Abstract
Dynamic contrast-enhanced magnetic resonance imaging (DCE–MRI) is increasingly used to estimate permeability in situations with subtle blood–brain barrier (BBB) leakage. However, the method's ability to differentiate such low values from zero is unknown, and no consensus exists on optimal selection of total measurement duration, temporal resolution, and modeling approach under varying physiologic circumstances. To estimate accuracy and precision of the DCE–MRI method we generated simulated data using a two-compartment model and progressively down-sampled and truncated the data to mimic low temporal resolution and short total measurement duration. Model fit was performed with the Patlak, the extended Tofts, and the Tikhonov two-compartment (Tik-2CM) models. Overall, 17 healthy controls were scanned to obtain
INTRODUCTION
Dynamic contrast-enhanced T1-weighted magnetic resonance imaging (DCE–MRI) was originally developed to estimate the blood–brain barrier (BBB) permeability in multiple sclerosis (MS) lesions and in brain tumors,1,2 and was later used to estimate capillary permeability in other organs, where permeability is several order of magnitudes higher. In both situations, the so-called Tofts model and the extended Tofts model (eTofts)
3
have been used extensively. Recently, it has been shown that if the prerequisites for the use of the Tofts model are not fulfilled or the sampling frequency is too low, severe errors in the estimated parameters will occur even when model fit to data seems perfect.4,5 We have previously used the Patlak method
6
with the aim of estimating the BBB permeability from DCE–MRI data.
7
The low permeability and hence low likelihood of back-diffusion (transport of contrast agent from tissue back to blood) are ideal prerequisites for the use of the Patlak method. Using simulations we showed that the Patlak method correctly estimated permeability if it was <3 ml/100 g/min (
Generally, DCE–MRI data sampling frequency has been low, typically in the order of 10 to several hundred seconds, mainly because high sampling frequency is not compatible with the necessary spatial image resolution and general image quality, resulting in under sampling of the arterial input function and the tissue enhancement curve. This is likely the reason that brain perfusion is normally not estimated using DCE–MRI because reliable estimates of perfusion based on an intravascular first-pass bolus passage, is hampered by such a low sampling frequency and also by the fact that a pure intravascular contrast agent does not enhance the T1-weighted MR signal sufficiently at a typical field strength of 1.5 T
8
However, at higher field strength, e.g. 3 T, the intravascular MR signal enhancement during the bolus passage of a contrast agent is clearly detectable. We and others have previously shown that realistic perfusion values can be obtained with DCE–MRI at 3T, using a high sampling frequency of ~1 to 5 seconds, where at least 1 second is normally considered fast enough to capture the fastest components of the contrast agents through the capillary bed.9–12 The total data sampling duration can be left short in the case of perfusion estimation, because one in principle only needs to monitor the wash-in of a contrast agent in the tissue together with the corresponding input function. In our previous implementation we monitored the contrast bolus passage and washout phase over a period of 5 minutes. Logically, we also wanted to estimate the BBB permeability from the same bolus passage, and the Patlak method was chosen, because we expected low permeability and negligible back-diffusion over a relative short measurement of 5 minutes. Using this method, we found that the BBB permeability in apparently healthy tissue in tumor patients was not zero, as normally assumed. Values obtained in gray and white brain tissue were in the order of (K
No previous study has looked into the required total measurement time and sampling frequency in case of slightly increased permeability as encountered in healthy brain tissue or in brain diseases, such as MS, infarctions or benign brain tumors. So far no studies have validated the methods employed and a gold standard is non-existing. Therefore, the aim of this study was to explore the optimal conditions required for accurate estimation of low permeability with regard to total measurement time and sampling frequency. We also wanted to explore and compare the performance of (1) the Patlak model, (2) the eTofts model, and (3) our newly proposed method of combining a model-free estimation of perfusion and the two-compartment model, from which permeability can be estimated. To this end we used both simulation and actual measurements on healthy volunteers and patients with visibly contrast-enhancing brain lesions. Ultimately, we wanted to estimate the lowest measurable permeability significantly different from zero, when using a conventional MR contrast agent.
MATERIALS AND METHODS
Simulation
Simulated data were generated using a two-compartment model having a biexponential residue impulse response function with four free parameters:7,15 perfusion (F), unidirectional transfer constant (K
Healthy Volunteers and Patients
For
Ethics
This study was approved by the Ethics Committee of Copenhagen County according to the standards of The National Committee on Health Research Ethics, protocol number H-D-2008-002. All experiments were conducted in accordance with the Declaration of Helsinki 1975 and all subjects gave written informed consent.
Dynamic Contrast-Enhanced MRI
Magnetic resonance imaging was performed on a 3T MR unit (Philips Achieva, Best, The Netherlands) using a 32-element phased-array head coil. Dynamic contrast-enhanced imaging used a previously described7,11 T1-weighted saturation-recovery gradient-echo sequence that was used both for the initial
Permeability Estimation
DCE-MRI data were analyzed with a semi-automatic procedure using in-house created MatLab-based Software (MathWorks, Natick, MA, USA). The DCE time series was converted to units of contrast agent concentration using
Generated tissue concentration curves from the simulation and real
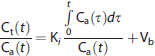
where the slope and the intercept gives K
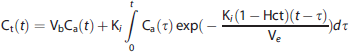
In this case, K
If the two last criteria are not met, a two-compartment model might be more suitable. A two-compartment model incorporates a loss of a tracer from the vascular system and in addition takes a significant intravascular transit time in account. However, a two-compartment model has twice as many free parameters to fit compared with a one-compartment model, resulting in a decrease in the accuracy and precision of the parameter estimates. Therefore, reducing the number of fitted parameters and optimal initialization of the free parameters entering the fitting routine is essential to achieve a robust and accurate result. In a previous study,
11
we suggested to estimate the perfusion (F) from model-free deconvolution of the tissue concentration with the arterial input function, and then passing this value as a fixed value to the two-compartment model, reducing the number of the free parameters then to be estimated. Perfusion estimation is performed using Tikhonov's deconvolution, which is a general form of singular value decomposition having a regularization term.
11
Optimal initialization of K
In this study, we present a detailed analysis and comparison of the feasibility of these three different methods to accurately estimate low values of permeability as encountered in healthy or apparently healthy brain tissues.
RESULTS
Figures 1–3 show results of the K
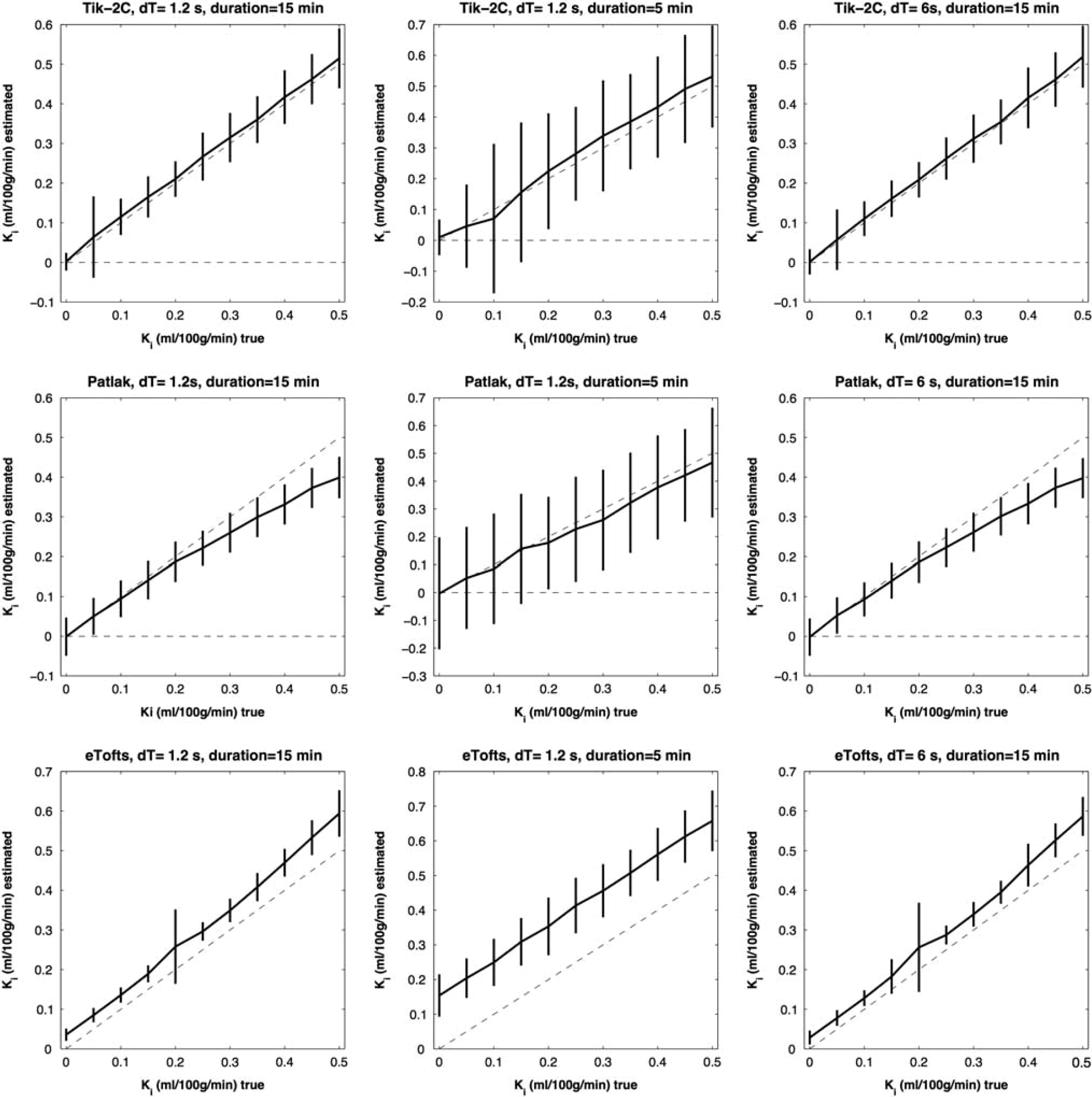
F = 20 ml/100 g/min;Vb = 2 ml/100 g/min. Estimated K
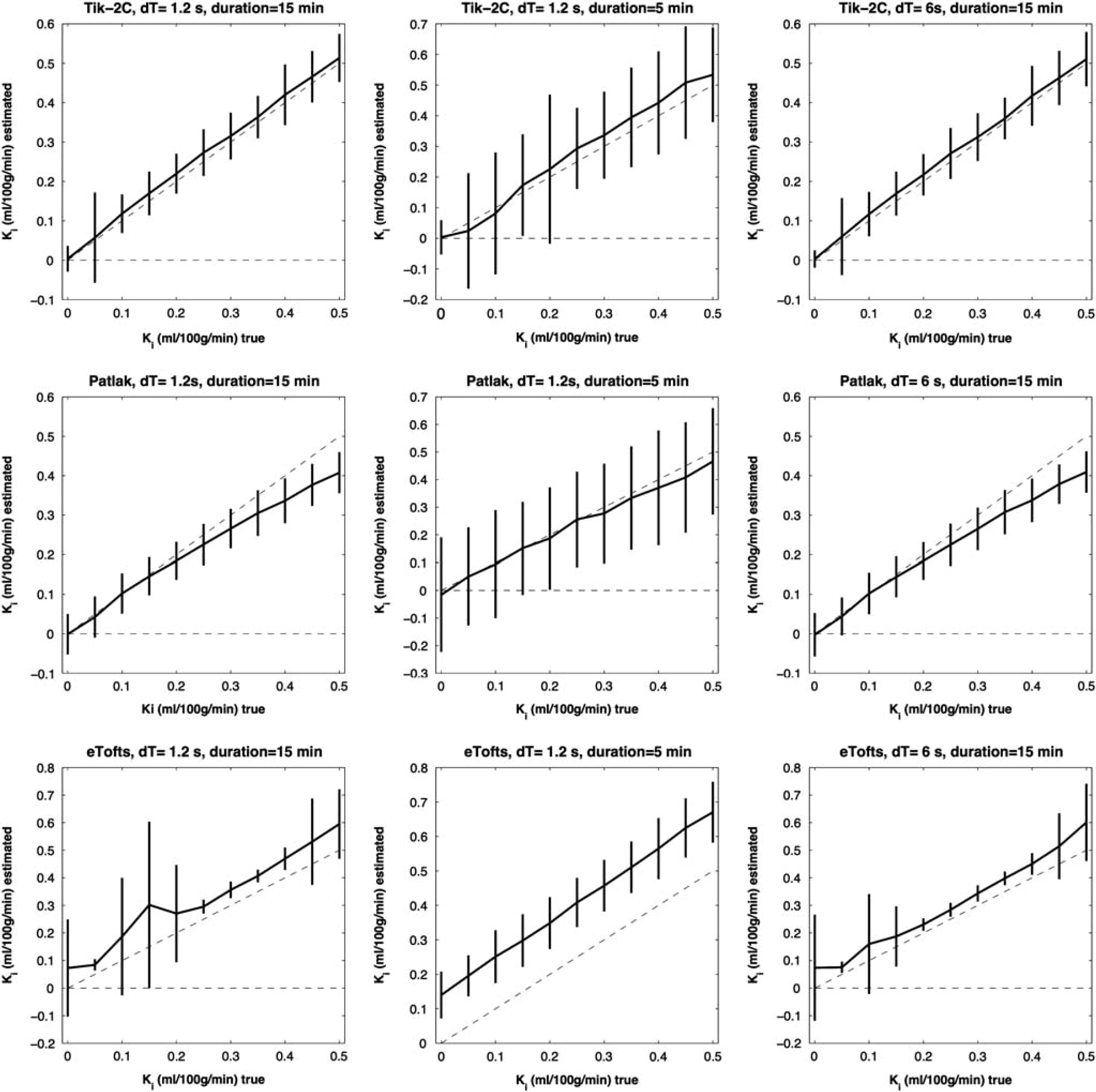
F = 50 ml/100 g/min; Vb =3 ml/100 g/min. Estimated K
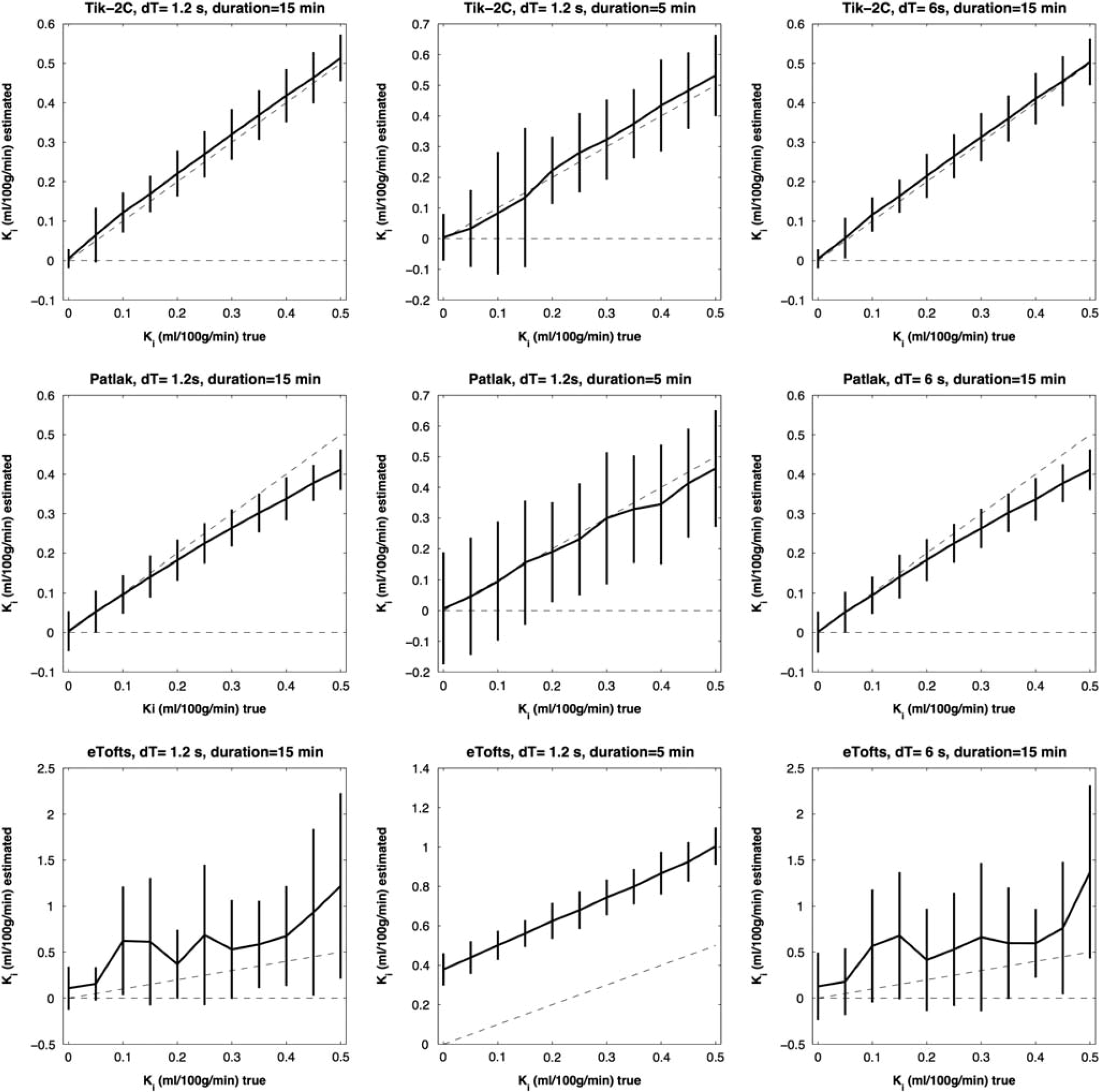
F = 80 ml/100 g/min; Vb = 6 ml/100 g/min. Estimated K
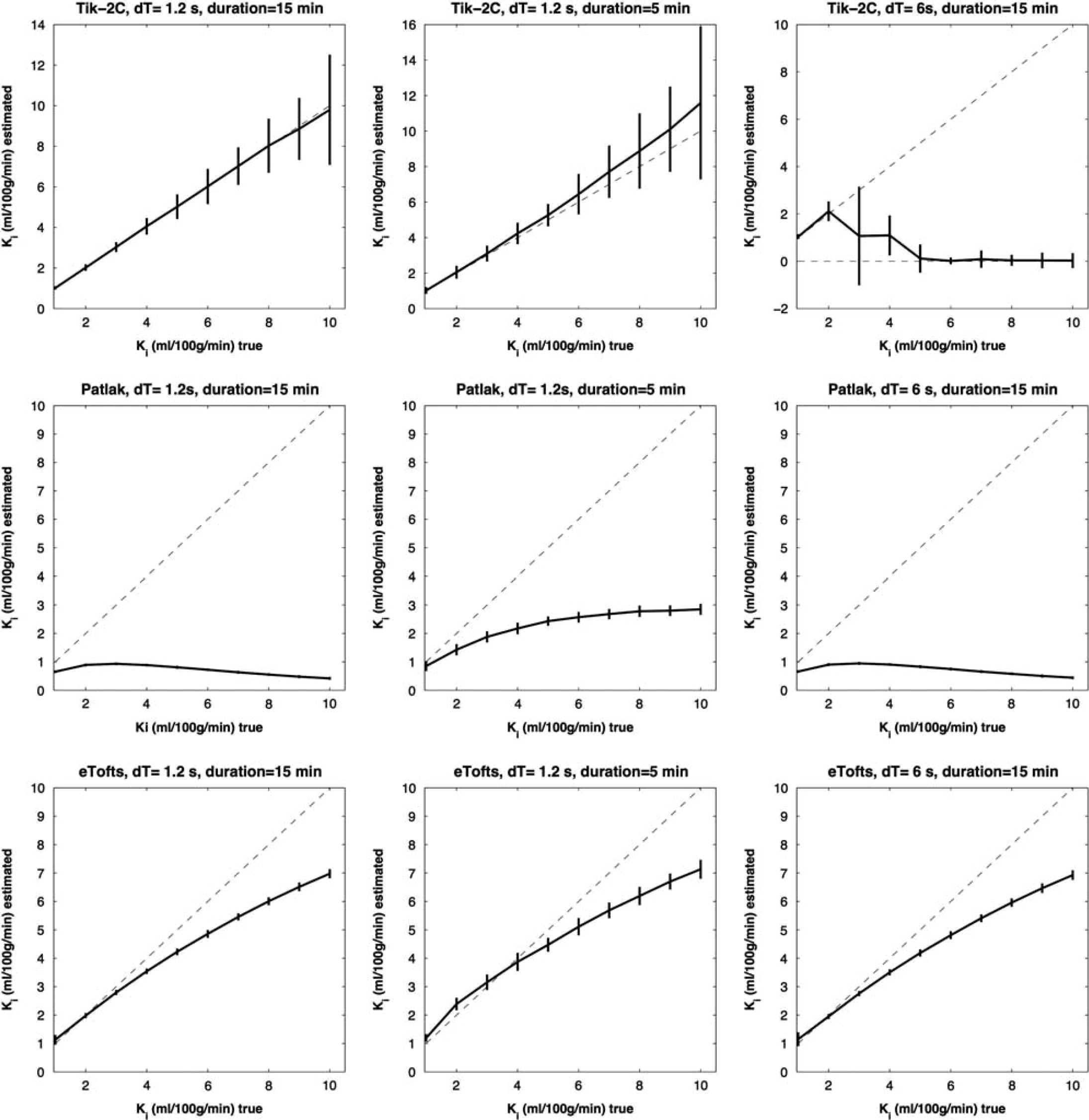
F = 20 ml/100 g/min; Vb = 2 ml/100 g/min. Estimated K
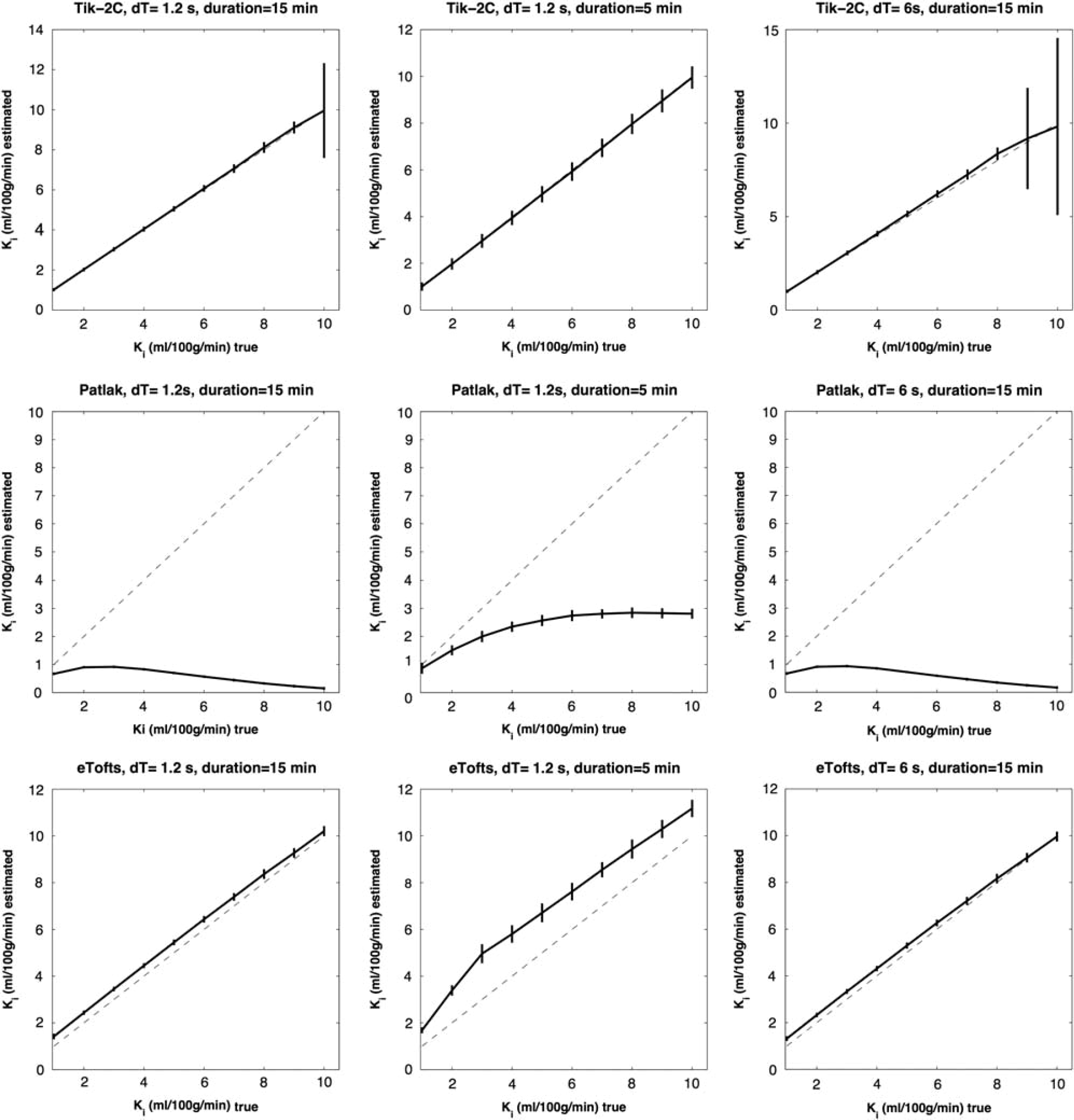
F = 80 ml/100 g/min; Vb = 6 ml/100 g/min. Estimated K
Figures 1–3 show that Tik-2CM is accurate and reproducible when using high time resolution and long total measurement time with no dependency of the perfusion level, and the method is clearly able to differentiate a permeability of 0.1 ml/100 g/min from zero. This is also true for the Patlak method (middle rows), except that a systematic underestimation of the permeability ensues at a permeability of 0.3 ml/100 g/min and higher values. The eTofts method (bottom row) seems acceptable for low perfusion and low blood volume at high sampling frequency and long total measurement time, but gives more unpredictable results as perfusion and blood volume increase. All methods are dependent on time resolution and total measurement duration. For example, the uncertainty of results of the Tik-2C method increases if sample frequency or measurement duration decreases (Figure 3, top row). Either situation results in a doubling of the detection limit of a permeability value significantly different from zero. The eTofts method shows a systematic overestimation as total measurement duration decreases from 15 to 5 minutes, which might be related to lack of information of the elimination phase of the contrast agent. If perfusion and blood volume are low, the eTofts method perform quite well and is not much affected by a reduction of the temporal resolution, as long as the measurement duration is kept sufficiently long.
Results for the K
In Vivo Generated Data
Figure 6 shows
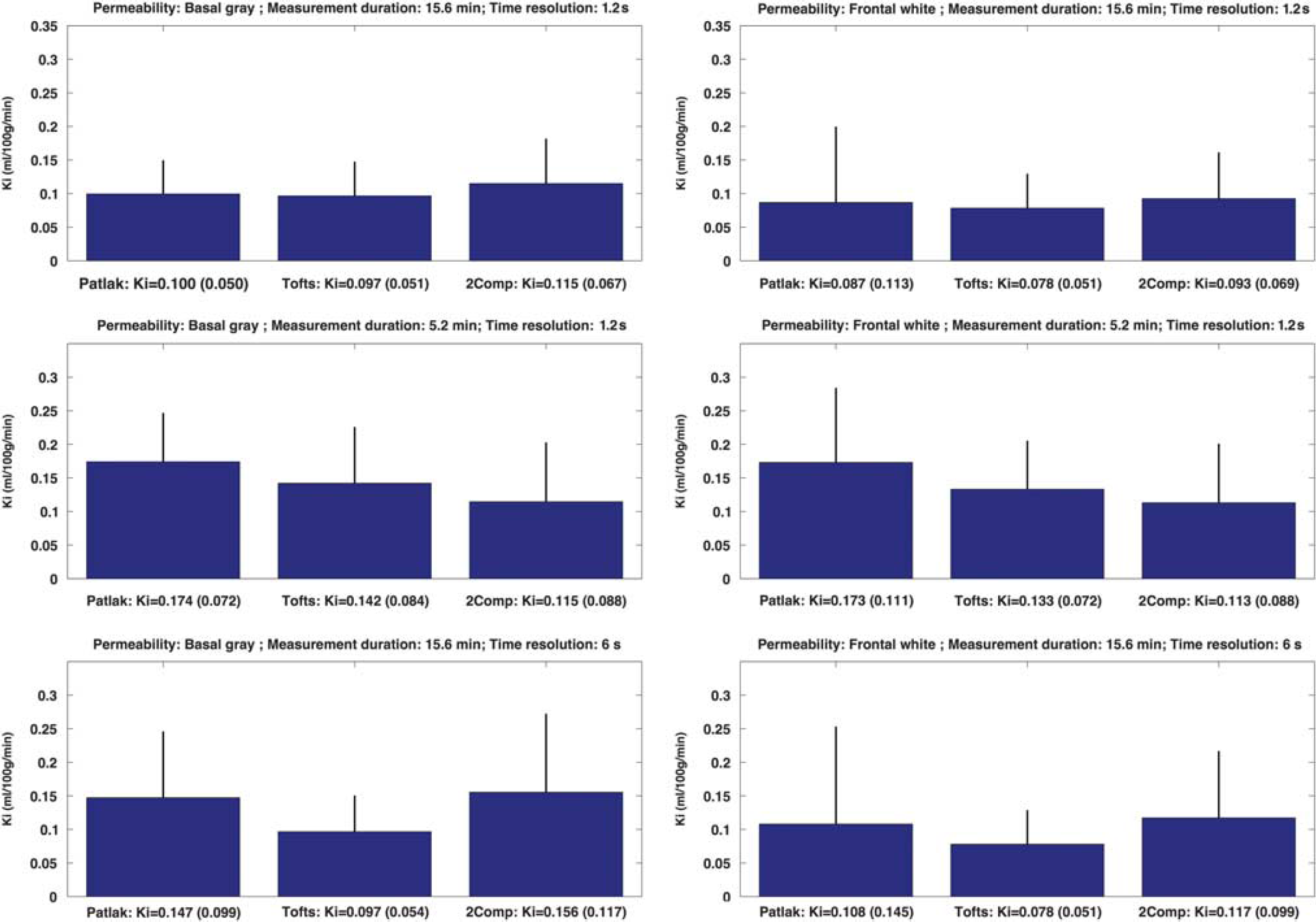
Permeability in basal gray matter (left column) and frontal white matter (right column) in 17 healthy controls estimated with Patlak, eTofts, and Tik-2C models with decreasing total measurement duration and time resolution. eTofts, extended Tofts model; K
Figure 7 shows the results of estimating the permeability using the three methods in nine MS lesions showing visible contrast enhancement on postcontrast T1-weighted MRI. For a high time resolution (1.26 seconds), the Patlak method underestimates the permeability, compared with the eTofts and the Tik-2C methods, a situation that is accentuated as permeability increases, as expected. However, this underestimation is attenuated if the measurement duration is reduced from 15 to 5 minutes. The eTofts method is not very sensitive to a reduction of the sample frequency, while the Patlak method and the Tik-2C method both are sensitive to a reduction in this parameter.
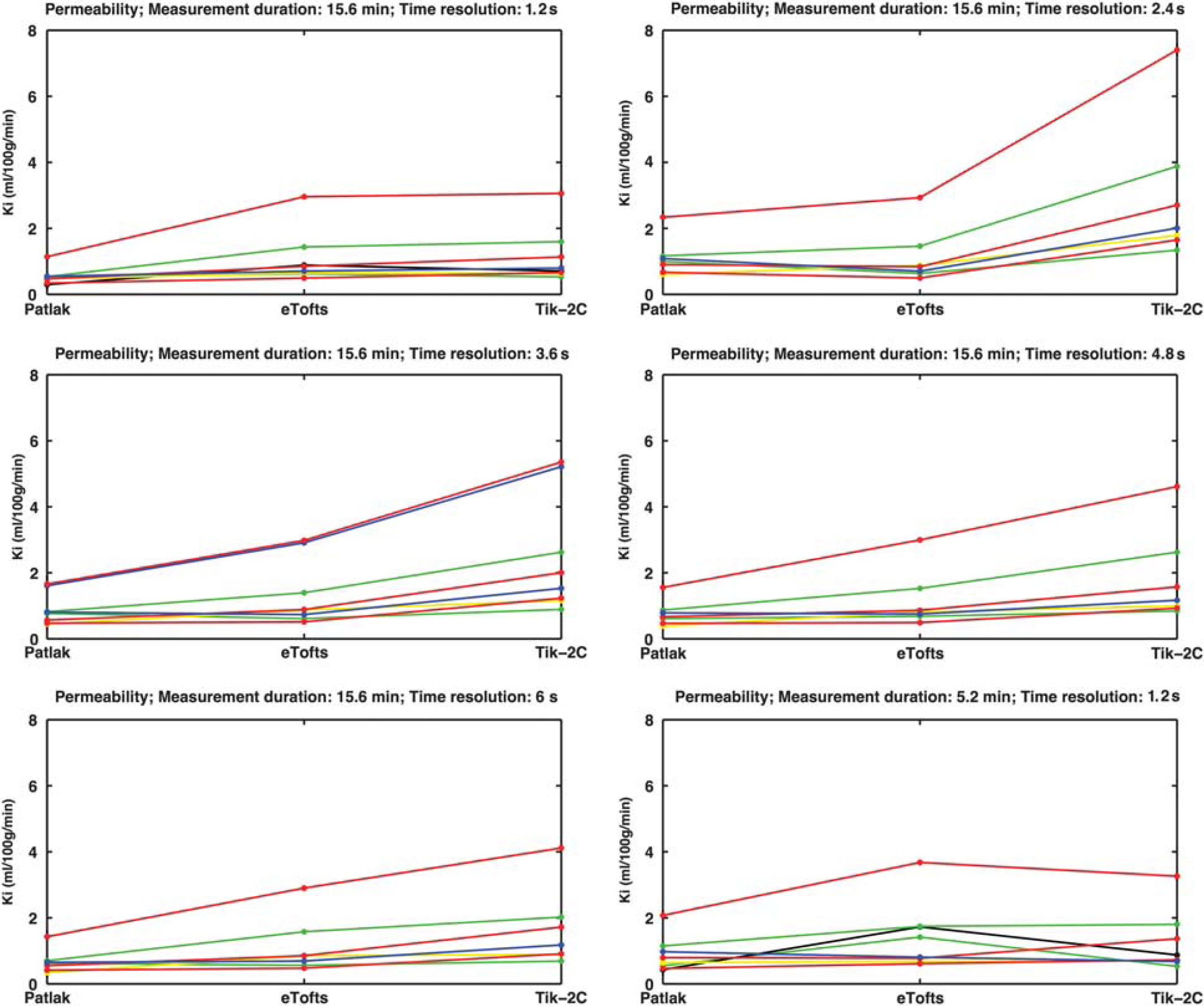
Permeability in eight visibly contrast-enhancing lesions (each line representing one lesion) estimated with Patlak, eTofts, and Tik-2C models with decreasing time resolution and total measurement duration. As true permeability increases, the Patlak model progressively underestimates permeability, hence supporting the findings from our simulations. All enhancing lesions were located in the periventricular region, with a mean area size of 29.3 mm2 (s.d. 21.3 mm2). There was no correlation between area size and permeability values. eTofts, extended Tofts model; K
DISCUSSION
Accurate determination of the BBB permeability might be of importance in certain diseases such as MS or brain tumors. The ability to monitor the permeability over a time course during a treatment trial is obviously beneficial and could strengthen the evaluation procedure of treatment efficacy. In addition, subtle changes of the BBB permeability has been suggested in MS,16–18 epilepsy,19,20 Alzheimer's dementia,21,22 vascular cognitive impairment, 23 migraine,24,25 and cortical spreading depression, 26 but in most of these conditions no conclusive evidence has yet been obtained, likely because of difficulties of measuring small changes of the permeability. To our knowledge, no previous study has looked into the methodological requirements for accurate estimation of low permeability as may be encountered in healthy or apparently healthy tissues in the brain. In a broader context, the measurement of BBB permeability might have implications for the study of the pericytes, which are known to govern both the integrity of the BBB as well as the regulation of cerebral blood flow. 27
In this study, we have shown that the level of permeability, even in a narrow range just above zero, is of importance for the choice of tracer kinetic model employed to estimate the permeability correctly. In addition, total measurement duration and time resolution has a pronounced impact on the accuracy and precision of obtained results. The three methods give the same results for normal brain tissue
Tik-2CM is also very dependent on the time resolution. This is conceivable because the method is based on an initial calculation of perfusion, which is then saved and used unmodified in the two-compartment model, and it is generally accepted that time resolution is pivotal in perfusion estimation, especially when employing intravascular contrast agents. Using long measurement duration and high sampling frequency, the Tik-2C method is the most robust method, showing the highest level of accuracy and precision for the entire range of permeability values investigated, without much dependency on the cerebral blood flow or cerebral blood volume level.
The eTofts model seems to be very dependent on the true permeability, blood volume and measurement conditions, and one should therefore be cautious when using the eTofts method. The eTofts method yields similar results as obtained for the Patlak and Tik-2C methods for normal brain tissue
In a previous positron emission tomography study by Iannotti
Other studies have looked into the precision of measurement of leakage when using the eTofts method, focusing on much higher permeability than the present study. Aerts
It is well known that the eTofts model is an approximation of the two-compartment model, sometimes called the two-compartment exchange model, but a thorough description of the connection in a strict mathematical sense has only lately been provided.
4
Sourbron and Buckley
4
show that the Tofts and the eTofts methods are not appropriate in several typical
Our approach of using two distinct modeling methods depend on whether back-diffusion can be ignored or not, is in line with the results of Sourbron. 12 In this study, the basic set of differential equations corresponding to the two compartments, blood and extravascular tissue, were solved under the condition that back-diffusion from the interstitial space to blood could be (1) neglected, giving the so-called two-compartment uptake model that allows estimation of perfusion, blood volume, and permeability; or (2) included, giving the full two-compartment exchange model allowing estimation of the interstitial space in addition. The choice of which model to be used for a given voxel was based on the Akaike information criterion. Our approach is somewhat different in that we abstain from modeling perfusion explicitly and instead used a model-free regularized deconvolution approach to estimate perfusion. This is because an intravascular contrast agent does not necessarily have an exponential impulse response, 34 as is implicit in the two-compartment exchange model. However, we completely agree that when selecting the actual model to be used for a given data set one has to carefully consider the possibility of back-diffusion, and whether to incorporate back-diffusion seems to be important for obtaining accurate results.
The MR signal obtained from brain tissue when using T1-weighted imaging generally suffers from low C/N, especially if significant leakage is not present, because the MR contrast agent primarily affects the relative small vascular volume. This inherently limits the generalization of the specific results of the present study, because C/N is critically dependent on dose of MR contrast agent, voxel size, MR sequence, and field strength. However, the principal finding may still be valid. An increase in C/N can be achieved by replacing the two-dimensional MR sequence with a three-dimensional sequence, without hampering time resolution. Also preliminary results suggest more than a doubling of C/N by increasing the field strength from 3 to 7 Tesla.
In conclusion, detection of subtle changes of the BBB permeability require optimization of measurement conditions such as time resolution and total measurement duration, in conjunction with selection of an appropriate tracer kinetic model. Here, we suggest the use of the Patlak method for measurement of normal and slightly increased permeability, where back-diffusion can be ignored. If the permeability is higher than a certain threshold, based on our results we suggest a limit of 0.3 ml/100 g/min, then a more complex method should be added. Based on simulations we suggest this method consisting of a two-compartment model being highly accurate and reproducible. The normal BBB has permeability larger than zero, when using a typical MR contrast agent, and can be reliably estimated with a careful optimized measurement strategy at 3 Tesla.
Footnotes
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
We would like to thank radiographers Bente Sonne Møller, Helle Simonsen and Karina Elin Segers at Department of Diagnostics, Glostrup Hospital for scanning assistance. Last we would like to thank the Research Foundation of the Capital Region of Denmark, Biogen Idec and the Danish Multiple Sclerosis Society, for their financial support, which made this project possible.
