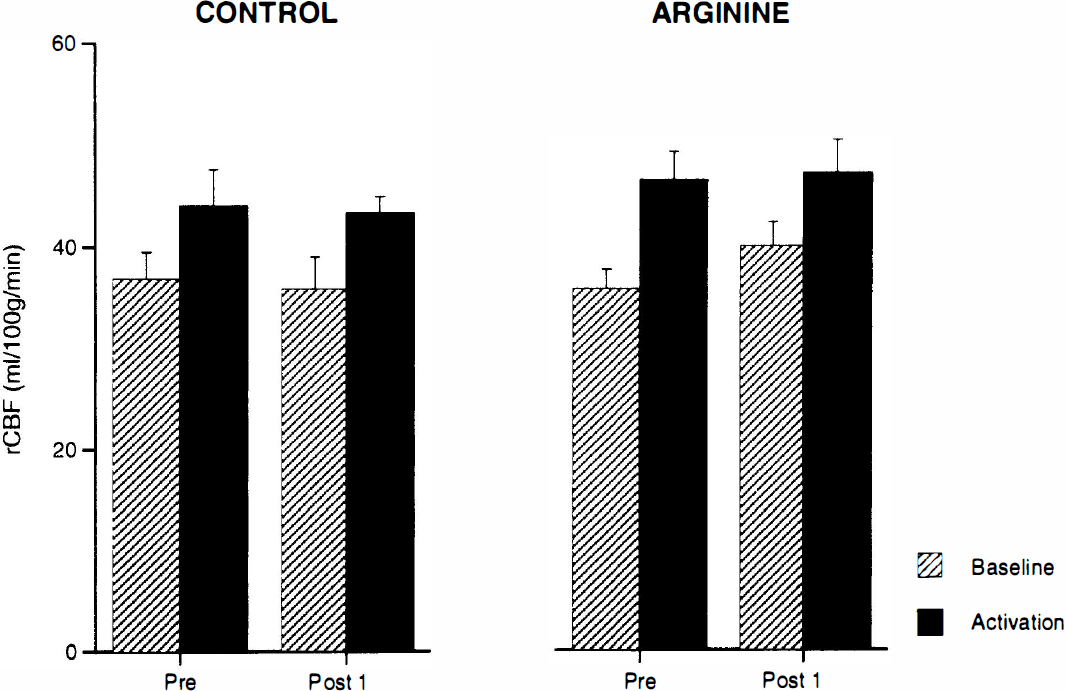
CBF in a volume of interest containing all voxels within 8-mm of the t statistic peak corresponding to the focus of activation in the left somatosensory cortex. Results for baseline and activation scans performed before (Pre) and ∼30 min after (Post 1) infusion are shown. In both groups, CBF in the activation focus was higher with vibrotactile stimulation than in the baseline state but L-arginine infusion had no additional effect on the increases in CBF observed with vibrotactile stimulation.
L-Arginine infusion had no additional effect on the increases in CBF observed with vibrotactile stimulation. With vibrotactile stimulation, CBF in the activation focus was 47.2 ± 6.4 ml 100 g–1 min–1 before and 47.5 ± 7.7 ml 100 g–1 min–1 after L-arginine infusion.
Blood pressure, arterial pH, and PCO 2, amino acids and hormones
L-Arginine infusion was associated with significant increases in the plasma levels of L-arginine (p < 0.001), L-citrulline (p < 0.05), and growth hormone (p < 0.05; Table 2; Fig. 4); however, 45 min after the start of the L-arginine infusion, when global CBF had returned to its initial values, plasma levels of growth hormone had increased further (10.3 ± 5.5; p < 0.01). The observed increase in insulin levels with L-arginine infusion was not statistically significant (Table 2). In two subjects, an increased global CBF in the first postinfusion scan was associated with plasma insulin levels below preinfusion values. Following L-arginine infusion, no significant changes in PCO2 were observed, but arterial pH decreased from 7.40 ± 0.03 to 7.36 ± 0.01 (p < 0.01). This change persisted (7.36 ± 0.02), even when whole-brain CBF had returned to its initial values 45 min after the start of the infusion. No significant changes in pulse rate and blood pressure were observed with L-arginine infusion.
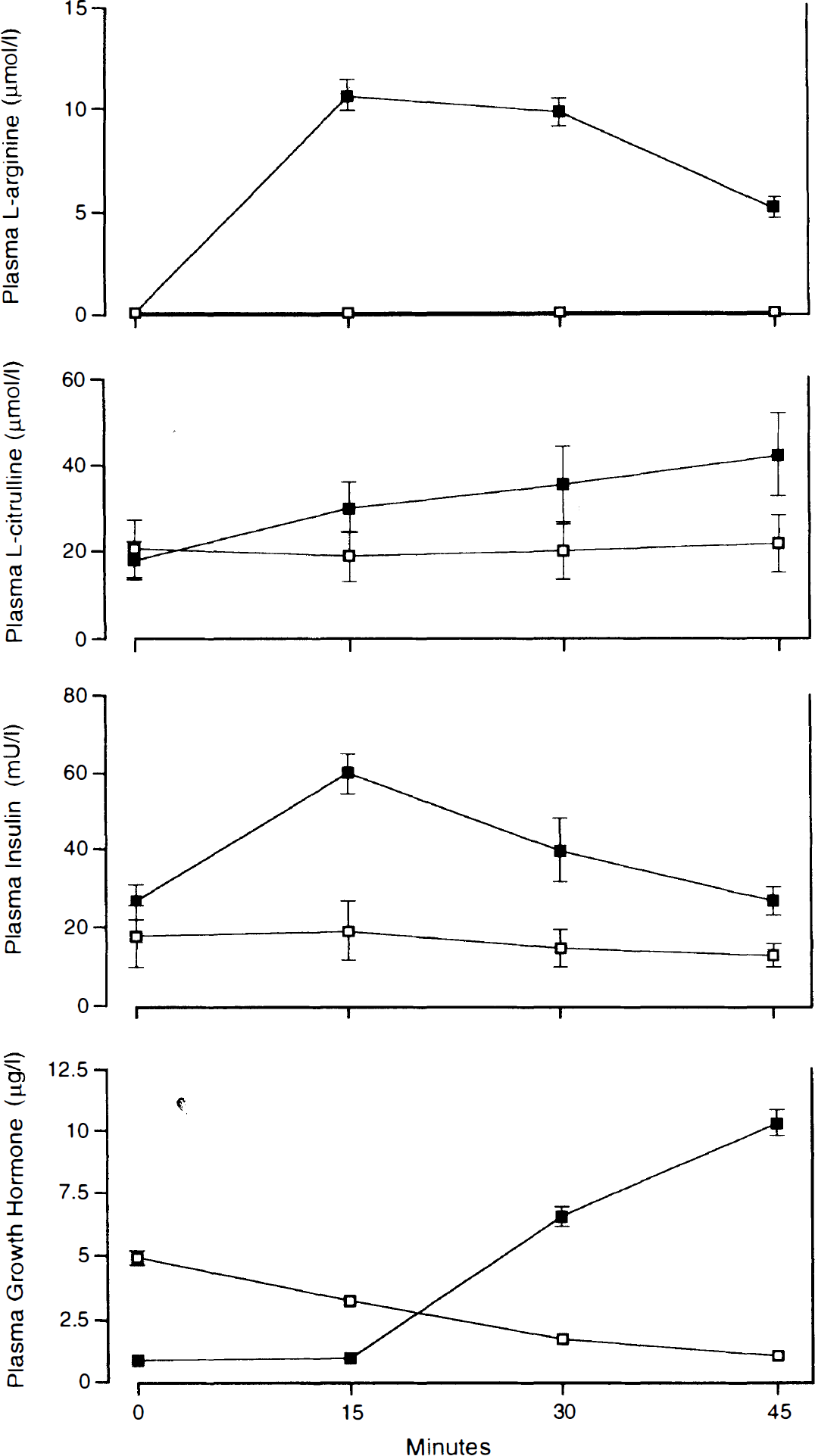
Plasma levels of L-arginine, L-citrulline, insulin, and growth hormone. The abscissa is time (min) after infusion of L-arginine (▪) or saline (□).
DISCUSSION
We observed that infusion of L-arginine produced a 9.5% increase in basal whole-brain CBF; CBF increased more dramatically in gray matter than in white matter. Activation-related blood flow was not altered by L-arginine.
A vascular effect has been demonstrated after L-arginine infusion in several vascular beds, both in vivo (Hishikawa et al., 1991; Morikawa et al., 1992; Bode-Böger et al., 1994) and in vitro (Rosenblum et al., 1990; Sun et al., 1992), although some preparations appeared unresponsive to similar treatment (Faraci, 1990; Faraci and Heistad, 1992; Kovach et al., 1992). L-Arginine induced vasodilation is likely to depend on enhanced synthesis of NO, a potent vasodilator synthesized from L-arginine by NO synthase located in endothelial cells (Gabbott and Bacon, 1993), perivascular nerves (Estrada et al., 1993), glial cells (Murphy et al., 1993), or neurons (Tomimoto et al., 1994).
Evidence for this is threefold: First, as in our study, others have observed that L-arginine infusion is associated with an increase in plasma L-citrulline, a byproduct of NO synthesis (Hishikawa et al., 1991; Smulders et al., 1994). Second, increased plasma levels and urinary excretion of cyclic GMP, the second messenger for NO, have been reported (Hishikawa et al., 1991; Bode-Böger et al., 1994; Smulders et al., 1994) after L-arginine infusion. Finally, Morikawa et al. (1992, 1994) observed that inhibition of NO synthase attenuated the vasodilator response to L-arginine in the cerebral circulation of rats. Our observations suggest that the L-arginine-NO system may play a part in the autoregulation of CBF; however, direct proof of a NO-mediated mechanism for the effect of L-arginine on CBF in humans would require administration of a NO synthase inhibitor, a procedure not currently possible.
The possibility that hyperinsulinemia mediates the vascular effects of L-arginine (Smulders et al., 1994) is not supported by our results. Blood flow increases were observed in subjects irrespective of the presence of hyperinsulinemia. This finding agrees with previous findings that L-arginine exerts vascular effects in insulin-dependent diabetics (Smulders et al., 1994). Similarly, the discordance between the time course of changes in CBF and of growth hormone secretion militates against the involvement of this hormone in the vascular effects of L-arginine. In our study, L-arginine infusion had no significant effect on arterial PCO2 but was associated with a mild reduction in arterial pH, which was still present when CBF had returned to its initial level. Changes in arterial pH in the absence of changes in PCO2 do not affect CBF significantly (Harper and Bell, 1963).
The most likely site of action of infused L-arginine is the endothelium; L-arginine does not cross the blood–brain barrier readily (Oldendorf, 1971), and endothelial damage attenuates the response to L-arginine in pial arterioles (Rosenblum et al., 1990). Furthermore, L-arginine-induced pial vasodilation can be blocked by topical nitro-L-arginine methyl ester at concentrations and time intervals that do not reduce neuronal NO synthase activity (Morikawa et al., 1992). Normal plasma concentrations of L-arginine and estimated concentrations in endothelial cells (2–4 mmol/l; Hecker et al., 1990, 1991) far exceed the Km for constitutive endothelial NO synthesis (2.9 μmol/L; Pollock et al., 1991). Thus, the suggestion that NO synthesis in endothelial cells can be increased by increasing the concentration of substrate appears paradoxical. However, by a mechanism that is presently unknown, substrate-dependence of constitutive endothelial NO synthase may be induced by physiological levels of L-glutamine (Hecker et al., 1990; Arnal et al., 1995). There may also be compart-mentalization of L-arginine in the endothelial cell, resulting in a low L-arginine concentration in the vicinity of NO synthase.
In this study, the relative increase in blood flow after L-arginine infusion was greater in gray matter than in white matter. Whether this relates simply to differences in vascular density (Göbel et al., 1990) or to regional differences in the concentration of NO synthase or the amino acid carrier in the endothelial cell (Abbott and Schachter, 1994) remains to be determined; however, a disproportionate increase in regional CBF in gray matter compared with white matter has also been described in hypercapnia (Ramsay et al., 1993; Rostrup et al., 1994).
In some experimental preparations, NO appears to mediate the increases in CBF that occur with neuronal activation (Dirnagl et al., 1993; Akgören et al., 1994; Irikura et al., 1994). In our study, L-arginine infusion did not augment activation-related blood flow despite a significant increase in global CBF. This finding is in keeping with the observation by Ramsay et al. (1993) that activation-related increases in CBF in the visual cortex show no systematic dependence on the level of global CBF. We raise three possible explanations for our observation. First, an L-arginine mechanism may be fully saturated during physiological activation. Second, neuronally derived NO, probably unaffected by L-arginine infusion, may mediate the blood flow increases with functional activation (Dirnagl et al., 1993; Akgören et al., 1994). Knockout mice, lacking neuronal NO synthase, show normal vasodilatory responses mediated by endothelial NO synthase (Irikura et al., 1995) and CBF responses to vibrissal stimulation that are no longer attenuated by NO synthase inhibitors (Ma et al., 1996). This finding led Ma et al. (1996) to conclude that endothelial NO synthase does not mediate CBF responses to neuronal activation. Third, as suggested by Adachi et al. (1994), increases in local CBF associated with somatosensory activation may not be mediated by NO at all. It is also possible that NO acts as a permissive factor rather than a mediator; NO may be necessary but not sufficient to mediate CBF increases with neuronal activation (Ma et al., 1996).
In this study, plasma L-arginine levels were still increased 45 min after infusion; yet global CBF had returned to baseline. In addition, the increase in CBF with activation was reduced in the second activation scan after L-arginine infusion. These findings may reflect the changing balance between homeostatic vasoconstrictor mechanisms and residual vasodilation after infusion. Vasoconstriction induces dissociation between glucose utilization and regional blood flow in a number of other circumstances. McBean et al. (1989) observed a reduction in CBF but not in glucose metabolic rate after administration of an exogenous vasoconstrictor. Vasoconstriction induced by hypocapnia is also associated with reductions in the CBF response to activation (Ramsay et al., 1993; Shimosegawa et al., 1995). Furthermore, during migraine attacks, both global CBF and the increase in regional CBF with functional activation are attenuated (Olesen et al., 1981; Lauritzen et al., 1983). Spreading depression in the rat is also followed by long-lasting reductions in cortical blood flow, normal glucose consumption, and reduced vascular reactivity to CO2 (Lauritzen, 1984; Lauritzen and Diemer, 1986). It is interesting to note that rebound myocardial ischemia or hypertension may occur with abrupt cessation of therapy with the NO donors nitroglycerin (deMots and Glasser, 1989) and sodium nitroprusside (Packer et al., 1979). Münzel and Bassenge (1996) observed rebound constriction in large coronary arteries after abrupt cessation of nitroglycerin infusion in dogs and also postulated that the vasodilatory effects of nitroglycerin had been removed so rapidly that counterregulatory vasoconstrictor effects, which were still operative, had become unopposed.
Our study demonstrates that L-arginine infusion can modulate basal CBF in humans. The demonstration that L-arginine infusion promotes CBF-dependent functional recovery and reduces infarct size in an experimental stroke model (Morikawa et al., 1992; Dalkara et al., 1994; Morikawa et al., 1994) raises prospects of a novel therapeutic strategy in human cerebral ischemia. Although the duration of the effect of infused L-arginine may be too short to be clinically useful, the effects of other NO precursors and of oral L-arginine supplementation certainly warrant further examination.
Footnotes
Acknowledgment:
Dr. Reutens was supported by a Neil Hamilton Fairley Fellowship of the National Health and Medical Research Council of Australia. We thank the technical and cyclotron staff of the McConnell Brain Imaging Centre for their tireless efforts and Dr. Edith Hamel for reviewing the manuscript.
