Abstract
The perfusion-/diffusion-weighted imaging (PWI/DWI) mismatch and the diffusion/fluid attenuated inversion recovery (DWI/FLAIR) mismatch are magnetic resonance imaging (MRI) markers of evolving brain ischemia. We examined whether the DWI/FLAIR mismatch was independently associated with the PWI/DWI mismatch. Furthermore, we determined whether the presence of the DWI/FLAIR mismatch in patients with the PWI/DWI mismatch would provide additional information regarding last seen normal time (LTM). We used data from the ‘AX200 for ischemic stroke’ trial (AXIS 2 study NCT00927836). We studied the association between the presence of the DWI/FLAIR and PWI/DWI mismatch, baseline National Institute of Health Stroke Scale (NIHSS), age, ischemic-core volume, gender, intravenous (IV) tissue plasminogen activator (tPA), and perfusion-mismatch volume in univariate analysis.
Significant variables (
INTRODUCTION
Patients with acute stroke can be treated with intravenous (IV) thrombolywithin a 4.5-hour time window after stroke onset. 1 However, in up to 25% of patients, the exact time of stroke onset cannot be obtained. 2 Patterns on magnetic resonance imaging (MRI) may permit an assessment of the time since stroke onset. Patients with an acute ischemic lesion detected with diffusion-weighted imaging (DWI) but not visible on fluid attenuated inversion recovery (FLAIR) imaging, are likely to be within a therapeutic time window in which thrombolysis is safe and effective. 3 The perfusion-weighted imaging (PWI)/DWI mismatch pattern on MRI has been studied as a marker of the ischemic penumbra. Patients with a particular PWI/DWI mismatch seem to benefit more from early reperfusion.4,5 Some studies show the presence of the PWI/DWI mismatch in up to 44% of patients even as long as 18 hours after symptom onset suggesting this mismatch to be variable between individual stroke patients and less time dependent.5,6 The relationship between the PWI/DWI mismatch and the DWI/FLAIR mismatch has not been studied in detail. The aim of this study was to assess whether there was a significant association between these two mismatch patterns. Furthermore, in current practice, most sites and clinical trials that rely on MRI to select patients for thrombolysis or endovascular treatment focus on the presence of the PWI/DWI mismatch.4,5,7,8 We wanted to determine whether the additional presence of the DWI/FLAIR mismatch would provide extra supplementary information regarding the time of symptom onset or more specifically in our study population the time since they were last seen normal till MRI (LTM).
MATERIALS AND METHODS
Patients
We analyzed neuroimaging and clinical data from patients included in the AX200 for ischemic stroke (AXIS 2) trial. 9 This was a large phase IIb, multicenter, placebo-controlled, randomized and double-blinded trial intended to determine the clinical efficacy of recombinant granulocyte colony-stimulating factor (Filgrastim, AX200) in acute ischemic stroke patients. 9 Granulocyte colony-stimulating factor treatment failed to meet the primary and secondary end points of the trial. The study was performed according to the International Conference of Harmonization Good Clinical Practice and was approved by the respective regulatory authorities (first approval received from the lead ethics committee of Linz, Austria on 16 March 2009; first approval for a whole country was received for Austria on 31 March 2009), and by the local ethics committees of all study sites. Informed consent of patients was required before they were entered into the study. Design and content of the consent form were according to country regulations and approved by the lead and local ethics committees. In total, 324 patients were included. The full methodology of this study has been described previously. 9 In brief, the main inclusion criteria were initial National Institute of Health Stroke Scale (NIHSS) score from 6 to 22, age 18 to 85 years, a time window of ≤ 9 hours, and stroke localization in the middle cerebral artery territory. Magnetic resonance imaging (FLAIR, T2, T2*, DWI, time-of-flight magnetic resonance angiography, PWI) was mandatory before inclusion. A minimum DWI lesion size of 15 mL was required. There were no prespecified PWI inclusion criteria. Prior treatment with IV tissue plasminogen activator (tPA) was allowed. No exact timing between the administration of IV tPA and the start of the MRI scan is known. However, if IV tPA was administered, then the infusion was finished before starting the MRI. The main exclusion criteria were signs of severe stroke on imaging (carotid T occlusion, ischemic lesions larger than two-thirds of the middle cerebral artery territory, signs of midline shift), hemorrhagic, and lacunar strokes. The NIHSS score was obtained at baseline and at 90 days after inclusion. 9
Image Analysis
The presence of the DWI/FLAIR mismatch was visually rated by two independent readers (VT and AW), blinded to clinical information. The visibility of a FLAIR hyperintensity in the region of the DWI lesion was appraised to determine the DWI/FLAIR mismatch. The same rating of the FLAIR hyperintensities was applied as is currently used in the ongoing WAKE-UP trial. 10 This liberal assessment of the FLAIR images does not consider subtle FLAIR changes but only marked hyperintensities are considered FLAIR positive (Figure 1). To assess the PWI/DWI mismatch, we calculated the perfusion maps with the software ‘perfusion mismatch analyzer’ (PMA version 3.4.0.6, Advanced Medical Science Center, Iwate Medical University, Iwate, Japan). The PMA software selected the arterial input function automatically based on histograms of peak concentration, time-to-peak, and mean transit time. We used the block- circulant singular value decomposition method. 11 Tmax (the time to maximum of the residue function obtained by deconvolution) maps were calculated. The ischemic core was defined semi-automatically on the apparent diffusion coefficient (ADC) maps. We determined the volumes of ADC lesions by applying an absolute threshold. Ischemic lesions here were defined as voxels with a value of below 600.10−6 mm 2 /s.12,13 The DWI maps were used as a mask for defining brain tissue voxels. From the resulting set of voxels, only those clusters which were likely to be part of the ischemic lesion based on visual assessment were selected. A PWI/DWI mismatch was defined after coregistration of the perfusion and ADC maps. The native perfusion image was used as a reference image. The coregistration was performed by Statistical parametric mapping 8 (SPM 8, Welcome Institute of Neurology, University College of London, UK), using mutual information. The PWI/DWI mismatch was calculated by dividing the perfusion volume by the volume of ADC voxels overlaying the perfusion volume. A ratio of 180% or more and a mismatch volume of more than 15 mL were defined as a mismatch, by analogy with the DEFUSE 2 study. 5 We used perfusion lesions with a threshold of Tmax of 6 seconds to determine the PWI/DWI mismatch.14,15 In a secondary analysis, the PWI/DWI mismatch was defined as the ratio of the perfusion volume (Tmax > 6 seconds) and the ADC volume with a volumetric, non-coregistered method. Intracranial vessel occlusion was rated by an experienced neuroradiologist (GW) on MR angiography.
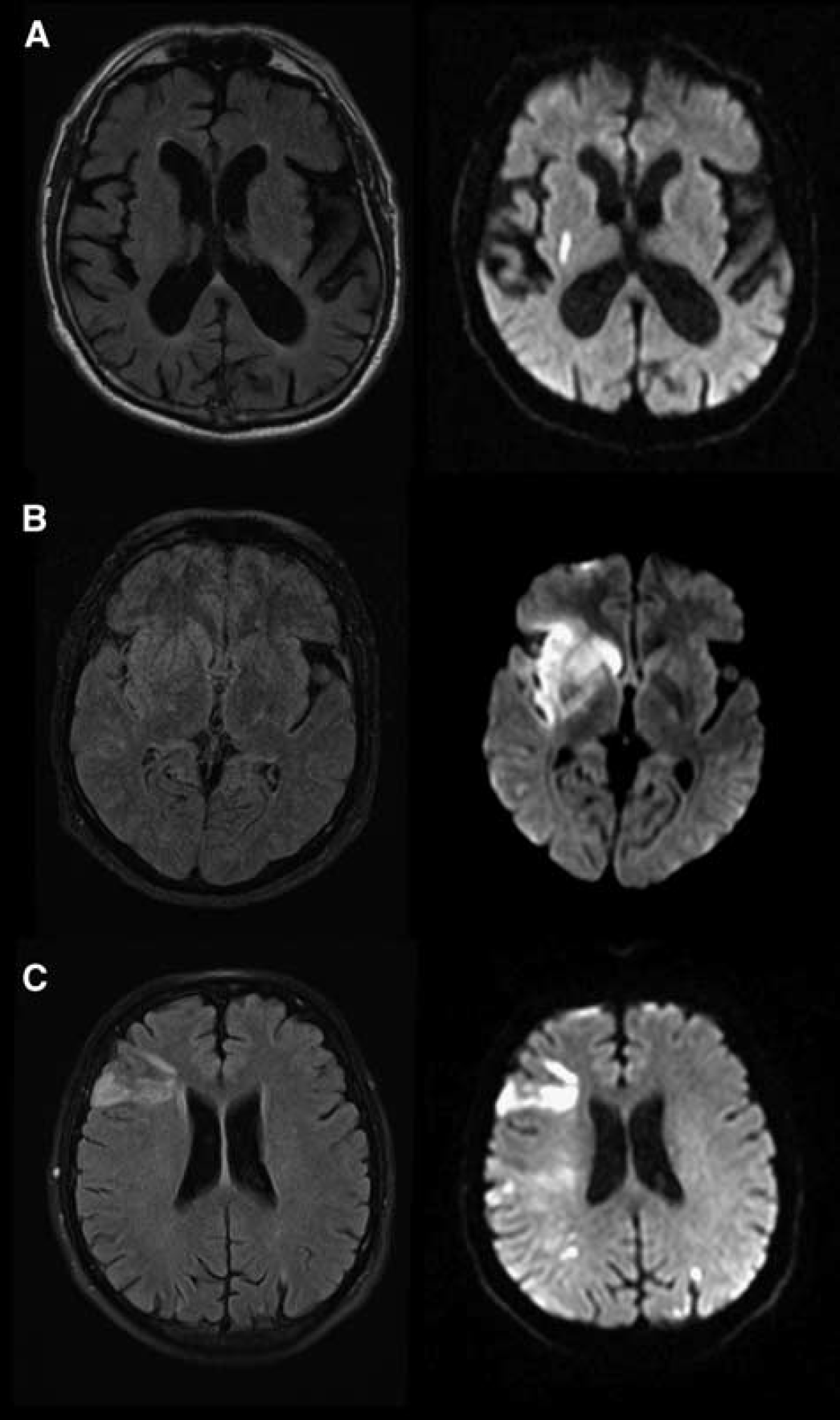
Assessment of diffusion-weighted imaging (DWI)/fluid attenuated inversion recovery (FLAIR) mismatch profile. An example of a FLAIR lesion that is absent (
Statistical Analysis
Two independent raters (AW and VT) visually judged the presence of the DWI/FLAIR mismatch. The interobserver agreement for the identification of the DWI/FLAIR mismatch was calculated, using Cohen's kappa. Discrepancies were resolved by consensus. Sensitivity, specificity, positive predictive value, and negative predictive values were calculated to predict LTM ≤ 4.5 hours for DWI/FLAIR mismatch as well as for PWI/DWI mismatch. Chisquared statistics and univariate logistic regression were performed to test the association between the presence of the DWI/FLAIR mismatch (dependent variable), LTM, age, NIHSS, DWI lesion volume, perfusion-mismatch volume, and the presence of the PWI/DWI mismatch. Significant variables (
RESULTS
Patient Population and Characteristics
In the AXIS 2 study, 324 patients were treated with Filgrastim (G-CSF) or placebo of whom 283 patients were included in the DWI/FLAIR mismatch analysis. Fifteen patients were excluded due to poor image quality, ten patients due to lack of proper image-data, nine due to extensive white-matter disease precluding analysis of the DWI/FLAIR mismatch, and three patients because no DWI lesion was found. Two patients had an infratentorial stroke and one patient had a subarachnoid hemorrhage. In one patient, no stroke onset time was recorded.
There were no differences in baseline characteristics between the groups with and without the DWI/FLAIR mismatch, except for LTM. In all, 32 patients (11%) were scanned within 3 hours after last seen normal, 65 (23%) between 3 and 4.5 hours, 84 (30%) between 4.5 hours and 6 hours, and 102 (36%) patients after 6 hours. In the original publication, 9 PWI results have been reported for 259 patients. In our analysis the PWI data could be analyzed and coregistered using the PMA software from 197 patients only, since 62 patients had to be excluded due to compatibility and quality reasons. The clinical characteristics of the included patients are summarized in Table 1. A comparison of the characteristics of the excluded and included patients can be found in Supplementary Table 1.
Clinical characteristics
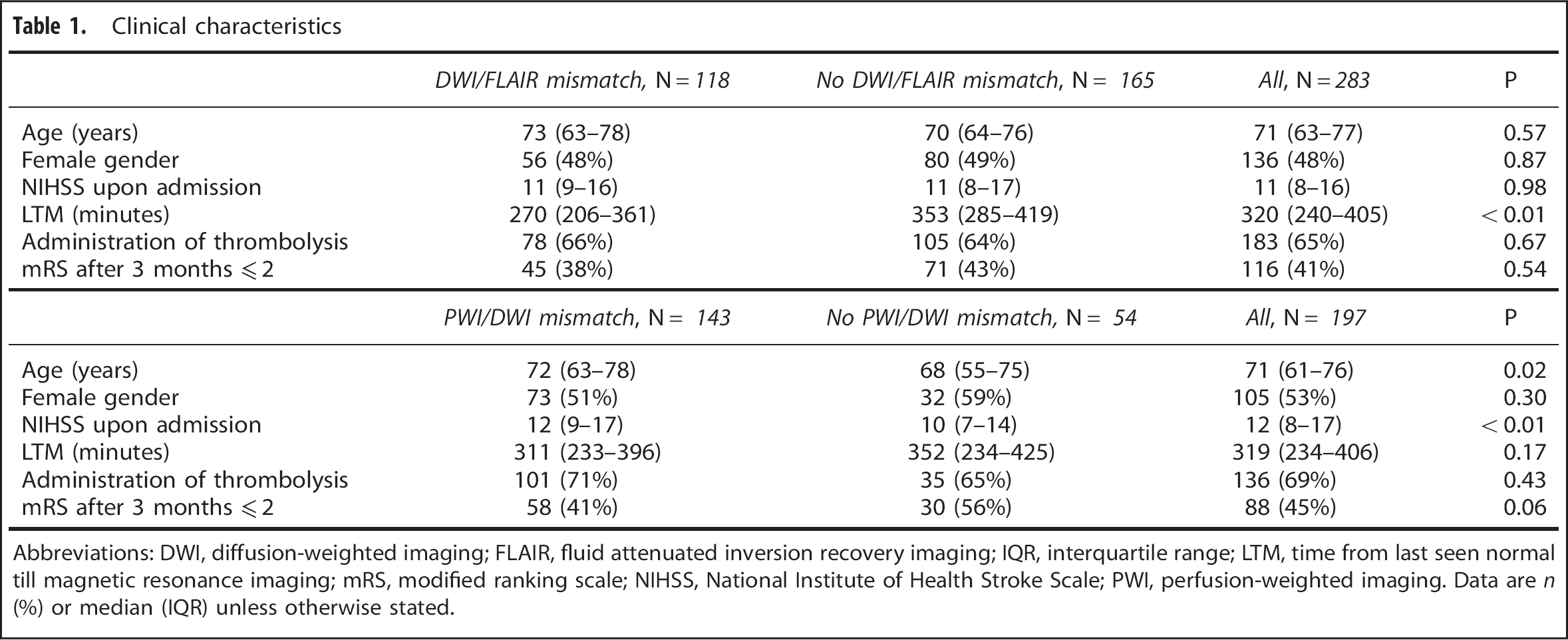
Abbreviations: DWI, diffusion-weighted imaging; FLAIR, fluid attenuated inversion recovery imaging; IQR, interquartile range; LTM, time from last seen normal till magnetic resonance imaging; mRS, modified ranking scale; NIHSS, National Institute of Health Stroke Scale; PWI, perfusion-weighted imaging. Data are
Diffusion-Weighted Imaging/Fluid Attenuated Inversion Recovery Mismatch
The interobserver reliability was high (kappa value = 0.88). The DWI/FLAIR mismatch was present in 118 (42%) patients. An LTM less than 4.5 hours was present in 97 (34%) patients. The mean DWI-lesion volume was 34 mL. There was a strong association between LTM and the presence of the DWI/FLAIR mismatch (
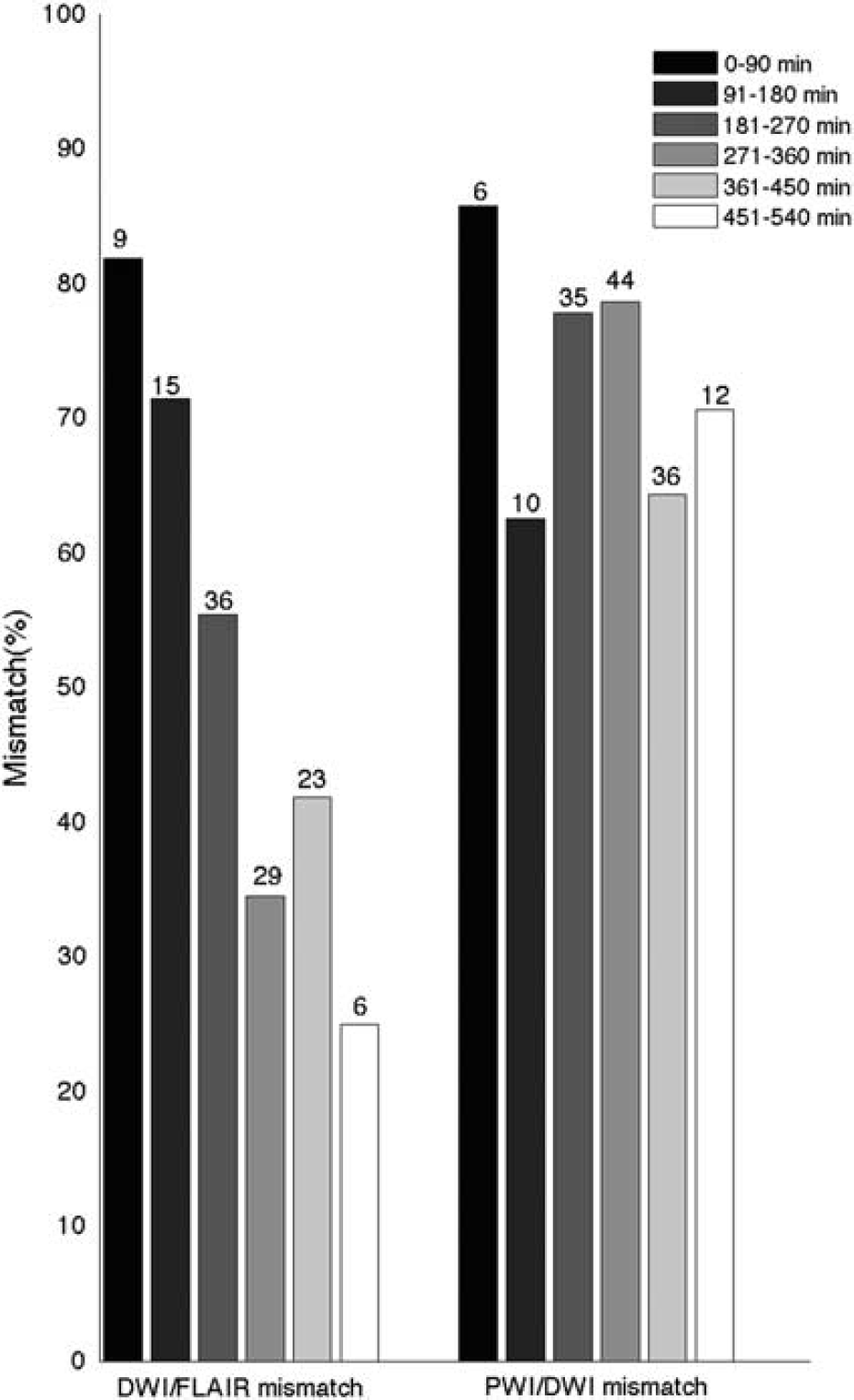
Distribution of mismatch patterns over time. Distribution of diffusion-weighted imaging (DWI)/fluid attenuated inversion recovery (FLAIR) (
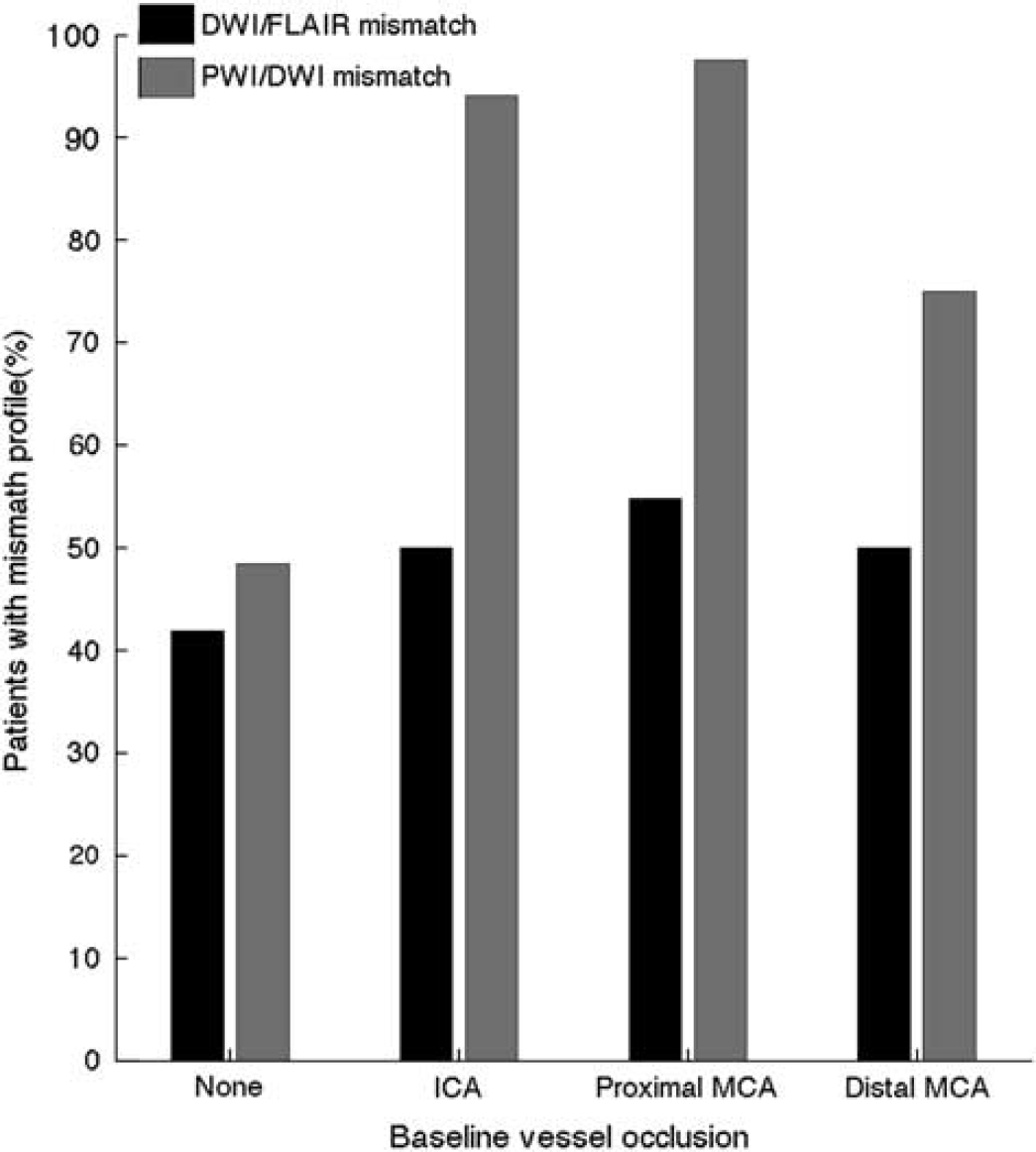
Distribution of mismatch patterns by baseline vessel occlusion. Magnetic resonance angiography (MRA) was present and readable in 154 patients. A four-scale rating was used: no occlusion, internal carotid artery (ACI), proximal medial cerebral artery (MCA), distal medial cerebral artery (MCA). Figure shows the distribution of DWI/FLAIR mismatch (black) and PWI/DWI mismatch (gray). DWI, diffusion-weighted imaging; FLAIR, fluid attenuated inversion recovery; PWI, perfusion-weighted imaging.
Perfusion-Weighted Imaging/Diffusion-Weighted Imaging Mismatch
Coregistration failed in 26 patients (12%), mainly due to poor quality of the original perfusion images, so that 197 patients remained for further analysis. In this group, the mean PWI/DWI mismatch volume was 74 mL. There was no association between the presence of the PWI/DWI mismatch and the LTM (
Association Between Diffusion-Weighted Imaging/Fluid Attenuated Inversion Recovery and Perfusion-Weighted Imaging/Diffusion-Weighted Imaging Mismatch
In 72 patients (37%), both the DWI/FLAIR mismatch and the PWI/DWI mismatch were present, a so-called double mismatch. We compared baseline and imaging characteristics in four groups of patients classified based on their mismatch profile (Table 2): patients with a double mismatch pattern, patients with only the DWI/FLAIR mismatch or the PWI/DWI mismatch, and patients with no mismatch. Patients with a double mismatch had a shorter LTM (270 minutes) when compared to patients with only the PWI/DWI mismatch (360 minutes,
Comparison of clinical and imaging variables between patients with four different mismatch patterns
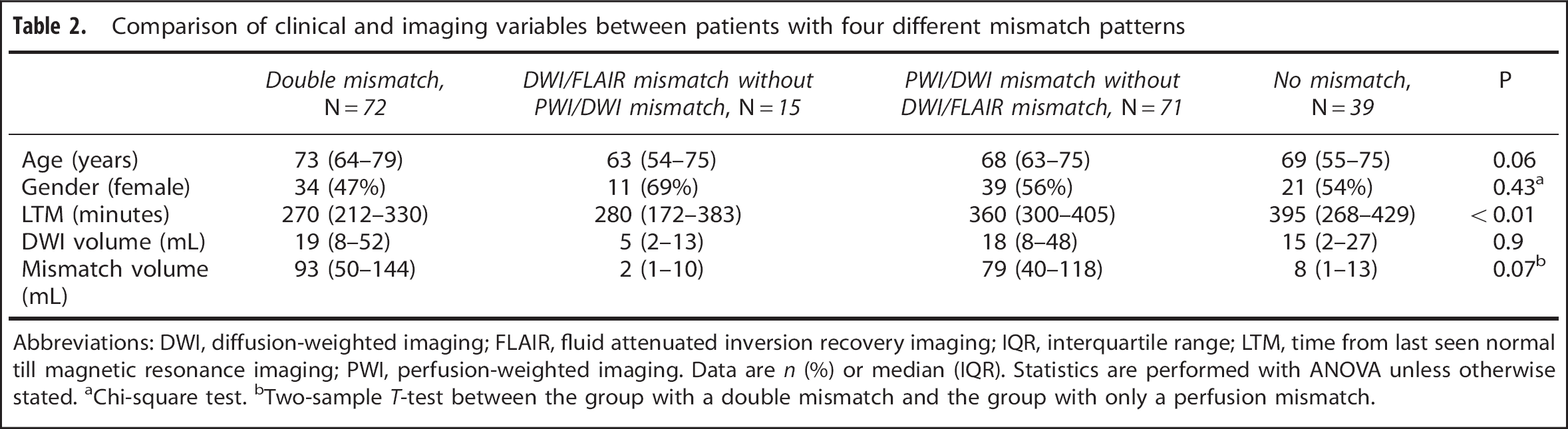
Abbreviations: DWI, diffusion-weighted imaging; FLAIR, fluid attenuated inversion recovery imaging; IQR, interquartile range; LTM, time from last seen normal till magnetic resonance imaging; PWI, perfusion-weighted imaging. Data are
Chi-square test.
Two-sample
In univariate analysis, the presence of the DWI/FLAIR mismatch was associated with the PWI/DWI mismatch, LTM and perfusion-mismatch volume (Table 3). Multivariate analysis confirmed the PWI/DWI mismatch (odds ratio (OR) 2.56; 95% CI 1.24 to 5.30) or alternatively mismatch volume (increase per 10 mL of mismatch volume, OR 1.06; 95% CI 1.01 to 1.12), to be predictors of the presence of the DWI/FLAIR mismatch, independent of LTM.
Significant odds ratios for univariate logistic regression with DWI/FLAIR as dependent variable
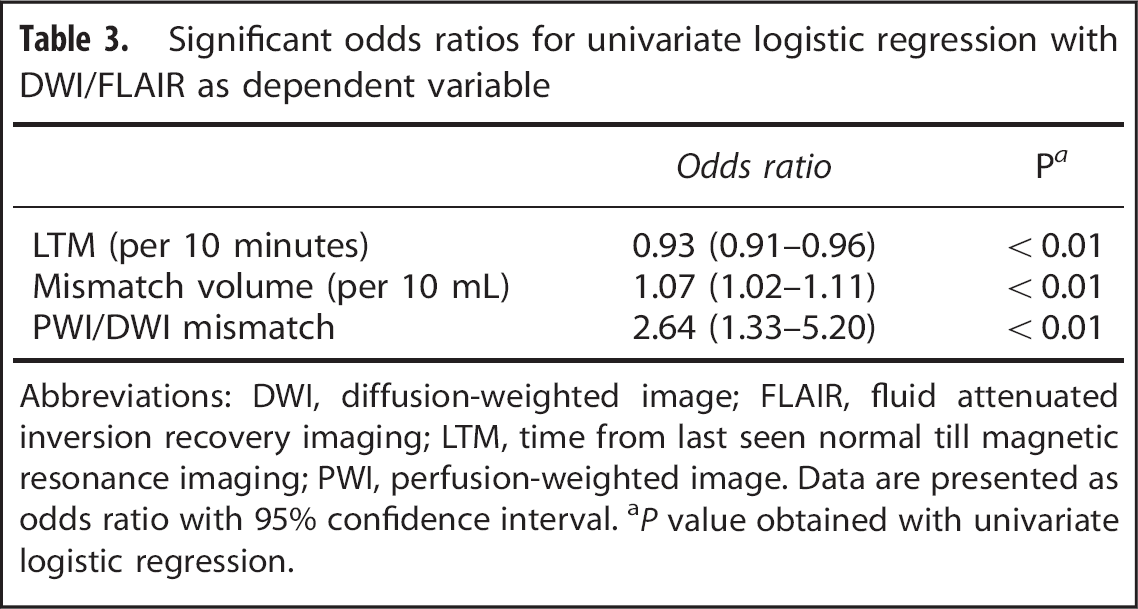
Abbreviations: DWI, diffusion-weighted image; FLAIR, fluid attenuated inversion recovery imaging; LTM, time from last seen normal till magnetic resonance imaging; PWI, perfusion-weighted image. Data are presented as odds ratio with 95% confidence interval.
In a secondary analysis with the volumetric PWI/DWI mismatch method, 30 patients less than with the co-registered method were identified with the PWI/DWI mismatch. Using the volumetric-calculated PWI/DWI mismatch for the multivariate analysis, no independent association was found between the presence of the PWI/DWI mismatch and DWI/FLAIR mismatch (OR 1.66 95% CI 0.94 to 2.93), but the mismatch volume remained an independent predictor (OR per 10 mL of mismatch volume, 1.06 95% CI 1.01 to 1.11).
DISCUSSION
The major finding of our study is the association between the DWI/FLAIR and the PWI/DWI mismatch pattern, independent from the time the patients were last seen normal (LTM). The DWI/FLAIR mismatch was more common when patients had the PWI/DWI mismatch independent of LTM suggesting a common pathophysiologic mechanism. In the PRE-FLAIR study, the visibility of a FLAIR lesion was shown to be increased when DWI volumes were larger.
3
Based on this finding and the association between the two mismatch profiles, present in our study, it could be assumed that DWI-lesion volumes might be smaller in patients with the PWI/DWI mismatch. This hypothesis was assessed in our study, but not confirmed suggesting this not to be a common characteristic of the two mismatch patterns. In this study the presence of the PWI/DWI mismatch and increasing volumes of critical hypoperfusion were associated with less intense FLAIR signal abnormalities. Hyperacute FLAIR abnormalities in the context of stroke are generally thought to reflect vasogenic edema occurring as a result of breakdown of the blood–brain barrier. Larger areas of hypoperfusion may reflect better collateral flow,
16
which in turn might prevent more significant damage to constituents of the blood–brain barrier. This statement is rather controversial as a direct relationship between the size of critical hypoperfusion and the presence of collaterals has not been consistently determined in the previous MR studies.16–18 Dissecting the relationship between the status of collaterals and the development of FLAIR signal abnormalities will be one of the aims in future studies. The association between perfusion and DWI/FLAIR mismatch has not been extensively reported on. One study of 48 patients found no significant difference in perfusion lesion volumes (defined as a time to peak delay of 4 seconds) in patients with the DWI/FLAIR mismatch compared with patients without a FLAIR abnormality.
19
This finding is actually in line with our study, since we found an association only with the mismatch volume, which is not reported in the study of Cheng
In addition, our study suggests that patients with the double mismatch pattern have an earlier stroke onset compared to patients with only the PWI/DWI mismatch profile. Identification of the PWI/DWI mismatch is used to select patients eligible for stroke treatment even outside the 4.5-hour time window.4,5,7,8 The presence of a double mismatch pattern might identify patients who present relatively short after stroke onset. At this moment, the effect on clinical outcome of achieving reperfusion, by an intervention, is expected to be substantial. Although caution is needed in interpreting these results since we cannot exclude bias due to the fact that not for every patient the exact time from symptom onset was known. Further studies are necessary to confirm this finding and to investigate the exact response to treatment of these double mismatch patients.
We were further able to confirm the association between LTM and the presence of the DWI/FLAIR mismatch. In our study, we found a sensitivity of 0.62 and a specificity of 0.69 to predict stroke onset before 4.5 hours. In a substudy 20 of the PRE-FLAIR study in which they used the same liberal rating scale as in our study, they found a sensitivity of 0.86 and a specificity of 0.48. In the AXIS 2 trial, stroke LTM was defined based on the moment the patient was last seen normal whereas in the PRE-FLAIR study 3 the symptom onset was known exactly in all patients. In theory, we predicted the sensitivity to be higher and the specificity to be lower as a consequence of not knowing the exact time from symptom onset. Our results actually show the exact opposite, which implies that other factors influence the sensitivity and the specificity. It has been shown previously that differences in study populations could lead to high variability in the accuracy of DWI/FLAIR mismatch in relationship to time. 21 In other studies, the sensitivity of DWI/FLAIR mismatch prediction of stroke onset before 4.5 hours ranges from 0.38 to 0.79, while the specificity ranges from 0.78 to 0.96.3,22–25 For example with every 10 mL increase in diffusion volume, the odds to find a FLAIR-positive lesion increases with 7%. 3 Sensitivity and specificity are additionally dependent on the prevalence of stroke onset before 4.5 hours. This differed between our study (0.34) and the PRE-FLAIR 3 trial (0.64).
The PWI/DWI mismatch in our study was not associated with LTM. To exclude that this was due to our mismatch definition, we also used a volumetric mismatch method, and again failed to show an association. This finding is similar to recent publications that found a lack of association between PWI/DWI mismatch and LTM6,26 or time from symptom onset. 27 In addition, positron emission tomography studies confirm the existence of hypoxic but still salvageable tissue up to 48 hours after stroke onset. 28 In light of the ‘time is brain’ concept, the lack of time dependency of the PWI/DWI mismatch in the 9-hour time window is counterintuitive. A selection bias cannot be ruled out completely. Investigators could have included more patients with the PWI/DWI mismatch profile in the later time window. In the AXIS 2 study, we deem this rather unlikely since perfusion imaging was not used as an entry criterion into the trial. Of course one cannot exclude a selection bias due to the prespecified inclusion and exclusion criteria.
We found no association between the site of vessel occlusion and the presence of the DWI/FLAIR mismatch, as opposed to the PWI/DWI mismatch. 26 We are not aware of studies investigating the relationship between vessel occlusion and DWI/FLAIR mismatch.
We used the voxel-based PWI/DWI mismatch definition in our study. Previous studies29,30 have proposed to define the PWI/DWI mismatch by this method, which is presumably superior because it does not take into account areas that are already reperfused. The PWI/DWI mismatch as assessed by the most commonly used volumetric approach was no longer associated with the DWI/FLAIR mismatch profile although the relationship between mismatch volume and the presence of the DWI/FLAIR mismatch remained present.
Our study has some limitations. As mentioned earlier when the time of stroke onset was unknown it was defined as the last time the patient was seen normal. This may have resulted in reporting longer times between stroke onset and MR in some patients. As a result, our study population was skewed towards a later time interval from stroke onset to MR compared with previous studies as the PRE-FLAIR study. 3 In other studies6,26 this problem occurred as well and we believe that in clinical practice only a minority of patients would be affected. Second, AXIS 2 patients were allowed to receive tPA before MR of the brain was performed. In the interval between tPA treatment and the MR, reperfusion was likely achieved in some patients resulting in an underestimation of the amount of patients with a perfusion mismatch.
In conclusion, our study shows an association between the DWI/FLAIR and PWI/DWI mismatch. This might point toward a common pathophysiologic pathway between the DWI/FLAIR mismatch and the PWI/DWI mismatch, independent from time after stroke onset. As a result, trials that enroll patients based on the presence of the DWI/FLAIR mismatch are enriched in patients with the PWI/DWI mismatch, which could influence the clinical outcomes after reperfusion. Second, patients with the double mismatch pattern seem to represent a group with an early onset of stroke, a finding that could add a significant value to the use of PWI/DWI mismatch in selection of patients eligible for reperfusion therapy.
Footnotes
AW conceived and designed the research, analyzed and interpreted the data, performed statistical analysis, drafted the manuscript. PD conceived and designed the research, analyzed and interpreted the data, made critical revision of the manuscript. EBR acquired the data, made critical revision of the manuscript. BN, AC, MG, RL, and AS acquired the data, made critical revision of the manuscript. GW analyzed and interpreted the data. GT made critical revision of the manuscript. RLe conceived and designed the research, analyzed and interpreted the data, made critical revision of the manuscript. VT acquired the data, conceived and designed the research, analyzed and interpreted the data, performed statistical analysis, made critical revision of the manuscript.
Dr Wouters reports grants from Rahmenprogramm der Europäischen Union (FP7/2007–2013) gefördert, Grant Agreement Nr. 278276 (WAKE-UP). Dr Chamorro and Prof Ringelstein report being a member of the steering committee of the AXIS 2 trial during conduct of the study. Dr Norrving reports personal fees from SYGNIS during the conduct of the study and personal fees from Bayer, AstraZeneca, and Daiichi Sankyo outside the submitted work. Dr Grond reports personal fees of SYGNIS during the conduct of the study. Dr Schneider and Dr Laage were employees of SYGNIS Bioscience GmbH during the conduct of the study. Dr Thomalla reports grants from European Union. Dr Lemmens is a Senior Clinical Investigator of FWO Flanders and is supported through Fonds Annie Planckaert-Dewaele. Dr Thijs reports being a member of the steering committee of the AXIS 2 trial during conduct of the study. Outside the study he reports speaker's fees paid to his institution by Boehringer Ingelheim, Pfizer, Bayer, and Medtronic. He is also Senior Clinical Investigator of FWO Flanders and is supported by grants from the European Union.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
