Abstract
This study presents the first in vivo measurement of transcytolemmal water exchange in the brain using a novel Magnetic Resonance technique. We extend previous applications of Chemical Exchange Saturation Transfer (CEST) to examine water exchange across cellular membranes in late-stage chicken embryo brains. The immature blood-brain barrier at this stage allows Gadolinium-Based Contrast Agents (GBCAs) to penetrate the brain’s interstitial space, sensitizing the CEST effect to water exchange between intra- and extracellular environments. Exchange rates were measured in the awake brain and under different anaesthetic regimens, including isoflurane and ketamine/xylazine. Results show that brain water exchange is dominated by activity-dependent mechanisms, with anaesthesia reducing exchange rates by over an order of magnitude. These findings suggest that anaesthetics may impact neuronal and glial function by interfering with active transport mechanisms, potentially altering brain water homeostasis. This study highlights the utility of CEST MRI for studying dynamic biological processes in vivo.
Introduction
The brain is composed of approximately 80% water and relies on complex mechanisms to balance and transport water to support tissue function and maintain homeostasis.1 –3 This interplay between water flow and exchange across various brain compartments, including blood, cerebrospinal fluid, and intracellular and interstitial spaces, is crucial for vital neurological processes such as cell volume regulation, ion balance, and modulation of neuronal activity. Dysregulation of water transport pathways is implicated in numerous brain pathologies, ranging from cerebral edema to traumatic injuries.4 –7
Transcytolemmal water cycling, i.e. the exchange of water across cell membranes, involves a combination of passive and active mechanisms.8,9 The traditional view of passive water diffusion across the phospholipid membrane has evolved with the discovery of aquaporins, membrane proteins that facilitate water exchange. 10 Additionally, membrane proteins actively transfer water molecules alongside ions or metabolites.11 –15 By way of example, it is estimated that each cycle of the Na+-K+-ATPase pump (NKA).16,17 co-transports >100 water molecules. Hence, water cycling in neuronal tissue also involves energy-consuming, activity-dependent processes.
Despite the critical role of transmembrane water exchange in the maintenance and regulation of brain function, our ability to assess this process in the living brain is still constrained. Indeed, non-invasive techniques such as positron emission tomography (PET)18 –20 and magnetic resonance (MR)21 –23 are primarily sensitive to net flows or slowly exchanging water pools, but less sensitive to fast or ultra-fast exchange occurring across cellular membranes. PET tracer kinetic studies using 15O-water make it possible to study brain perfusion and water transport through the Blood Brain Barrier (BBB),20,24 but are limited in their sensitivity to transmembrane exchange. Magnetic resonance imaging techniques, traditionally based on detection of water protons in the various compartments, characterized by different physical and chemical environments, are more promising. Quantitative blood-oxygen-level-dependent (qBOLD) MRI 25 exploits the slight difference between the intrinsic transverse relaxation times (T2*) of the intra- and extracellular pools, often in combination with arterial spin labelling (ASL). 26 However, the small difference in relaxation times between the different microenvironments limits the sensitivity to exchange rates of the order of 10 s−1 ref. Other MR methods differentiate between intracellular and extracellular water by measuring their distinct apparent diffusivities.27 –29 A recent MR approach dubbed Diffusion Exchange SpectroscopY (DEXSY) enabled measurements of water exchange rates of the order of 100 s−1 in ex vivo tissue preparations, with a promising prospect of extension in vivo.
Alternative MR methods have relied on the introduction of exogenous gadolinium-based contrast agents (GBCAs) to amplify the difference in relaxation times, specifically T1, between intra- and extracellular water, 16 thus increasing the sensitivity to fast water exchange. Although this strategy is highly effective, it is challenging to apply in vivo in brain studies because GBCAs ordinarily do not cross the blood-brain barrier (BBB). Application of these methods in brain studies involve invasive procedures, like direct infusion into the cerebral ventricles 30 or osmotic breakdown of the BBB or transient opening of BBB by ultrasound stimulation, 31 to allow diffusion of GBCAs into the extracellular space in the parenchyma.
An MRI application highly sensitive to very rapid exchanges in the hundreds Hz range is Chemical Exchange Saturation Transfer (CEST).32,33 It operates by selectively saturating resonances of molecules that exchange protons with surrounding water. This exchange alters the water signal, enabling indirect detection of the target molecules. Within cells, various endogenous species contain exchangeable protons exploitable in CEST experiments. Two signals commonly observed in biological tissues resonate at 3.5 ppm away from the water due to protein amide groups (amide proton transfer, APT) and at 2.2 ppm attributed to creatine and generic amines.34,35 The longer the relaxation time of protons involved in species exchange, the stronger the CEST effect.
Recently, a Chemical Exchange Saturation Transfer (CEST)-based approach has been proposed for measuring water exchange across the membranes of red blood cells and cancerous cells. 36 This method leverages the compartmentalization of contrast agents, which are confined to the extracellular space. The influence of the contrast agent on the T1 relaxation time of intracellular water is contingent upon transmembrane water exchange. Consequently, the intracellular CEST effect is directly modulated by transmembrane water cycling (Figure 1, panel a). The frequency-dependent saturation of the water signal can be plotted as a function of the irradiation frequency, with the resonance frequency of water protons conventionally set to 0 (the z-spectrum, shown in Figure 1, panel b). The presence of exchangeable proton pools results in an asymmetrical z-spectrum (Figure 1, panel b). The asymmetry of the spectrum can be quantified by an asymmetry parameter (MTRasym), obtained by subtracting the signal intensity ratios at positive and negative frequency offsets from the central water resonance. This procedure makes it possible to measure the CEST effect while mitigating potential non-CEST contributions and radiofrequency field strength variability. A three- compartment model (comprising a pool of exchangeable protons, and two pools of intra- and extracellular water, respectively) makes it possible to extract quantitative information about water exchange rates.
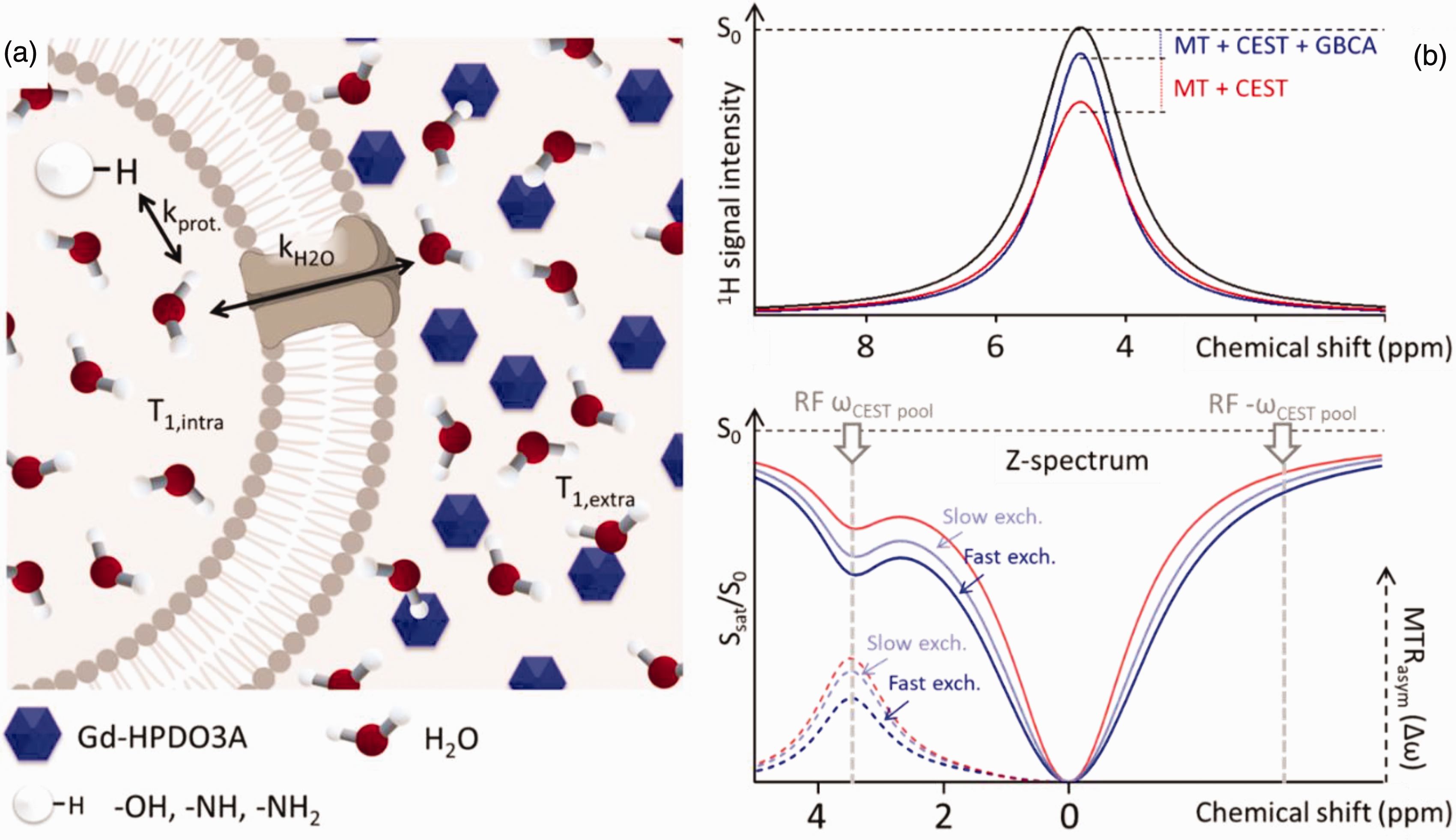
(a) Schematic representation of the three compartments (pool of exchangeable protons, intracellular water, extracellular water). Gadolinium is confined to the extracellular space, where the T1 of water protons is shortened by interactions with the paramagnetic agent. The T1 of the intracellular water is affected by extracellular gadolinium due to water exchange across the membrane. Hence, the intracellular water T1 depends on the trancytolemmal water exchange rate kH2O. The CEST response from the intracellular water and the pool of exchangeable protons is strongly dependent on water T1 and can be exploited to indirectly measure kH2O and (b) The CEST contrast enhancement mechanism is illustrated with a 3-site exchange between a CEST pool, intracellular water, and extracellular water with GBCA (blue and light blue line) with and without GBCA (red line). Radiofrequency (RF) saturation is applied at the resonance frequency of the labile CEST protons (ΔωCEST pool), leading to a loss of net magnetization. These saturated protons (red lines) from the CEST pool then exchange with unsaturated protons (black line) from the much larger water pool with an exchange rate kprot, resulting in an accumulation of saturated protons in the intracellular water pool. The accumulation of zero net magnetization of CEST protons in water causes a decrease in the total water signal. While the saturation pulse is applied, this process continues to decrease the water magnetization through the CEST effect as well as through magnetization transfer (MT) and direct water saturation effects. A saturation pulse applied at the corresponding reference frequency symmetrically at the opposite side of the water resonance (−ΔωCEST pool) will decrease the water magnetization through MT effects only. Saturation transfer effects can be assessed using a z-spectrum where the water signal is plotted as a function of saturation frequency. Asymmetry analysis (MTRasym) is performed by subtracting the water signal from one side of the z-spectrum from the other side to emphasize the effects of chemical exchange (dotted lines). Here the water resonance frequency is used as the centre frequency and assigned the chemical shift of 0 ppm as opposed to in NMR spectra, where water protons have a chemical shift of 4.7 ppm. When extracellular water undergoes the effect of Gd-HPDO3A (blue and light blue lines), its relaxation time shortens causing its magnetization, saturated by the radiofrequency pulse of the CEST sequence, to reconstitute more rapidly. The more rapidly water exchanges across the membranes, the stronger the effect of extracellular Gd on the intracellular CEST effect. The dependence on transmembrane water exchange rate is shown by comparing the two curves where GBCA is present in the extracellular compartment. Under otherwise identical conditions, the slower exchange is represented by the light blue line, while the faster exchange is depicted by the blue line.
In this study, we have extended this novel Chemical Exchange Saturation Transfer (CEST) technique to investigate transmembrane water exchange in the brains of chicken embryos (Gallus gallus domesticus) in ovo, at a late developmental stage (day eighteen). The chicken, being a precocial species, has a brain that is already well-developed and structured at this stage (Figure 2, panel a). 37 However, the blood-brain barrier (BBB) is not yet fully closed, allowing systemically administered GBCAs to penetrate the brain parenchyma and disperse into the extracellular space. Contrast agents and other pharmaceuticals can be readily introduced through the chorioallantoic membrane (CAM), an extensively vascularized extraembryonic membrane that serves multiple functions during embryonic development, including gas exchange (Figure 2, panel b). Hence, the chick embryo represents an ideal model to study transcytolemmal water cycling with our CEST approach.
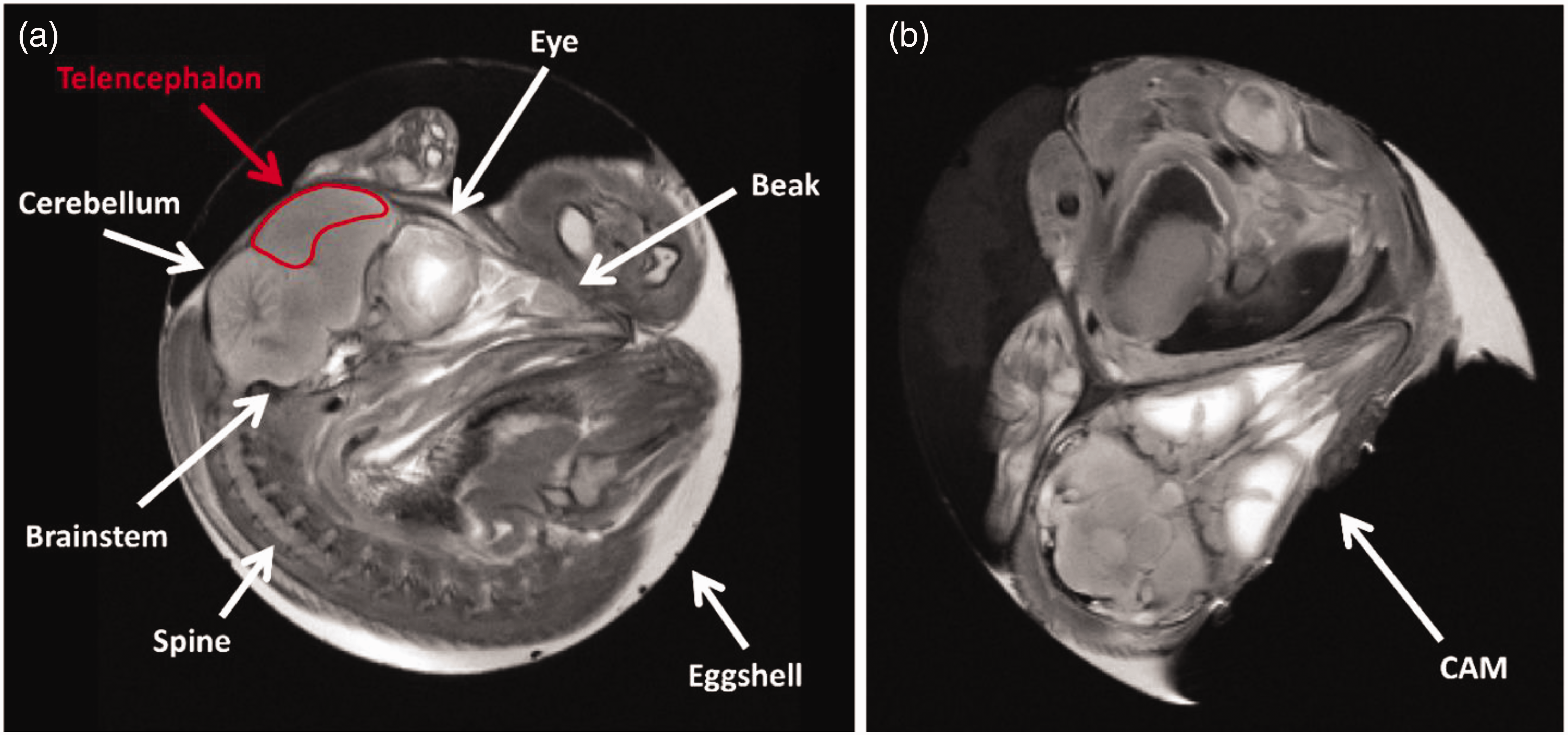
(a) Morphological images (sagittal view) of a chicken embryo inside the egg, obtained with a T2-weighted sequence acquired using 7 Tesla MRI on embryonic day 18 (E18). The red ROI indicates the area from which data for the z-spectrum were obtained, the telencephalon. The ROIs designed for signal analysis were delineated conservatively to ensure the observation of contributions solely from the telencephalon and (b) T2-weighted morphological images (coronal view) of a chicken embryo inside the egg acquired at 7 Tesla MRI on E18. The chorioallantoic membrane (CAM) is visible in the bottom right portion, corresponding to the air sac.
Previous research utilizing ex vivo models has highlighted the significant role of active, ATP-consuming mechanisms in facilitating water transport across cellular membranes. Hence, general anesthetics, which are known to modulate baseline neuronal activity, may influence water cycling dynamics. However, the impact of these substances on transmembrane water exchange has not yet been quantified. To address this gap, we have employed our CEST to investigate the effects of various anesthetic protocols on transcytolemmal water cycling in the chick embryo. The anaesthetic agents hereby investigated include both gaseous and injectable agents, specifically isoflurane and a combination of ketamine/xylazine, respectively.38,39 These compounds, designed to temporarily alter consciousness and diminish pain perception, act through different mechanisms involving specific receptors, ion channels, and metabolic pathways within neurons and glial cells. The present study provides, for the first time, a quantitative assessment of their effects on water cycling and homeostasis under in vivo conditions.
Material and methods
Embryonic manipulations and treatments
Fresh fertilised eggs of the Aviagen Ross 308 strain (Gallus gallus domesticus; provider: Azienda Agricola Crescenti Brescia, Italy) were incubated in the laboratory under constant temperature and humidity (37.7°C; 60%; incubator: FIEM CIPCIP 28 MINILED). This study used pre-hatch avian embryos (chicken embryos), which do not require specific authorization for animal experimentation under Italian regulations.
Initiating around day 7 post-fertilization and maturing by day 12, the CAM's high vascularization allows convenient substance administration, including drugs, dyes, or other chemicals, for studying embryonic development. Various methods have been employed since 1938 for precise drug delivery into the vascular system, such as injection into chorioallantoic veins40 –42 or directly through the CAM, 43 the latter involving puncturing the eggshell at the air sac location to allow substances to drip onto the membrane. Sealing the puncture with paper tape, eggs can be repositioned in the incubator, facilitating substance absorption into the developing embryo's bloodstream.
In the recently established MEMRI protocol by Lorenzi et al. 43 for detecting asymmetric embryonic brain activity in ovo, the manganese is administered through droplets onto the CAM. Following the same protocol, varying amounts of Gd-HPDO3A were administered on E17. the eggshell was pierced in correspondence to the air sac. 100 μL of Gd-HPDO3A 0.5 M (Gadoteridol, ProHance®, Bracco Imaging S.p.A.) were dropped on the CAM. Paper tape was used to seal the eggshell again and eggs were placed back in the incubator.
All MRI measurements were performed at E18. The measurements included the acquisition of anatomical reference images, quantitative T1 maps, and CEST z-spectra. The experimental groups consisted of 6 chicks each.
Control groups
One group did not receive GBCAs and was measured under awake conditions (Ctrl group).
The same group was then measured post-mortem after being sacrificed with CO2 (Ctrl CO2 group).
Gadolinium-based contrast agents (GBCA) groups
One group of 6 chicks received GBCAs and was measured under awake conditions (Gd group).
One group received GBCAs and was anesthetized with inhalational anaesthesia (isoflurane, Gd iso).
Another group received GBCAs and was anesthetized with systemic anaesthesia (xylazine and ketamine, Gd x + k).
Post-Mortem measurements
From the Gd iso and Gd x + k groups, 6 subjects were randomly selected, sacrificed with CO2, and measured again (Gd CO2 group).
Recovery
Four subjects from the Gd iso group were allowed to recover after anaesthesia prior to an additional MRI session.
In the case of experiments indicated with Ctrl and Gd, the eggs were moved to a 4°C refrigerator for 30 minutes before being placed in the MRI scanner; this procedure ensures that the slight hypothermic state of the embryo precludes movement during scanning.
For isoflurane anaesthesia, the commercial veterinary anaesthesia system Vevo Compact Dual Anesthesia System® (Fujifilm|Visualsonics, Toronto, Ontario Canada) was utilized. The vaporizer was adjusted to deliver an isoflurane concentration of 5% with oxygen as the carrier gas at a flow rate set to 0.6 L/min. 38 To induce anaesthesia, the egg was placed inside a transparent food freezing bag (Cuki GELOPIÙ®) connected to the Anaesthesia System base system via two gas inlet and outlet tubes, and the bag was then sealed with paper tape. The narcosis induction chamber has one inlet and one charcoal-filter-connected outlet to maintain a constant narcotic concentration. To achieve the desired isoflurane concentration of 5% within the bag before initiating anaesthesia induction, it was flushed for 5 minutes with an oxygen flow of 5 L/min and an isoflurane concentration of 5%.
The xylazine + ketamine anaesthetic (0.125 mg of xylazine and 5 mg of ketamine administered per egg) was applied in a liquid form directly onto the chorioallantoic membrane (CAM). 39 A hole (approximately 2.5 mm in diameter) was drilled into the eggshell, and the CAM was exposed to enable administration. Subsequently, the hole was sealed with paper tape. In the case of eggs that had already been administered gadolinium, the same hole created the day before was used for the administration of the anaesthetic. The anaesthetic regimen was based on the dose used in chick embryos38,39 and other birds,38,44 considering the weight of an embryonated egg of 55–60 g and a chicken embryo of 20–22 g.
In experiments conducted with isoflurane and xylazine+ketamine, the egg was placed on an MRI-compatible heated pad to maintain the embryo at physiological temperature during the acquisitions.
Before and after each experiment, the eggs were candled to visually check for embryo motion. The impact of anaesthesia was assessed based on both MRI image quality, which is very sensitive to motion, and embryo recovery after the experiment. The image quality criterion required a stable embryo position within the egg, allowing for minimal motion artifacts such as those caused by beak opening/closing.
After completing the acquisitions for live embryos, the eggs were placed in a Small Animal Rodent Euthanasia Chamber and exposed to 90% CO2 for at least 20 minutes before being used for post-mortem acquisitions.
Image acquisition
Images were acquired at 7 T on a Bruker Pharmascan with a 72 mm i.d. quadrature transmitter-receiver birdcage resonator.
First, fast positioning imaging was conducted using a T2-weighted fast spin echo sequence with a large FOV and thick slice (TR/TE 1500/60 ms, FOV 54 mm, matrix 128 × 128, 15 slices with a thickness of 1.5 mm, lasting 43 seconds), to obtain standard axial, sagittal, and coronal brain planes.
Prior to the acquisition of CEST and T1 maps, shimming and flip angle local adjustment were performed on the region of interest assigned to the telencephalon of the chick.
For CEST-MRI analysis, a typical RARE sequence was employed (TR/TE 5000/6.5 ms) with an isotropic 120 × 120 acquisition matrix, FOV 54 mm, and a slice thickness of 1.5 mm, using 64 RF excitations. The entire sequence was preceded by a saturation scheme comprising a continuous rectangular wave pulse lasting 3 s, with an RF intensity corresponding to B1 = 3 μT (other TR and B1 powers were tested). A frequency offset range of ±100 ppm relative to the water signal was investigated.
T1 maps images were obtained using a saturation recovery sequence with 6 TR values (RARE VTR sequence, TR = 0.1, 0.5, 1.5, 2.5, 4.5, 7.0 s, TE = 6 ms, RF = 4), and the T2 maps images were obtained using (MSME sequence, TR/TE 5000/6.5 ms, with 256 variable TE images), with an isotropic 64 × 64 acquisition matrix, FOV 54 mm, and a slice thickness of 1.5 mm. Anatomical reference images were acquired with a RARE sequence (TR/TE 2500/60 ms) with an isotropic 256 × 256 acquisition matrix, FOV 54 mm, and a slice thickness of 1.5 mm, using 16 RF excitations. The CEST, T1, T2 maps, and the anatomical images of each embryo were spatially co-registered.
Each type of experiment required approximately 20–30 minutes from the insertion of the egg into the MRI scanner to the completion of the acquisitions. In this time window, the egg was placed in a foam pad enclosure to further attenuate mechanical vibrations induced by imaging gradient switching and inserted into the birdcage coil for MR scanning procedures.
Image analyses
The region of interest (ROI) was manually segmented to include the entire chick telencephalon on the anatomical reference image of each animal using the software Fiji – ImageJ (https://imagej.nih.gov/ij/download.html and https://imagej.net/Fiji/Downloads). The signal intensity and standard deviation of the signal were then extracted and imported into OriginPro 2016 for data fitting.
CEST z-spectra model for simulations and fitting
For simulating and analyzing the z-spectra data, a modified version of the full analytical solution of the underlying Bloch–McConnell equations for both magnetization transfer (MT) and chemical exchange saturation transfer (CEST) was utilized, which includes an additional pool representing extracellular water. This approach, proposed in 2014 by Moritz Zaiss et al. 45 and implemented in Mathematica (Wolfram Research, Inc., Mathematica, Version 11, Champaign, IL) and OriginPro (OriginLab Corporation, Northampton, MA, USA, 2016), provides insights into their interaction and suggests a simple means to isolate their effects. The analytical solution, based on the eigenspace solution of the Bloch–McConnell coupled differential equations, describes the prototropic exchange between the CEST pool and the intracellular water, and the simultaneous exchange of intra- and extracellular water (see Figure 5).
Prior to fitting, all z-spectra were normalized to the signal obtained in the ROI at +/− 100 ppm from water.
As shown in Figure 5, the system under study was simplified into three compartments. The first is an extracellular water pool, where no prototropic exchange is assumed, and where the water exhibits distinct T1extra and T2extra relaxation parameters that can be influenced by the presence of gadolinium (Gd).
The second compartment is the intracellular water pool, characterized by T1intra and T2intra, and subjected to two distinct exchange mechanisms: the transmembrane exchange of water molecules and the prototropic exchange with protonic groups of molecules within the intracellular space. The resonance frequencies, or chemical shifts, of intra- and extracellular water are considered identical.
The third pool, the CEST pool, represents the various macromolecules within the intracellular volume that contain exchangeable protons with surrounding water molecules. Three types of prototropic exchanges were identified in the CEST pool: groups resonating at approximately 3.5 ppm from the water resonance due to protein amide groups (amide proton transfer: APT); groups resonating at 2.2 ppm, attributed to creatine and generic amines; and the –OH exchange group resonating at 0.8 ppm from water. Additionally, a fourth fictitious pool was introduced to account for the observable NOE effect at −3.5 ppm from the water signal.
Each Z-spectrum curve was obtained by averaging measurements from 6 embryos. The curves were simultaneously fitted, sharing parameters related to the CEST pools (T1,CEST, T2,CEST, kprot, proton fraction), and fixing the values related to the relaxation characteristics of intracellular and extracellular water, while independently varying kH2O for the various curve, each corresponding to a different condition. In this context, we have defined kH2O as the water exchange rate across cell membranes and kprot as the proton exchange rate within the CEST pool.
T1 and T2 values were measured for each chick, and the averages of these values were used to establish T1 and T2 in the fitting equation. The intracellular T1intra and T2intra values for all curves and the extracellular T1extra and T2extra values for embryos not treated with GBCA were set to the average T1 and T2 values obtained from the telencephalons of control embryos (not administered with GBCA).
To establish the extracellular T1extra of embryos administered with GBCA at E17, the parenchyma T1 value was measured post GBCA administration, and the effects of Gd on the extracellular water pool were estimated assuming an extracellular volume of approximately 20%.
Results
GBCA penetrates in the cerebral tissue of the chick embryo at E18
All experiments were conducted on chicken embryos in the final stages of development, on the eighteenth embryonic day (E18) of a 21-day developmental period. 37 Details on the experimental design are reported in the Methods section.
For quantifying the penetration of GBCA into the extracellular space of the telencephalon, 12 embryos were divided into two cohorts. Six chicks were included in the control group (Ctrl) and did not receive Gd. In the remaining chicks (Gd), 100 µL of Gd-HPDO3A (500 mM) was administered into the CAM, leading to an average T1 reduction across the telencephalon of approximately 60% (from 2.0 ± 0.1 s to 0.9 ± 0.1 s). Assuming a 20% extracellular volume of the total observed ROI 46 and considering a relaxivity of 3.3 mM−1 s−1 reported for this contrast agent at 7 Tesla in human plasma, 47 a Gd-HPDO3A concentration in the extracellular space of 0.71 ± 0.21 mM can be estimated.
A significant finding is the observation that the concentration of Gd-HPDO3A in the extracellular space in the telencephalon remains constant for at least one day after administration in the CAM. This observation contrasts with prior research on adult mammals, where continuous infusion of gadolinium-based contrast agents (GBCAs) into the cerebral ventricles was necessary during MRI. This was due to the rapid renal excretion of gadolinium, which decreased its concentration in the brain parenchyma.
Exchange rates are linked to membrane permeability and cells activity in brain tissue
To measure water exchange rates, chick embryos were examined on E18 after administration of GBCA under different conditions of brain activity. The embryos were randomly assigned to different experimental groups (6 subject each), either awake, anaesthetized with inhalational anaesthesia (isoflurane), anaesthetized with systemic anaesthesia (xylazine and ketamine), and after sacrifice with CO2. These conditions are referred to as Gd, Gd iso, Gd x + k, Gd CO2, respectively. The T1s measured in the telencephalon for all experimental groups are reported in Figure 3(a). Z-spectra were acquired for all conditions across the same region (Figure 4(a)), and the asymmetry spectra were calculated (Figure 4(b)).
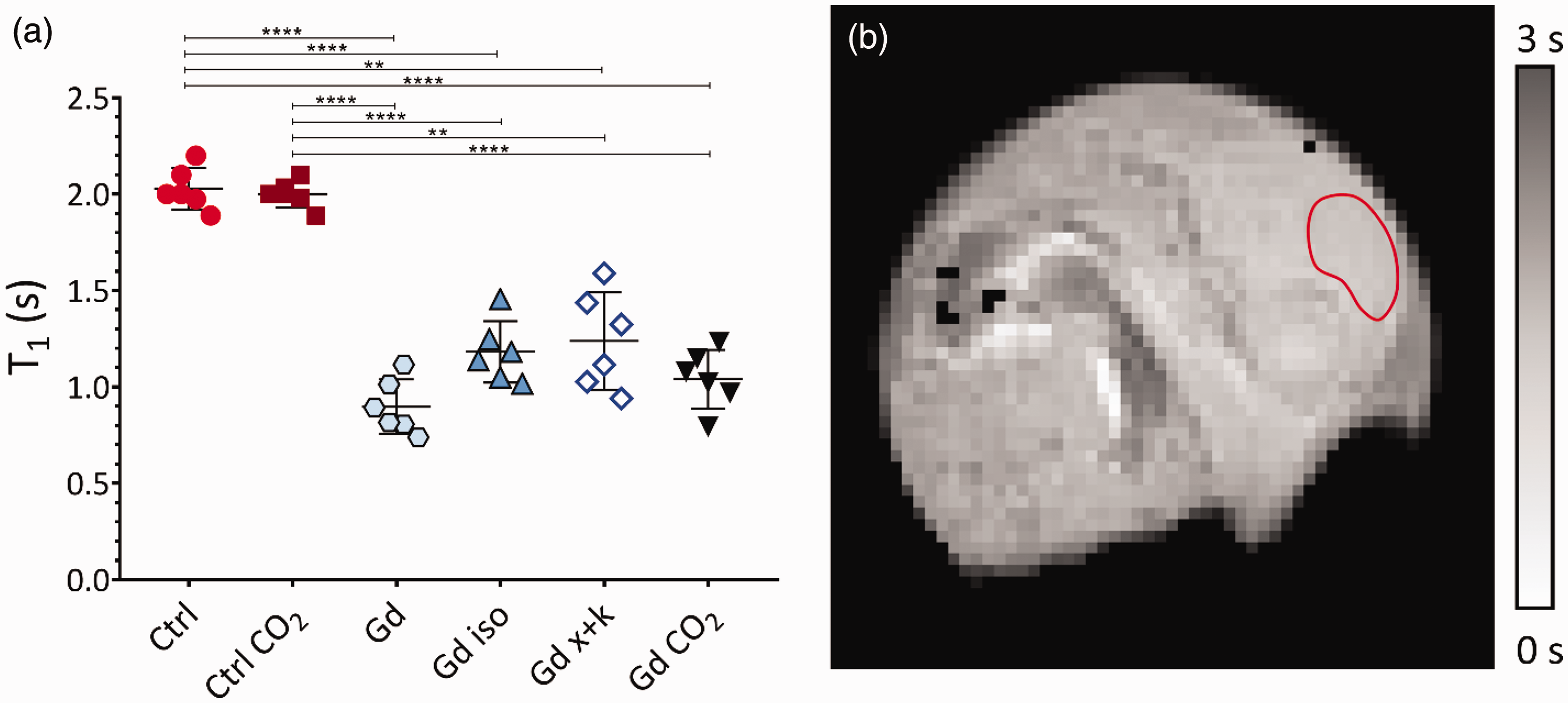
(a) Graph displaying the T1 values obtained through T1-map acquisition with and without the administration of Gd-HPDO3A, acquired immediately after the CEST image acquisition. Individual values, mean, and standard deviation for the six analysed conditions are depicted. The asterisks denote the significance of the difference between the groups. For P > 0.05, no symbol is shown; ** indicates P < 0.005, and *** indicates P < 0.0001 and (b) A T1 maps obtained for an embryo after administration of Gd-HPDO3A. The T1 appears homogeneous across the telencephalon thus suggesting a uniform distribution of Gd in that region. The red ROI, conservatively delineated, indicates the telencephalon area from which data for the T1 values and z-spectrum were obtained.
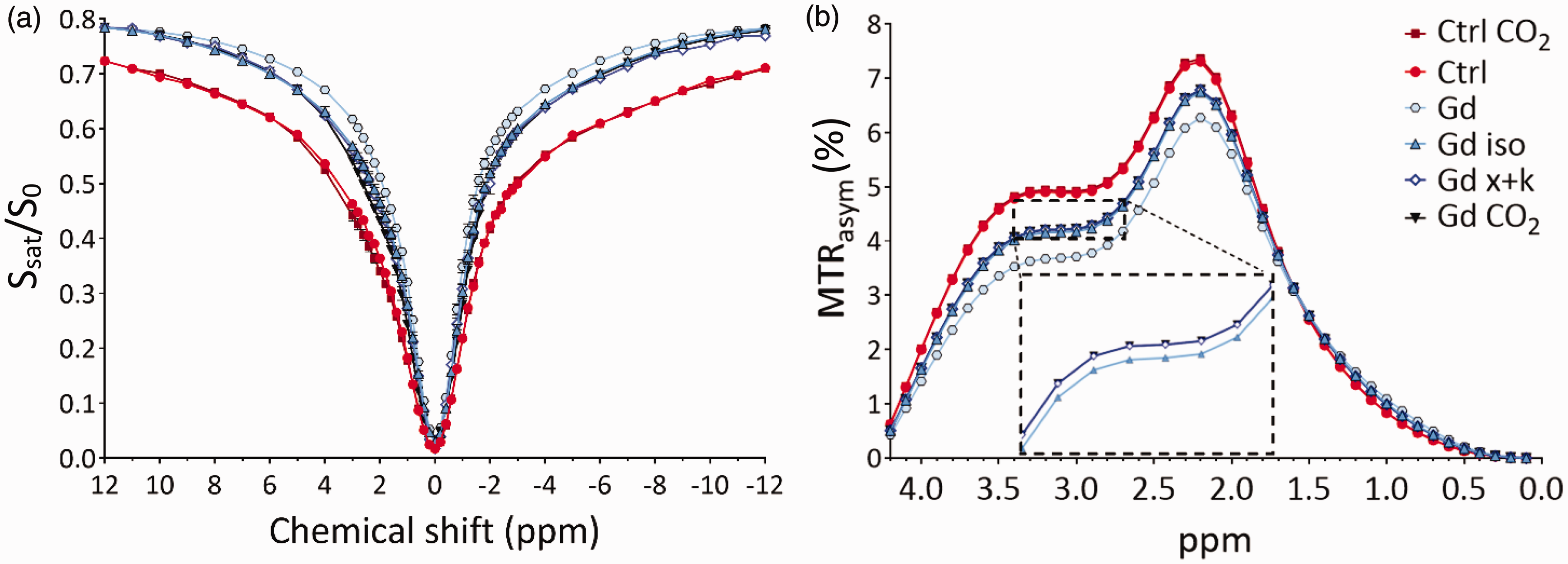
(a) Z-spectra obtained for the telencephalon of chicks without the administration of Gd-HPDO3A in vivo and post-mortem (red and dark red lines), and then with the administration of Gd-HPDO3A in vivo, under the effect of two different anaesthetics and post-mortem (light blues, blue, and black lines). n = 6 for each condition and (b) Corresponding MTRasym(%)-spectra highlighting the effect of water exchange rate across the cell membrane in the presence of Gd-HPDO3A and under different conditions. The inset shows magnification of a portion of the spectrum to highlight the small differences between the Gd iso (light blue upward triangles) and Gd x + k groups (empty blue diamonds). The data from the Gd CO2 group (black downward triangles) nearly overlap with those from the Gd x + k group (empty blue diamonds). The difference between the Ctrl and Ctrl CO2 groups, when Gd is absent, is within the margin of the symbol size used to represent the data points.
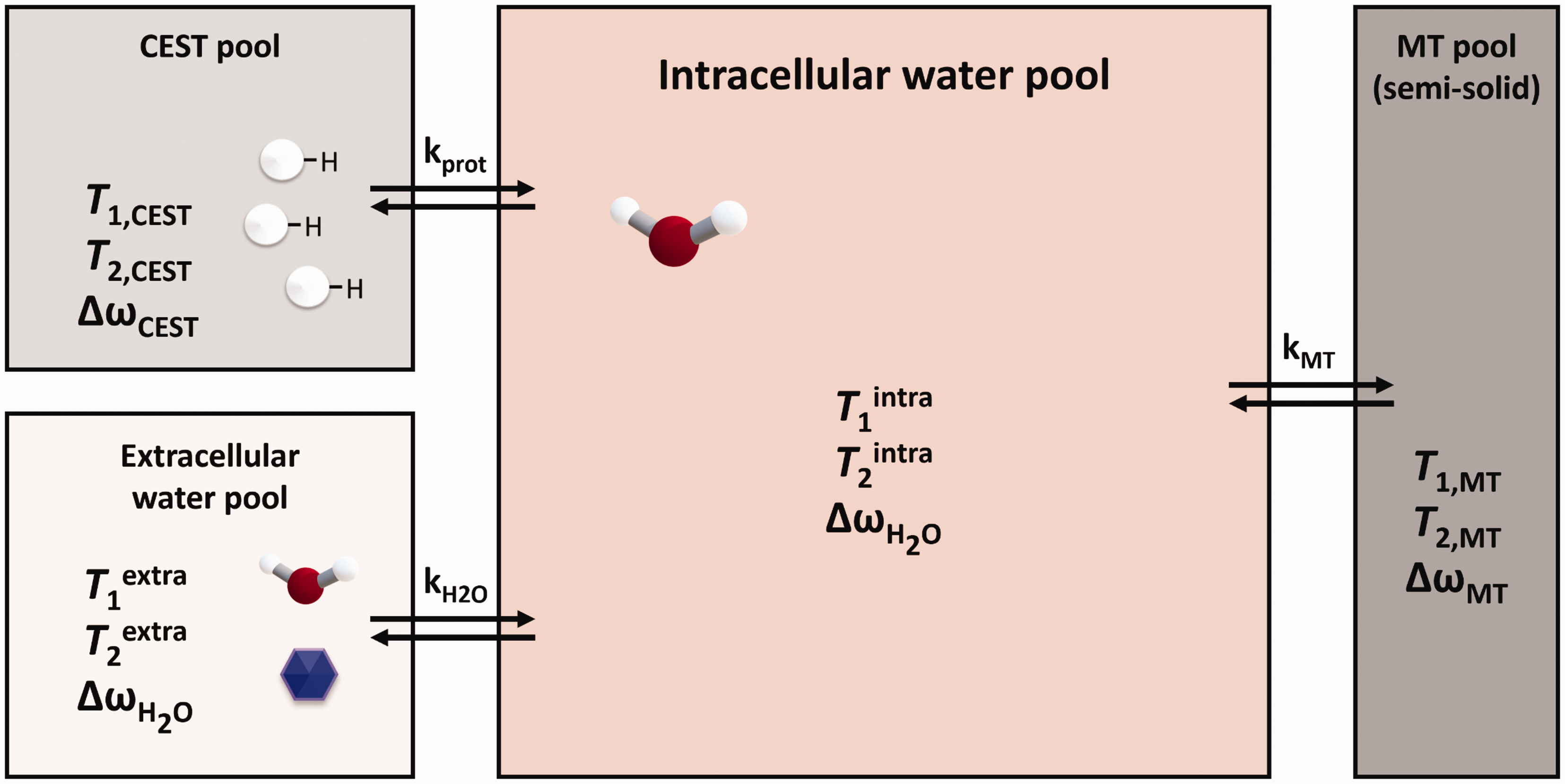
The z-spectra (referenced in Figure 4, Panel a) were analysed using the Bloch-McConnell equations. The system was conceptualized into three compartments: extracellular and intracellular water pools, and the CEST pool. The CEST pool comprises different species involved in intracellular prototropic exchange with water: amide proton transfer (APT) at 3.5 ppm, amines/creatine at 2.2 ppm, and –OH groups at 0.8 ppm. Additionally, to prototropic exchange, the model considers magnetization transfer by the Nuclear Overhauser Effect (NOE). The presence of gadolinium-based contrast agents (GBCA) in the extracellular space was accounted for using experimentally obtained variations in T1 and T2 values induced by the presence of Gd, as determined from relaxation time maps.
The six control chicks that had not received GBCA were examined to obtain telencephalon z-spectra without the influence of Gd, both awake and post-sacrifice with CO2 (referred to as Ctrl and Ctrl CO2).
The normalized z-spectra reported in Figure 4 were analysed using a full analytical solution of the underlying Bloch-McConnell equations for both magnetization transfer (MT) and chemical exchange saturation transfer (CEST), taking into account three pools of intracellular prototropic exchange (APT at 3.5 ppm, amines and creatine at 2.2 ppm, and –OH groups at 0.8 ppm from water), the Nuclear Overhauser Effect (NOE), and the presence or absence of GBCA in the extracellular space using the T1 and T2 values obtained from the relaxation time mapping experiments. A description of the model il reported in the Method section. This model was used to extract the water exchange rates across the cell membranes for all conditions, reported in the following table (Table 1). Interestingly, both anaesthetic regimens resulted in a drop-in exchange rate by an order of magnitude. Immediately after sacrifice with CO2, the post-mortem exchange rate was reduced by two orders of magnitude compared to the baseline value.
Water exchange rates in the telencephalon of chicks under different conditions.

The table presents the water exchange rates across the cell membrane, kH2O, measured in the telencephalon of non-anaesthetized chicks (Gd), chicks anaesthetized with isoflurane (Gd iso), chicks anaesthetized with xylazine and ketamine (Gd x + k), and post-mortem chicks (Gd CO2). For each experimental condition, the mean value and standard error for a group of n = 6 animals are reported. An ANOVA test was performed and the comparison between the Gd group and the other groups shows a P value < 0.0001 for all cases.
A saturation power of 3 μT was selected for this study based on preliminary experiments and simulations to achieve sufficient CEST contrast while minimizing the effects of direct water saturation. Indeed, excessively high saturation powers may reduce sensitivity to T1 due to the offsetting influence of direct water saturation and relaxation, and insufficient power may fail to provide adequate contrast and sensitivity for detecting fast transmembrane water exchange.48,49 Our results indicate that this power level was optimal for capturing exchange dynamics while minimizing non-specific contributions.
Reversibility of the effects of anaesthetics on water exchange rates
A subgroup of four chicks, subjected to isoflurane anaesthesia, was allowed to recover for 2.5 hours. The reversal of the anaesthetic effects was assessed by monitoring the animals' mobility and responsiveness to light stimuli. After full recovery, the CEST experiment was repeated. The water exchange rate returned to a value statistically equivalent to the baseline measured in the same awake group (refer to the Supplementary Information document for further details).
Discussion
This study enabled the first measurement of fast water exchange rates across the cellular membrane in the living brain. By administering gadolinium into the extracellular space, we could distinguish between water pools on either side of the membrane. A novel CEST technique, highly sensitive to rapid water transmembrane exchange, provided precise measurements of exchange rates ranging from a few Hz to hundreds of Hz.
MRI has been employed in various ways over the past twenty years to attempt to measure transmembrane water cycling. Early studies yielded an estimation of exchange rates of a few Hz, with reported values ranging from 1 to 5 Hz.8,16,26,30,50 These reported values can be attributed to other methods being sensitive to slowly exchanging water pools associated with larger or less permeable membrane structures, such as cell bodies and myelinated axons, but insensitive to rapidly exchanging pools.
It was only in 2023, 51 in mouse spinal cords isolated and placed in a dissecting chamber perfused with cold low-calcium high-magnesium artificial cerebrospinal fluid, that exchange rates greater than 100 Hz were reported. These rates can be associated with the activity of neuronal and glial cell processes that comprise the majority of grey matter tissue volume. In the same study, the highest exchange rate measured was 228 Hz for samples at 35°C, with the expectation of even greater exchange rates at 37°C in vivo. The exchange rates of water measured in the present study in vivo in awake subjects are of the same order of magnitude, albeit faster by a factor 2.
As expected, exchange rates drop drastically after sacrifice, consistently with the idea that transcyotolemmal water exchange is dominated by active transport mechanisms. A reduction in water cycling rates by an order of magnitude was observed under the influence of two different anaesthetic agents. One plausible explanation for this sharp reduction in water cycling rates is the decrease in active transmembrane transport of ions induced by general anaesthetics.52 –54 General anaesthetics significantly reduce neuronal activity, though the extent of this reduction varies depending on the type of anaesthetic and its concentration. Volatile anaesthetics like halothane, isoflurane, and enflurane can reduce neuronal firing rates in organotypic slice cultures by approximately half at concentrations about one-half the EC50 for general anaesthesia. Decreased spontaneous firing of neurons via intracortical mechanisms was also found in vivo; concentrations of isoflurane inducing hypnosis in humans reduced discharge rates by approximately 50%. 55
Intravenous anaesthetics have also been found to reduce the neuronal firing rate by approximately 40–60% in rat lumbar dorsal horn neurons. 56 Under pentobarbital/chloral hydrate anaesthesia, only 29% of neurons in the rat auditory cortex remained active. 57 Another study found that neuronal firing rates in the rat auditory cortex can be reduced by 6–60% in a dose-dependent manner with increasing doses of propofol injected intravenously, further supporting the significant impact of anaesthetics on neural activity. 58
Ketamine's effects on neuronal activity are more diverse and strongly dose-dependent, reflecting its complex pharmacological profile and the variety of neural circuits it influences.
One study showed that most neurons in the somatosensory cortex either showed reduced or unchanged firing rates following administration of ketamine at anaesthetic doses. 59 Another study noted that ketamine can reduce N-methyl-D-aspartate (NMDA)-activated responses by 50%, supporting the hypothesis that its anaesthetic effect arises from a specific interaction with the NMDA receptor. 60
The conclusion drawn from our findings is that water cycling rates within the brain are primarily governed by activity dependent mechanisms. This aligns with the understanding of water transport through metabolite and ion channels, where the solvation of the transported molecules facilitates the movement of water across cell membranes. The extent of the effect measured in the present study is somewhat surprising, and consistent with the idea that transcytolemmal water exchange in brain parenchyma is strongly activity dependent.
An important additional finding is the reversibility of the decrease in water exchange rate through membranes following isoflurane anaesthesia. This evidence eliminates the possibility that the anaesthetic regimen used in these experiments may have caused permanent changes in embryo physiology. It is also consistent with experimental evidence of the reversibility of anaesthesia under the regimes used in this and previous studies.38,39
We note that anaesthetics can also alter the relative distribution of intra- and extracellular space. This could affect the measured CEST effect through the resulting changes in extracellular T1s and relative compartment size. However, this mechanism is unlikely to play a significant role in our observations.
Indeed, isoflurane and a mixture of ketamine and xylazine have opposite effects on compartment ratio. It has been observed that the administration of isoflurane leads to a decrease in the percentage of extracellular water to approximately 17% 61 in Adult male Sprague-Dawley rats, while the co-administration of ketamine and xylazine increases the interstitial space, reaching up to 32% in mice, 62 where the normal extracellular volume fraction is 20%. 46 Moreover, if we consider plausible relative changes in compartment size in our theoretical model, on the order of a few percent, the variation in the resulting exchange rate is much smaller than what is observed experimentally. Thus, the observed reduction in membrane exchange rate cannot be attributed to alterations in the extracellular space volume alone.
We note that our measurements are not specific to neuronal cells, as the z-spectra are integrated over large ROIs comprising the entire telencephalon. Hence, the exchange rate is the average of the exchange rates between intra- and extracellular spaces for all cell types in the parenchyma, including neurons and glia. The glial cells are approximately 10-times more numerous than neurons, and they occupy approximately 50% of parenchymal volume. 63 If the effects of anaesthetics were limited solely to modulating exchange rates in neurons, we could not account for the sharp reduction in average exchange rate observed over the parenchyma. Hence, these results raise the interesting hypothesis that anaesthetic agents also affect water exchange in other cell populations in the brain.
Very few studies have examined aspects of glial cell function under the influence of general anaesthetics, which may directly or indirectly relate to water cycling. General anaesthetics have been found to have differential inhibitory effects on gap junction channels and hemichannels in astrocytes, with implications for intercellular communication and possibly affecting the astrocytic regulation of water balance. 64 General anaesthetic agents can also affect Ca2+ signalling, 65 astrocytes regulate water homeostasis mechanisms that involve intracellular calcium levels. Thus, alterations in calcium dynamics within astrocytes, such as those caused by general anaesthesia, might impact their ability to regulate water transport and influence brain water homeostasis. However, direct evidence specifically addressing water cycling in astrocytes and glial cells under the influence of general anaesthetics appears to be limited based on the current literature. Further research is needed to specifically investigate how general anaesthetics impact water cycling in these cell types within the central nervous system.
This study was carried out using the chick embryo in ovo as an animal model, which provides significant advantages for the applied methodology. The chick is a precocial species, and its embryo presents a relatively permeable BBB, even in the final stages of development, just before birth, which allows for a high concentration of GBCA in the brain's extracellular space. This was an enabling point to measure high-frequency exchange rates, like those involved in transcytolemmal water exchange. Importantly, we found that concentration of GBCA in the extracellular space once absorbed by the tissues was constant throughout the experimental time, with no appreciable washout. This characteristic ensures that any variation observed in the CEST effect can be attributed exclusively to alterations in the water exchange rate across cellular membranes, rather than potentially confounding effects related with variations of GBCA concentration over time.
It is important to emphasize that regional variations in solute concentrations, particularly proteins and metabolites involved in CEST, may influence the quantification of transmembrane water exchange rates. In this study, we estimated an average exchange rate over a large ROI that includes the entire telencephalon, encompassing a variety of cell types. For spatially resolved mapping of exchange rates, acquisition and co-registration of Z-spectra and T1 maps before and after Gd administration would enable the quantification of solutes and contrast agent concentrations on a voxel-by-voxel basis, allowing for the normalization of water exchange rate measurements across different brain regions.
In conclusion, this study represents the first direct in vivo measurement of transmembrane water exchange rates in an intact brain. Our findings reveal a significant reduction in water exchange rates under general anaesthesia conditions, regardless of the specific molecular mechanisms of the anaesthetic agent used. This observation strongly suggests that water cycling in the brain is predominantly governed by ion and metabolite transport mechanisms, and by activity dependent mechanisms. Furthermore, our results indicate a potential effect of anaesthetics on astrocytic activity, highlighting the complex interplay between anaesthesia and brain water homeostasis.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X241309783 - Supplemental material for In vivo assessment of the influence of general anaesthetics on transmembrane water cycling in the brain
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X241309783 for In vivo assessment of the influence of general anaesthetics on transmembrane water cycling in the brain by Eleonora Cavallari, Elena Lorenzi, Enza Di Gregorio, Giuseppe Ferrauto, Silvio Aime, Giorgio Vallortigara and Angelo Bifone in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The MR imaging work was supported by FOE contribution to the EuroBioImaging MultiModal Molecular Imaging Italian Node (![]() ), the PNRR PoC Nodes REDiRECt-Gd Project and the PNRR PoC Nodes ROSEWATER Project. Giorgio Vallortigara acknowledges support from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation program (grant agreement SPANUMBRA No. 833504).
), the PNRR PoC Nodes REDiRECt-Gd Project and the PNRR PoC Nodes ROSEWATER Project. Giorgio Vallortigara acknowledges support from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation program (grant agreement SPANUMBRA No. 833504).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
E.C. and A.B. conceived and planned the study. E.C. and E.L. performed the experiments. E.C. analyzed the images and conducted the data analysis. E.C. and A.B. wrote and edited the manuscript. E.C., E.L., E.D.G., G.F., S.A., G.V., and A.B. contributed to reviewing, commenting, and editing the manuscript.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
