Abstract
The blood–brain barrier (BBB) is involved in brain water and salt homeostasis. Blood osmolarity increases during dehydration and water is osmotically extracted from the brain. The loss of water is less than expected from pure osmotic forces, due to brain electrolyte accumulation. Although the underlying molecular mechanisms are unresolved, the current model suggests the luminally expressed Na+-K+-2Cl− co-transporter 1 (NKCC1) as a key component, while the role of the Na+/K+-ATPase remains uninvestigated. To test the involvement of these proteins in brain electrolyte flux under mimicked dehydration, we employed a tight in vitro co-culture BBB model with primary cultures of brain endothelial cells and astrocytes. The Na+/K+-ATPase and the NKCC1 were both functionally dominant in the abluminal membrane. Exposure of the in vitro BBB model to conditions mimicking systemic dehydration, i.e. hyperosmotic conditions, vasopressin, or increased [K+]o illustrated that NKCC1 activity was unaffected by exposure to vasopressin and to hyperosmotic conditions. Hyperosmotic conditions and increased K+ concentrations enhanced the Na+/K+-ATPase activity, here determined to consist of the
Introduction
Maintenance of the water balance is a fundamental requirement for the organism to function properly. In case of systemic dehydration, the blood volume drops and the plasma becomes hyperosmotic, thus causing osmotic extraction of water from various tissues including the brain. Although aquaporins are absent from cerebral endothelium
1
and its osmotic water permeability low,
2
significant amounts of brain water are extracted during systemic hyperosmolarity due to the large surface area of the cerebral vascular bed.
3
However, this water loss is counteracted by cerebral accumulation of the osmolytes Na+ and Cl−, which enter the brain via unresolved volume regulatory mechanisms.4,5 A proposed model suggests the endothelial Na+/K+/2Cl− cotransporter 1 (NKCC1), expressed predominantly at the luminal endothelial membrane, as a key component in brain electrolyte regulation.6,7 NKCC1 could, in this capacity, and via its ability to cotransport water independently of osmotic forces,
8
serve as a molecular transport mechanism underlying dehydration-induced brain accumulation of Na+ and Cl− with associated water influx. However, as NKCC1 is notoriously upregulated in cultured cells9,10 and its endothelial expression debated,11,12 its precise role in brain microvasculature-dependent electrolyte flux is unresolved. An alternative endothelial ion transport mechanism involved in brain ion homeostasis is the Na+/K+-ATPase, which appears to be predominantly located on the abluminal side of the brain endothelium.13,14 With this polarized localization, the Na+/K+-ATPase could partake in dehydration-induced Na+ influx into the brain parenchyma. The Na+/K+-ATPase consists of one
In addition to the cell shrinkage expected to occur with the increased plasma osmolarity during systemic dehydration, the antidiuretic hormone, vasopressin (AVP), is released both systemically and centrally.18,19 Although the kidney is a key target, 20 vasopressin also leads to increased brain water accumulation through activation of the vasopressin V1a receptor (V1aR).21–23 V1aR may be expressed in the brain microvasculature 24 although its expression in the cerebral endothelium remains disputed.12,25,26 The current model includes V1aR activation of the luminal NKCC1, 27 thus promoting dehydration-induced brain electrolyte accumulation.
Here we set out to determine the response of brain capillary endothelial NKCC1 and Na+/K+-ATPase to factors present during systemic dehydration and resolve their proposed contribution to dehydration-induced brain electrolyte accumulation.
Materials and methods
Cultivation of bovine brain endothelial cells, the bEnd3 cell-line, and rat astrocytes
Primary culture of brain microvasculature and astrocytes was used to approximate in vivo conditions in the in vitro co-culture BBB model, while an immortalized cell line was used for comparison. Astrocytes obtained from newborn rats potently induce barrier tightness in bovine endothelium.28,29 The procedures for isolation of bovine capillaries and rat astrocytes and the culturing procedures were performed as previously described. 30 The protocol, by which the primary cells were obtained, complies with the European Community guidelines for the use of experimental animals and the results are reported in compliance with the ARRIVE guidelines. The bovine cerebral endothelial cells (BCECs) were passaged with a brief trypsinization and seeded (30,000 cells/cm2) either in collagen/fibronectin-coated 96-well plates (Ca2+ signaling) or 24-well plates (radiotracer experiments), or (90,000 cells/cm2) on collagen/fibronectin-coated transwell polycarbonate filter inserts (area = 1.12 cm2, pore radius = 0.4 µm, Corning Life Sciences, NY) in a non-contact co-culture with astrocytes. 30 The murine endothelial cell line (bEnd3, ATCC® CRL-2299™) was cultured to confluency in DMEM (30-2002, ATCC) supplemented with 10% FBS (37℃, 5 % CO2) and passages 30–37 were used for experiments.
Light and electron microscopy
To verify proper cell origin and culture purity in the in vitro BBB model, immunocytochemistry was performed on endothelial cells grown in the co-cultured BBB model, according to standard protocols. Employed primary antibodies were: rabbit
Oocyte preparation and in vitro transcription
To obtain a scenario in which we could determine the activity of a given protein in isolation, we employed
Radioactive flux experiments
K+ transport is used as the functional read-out of NKCC1 and Na+/K+-ATPase activity. Their activity was measured at 37℃ (oocytes at room temperature, RT) as bumetanide-sensitive (10 µM
33
) and ouabain-sensitive (1 mM
34
) K+ uptake using (2–6 µCi/ml) 86Rb+ (NEZ072, PerkinElmer) as a tracer for K+. 86Rb+ quantification was determined with Ultima Gold XR scintillation liquid (PerkinElmer, Skovlunde, Denmark) using a Tri-Carb Liquid Scintillation Analyzer (PerkinElmer). The K+ uptake rates were calculated from the 86Rb+ flux data and the tracer dilution, where indicated. Primary endothelial monocultures or bEnd3 cells cultured in 24-well plates were pretreated in ((in mM): 145 or 139 NaCl (primary culture or bEnd3), 3 KCl, 1.2 CaCl2, 1 MgCl2, 5 D-glucose, 20 Hepes; pH 7.4; 317 or 307 mOsm) for 5–30 min prior to 5 min exposure to identical media containing 86Rb+ (± ouabain, ± bumetanide, or ± [Arg
8
]-vasopressin (AVP), (Sigma-Aldrich), or vehicle). Osmolarities of the test media were determined with an osmometer, Type 15 (Löser; Berlin, Germany). For studies of Na+/K+-ATPase K+ affinity, cells were washed in K+-free medium (containing in mM: 127 NaCl, 20 choline chloride, 1.2 CaCl2, 1 MgCl2, 5 D-glucose, 20 Hepes; pH 7.4, 317 mOsm) prior to exposure to isosmotic medium with different concentrations of KCl (equimolar substitution with choline chloride). The assays were terminated by rapid wash with cold assay buffer containing 10 µM bumetanide, 1 mM ouabain and 1 mM BaCl2. The cells were lysed with 1 mM NaOH and protein concentration determined.
35
For the co-cultured endothelial cells, the transendothelial electrical resistance (TEER) was measured at RT prior to all experiments, using an Endohm-12 cup electrode chamber (World Precision Instruments, Sarasota, FL) connected to a Millicell-ERS device (Millipore, MA). The uptake experiments were carried out in TES-buffered DMEM-comp without FBS
30
and the 86Rb+ was added to the luminal and/or abluminal side by media change. In hyperosmotic experiments, the endothelial cells were pretreated for 30 min, in absence of astrocytes, with glucose-containing Hepes and bicarbonate-buffered Hank’s balanced salt solution (HBSS, Thermo-Fisher) supplemented with mannitol (70 mM) in both chambers for an isosmotic treatment, or with 170 mM mannitol in both chambers for hyperosmotic (Δ100 mOsm) treatment, prior to introduction of the tracer. The assays were terminated by rapid wash of the filter support with cold isosmotic HBSS containing 10 µM bumetanide, 1 mM ouabain and 1 mM BaCl2, and the radioactive content of the filter determined. The uptake assays in
Ca2+ signaling
To determine the activity of the G protein-coupled vasopressin receptor, intracellular calcium measurements were performed on primary monoculture of bovine endothelial cells grown in 96-well plates with Fura2-AM (5 µM, Life Technologies, Carlsbad, CA). 36 Excitation was conducted alternatingly at 340 nm and 380 nm and emission was measured at 510 nm with a NOVOstar microplate reader (BMG Labtech, Germany). Vasopressin (Sigma-Aldrich) and ATP (Tocris Bioscience, Bristol, UK) was added automatically from stock solutions to final concentrations of 1 µM and 100 µM. Autofluorescence values were subtracted from the experimentally achieved emission values and changes in [Ca2+]i were calculated as 340:380 nm ratios.
Oocyte volume measurements
The experimental setup for measuring and calculating water permeability of oocytes has been described in detail previously. 37 Briefly, the oocytes were placed in a small chamber with a glass bottom and perfused with a control solution (in mM: 100 NaCl, 2 KCl, 1 CaCl2, 1 MgCl2, 10 Hepes, pH 7.4, RT). Oocyte images were captured from below at a rate of 25 images/s. The oocytes were challenged with a hyperosmotic solution (addition of 100 mOsm mannitol) for determination of water permeability and cell volume changes.
Quantitative PCR
Total RNA from primary endothelial cells cultured as monolayers or as co-cultures on filter inserts was purified using the RNeasy mini and micro kit (Qiagen, Copenhagen, Denmark), treated with DNase, reverse transcribed (0.2 µg RNA) and amplified by qPCR as previously described. 38 Primers were from Baddela et al. 39 or designed using NCBI’s pick primer software: ATP1A1 (NM_001076798.1); F: GGAATGGGTGTTGCCCTGAG; R: TGAGCTTCCGGACTTCGTCA (106 bp), ATP1A2 (NM_001081524.1); ATCCCCGAGATCACCCCTTT; R: TTCACCAGCTTGTCCGTCTG (188 bp), ATP1A3 (XM_002695074.5); F: ATTGTCCTCTGGCTCCGTGA; R: GGTACCGGTTGTCATTGGGG (128 bp), ATP1B1 (NM_001035334.1); F: GGAGTTTTTGGGCAGGACCG; R: CCGCCAGGCAGCCATAAAAA (77 bp), ATP1B2 (NM_174677.2); F: TGGACAGCCCTGTGTCTTCA; R: TTGCCGTTTGCAGGGAACAT (147), ATP1B3 (NM_001035393.2); F: TACAACCGGACAACCGGAGA; R: GAGTCTGAAGCATGGCCCAC (133 bp), GFAP (NM_174065.2); F: CACAGGAAAGGAAGAGGCTG; R: AGTGTTTCCTGGGTCAGGTG (138 bp). For reference genes, we employed GAPDH (NM_001034034.2); F: GTGAAGGTCGGAGTGAACGG; R: TTGATGGCGACGATGTCCAC (92 bp) and RPS18 (NM_001033614.2); F: GAGGTGGAACGTGTGATCACCATT; R: TGTATTTCCCGTCCTTCACGTCCT (100 bp). The primer annealing temperature was 60℃. The optimum concentration for each primer set was 200 nM, except for GAPDH (300 nM) and the amplification efficiencies were 90–99% for all primer sets. GenEx (MultiD Analyses AB, Sweden) was used for data analysis, including testing for best reference gene combinations.
SDS-PAGE and Western blot
Bovine grey matter tissue, purified capillaries, and co-cultured endothelial cells were lysed in RIPA buffer (150 mM NaCl, 1% Triton X-100, 0.5% Na deoxycholate, 0.1% SDS, 5 mM EDTA, 50 mM Tris, pH 8, supplemented with 0.4 mM Pefabloc and 8 µM Leupeptin (Sigma-Aldrich)) and sonicated with Sonoplus mini20 (Bandelin electronic, Berlin, Germany). Protein concentration was determined using DC Protein Assay (Bio-Rad, Hercules, CA). The expression of Na+/K+-ATPase isoforms was analyzed by SDS-PAGE and immunoblotting on Immobilon-FL PVDF membranes (Millipore, MA) using: mouse anti-
Data analysis
All functional assays were performed on batches of cells obtained from at least three different animals (primary cell culture and
Results
Brain endothelial cells, co-cultured with astrocytes, generate electrically tight monolayers and express endothelial cell marker proteins
To approximate the in vivo blood–brain barrier with proper endothelial polarization and monolayer tightness, we employed a co-culture in vitro BBB model. A non-contact co-culture configuration (Figure 1(a)) was chosen to facilitate determination of endothelial ion transport from both the luminal and the abluminal compartment.
30
The primary endothelial cultures were immunolabelled with antibodies towards an endothelial marker (vWF), an astrocyte marker (GFAP), and a pericyte marker (PDGFR (a) Illustration of the primary cultured bovine capillary endothelial cells (rectangles) and astrocyte (stars) in vitro non-contact co-culture model of the BBB. (b) Immunocytochemistry with antibodies targeting filamentous actin, von Willbrand’s factor (vWF), glial fibrillary acidic protein (GFAP) and platelet-derived growth factor recetor β (PDGFRβ) in the co-cultured endothelial cells (green). Visualization of cell nuclei with propidium iodide (red). Scale bars: 20 µm. (c) TEER values for the co-cultured endothelial cells (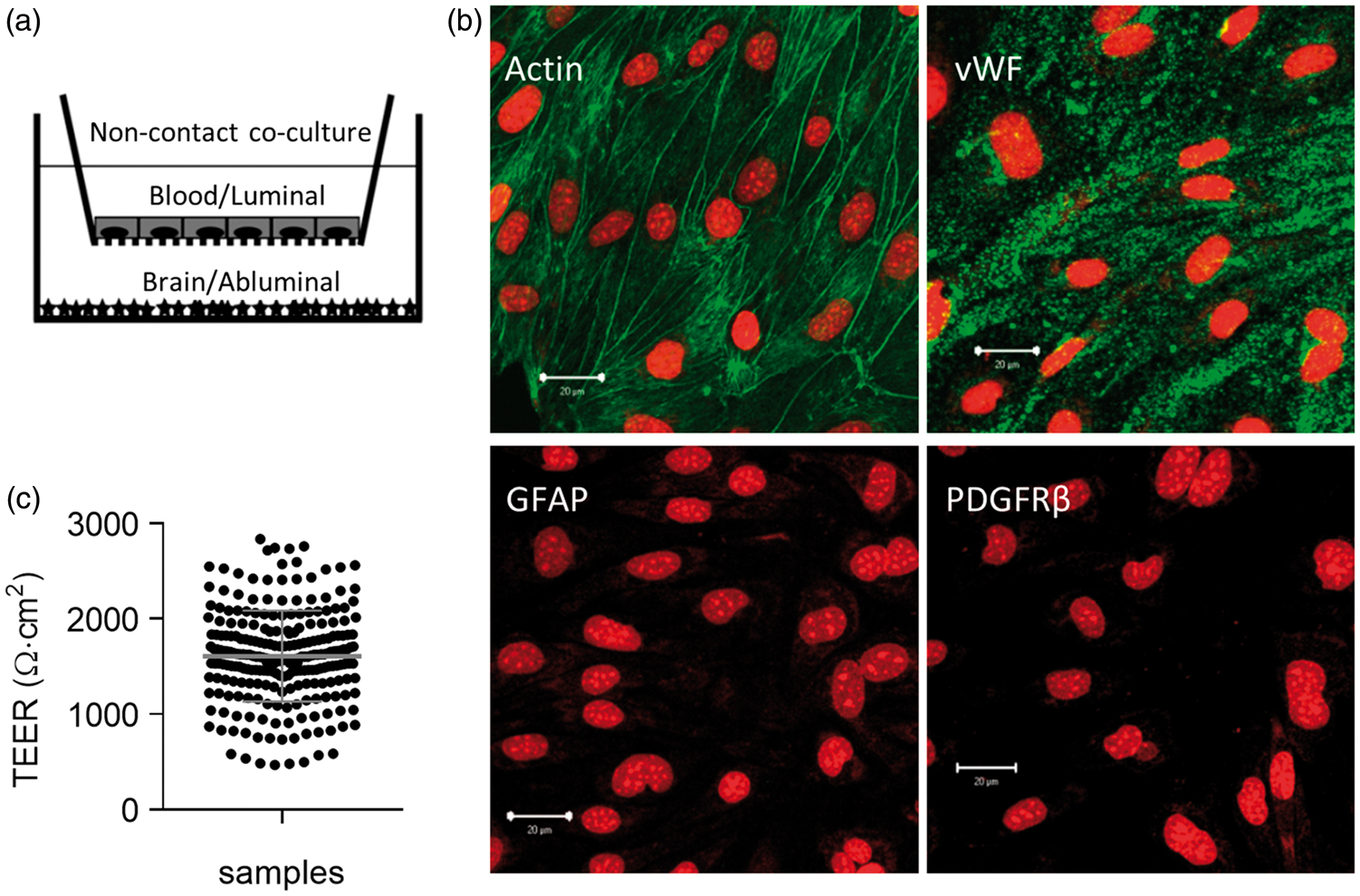
We initially determined the transendothelial flux of K+ (in the form of its radioactive congener 86Rb+) in the luminal-to-abluminal and abluminal-to-luminal direction. The bidirectional fluxes were of equal magnitude and dependent on the TEER (Figure 2(a)), a phenomenon commonly observed for small paracellular tracers like mannitol.42,43 The transendothelial K+ flux was not affected by addition of inhibitors of NKCC1 (bumetanide) and the Na+/K+-ATPase (ouabain) to the luminal or abluminal side (Figure 2(a)). These results indicate that transendothelial K+ flux occurs predominantly via the paracellular pathway in the co-cultured endothelial monolayers.
K+ flux studies in the in vitro co-culture BBB model using 86Rb+ as a tracer for K+. (a) Transendothelial K+ flux as a function of TEER. A–L: Abluminal to luminal flux; L–A: Luminal to abluminal flux. Flux was measured for 2–6 min in the presence (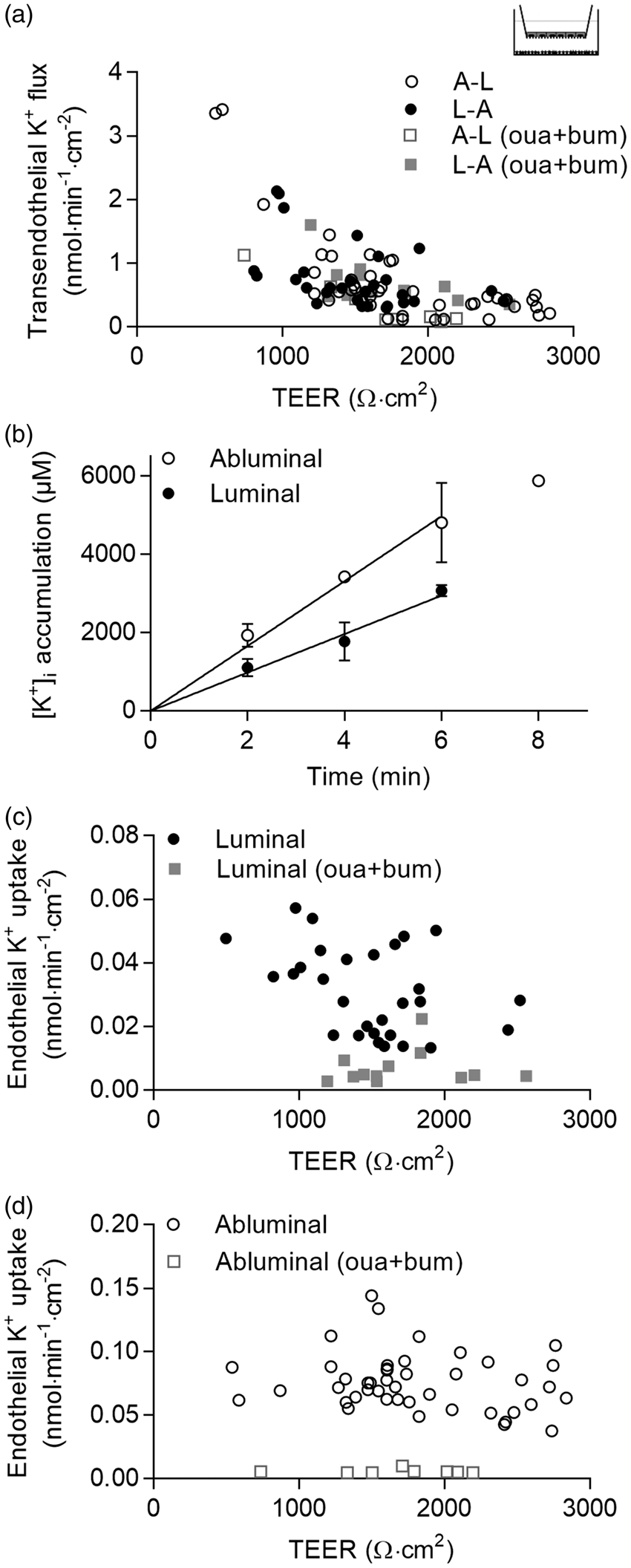
We therefore, instead, determined the uptake rates across each endothelial membrane by adding 86Rb+ to either the luminal or the abluminal compartment, followed by determination of the intracellular 86Rb+ accumulation. The intracellular accumulation of the radioisotope occurred as a linear function of time, at least until 6 min, regardless of whether the isotope was added to the luminal or abluminal side (Figure 2(b)). All subsequent uptake assays were therefore performed in a 5-min experimental window. In this time window, the amount of total transendothelial isotope crossing was negligible (< 0.1% of total isotope) in endothelial cells with TEER above 500 Ω·cm2 and this TEER value was therefore employed as the cut-off value for measuring luminal and abluminal K+-transporting mechanisms.
The luminal and abluminal endothelial K+ uptake rates showed no TEER dependency and were completely blocked by simultaneous addition of ouabain and bumetanide (Figure 2(c) and (d)), indicating that NKCC1 and the Na+/K+-ATPase act as the major endothelial inwardly directed K+-transporting mechanisms in the in vitro co-culture BBB model.
The differences in luminal and abluminal transport rates indicate dominant abluminal expression of NKCC1 and the Na+/K+-ATPase
The luminal and abluminal K+ uptake rates were determined to estimate the relative contributions of NKCC1 and the Na+/K+-ATPase at the two membranes. The abluminal NKCC1 activity was 7-fold higher than the luminal NKCC1 activity ( NKCC1 and Na+/K+-ATPase activity in the in vitro co-culture BBB model. (a) Luminal and abluminal NKCC1 activity assessed as bumetanide-sensitive K+ uptake. The co-cultured endothelial cells were assayed for co-transporter activity for 5 min in medium containing bumetanide (0 or 10 µM) at the same side as tracer. (b) Luminal and abluminal Na+/K+-ATPase activity assessed as ouabain-sensitive K+ uptake in co-culture models exposed to medium containing ouabain (0 or 1 mM) at the same side as the tracer for 5 min. Data are shown as means ± SEM with 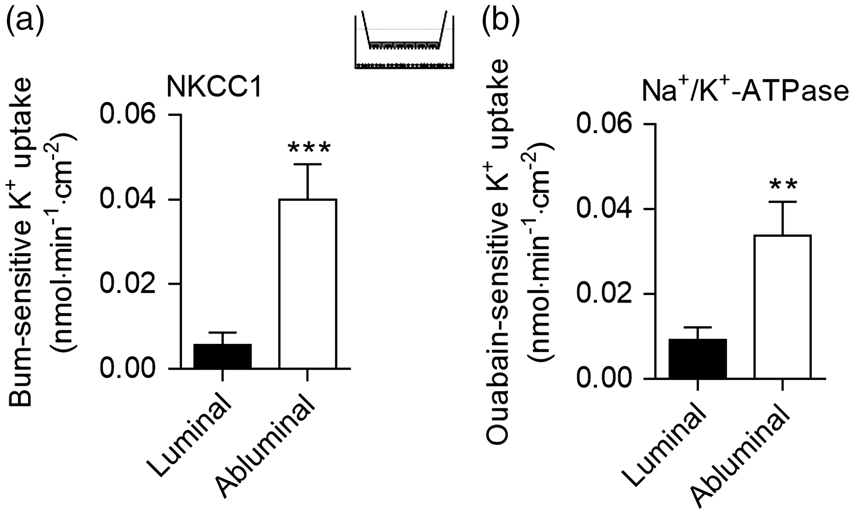
Vasopressin does not affect NKCC1 and Na+/K+-ATPase activity in the endothelial cells
To resolve if a vasopressin-dependent regulatory pathway induces NKCC1-mediated activity, vasopressin was applied to both sides of the in vitro co-culture BBB model prior to measuring the bumetanide-sensitive K+ uptake from both the luminal and the abluminal face of the endothelial cell layer. Vasopressin did not influence neither luminal NKCC1 activity ( Effects of vasopressin on NKCC1 and Na+/K+-ATPase activity. (a) Luminal and (b) abluminal NKCC1 activity in bovine endothelial cells of the in vitro co-culture BBB model treated for 5 min with bumetanide (0 or 10 µM) on the same side as 86Rb+ ± vasopressin (AVP) (1 µM) in both chambers (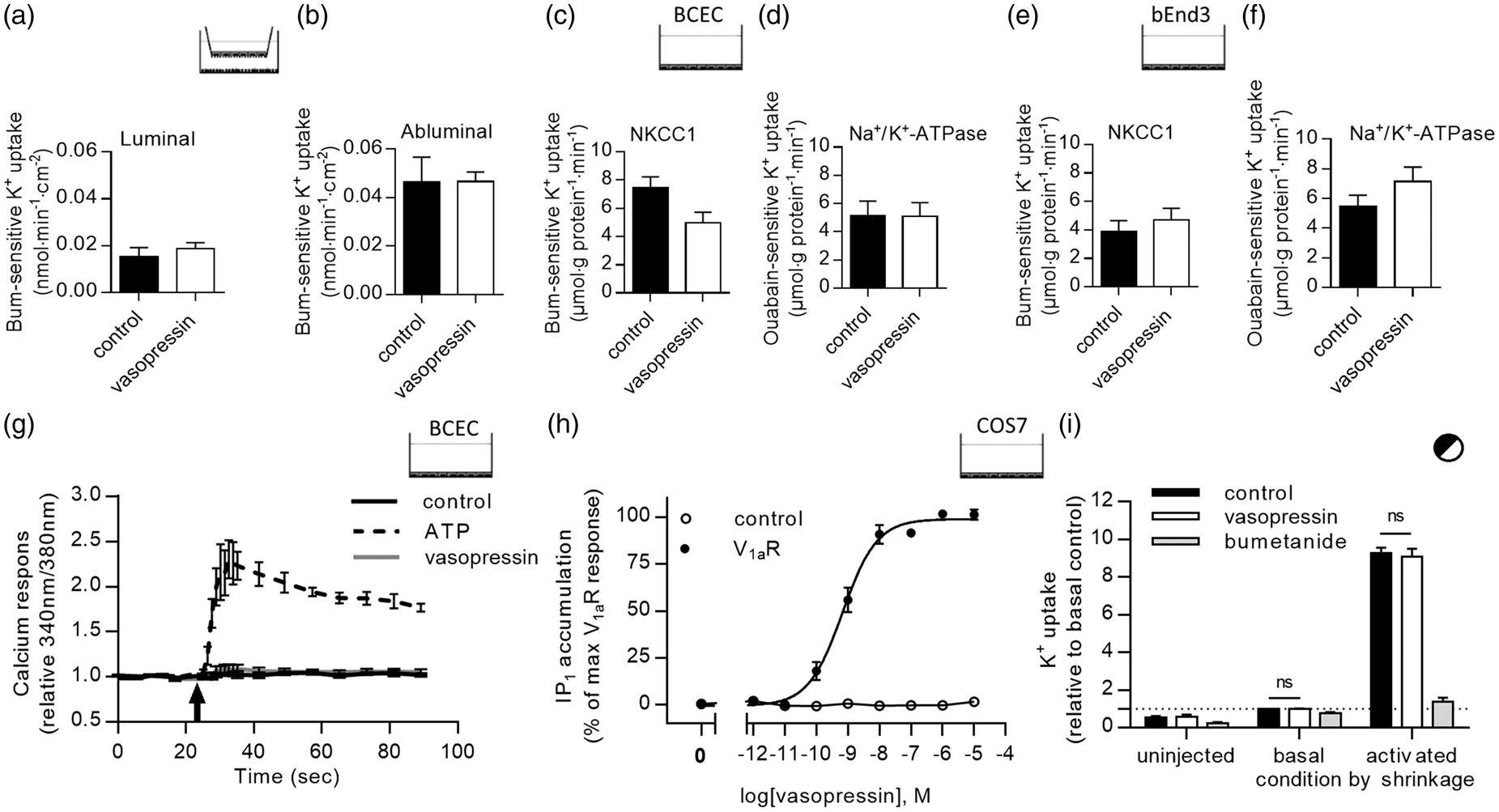
To test if V1aR is functionally present in primary monoculture of bovine endothelium pre-loaded with the Ca2+ indicator Fura2-AM, the intracellular Ca2+ response was monitored upon addition of vasopressin to the test medium. No vasopressin-dependent intracellular Ca2+ response was observed, whereas addition of the positive control, ATP, to the extracellular medium produced a robust Ca2+ response (Figure 4(g)). To ensure biological activity of the employed batch of vasopressin, we (in another project carried out simultaneously 44 ) tested the vasopressin-mediated inositol phosphate (IP) production in hV1aR-expressing COS-7 cells. As illustrated in Figure 4(h), the V1aR responded correctly to addition of vasopressin to the test medium with an EC50 of 0.68 nM (figure adapted from Lykke et al. 44 ). These data indicate that the V1aR is not functionally expressed in the cultured endothelial cells employed in these experiments.
To obtain an experimental scenario in which we could ensure co-expression of V1aR and NKCC1, we co-expressed these in the
Cell shrinkage activates NKCC1 differentially depending on the experimental system
To test if cell shrinkage could directly affect the two transport systems and thus lead to brain ion and water influx, the in vitro co-culture BBB model was exposed to an isosmotic or hyperosmotic (+100 mOsm) solution (luminal and abluminal) prior to determination of NKCC1- and Na+/K+-ATPase-mediated activity in the respective test solutions. The transporter activity determined following the hyperosmotic challenge was not significantly different from the control situation for NKCC1 ( Hyperosmotic regulation of NKCC1 and Na+/K+-ATPase. (a) Bumetanide-sensitive and (b) ouabain-sensitive K+ uptake in the endothelial cells of the in vitro co-culture BBB model pretreated with isosmotic or hyperosmotic test solution (+100 mOsm mannitol) for 30 min before assayed in identical solution containing 86Rb+ ± bumetanide (0 or 10 µM) or ouabain (0 or 1 mM) for 5 min, 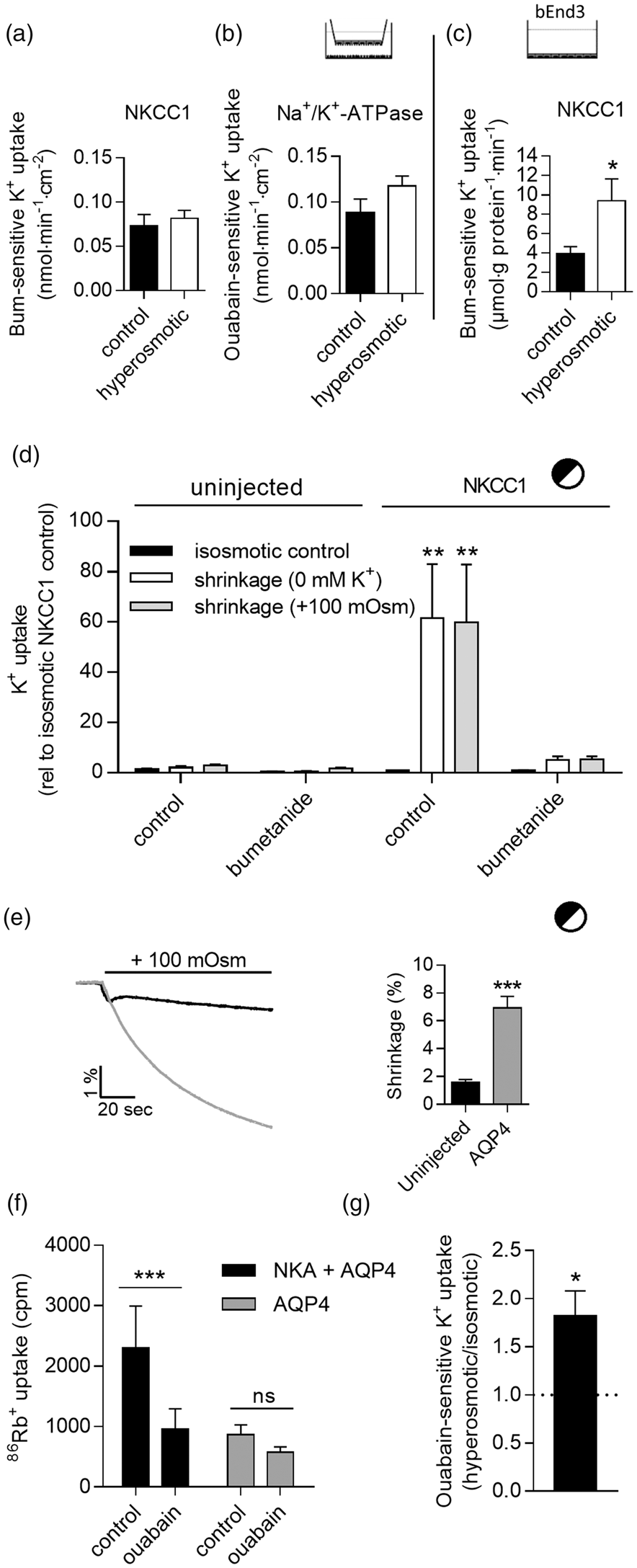
The Na+/K+-ATPase is activated by hyperosmolar-induced cell shrinkage
We expressed the Na+/K+-ATPase in
K+ affinity of the Na+/K+-ATPase suggests physiologically relevant regulation
To investigate if brain endothelial Na+/K+-ATPase activity is regulated by the expected increase in parenchymal K+ during osmotic extraction of brain water and, as a consequence, regulate Na+ flux into the brain, we determined the ouabain-sensitive apparent K+ affinity in the primary-cultured endothelial monolayer (Figure 6). The measured KM of 2.1 ± 0.7 mM, K+ affinity of the Na+/K+-ATPases in primary cultured endothelial monocultures (BCEC) assessed as ouabain-sensitive K+ uptake (with 86Rb+ as the tracer). Cells were pretreated in Hepes-buffered medium containing 3 mM K+ for 5 min and then rapidly washed in K+-free buffer before assayed in isosmotic buffer containing K+ (0.5 to 20 mM) ± ouabain (1 mM). Data and KM are shown as means ± SEM (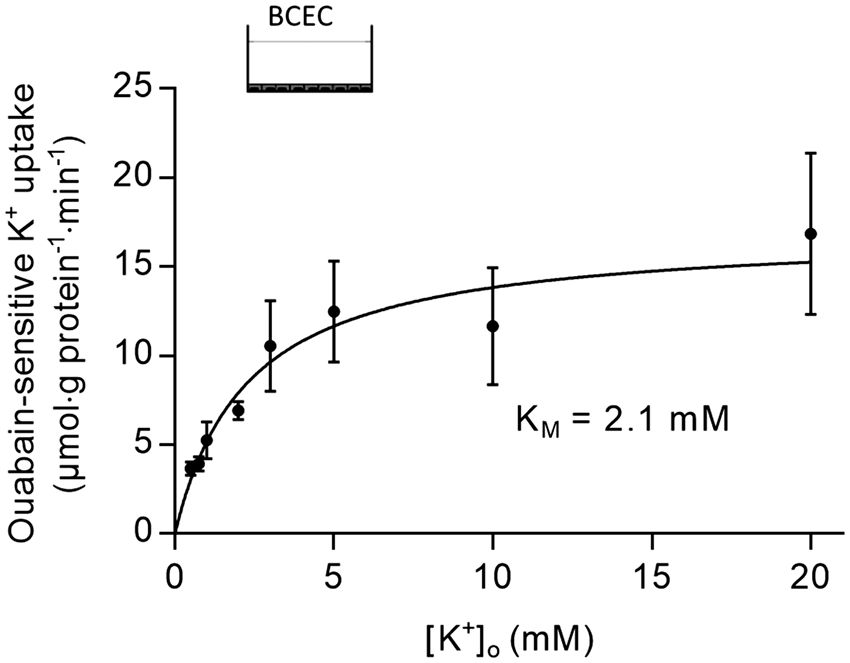
Na+/K+-ATPase of the α 1β 1 and α 1β 3 isoform combinations dominate in brain capillaries
To determine the endothelial Na+/K+-ATPase isoform distribution in the endothelial compartment, the mRNA encoding the Na+/K+-ATPase subunit isoforms were quantified by qPCR in freshly isolated bovine cerebral capillaries. mRNA from the two Na+/K+-ATPase subunit expression in the cerebral endothelium. (a) qPCR quantification of Na+/K+-ATPase subunit mRNA in purified bovine cortical capillaries normalized to reference genes (see materials and methods). (b) mRNA levels in primary cultured cerebral endothelial cells (monoculture (BCEC) or co-culture), normalized to reference genes and quantified relative to the corresponding capillary preparation (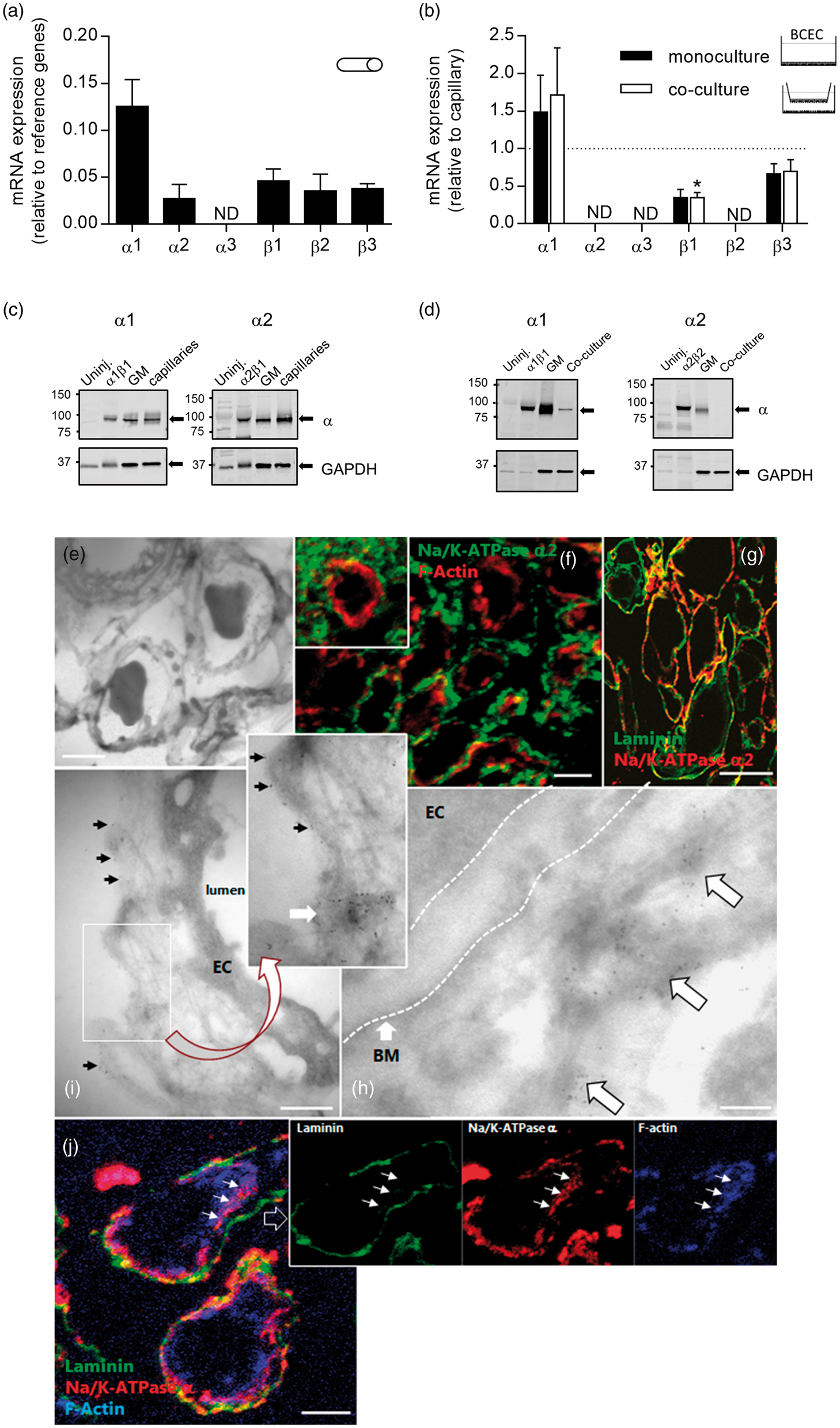
The transcript data were substantiated by Western blotting, which showed expression of Na+/K+-ATPase
The difference in expression of Na+/K+-ATPase isoforms between isolated capillaries and cultured endothelial cells derived from them suggests astrocytic remnants in the isolated capillaries, which would be absent from cultured cells. To answer this question, we applied morphological analysis to resolve the endothelial distribution of Na+/K+-ATPase in vivo by immunofluorescence and cryo-immunogold labeling of in situ or isolated bovine capillaries. Na+/K+-ATPase
Discussion
In the present study, we have demonstrated that the Na+/K+-ATPase, but not the NKCC1, is subject to functional regulation upon treatments mimicking dehydration in a tight in vitro co-culture model of the BBB. To access both membranes of the endothelial cell layer, we employed a non-contact in vitro endothelial/astrocytic co-culture model of the blood–brain barrier with TEER similar to estimates of the in vivo BBB TEER (1500-1870 Ω·cm2).40,41 This tightness of the endothelial cell layer is crucial for proper induction of the BBB properties and allowed us to determine the transport rate from each side of the endothelium in isolation. Functional expression of NKCC1 and the Na+/K+-ATPase was primarily detected at the astrocytic compartment-facing side of the endothelial cell layer, thus denoted the abluminal side of the in vitro BBB model. This abluminally located NKCC1-mediated activity is in agreement with a previous study, 46 while others have found predominantly luminal NKCC1 expression.7,47 The contrasting localization of NKCC1 may arise from differences in the monolayer tightness of the in vitro BBB model and underscores the importance of employing models with high TEER values to ensure proper BBB characteristics. Immunohistochemical studies of rat brain indicated scarce expression of NKCC1 in the cerebral endothelium in vivo, 11 which aligns well with transcriptome studies detecting limited mRNA encoding NKCC1 in this cell type. 12 With NKCC1 notoriously upregulated upon cell cultivation 10 and the limited specificity of a range of commercially available NKCC1 antibodies, it remains unresolved to what extent NKCC1 is functionally expressed in brain microvascular endothelium in vivo under certain conditions. Nevertheless, the dominant expression of NKCC1 at the abluminal side of the endothelium and a predicted inwardly-directed ion transport is challenging to reconcile with the current model based on NKCC1 serving as the molecular mechanism underlying dehydration-induced ion and water accumulation in the brain.
The antidiuretic hormone vasopressin increases in the plasma of patients experiencing stroke and intracranial injuries48,49 and promotes enhanced endothelial water flux
50
and edema formation
51
in a manner sensitive to V1aR antagonism.21,22 The vasopressin-mediated endothelial water flux could thus occur via the V1aR-dependent NKCC1 activation, proposed in the current model.
27
We were, however, unable to detect vasopressin-mediated activation of NKCC1 activity, whether determined in monocultures or co-cultures of brain endothelial cells (
Ultrastructural determination of distribution of Na+/K+-ATPase reaction products at the microvascular endothelium has revealed predominant accumulation at the abluminal membrane13,14,54 although luminal membrane Na+/K+-ATPase has been proposed as well.55–57 A possible reason for this discrepancy is the variable tissue fixation, which has been shown to alter the levels of the Na+/K+-ATPase reaction products at the two endothelial membranes,
55
while the enzyme distribution may be rearranged upon cultivation in vitro.
54
These biochemical methods, however, does not reveal the isoform-specific distribution of the Na+/K+-ATPase. Western blot analysis of isolated brain microvessels (possibly including remnants of astrocytic endfeet and nerve endings) has previously detected expression of all tested isoforms (
Different isoform combinations of the Na+/K+-ATPase display distinct kinetic characteristics, such as apparent ion affinities.9,15,16,61 For the endothelial Na+/K+-ATPase to increase its activity in response to dehydration-induced osmotic extraction of water and associated predicted fluctuations in parenchymal [K+]o (and other ions) and thus maintain the cerebral K+ homeostasis, its isoform expression should be of a combination which renders the Na+/K+-ATPase sensitive to [K+]o in the parenchyma.
62
Such K+-mediated increase in Na+/K+-ATPase activity is accompanied by Na+ flux from the endothelium to the brain and this net parenchymal accumulation of electrolytes thus counteracts the osmotic brain water loss, given parallel accumulation of a counter ion. With a KM for K+ of 2.1 mM in the primary endothelial cultures, in agreement with previous reports,59,62,63 the Na+/K+-ATPase displays the ability to react to increased [K+]. In contrast, the neuronal Na+/K+-ATPase activity saturates at the basal [K+]o of around 3 mM.
63
Of the two isoform combinations predicted to dominate the endothelial Na+/K+-ATPase activity, the
Endothelial cell shrinkage is predicted to occur during systemic dehydration and the Na+/K+-ATPase activity in the in vitro co-culture BBB model displayed a tendency towards hyperosmolar activation. Such shrinkage-induced activation reached statistical significance in heterologously expressed Na+/K+-ATPase
In conclusion, in an in vitro co-culture BBB model with TEER values resembling that of the in vivo BBB, we detected abluminal localization of NKCC1 and lack of hyperosmolar- and vasopressin receptor-mediated NKCC1 activation. This finding, taken together with low expression of NKCC1 and V1aR in cerebral endothelium, suggests a limited role of NKCC1 in dehydration-induced brain electrolyte accumulation and thus contrasts with the current model of dehydration-induced brain electrolyte accumulation. The abluminal localization of the Na+/K+-ATPase (of the
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Sapere Aude program under the Danish Medical Research Council (grant number 0602-02344B FSS) to NM, Læge Sofus Carl Emil Friis og hustru Olga Doris Friis’ legat (to NM), the Carlsberg Foundation (CF15-0070) to NM and the Faculty of Health and Medical Sciences, University of Copenhagen (to NM).
Acknowledgements
We thank Carina Lynnerup Pedersen, Charlotte Goos Iversen for technical assistance and Professor Kjeld Møllgård for expert histological guidance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
KL, MA, SH, HCCH, AS, TLTB, KT, FV, BB, NM Made a substantial contribution to the concept and design, acquisition of data or analysis and interpretation of data. KL, MA, SH, HCCH, AS, KT, FV, BB, NM Drafted the article or revised it critically for important intellectual content. KL, MA, SH, HCCH, AS, TLTB, KT, FV, BB, NM Approved the version to be published.
