Abstract
We combined electrical perforant pathway stimulation with electrophysiological and fMRI recordings in the hippocampus to investigate the effects of neuronal afterdischarges (nAD) on subsequent fMRI BOLD signals in the presence of isoflurane and medetomidine. These two drugs already alter basal hemodynamics in the hippocampus, with isoflurane being mildly vasodilatory and medetomidine being mildly vasoconstrictive. The perforant pathway was stimulated once for 8 seconds with either continuous 20 Hz pulses (continuous stimulation) or 8 bursts of 20 high-frequency pulses (burst stimulation). Burst stimulation in the presence of medetomidine elicited long-lasting nAD that coincided with a brief positive BOLD response and a subsequent long-lasting decrease in BOLD signals. Under isoflurane, this stimulation elicited only short-lasting nAD and only a short-lasting decline in BOLD signals. In contrast, continuous stimulation under isoflurane and medetomidine caused a similar duration of nAD. Under isoflurane, this caused only a sharp and prolonged decline in BOLD signals, whereas under medetomidine, again, only a brief positive BOLD response was elicited, followed by a shorter and moderate decline in BOLD signals. Our results suggest that nAD simultaneously activate different neurovascular coupling mechanisms that then independently alter local hemodynamics in the hippocampus, resulting in an even more complex neurovascular coupling mechanism.
Keywords
Introduction
BOLD-fMRI is a commonly used technique to non-invasively image changes in neuronal activity throughout the brain with high spatial resolution. Nevertheless, BOLD-fMRI still remains only an indirect measure of neuronal activity because it reflects only hemodynamic parameters, namely blood oxygenation and/or blood flow/volume, which in turn are controlled by neuronal activity through a complex mechanism referred to as neurovascular coupling (NVC). As the time course of neuronal processes and the resulting hemodynamic responses are markedly different, direct causal dependencies between the two processes are difficult to confirm, especially when inferring underlying neuronal activity from fMRI data. There is now ample evidence that neurovascular coupling processes are more of a feedforward process, i.e., an increase in neuronal activity causes the release of vasodilatory substances, 1 which in turn increase local blood supply to an extent that exceeds actual oxygen consumption, so that more oxygen-rich blood leaves the capillaries and enters the venous system. This change in blood oxygen saturation, represented by the BOLD signal, depends on both tissue oxygen consumption (the higher it is, the lower the BOLD signal becomes) and changes in blood flow due to the release of vasoactive substances (in which vasodilation leads to an increase and vasoconstriction to a decrease in the BOLD signal). While oxygen consumption is closely related to the instantaneous neuronal activity, the duration of the vascular response mainly depends on the kinetics of the released/activated vasoactive substances. In a recent study, we observed in the rat hippocampus a very long-lasting (i.e., longer than two hours) decline in BOLD signals after only a short period (i.e., 15–20 s) of neuronal afterdischarges (nAD). The nAD-induced sustained decline in BOLD signaling did not correlate with concomitant measured changes in neuronal activity, leading to the suggestion that an existing, normally permanently active vasodilatory mechanism is inhibited 2 as previously also assumed by others. 3 However, these experiments were performed in the presence of medetomidine, which itself may also have a vasoconstrictor effect in the long term.2,4 –6 Although nAD- and medetomidine-induced effects on vasoconstriction appeared to be additive, one cannot completely exclude the possibility that nAD-induced mechanisms may enhance only the vasoconstriction mediated by medetomidine acting on α2-noradrenergic receptors.
Therefore, we question whether the medetomidine- and nAD-induced long-lasting decline in BOLD signaling is mediated by the same or by different vascular mechanisms.
In addition to the vascular effects, α2-noradrenergic receptor activation has been reported to facilitate also the onset of seizure activity,7,8 although there is also evidence that it may inhibits epileptiform discharges. 9 Therefore, it is conceivable that medetomidine also affects nAD during and after electrical perforant pathway stimulation and eased the sustained subsequent decline of baseline BOLD signals in this way. Both the additive effect of medetomidine on hemodynamic (vasoconstriction) and seizure activity (generation of nAD) may indicate that the observed long-lasting decline of baseline BOLD signals critically depends on the co-activation of α2-noradrenergic receptors.
To test whether the continuous co-activation of α2-noradrenergic receptors is both required and necessary to generate the long-lasting decline in baseline BOLD signals after nAD, we performed similar experiments in the presence of isoflurane. This anesthetic was chosen because it does not target α2-noradrenergic receptors and also acts as vasodilator in the long-term.10,11 Considering that high-frequency stimulation of the perforant pathway may also activate the endogenous noradrenergic system 12 and thus α2-noradrenergic receptors, we also performed an experiment with atipamezole, a specific inhibitor of α2-noradrenergic receptors.
Material and methods
Animals
Animals were cared for and used according to a protocol approved by the Animal Experiment and Ethics Committee and in conformity with European conventions for the protection of vertebrate animals used for experimental purposes as well as institutional guidelines 86/609/CEE (November 24, 1986). The experiments were approved by the animal care committee of Saxony-Anhalt state (No. 42502-2-1406 DZNE) and performed according to the Animal Research: Reporting In Vivo Experiments (ARRIVE) guidelines. Male Wistar Han rats (age 9–13 weeks) were housed under conditions of constant temperature (23 °C) and maintained on a controlled 12 h light:12 h dark cycle. Food and tap water were provided ad libitum. A total of 35 rats were included in this study: all rats were used for BOLD fMRI measurements and 17 rats for local field potential (LFP) recordings. There was at least 1 week between the two measurements.
Surgical procedure
Electrode implantation was performed as previously described in detail.2,13 In brief, for electrode implantation, the rats were anesthetized with pentobarbital (40 mg/kg, i.p.) and placed into a stereotactic frame. A bipolar stimulation electrode (114 µm in diameter, made from Teflon-coated tungsten wire, impedance 18–20 KΩ) was placed into the perforant pathway in the right hemisphere at the coordinates AP: −7.4, ML: 4.1 mm from bregma, DV: 2.0 to 2.5 mm from the dural surface. A monopolar recording electrode (114 µm in diameter, made from Teflon-coated tungsten wire, impedance 18–20 KΩ) was lowered into the granular cell layer of the right dentate gyrus at the coordinates AP: −4.0 mm, ML: 2.3 mm from bregma, DV: 2.8 to 3.2 mm from the dural surface. Monosynaptic-evoked field potentials were measured during electrode implantation to control for the correct placement, especially with regard to electrode depth. Grounding and indifferent electrodes (silver wires) were set on the dura through the left side of the cranium and fixed to the skull with dental cement and plastic screws. Following surgery, the animals were housed individually and given 1 week for recovery, with ad libitum food and water.
Combined fMRI and electrophysiological measurements
The experimental setup for simultaneous fMRI and electrophysiological measurements during electrophysiological stimulations of the right perforant pathway in a 9.4 T animal scanner was recently described. 2 Animals were initially anesthetized with isoflurane (1.5%–2%; in 50:50 N2:O2, v:v) and the anesthesia was either maintained with isoflurane (1.0–1.2%), switched to deep sedation by application of medetomidine (Dorbene, Pfizer GmbH, bolus: 50 µg/kg s.c. and after 10 min 100 µg/kg/h s.c.) or continued with an combination of isoflurane (0.8–0.9%) and medetomidine (100 µg/kg/h s.c.) after animals were fixed into the head holder and connected to recording and stimulation electrodes (Figure 1(a)).
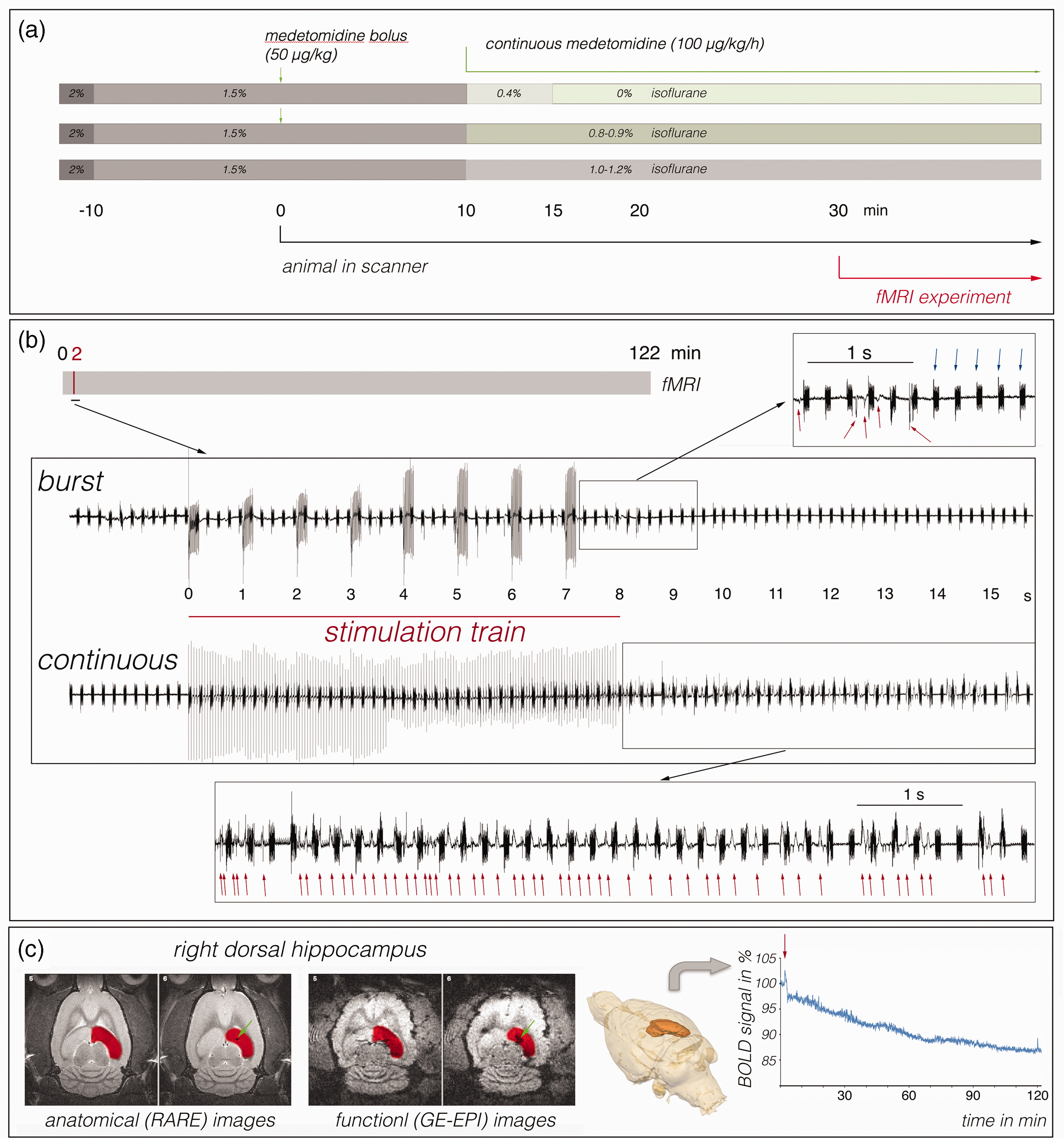
(a) Schematic view of the experimental design. All animals were initially anesthetized with 2% isoflurane. After fixation of the rat in the animal bed, the isoflurane concentration was decreased to 1.5% and all necessary connections (for electrophysiology, physiological measurements and drug injections) were prepared. Anesthesia was then either changed to deep sedation with medetomidine (upper part), maintained at a concentration of 1–1.2% isoflurane (lower part) or continued with a combination of the two treatments (middle part). (b) Example of an electrophysiological recording during an fMRI measurement in the presence of isoflurane. Electrophysiological recordings began just before the start of the stimulation period and lasted for 2 minutes. The upper line shows a recording during burst stimulation and the lower line during continuous stimulation. After cessation of electrical stimulation, clear synchronized neuronal activity (indicated by red arrows) was observed that was either short after burst stimulation or long-lasting after continuous stimulation. The duration of the synchronized neuronal activity was considered the duration of nAD. The occurrence of scanner-related gradient artifacts is indicated by blue arrows and (c) Example of MR-images. Left side: The examined VOI (right dorsal hippocampus) is highlighted in red in the anatomical and the corresponding functional image. A green arrow marks the localization of the recording electrode. Center: The entire analyzed VOI is also shown in the standard 3D brain used. Right side: Development of the BOLD signal intensities of all voxels in the entire VOI over the course of a 2-hour measurement. The perforant pathway was stimulated after 2 min (indicated by a red arrow).
The body temperature was maintained with the help of an animal cradle, which was heated by circulating water. Body temperature was measured in some animals before and after the fMRI session; the temperature remained stable between 37.5 and 38.5 °C in all measured animals. The heart rate and breathing rate were monitored during the entire experiment using an MR-compatible monitoring and gating system for small animals (Model1030, SA Instruments, Inc. Stony Brooks, NY, USA); the breathing rate (under medetomidine between 40 and 60 breaths/min; under isoflurane: 50–70 breaths/min, medetomidine/isoflurane 20–30 breaths/min) and heart rate (under medetomidine: between 220 and 300 bpm under isoflurane: 340–420 bpm, medetomidine/isoflurane 220 and 300 bpm) varied between individual animals but remained stable in all individual animals during the fMRI measurement.
To stimulate the right perforant pathway, electrical bipolar pulses (pulse width 0.2 ms) at 350 µA were used. The following two stimulation protocols were used for all experiments: 1) high-frequency pulse burst stimulation (burst stimulation) and 2) continuous 20-Hz pulse stimulation (continuous stimulation). The two stimulation protocols consisted of a single stimulation period lasting 8 s. During this stimulation period, 20 identical biphasic pulses were applied every second (160 pulses in total) either as burst of 20 pulses with an inter-pulse-interval of 10 ms (i.e., 100 Hz) at the beginning of every second, burst stimulation, or as continuous pulses with an inter-pulse interval of 50 ms (i.e., 20 Hz) during the entire period, continuous stimulation (Figure 1(b)).
The electrophysiological responses were filtered with an antialiasing filter, that is, a low-pass filter (5000 Hz) and a high-pass filter (0.1 Hz) using an EX1 amplifier (Science Products, Hofheim, Germany), transformed by an analog-to-digital interface (power-CED, Cambridge Electronic Design, Cambridge, UK), stored on a personal computer with a sampling rate of 5000 Hz.
All fMRI experiments were performed on a 9.4 T Bruker Biospec 94/20 scanner, equipped with a BGA12 HP (440 mT/m) gradient system. An 86-mm transmit/receive volume coil (Bruker Biospin MRI GmbH, Ettlingen, Germany) was used for radio frequency (RF) excitation and a 20-mm planar surface coil (Bruker Biospin MRI GmbH) was used for signal reception. Initially, a B0-field map was acquired, which was used for local shimming using an ellipsoid that covered the entire brain. BOLD-fMRI was performed by using a gradient-echo echo planar imaging (EPI) sequence with the following parameters: TR 2000 ms, TE 20.61 ms, flip angle 90°, bandwidth 326087 Hz, slice number: 10, slice thickness 0.4 mm, inter slice distance 0.1 mm, field of view (FOV) 25.6 × 25.6 mm, matrix 128 × 128 (in plane resolution 200 × 200 µm).
Trigger pulses that were generated by the scanner at the beginning of every volume—that is, every 2 s—were used to synchronize fMRI image acquisition and electrophysiological stimulation. Each fMRI measurement started with an initial 2-min period without any stimulation (to determine baseline BOLD signals) and then atipamezole or the appropriate stimulation protocol was applied. If atipamezole was administered, then stimulation started 30 minutes later.
After fMRI measurements, 10 horizontal anatomical spin-echo-images (T2-weighted) were obtained using a rapid acquisition relaxation enhanced (RARE) sequence 14 with the following parameters: TR 3000 ms, TE 33 ms, slice thickness 0.4 mm, FOV 25.6 × 25.6 mm, matrix 256 × 256, RARE factor 8, averages 4. The total scanning time was 6.4 min. The slice geometry, 10 horizontal slices, was identical to the previously obtained gradient-echo EPI (Figure 1(c) and supplemental_Figure S1).
LFP recordings
We also recorded LFP outside the scanner to monitor ongoing neuronal activity in the dentate gyrus during and after stimulation. Signals were filtered (high-pass filter: 0.1 Hz; low-pass filter: 5000 Hz) using EX1 amplifiers (Science Products, Hofheim, Germany) and transformed by an analog-to-digital interface (power-CED, Cambridge Electronic Design). Data were recorded by using a sampling rate of 10000 Hz with Spike2 (version 10).
VOI analysis
The functional data were loaded and converted to the BrainVoyager data format. A standard sequence of pre-processing steps implemented in the BrainVoyager QX 22.0 software (Brain Innovation, Maastricht, the Netherlands) such as slice scan time correction and 3D motion correction (trilinear interpolation and data reduction by using the first volume as a reference) were applied to each data set.
Each individual functional imaging data set was aligned to a 3D standard rat brain by using the 3D volume tool implemented in the BrainVoyager software. The 3D standard rat brain had previously been generated from a rat of the same age and strain. The right dorsal hippocampus was marked as volume of interest (VOI) in the 3D standard rat brain (Figure 2(a)). The averaged BOLD time series of all voxels located in this VOI was then calculated for each individual animal using the VOI analysis tool implemented in the BrainVoyager software. Each individual BOLD time series was normalized by using the averaged BOLD signal intensity of 100%. Normalized BOLD time series were then averaged and are depicted as mean BOLD time series ± SD.
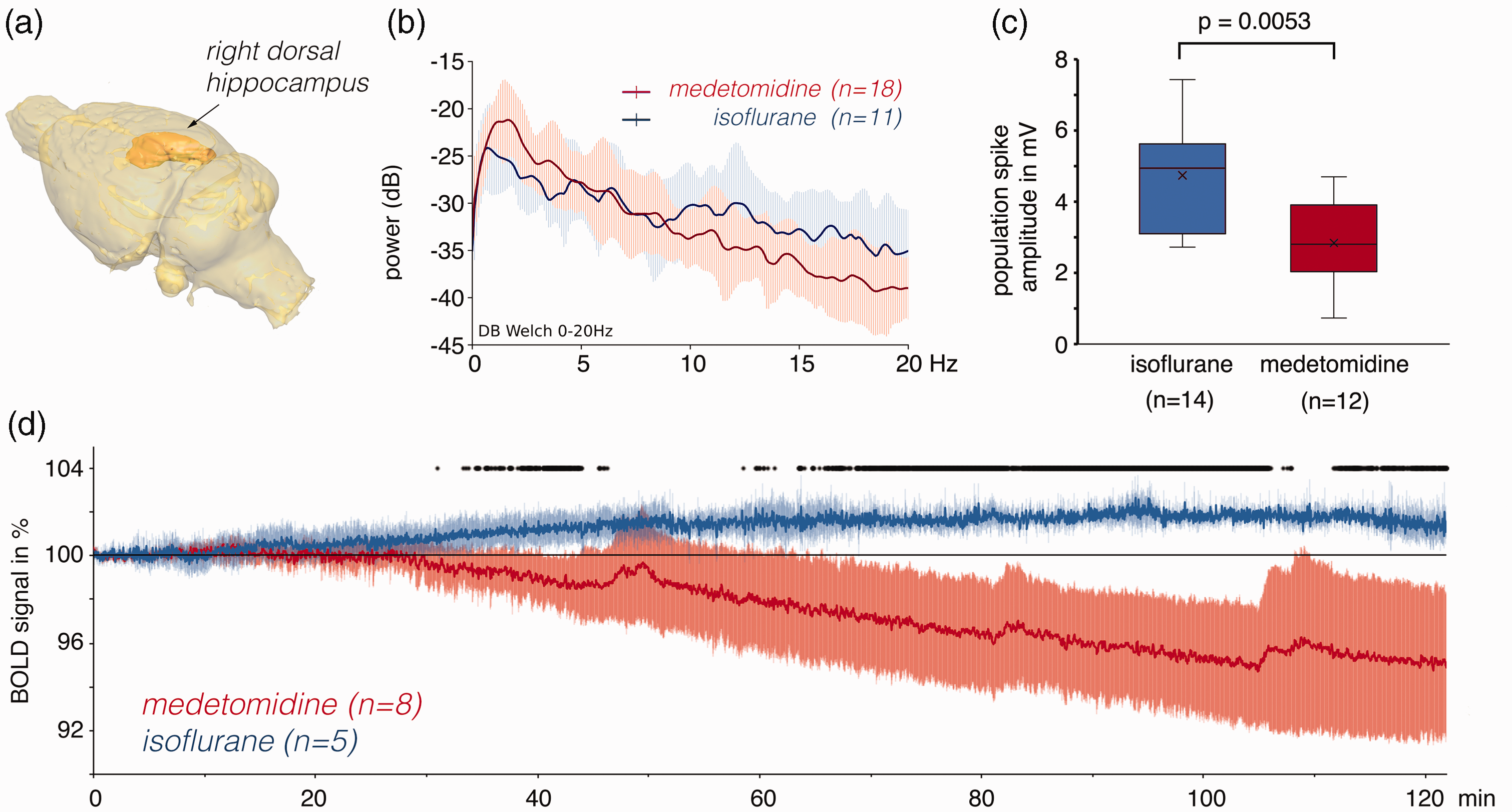
BOLD time series in the right dorsal hippocampus during an fMRI measurement without perforant pathway stimulation. (a) Analyzed region of interest (ROI, right dorsal hippocampus) in the rat brain. (b) Averaged power spectrum of LFP recordings in the right dorsal hippocampus during isoflurane anesthesia (blue line) and medetomidine sedation (red line). Under isoflurane, the delta frequency activation is shifted to the left and the power in the alpha band is increased. (c) A test pulse (biphasic pulse, 350 µA, 0.2 ms) applied immediately before the start of the fMRI measurement elicited in the presence of isoflurane a significantly stronger population spike amplitude in the dentate gyrus than in presence of medetomidine and (d) BOLD time series in the right dorsal hippocampus during a 2 h fMRI session without any electrical stimulation in the presence of isoflurane (blue line) or medetomidine (red line). Significant differences between the two BOLD time series are indicated by black dots at the top.
We calculated significant changes between two BOLD time series as described previously using paired t-tests. 15 We performed for each time point a two-sample equal variance t-test with Bonferroni correction. We considered differences to be significant when p < 0.01. Because we assumed relevant BOLD signal changes (δ) ≥ 0.5%, and baseline BOLD signal variation (σ) of 0.2%, we used a sample size of ≥5 animals per group (n = 16/Δ; 2 with Δ = δ/σ). No anonymizing was done.
Analysis of LFP recordings
The frequency band-specific power from the LFP recordings was estimated from the Hilbert transform of frequency band-filtered LFP recordings as previously described in. 2 Briefly: the data were first down-sampled to 1000 Hz and then they were band-pass filtered using the following ranges: delta (1–4 Hz), theta (4–8 Hz), alpha (8–13 Hz), beta (13–30 Hz), low gamma (30–49 Hz), middle gamma (51–90 Hz), fast gamma (90–140 Hz) and ultra-high gamma (140–170 Hz). The ranges were chosen to remove the 50 Hz line noise. The Hilbert transform was then applied to the filtered data and the instantaneous power at each time point was obtained by calculating the absolute value of the Hilbert transform raised to the second. Using the percentage change method, the power of each frequency band was normalized to the baseline (120 sec before the stimulus application). In this way, it was possible to average together the normalized power of all animals. To quantify the power changes in time from the baseline of each frequency band, the power was divided into 10 sec intervals starting from the stimulus application. For each frequency band, changes in normalized power between the baseline and the 10 sec intervals following the stimulation were tested using either the paired t-test or Bonferroni corrected Wilcoxon test. Normality was tested using the Shapiro–Wilk test. Differences were considered significant when p < 0.05. All data were expressed as mean ± SD. To better visualize the time course of the normalized power of the selected frequency bands, a smoothing window of 20 seconds was applied to the normalized data prior to plotting. The Welch’s method was used to estimate power spectral density in a 6-second interval before stimulus presentation (function: signal.welch, sample rate: 1000 Hz, window: Hann, length 2 sec, overlap: 25%). LFP analysis was performed using Spyder 4.2.5 (Python 3.8.8).
Analysis of stimulus-induced nAD recorded during fMRI
As previously observed,
2
electrical stimulation of the perforant pathway can trigger nAD in the hippocampus, i.e., immediately after stimulation, heavy spiking was observed with an appearance of frequent population spikes followed by large fEPSP (
Results
Isoflurane and medetomidine differentially affect basal neuronal activity and baseline BOLD signals in the dorsal hippocampus during a 2-hour fMRI measurement
When isoflurane was used instead of medetomidine to immobilize animals for fMRI measurement, baseline neuronal rhythmic activity in the delta frequency range shifted to a lower frequency and, in addition, an increase in power was present in the 9–20 Hz range (alpha, beta frequency range: Figure 2(b)). Furthermore, as previously described 16 in the presence of isoflurane, a biphasic test pulse (intensity: 350 µA, 0.2 ms width) elicited a significantly stronger synchronized neuronal response in the dentate gyrus than in the presence of medetomidine (Figure 2(c)). Thus, the neuronal activation state at the beginning of the fMRI measurements was different when isoflurane was used instead of medetomidine.
Also, baseline BOLD signals developed differently when a 2-hour fMRI measurement was performed in the presence of isoflurane instead of medetomidine. In the presence of isoflurane, BOLD signals in the dorsal hippocampus initially increase up to 101.59 ± 0.20% during the first 60 min and remained then relatively stable until the end of a 2 h measurement, 101.34 ± 0.22% (Figure 2(d)). The development of the BOLD baseline signals in the presence of isoflurane was region-specific (supplemental_Figure S3) and thus did not represent an unspecific hardware-related artifact. Thus, in the presence of isoflurane, BOLD signals developed in the opposite direction as under medetomidine, where it decreased to 95.3 ± 0.13% during the first 100 min and remained at this level until the end of the 2 h measurement (95.06 ± 0.13%). 2 In agreement with a previous study, 17 we also did not observe any occurrence of “spontaneous BOLD waves (SBW)” in the presence of isoflurane, as seen in the presence of medetomidine. 2 After the occurrence of SBW, the BOLD signals did not always immediately return to the pre-existing level. This led to the significantly greater variations in mean BOLD signals at later time points in the presence of medetomidine. No SBWs were observed in the presence of isoflurane, and consequently the variation in mean BOLD signals at later time points remained significantly smaller (supplemental_ Figure S4).
Isoflurane and medetomidine differentially affect stimulus-induced neuronal and BOLD responses in the dorsal hippocampus
Next, we checked whether an identical electrical stimulation of the perforant pathway would also induce identical BOLD signal changes under isoflurane anesthesia or medetomidine sedation. Under isoflurane anesthesia, electrical stimulation of the right perforant pathway with 8 high-frequency pulse bursts (i.e., burst stimulation) only elicited short-lasting neuronal afterdischarges (nAD, duration: 3.1 ± 1.2 s). Thus, only a small number of additional population spikes appeared during the end and after the stimulation ceased (Figures 1(b) and 3(a)). The result of the induced neuronal activity was only a minor non-significant positive BOLD signal increase in the right dorsal hippocampus which then transiently declined and returned to the initial level within 15 min before it gradually increased until the end of the measurement to about 102% (light blue lines, Figure 3(b)) as observed in the absence of any stimulations.
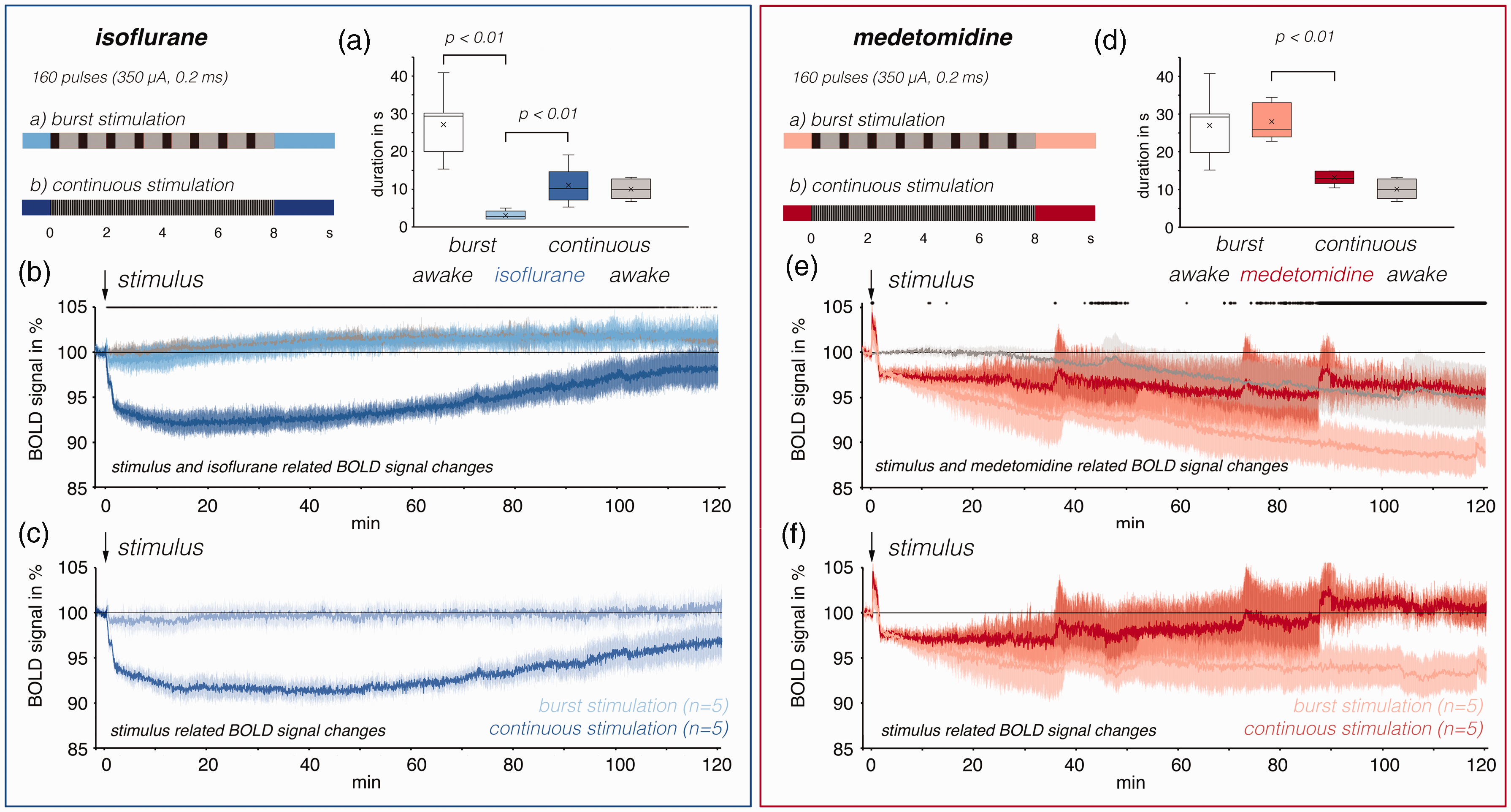
Development of BOLD signals in the right dorsal hippocampus during and after one stimulation period in presence of isoflurane or medetomidine. (a) Either a burst stimulation or a continuous stimulation protocol was applied in the presence of isoflurane. Both stimulation protocols elicited neuronal afterdischarges (nAD), recorded in the dentate gyrus, which were significantly longer after continuous stimulation (top panel). (b) Measured BOLD time series in the right hippocampus. One continuous stimulation train (indicated by the black arrow at 2 min) caused a long-lasting decline of BOLD signals, which almost recovered after 2 h (dark blue line). In contrast, one burst stimulation train only caused a subsequently minor transient decrease in BOLD signals (light blue line). For comparison, the development of BOLD signals when no stimulation was given is depicted as a gray line. Significant differences in the BOLD time series between the two stimulation conditions are indicated as a black line at the top. (c) For the calculation of only stimulus-related BOLD signal changes, BOLD time series without stimulation was subtracted from the BOLD time with stimulation. (d) Rerun of the same experiment in the presence of medetomidine. Under medetomidine significantly stronger nAD were induced by burst stimulation than by continuous stimulation. The duration of nAD after burst and continuous stimulation was similar to the duration of nAD in awake animals (white and gray box). (e) In agreement, the nAD-induce decrease in BOLD signals was stronger and more prolonged after burst stimulation. Under medetomidine, both stimulation protocols triggered a significant short-lasting positive BOLD response in the right dorsal hippocampus and (f) Calculation of only stimulus-related BOLD signal changes.
Stimulation with the same number of pulses but applied as continuous 20 Hz pulses for 8 sec (i.e., continuous stimulation) elicited nAD in the dentate gyrus lasting for 11.10 ± 4.15 s (Figure 3(a)). Even under this condition, averaged BOLD signals in the entire right dorsal hippocampus barely increased during stimulation but rapidly dropped significantly by more than 7% within 2 minutes after stimulation. BOLD signals remained at this low level for at least 30 minutes before slowly recovering thereafter (dark blue lines, Figure 3(b)). Two hours after stimulation, BOLD signals then reached a level of 98.2 ± 1.8%, which was still significantly lower than the level reached at the same time after burst stimulation (101.8 ± 1.3%) or without any stimulation (101.34 ± 0.22%). Taken together, the results obtained under isoflurane anesthesia show that continuous stimulation caused a strong decline of BOLD signals in the right dorsal hippocampus for about 2 hours, whereas burst stimulation had only a minor effect during the first 15 minutes (Figure 3(c)).
Similar to the different development of the BOLD signals after the two stimulation conditions, the ongoing neuronal rhythmic activity was also affected differently by the two stimulation protocols in presence of isoflurane. Whereas continuous stimulation significantly suppressed the power of various frequency bands (mainly in the theta (4–8 Hz), beta (13–30 Hz) and gamma, low to ultra-high) for about 80–90 min, burst stimulation only affected the power of the theta (4–8 Hz) and to a lesser extend of the alpha (8–13 Hz) frequency band. Thus, the decrease in BOLD signals after nAD coincides with the decreased power of the gamma frequency bands rather than with changes in the power of the low frequency bands (Figure 4, supplemental_Figure S5).
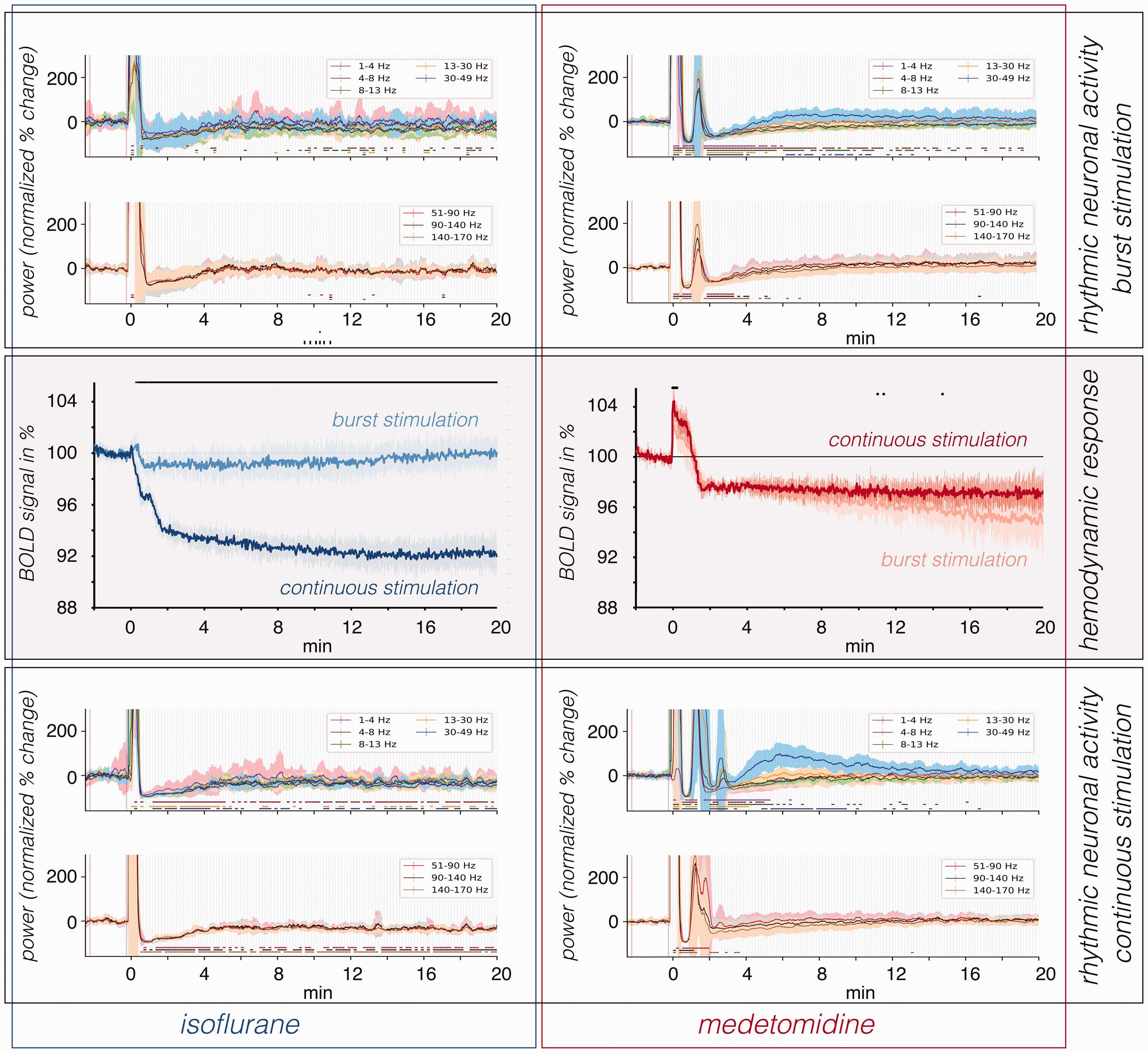
Summary of BOLD signal changes and corresponding changes in the power of various frequency bands in the dorsal hippocampus during all used stimulation conditions. Left side: In presence of isoflurane burst stimulation only caused a short and minor decrease in BOLD signals (light blue line) whereas continuous stimulation elicited an immediate and long-lasting strong decline of BOLD signals in the right dorsal hippocampus (middle panel, dark blue line). Both stimulation protocols caused sustained reduction of the average band power in low frequency bands but only continuous stimulation caused a sustained decline of neuronal activity in high-frequency bands. Right side: In the presence of medetomidine, the two stimulation protocols caused an initial transient positive BOLD response that is followed by a similar decline in BOLD signals (middle panel). The initial decline remained relatively stable after continuous stimulation dark red line) but decreased even further after burst stimulation (light red line). Again, the two stimulation protocols caused a reduction in the average band power in low frequency bands; however, there was no sustained decline in high-frequency bands. BOLD signal changes: significant differences between the two BOLD time series are indicated by the black dots on top. Power (normalized % change): significant differences in the average power from the baseline were indicated by lines at the bottom of the graph. The colors used to define each frequency band and the significant differences are shown in the legend. The vertical dotted gray lines mark the 10 sec intervals used for the statistical analysis.
Repeating the same experiments in the presence of medetomidine showed that the two stimulation protocols also elicited nAD with different durations. As previously observed, 2 burst stimulation elicited long-lasting (24.62 ± 8.26 s) nAD, whereas continuous stimulation elicited nAD that lasted for only 13.25 ± 1.66 s (Figure 3(d)).
Also, in the presence of medetomidine, the two simulation protocols elicited different hemodynamic responses in the right hippocampus. First, the two stimulation protocols elicited strong significant immediate positive BOLD responses that lasted for approximately 60 seconds. The stimulus-induced positive BOLD response consisted of an initial short strong positive component that was followed by a longer-lasting plateau-like component. Only the initial component was significantly stronger during continuous stimulation when compared to burst stimulation (continuous stimulation: 104.65 ± 0.89; burst stimulation: 102.90 ± 0.93; p = 0.016; Figures 3(e) and 4). Then, BOLD signals quickly declined below the initial level. The decrease in BOLD signals was initially similar during the two stimulation conditions, but then decreased more after burst stimulation (Figures 3(e) and 4). Thus, at the end of the fMRI experiment (i.e., after 120 minutes), the BOLD baseline signals were significantly lower after burst stimulation than after continuous stimulation. Although occasionally positive “spontaneous BOLD waves” 17 appeared, BOLD signals did not recover until the end of the measurement (i.e., during 2 h after stimulation). The final decrease in BOLD signals at the end of the measurement again related to the strength of stimulus-induced nAD, which was significantly longer after burst stimulation. Taking into account the intrinsic effect of medetomidine on the development of BOLD signals, continuous stimulation reduced BOLD signals for about 35 minutes, but burst stimulation reduced them for at least 2 hours (Figure 3(f)).
Although the BOLD time series differed after stimulation with the two stimulation protocols, the effects on ongoing rhythmic activity in the right hippocampus were quite similar. The two stimulation protocols caused a transient decline (<5 min) followed by a transient increase of the power in the low gamma frequency range. After burst stimulation, the transient decline in power for the theta and alpha band was longer (approximately 11 min) (Figure 4). Thus, in the presence of medetomidine, the long-lasting decline of BOLD signals after burst stimulation was not accompanied by a long-lasting reduction of rhythmic neuronal activity in the gamma frequency range, as observed in the presence of isoflurane.
In addition to a different sensitivity for the generation of nADs, the presence of isoflurane and medetomidine also caused qualitatively different neuronal activity after one stimulation period. LFP measurements performed outside the scanner revealed a second period of increased neuronal activity approximately 1 minute after stimulation only in the presence of medetomidine (Figure 5). This secondary increase in neuronal activity coincided with the period in which BOLD signals further declined; after this period of increased neuronal activity, BOLD signals remained at this low level. This secondary increase in neuronal activity was not present when nAD were induced in the presence of isoflurane. Nonetheless, a decrease in BOLD signals was also observed during this period so this may be more likely caused by the end of the silent period that began after the initial nAD.
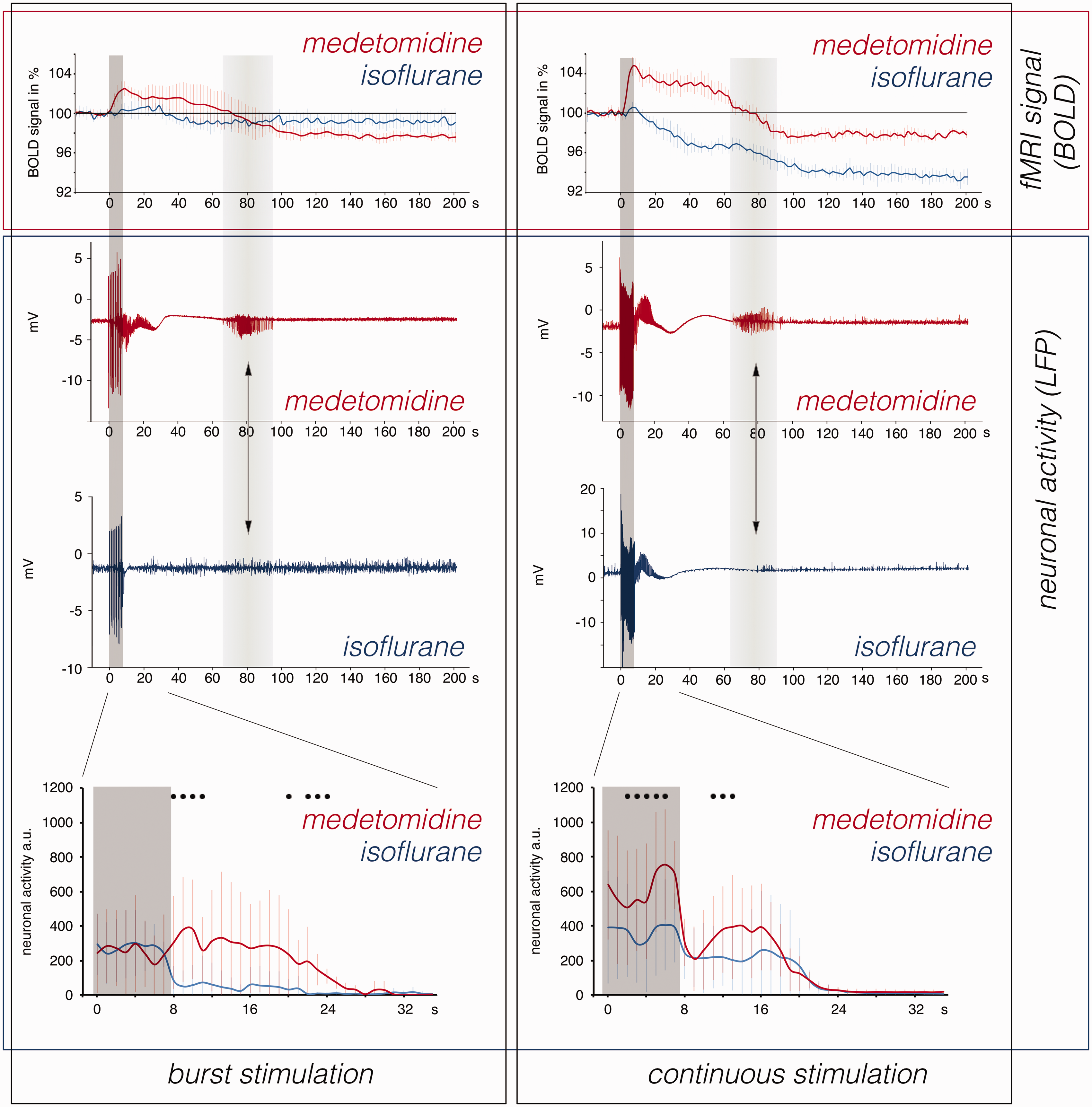
Temporal comparison of BOLD signals (upper panel) and raw LFP recordings (middle panel) in the right dorsal hippocampus recorded during the first 200 seconds after stimulation in the presence of isoflurane (blue lines) or medetomidine (red lines).
In summary, continuous stimulation of the perforant pathway in the presence of medetomidine or isoflurane caused in the right dentate gyrus nAD of similar duration. In the presence of medetomidine, these nADs elicited an immediate, short-lasting positive BOLD response in the right dorsal hippocampus, but no significant positive BOLD response in the presence of isoflurane. Thereafter, in the presence of medetomidine, there was only a moderate decrease in BOLD signals whereas in the presence of isoflurane, there was a strong decrease in baseline BOLD signals below the initial value. The decrease in BOLD baseline signals was (when subtracting the intrinsic effect of the used anesthetic/sedative) normalized after 2 hours in the presence of isoflurane and after about 35 min in the presence of medetomidine. In contrast, burst stimulation caused long-lasting nAD in the presence of medetomidine, but only very short nAD in the presence of isoflurane. In the presence of medetomidine, baseline BOLD signals decreased for more than 2 hours, but in the presence of isoflurane, they decreased only very slightly and only for a short period. Furthermore, an appearance of positive “spontaneous BOLD waves” 17 was only observed when the animals were sedated with medetomidine but not in presence of isoflurane.
Under isoflurane, the inhibition of noradrenergic α2 receptors does not affect baseline BOLD signals but attenuates nAD-related long lasting decline
In the presence of medetomidine, continuous activation of noradrenergic α2-receptors led to a decrease in BOLD basal signals. Therefore, we tested whether the long-lasting decrease in BOLD basal signals induced by nAD also depended on these receptors. This might be the case when the endogenous noradrenergic system becomes activated during generations of nAD.
Application of the noradrenergic α2 receptors antagonist atipamezole (1 mg/kg) in the presence of isoflurane did not significantly affect the development of baseline BOLD signals during a 2-hour fMRI measurement (Figure 6(a)), thus there is no obvious ongoing activity of noradrenergic α2 receptors that affects baseline BOLD signals.
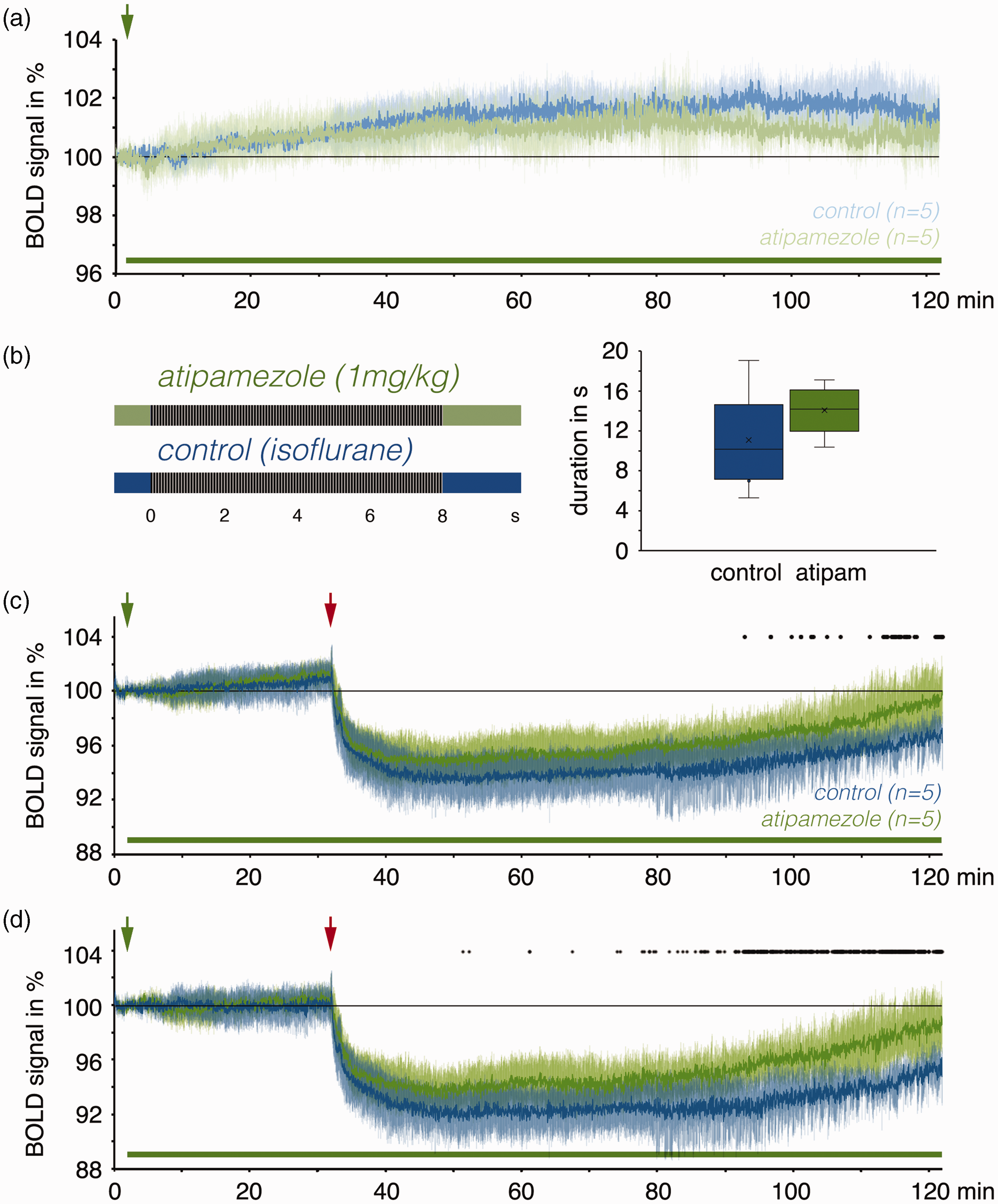
Effect of atipamezole on stimulus-induced BOLD signal changes. (a) Atipamezole was applied during isoflurane anesthesia. Atipamezole was injected 2 min after the start of the fMRI measurement (indicated by the green arrow and green line at the bottom) and the presence of atipamezole did not significantly affect baseline BOLD signals within the subsequent 2 h. (b) Atipamezole did not modify the duration of stimulus-induced nAD. (c) Presence of atipamezole did not affect the initial decline of BOLD signals after continuous stimulation (indicated by the red arrow) but shortened the recovery (control: blue line, atipamezole: green line). Significant differences between the two BOLD time series are indicated by black dots in the top area and (d) Calculation of stimulus-related BOLD signal changes (i.e., subtracting baseline BOLD time series shown in A from corresponding stimulus-related BOLD time series shown in C (significant differences between the two BOLD times series are indicated by black dots in the top area).
The presence of atipamezole had no significant effect on the generation and duration of nAD induced by continuous stimulation under isoflurane (Figure 6(b)). However, the sustained decline of BOLD signals after the stimulation period was shortened by atipamezole, indicating that the induction of the prolonged decline in BOLD signals after nAD-mediated is partly mediated by noradrenergic α2 receptors (Figure 6(c) and (d)).
Stimulation in the presence of medetomidine and isoflurane induced hemodynamic responses like those in the sole presence of isoflurane
Continuous stimulation elicited nAD of equal duration in the presence of isoflurane and medetomidine. However, the BOLD responses differed between the two conditions. Whereas the initial BOLD responses elicited by nAD were stronger in the presence of medetomidine, the subsequent sustained decrease in BOLD signals was stronger in the presence of isoflurane. To test how (I) the initial positive BOLD response and (II) the subsequent sustained decline in BOLD signals are modulated by isoflurane or medetomidine, we repeated the stimulation experiment in the simultaneous presence of medetomidine and isoflurane. Because the concomitant use of isoflurane and medetomidine strongly suppressed respiratory rate, we reduced the isoflurane concentration to 0.8–0.9% (instead of 1.0–1.2% with isoflurane treatment alone) and finished the fMRI measurements as early as 20 minutes after stimulation.
Under this condition, continuous stimulation elicited nAD that lasted 15.4 ± 4.4 s, a duration similar to that in the presence of isoflurane or medetomidine alone. The neuronal activity triggered by the stimulus elicited only a minor, nonsignificant initial positive BOLD response (as also occurred in the presence of isoflurane alone), and was followed by a sustained decrease in BOLD signals to approximately 93.6 ± 0.1%, which was also similar to the decrease seen in the presence of isoflurane alone (Figure 7). This suggests that isoflurane inhibited the initial positive BOLD response and medetomidine did not interfere with the sustained decline in BOLD signals.
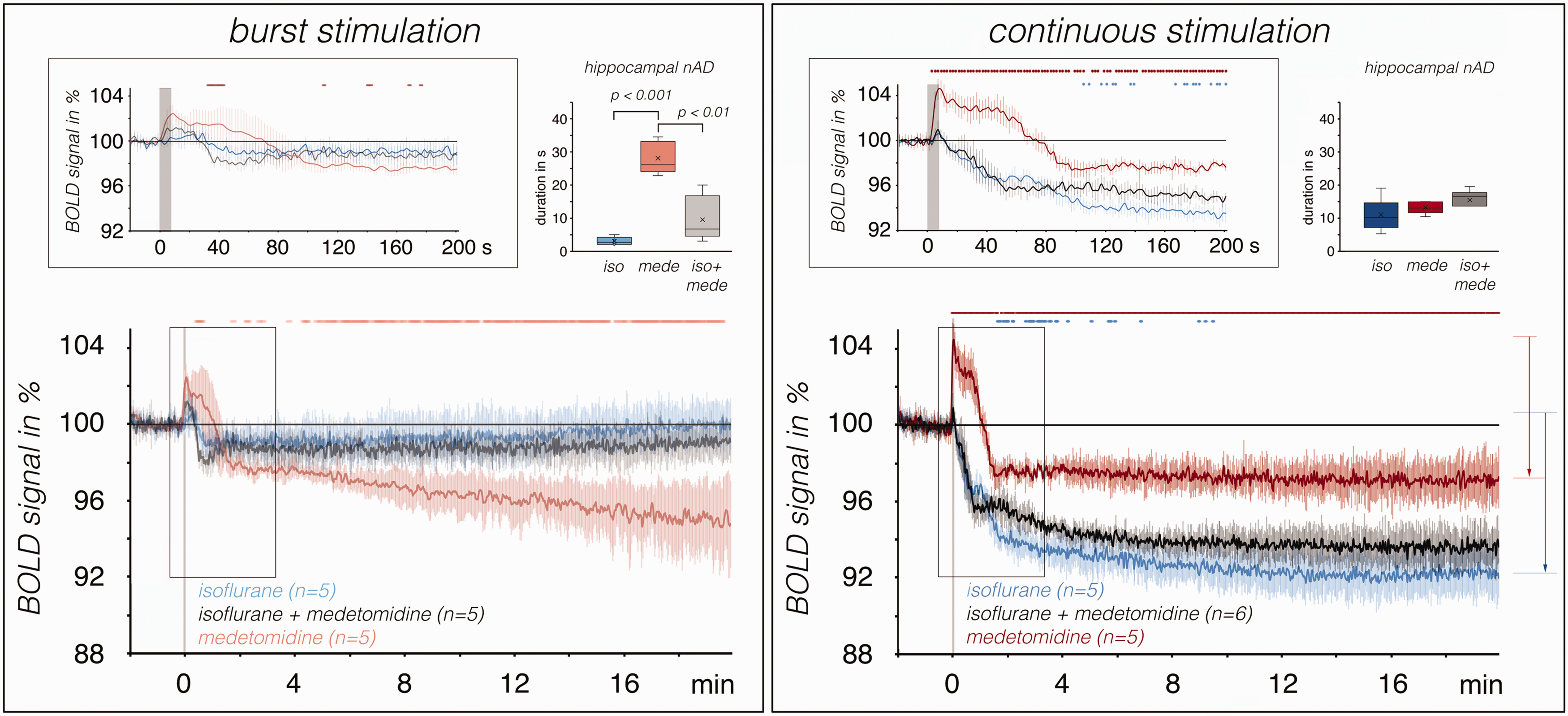
Comparison of BOLD time series recorded during burst or continuous stimulation in the presence of medetomidine (red lines), isoflurane (blue lines) or a combination of the two drugs (black lines).
When burst stimulation was performed in the presence of medetomidine and isoflurane, nAD were elicited, whose duration was similar to that in the presence of isoflurane alone. The associated BOLD time series was again similar to the BOLD time series observed in the presence of isoflurane. The main difference was that there was also an initial positive BOLD response, albeit in an attenuated form, similar to that observed in the presence of medetomidine alone. This again indicates that the additional administration of medetomidine during isoflurane anesthesia did not alter the stimulus-induced BOLD time series. Or, conversely, when isoflurane was additionally administered to medetomidine treatment, both the formation of nAD and the subsequent long-lasting decline in baseline BOLD signals were suppressed.
Discussion
Many sedative and anesthetic drugs affect not only the ongoing neuronal activity, as they are intended to do, but also general hemodynamics in the brain. Because local hemodynamics are eventually controlled by smooth muscle cell contractility in arterioles and to a lesser extent in veins and by pericytes in capillaries, 18 neuronal-activity-related via neurovascular coupling (NVC) and anesthetic/sedative-related mechanisms should converge at this point. Thus, a neuronal-activity-related effect on individual hemodynamic parameters (e.g., BOLD signals) is always superimposed on preexisting hemodynamic effects that are already altered by anesthetics/sedatives. If the two mechanisms would eventually converge at the same mechanism (i.e., Ca2+ induced myosin contraction) at the same place in the local microvasculature, then 2 general scenarios are feasible. First, an “additive effect”: stimulus-induced neuronal activity that causes vasoconstriction should be enhanced in the presence of a vasoconstrictive drug, like medetomidine, but may be attenuated in the presence of a vasodilative drug, like isoflurane. Alternatively, second, there could be a “saturation effect”: if the anesthetic/sedative used already causes vasodilation or vasoconstriction, then subsequently altered neuronal activities that would also cause the same hemodynamic response no longer result in a measurable hemodynamic response.
Although these two assumptions are partly supported by several studies describing different hemodynamic responses to identical stimuli in the presence of various anesthetics/sedatives,19 –23 they are, however, far too simplistic as our current results show.
Stimulation of the perforant pathway simultaneously activates distinct neurovascular coupling mechanisms
As depicted in Figure 5, continuous stimulation caused a clear immediate positive BOLD response in the presence of medetomidine but not in the presence of isoflurane, although neuronal responses to individual pulses were significantly higher under isoflurane and the duration of nAD was similar under both conditions (Figures 2 and 3). It could be argued that isoflurane already causes vasodilation before the onset of stimulation, so that an additional stimulus-induced NVC mechanism might no longer be effective (i.e., “saturation effect”). However, previous experiments revealed that continuous stimulations can trigger immediate positive BOLD responses in the presence of isoflurane, but only when these stimulations were repeated at least 3 times with an inter-stimulus interval of 1 min.13,24
Moreover, the subsequent decline in BOLD signals was significantly stronger in the presence of isoflurane than in the presence of medetomidine, although, again, the duration of nAD did not differ significantly under these two conditions. Thus, one might conclude that isoflurane inhibits an NVC mechanism that causes the immediate positive BOLD response and simultaneously activates an NVC mechanism that mediates the sustained negative BOLD response downstream of nADs, or alternatively, that medetomidine activates an NVC mechanism that mediates a positive BOLD response and simultaneously inhibits an NVC mechanism that causes the sustained BOLD signal decline. To clarify this unambiguously, one would have to perform these fMRI measurements also in awake animals, but this is not really practical in this experimental setup (i.e., after chronic electrode implantation and in combination with electrophysiology) so far. However, when the same stimulation was presented in the presence of isoflurane and medetomidine, the resulting BOLD time series was almost identical to that observed in the presence of isoflurane alone. Consequently, the presence of isoflurane inhibits an NVC mechanism that causes the initial positive BOLD response, and the presence of medetomidine does not inhibit the NVC mechanism responsible for the sustained decline in BOLD signals after nAD. Because isoflurane inhibited only the initial positive BOLD response, it became clear that stimulus-induced neuronal activity also simultaneously activated an NVC mechanism that led to the long-lasting decline in BOLD signals. Thus, these were not 2 consecutive processes but 2 different NVC mechanisms running in parallel. When these two opposite NVC mechanism were activated in parallel it also means that they were mediated by spatially distinct vascular elements, as suggested recently. 18 Why after continuous stimulation in the presence of isoflurane the sustained decline in BOLD signals was more pronounced than in the presence of medetomidine cannot be clearly answered. However, it is noticeable that the overall decrease in BOLD signals after the end of the stimulation period was very similar in the presence of isoflurane or medetomidine (Figure 7, supplemental_Figure S6). That is, in the presence of isoflurane, only the initial increase in BOLD signal was absent, but the subsequent decrease was as strong as in the presence of medetomidine. This again would indicate, that the two NVC mechanisms act in parallel and independently of each other.
When burst stimulation was applied in the presence of isoflurane and medetomidine, the initial positive BOLD response was only attenuated and not completely blocked as was the case in the presence of isoflurane alone (Figure 7). It should be noted that when isoflurane and medetomidine were used concomitantly, the isoflurane concentration was reduced to 0.8–0.9% (instead of 1.0–1.2%). Thus, during burst stimulation, NVC mechanisms responsible for the initial positive BOLD response were less affected by isoflurane than NVC mechanisms activated during continuous stimulation. Therefore, we assume that the two stimulation protocols activate partly different NVC mechanisms all of which mediate the initial transient BOLD response. The experiments with medetomidine, in which continuous and burst stimulation resulted in significantly different initial BOLD responses (Figure 4), already strongly suggested that the two stimulation protocols activate different NVC mechanisms. Therefore, we suggest that isoflurane effectively inhibits an NVC mechanism that is activated primarily during continuous stimulation and to a lesser extent during burst stimulation.
In summary, continuous stimulation simultaneously activates two different NVC mechanisms, with isoflurane inhibiting only the mechanism mediating the initial positive BOLD response. Both mechanisms appear to be additive in their action, which is due to the fact that they are mediated by different vascular components. In addition, burst stimulation appears to activate different NVC mechanisms than continuous stimulation, and these mechanisms seem to be less affected by the presence of isoflurane.
Medetomidine and isoflurane differently affect the generation of nAD in the hippocampus
If similar BOLD responses to an identical stimulus are elicited in the presence of different drugs, then it implies that the stimulus-induced neuronal activity and the resulting activated NVC mechanisms are not inhibited (or, very unlikely, identically affected) by these treatments. In our experiments, this was never the case. In contrast, if an identical stimulus generates different BOLD responses in the presence of a different anesthetic/sedative, then these drugs differently affect either NVC mechanisms, neuronal activity or both. In our experiments, this was the case for the stimulus-related immediate positive BOLD response and for the nAD-related sustained negative BOLD response. Medetomidine and isoflurane act on completely different neuronal mechanisms to sedate or anesthetize the animals. Medetomidine acts as a presynaptic noradrenergic α2 receptor agonist, thereby reducing transmitter release, i.e., neuronal transmission. In contrast, isoflurane acts at various targets; it is a positive allosteric GABAA receptor modulator, NMDA-receptor inhibitor, tandem pore (K2P9) potassium channel inhibitor as well as inhibitor of the plasma membrane calcium ATPase. 25 Because of these different modes of action, we observed different population spike amplitudes in responses to an identical test stimulus, different basal rhythmicity and different susceptibility to nAD generation in the presence of isoflurane or medetomidine (Figures 2 and 3). Whereas continuous stimulation produced nAD of equal duration in the presence of isoflurane or medetomidine or awake animals, it was completely different when burst stimulation was applied. Then, short nAD were elicited in the presence of isoflurane, but very strong nAD was elicited in the presence of medetomidine. In awake animals, the duration of nAD was similar to that observed with medetomidine. All of this suggests that there are at least two different mechanisms that trigger nAD in the hippocampus and that only one of them is inhibited by isoflurane. Because nAD activates the NVC mechanisms that mediate the long-lasting decline in BOLD signals, this decline was absent when burst stimulation was applied in the presence of isoflurane.
However, even if the duration of nAD during continuous stimulation in the presence of isoflurane or medetomidine was very similar, the subsequent BOLD responses still differed markedly, at least in terms of the time needed to return to the original BOLD baseline values (Figure 3). One could argue that it is not the duration but rather the quality of the nAD that determines the duration of the subsequent BOLD signal decline. After all, nAD in the presence of isoflurane and medetomidine differed by the presence or absence of a second phase of increased neuronal activity and severity (Figure 5), suggesting that different NVC mechanisms were activated. Strikingly, not only was the nAD-dependent BOLD signal drop (i.e., BOLD signal changes after onset of nAD) very similar under isoflurane and medetomidine (as mentioned above), but also the subsequent slow recovery (Figure 7, supplemental_Figure S6). Thus, this suggests that the nAD-induced NVC mechanisms underlying the sustained BOLD signal decline under isoflurane and medetomidine are actually the same, they just started at a different level.
In summary, our results indicate that at least 2 different NVC mechanisms are activated simultaneously during a continuous stimulation period. One NVC mechanism caused an immediate transient positive BOLD response and was inhibitable by isoflurane (but not by medetomidine), while a second caused a sustained negative BOLD response. Because both NVC mechanisms are additive and independent, they must be mediated by different components (cells) of the vasculature. Therefore, interpretation of BOLD responses should take into account the fact that NVC mechanisms are controlled not only by different neuronal activity but also by different vascular processes acting in parallel.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X231221039 - Supplemental material for Hemodynamic responses in the rat hippocampus are simultaneously controlled by at least two independently acting neurovascular coupling mechanisms
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X231221039 for Hemodynamic responses in the rat hippocampus are simultaneously controlled by at least two independently acting neurovascular coupling mechanisms by Alberto Arboit, Karla Krautwald and Frank Angenstein in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Deutsche Forschungsgemeinschaft (DFG) – Project-ID 425899996-SFB 1436.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
FA: Conception and design, Acquisition of data, Analysis and interpretation of data, Drafting and revising the article; AA: Conception and design, Acquisition of data, Analysis and interpretation of data, revising the article: KK: Acquisition of data, Analysis of data, revising the article
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
