Abstract
The stage of neurogenesis can be divided into three steps: proliferation, migration, and differentiation. To elucidate detailed relations between these three steps after ischemia, the authors evaluated the three steps in the adult gerbil dentate gyrus (DG) after 5 minutes of transient global ischemia using bromodeoxyuridine (BrdU), highly polysialylated neural cell adhesion molecule (PSA-NCAM), and neuronal nuclear antigen (NeuN) and glial fibrillary acidic protein (GFAP) as markers for proliferation, migration, and differentiation, respectively. Bromodeoxyuridine-labeled cells increased approximately sevenfold, and PSA-NCAM–positive cells increased approximately threefold in the subgranular zone (SGZ) with a peak 10 days after ischemia. Bromodeoxyuridine-labeled cells with PSA-NCAM expression were first detected both in the SGZ and the granule cell layer (GCL) 20 days after ischemia and gradually decreased after that, whereas BrdU-labeled cells with NeuN gradually increased in the GCL until 60 days after ischemia. A few BrdU-labeled cells with GFAP expression were detected in DG after ischemia; no PSA-NCAM–positive cells with GFAP expression were detected, but the radial processes of glial cells were partly in contact with PSA-NCAM–positive cell bodies and dendrites. These results suggest that neural stem cell proliferation begins at the SGZ, and that the cells then migrate into the GCL and differentiate mainly into neuronal cells. The majority of these three steps finished in 2 months after transient global ischemia.
Persistent neurogenesis occurs in discrete regions of the adult brain, including the subgranular zone (SGZ) of the hippocampal dentate gyrus (DG) (Altman and Das, 1965; Kaplan and Hinds, 1977; Bayer et al., 1982) and the subventricular zone of the lateral ventricle (Altman, 1969; Doetsch et al., 1997). After Eriksson et al. (1998) showed that the human hippocampus retains an ability to generate neurons throughout life, it is now expected that neurogenesis has a potential to compensate for and recover neural functions that were lost by central nervous system disorders. Neurogenesis in adult brain increases in such conditions as an exogenous neurotrophic factor supplement (Craig et al., 1996; Åberg et al., 2000), an enriched environment (Kempermann et al., 1997), antidepressant treatment (Malberg et al., 2000), seizure (Parent et al., 1997), and ischemia (Liu et al., 1998; Jin et al., 2001). It decreases under stress (Gould et al., 1998) and supplementation of excitatory amino acids (Cameron et al., 1995) or adrenal steroids (Cameron et al., 1998). Because ischemic stroke often causes loss of the neural functions because of neural cell death, neurogenesis after ischemia should be important for compensation for and recovery of those functions (Abe, 2000). The stage of neurogenesis in the DG can be divided into three steps: (1) proliferation, (2) migration, and (3) differentiation (Gage, 2000). Liu et al. (1998) showed that neurogenesis increased in postischemic DG of gerbils, and that 60% of newborn cells in the granule cell layer (GCL) expressed neuronal markers NeuN, calbindin, and MAP-2. Not clear, however, is how transient ischemia affects the three steps of neurogenesis, or the detailed relations between these three steps in DG.
PSA-NCAM is a well-known, highly polysialylated form of glycoprotein that mediates cell–cell adhesion and recognition. PSA-NCAM expression occurs during a period of development on the dendrites and cell bodies of the newly generated dentate granule cell. The expression decreases but is maintained in adult brain (Seki and Arai, 1993). PSA-NCAM is present on the surface of the migrating cell through the rostral migratory stream from subventricular zone to olfactory bulb, and deletion of the gene for NCAM or cleavage of the polysialic acid moiety results in defects in migration (Bonfanti and Theodosis, 1994; Ono et al., 1994). Our previous report showed that the PSA-NCAM–positive cell numbers increased in the DG after transient ischemia (Iwai et al., 2001). However, it was reported that PSA-NCAM could also be expressed in the glial cells after transient forebrain ischemia (Fox et al., 2001). However, Fox et al. did not carry out the double-staining of PSA-NCAM with neuronal marker; thus, the fate of such PSA-NCAM–positive cells was still unknown.
In the present study, therefore, we evaluated the three steps of neurogenesis in the gerbil DG after transient global ischemia using bromodeoxyuridine (BrdU), PSA-NCAM, and NeuN as markers for proliferation, migration, and differentiation, respectively.
MATERIALS AND METHODS
Surgical preparations
Eleven-week-old adult male Mongolian gerbils (SCL, Shizuoka, Japan) were used for the experiments. The animals were lightly anesthetized by inhalation of a nitrous oxide/oxygen/halothane (69%:30%:1%) mixture through a closely fitting face mask. After a neck incision, both common carotid arteries were exposed. When the animals began to awaken after anesthesia was stopped, both CCAs were occluded with aneurysm clips for 5 minutes, and the clips were then removed to restore cerebral blood flow. The rectal temperature was monitored and maintained at 37.0 ± 0.5°C with a heating pad (BRC, Aichi, Japan) during the operation. The surgical incision was then closed, and animals were allowed free access to water and food at ambient temperature. Sham-control animals were treated identically, except for the occlusion of common carotid arteries.
5-Bromodeoxyuridine labeling
A cell proliferation marker, 5-BrdU (Sigma, St. Louis, MO, U.S.A.), was dissolved in saline, and was intraperitoneally injected (50 mg/kg) three times at 8-hour intervals during the day. Four patterns of BrdU injection were performed in relation to the time the animals were killed after transient global ischemia (Fig. 1). Bromodeoxyuridine was injected at 9 days in the 10-d (n = 5) and sham-operated groups, at 9 and 19 days in the 20-d group (n = 5), at 9, 19 and 29 days in the 30-d group (n = 5), and at 9, 19, 29, and 59 days in the 60-d group (n = 5). In each group, the gerbils were killed within 24 hours of the last BrdU injection.
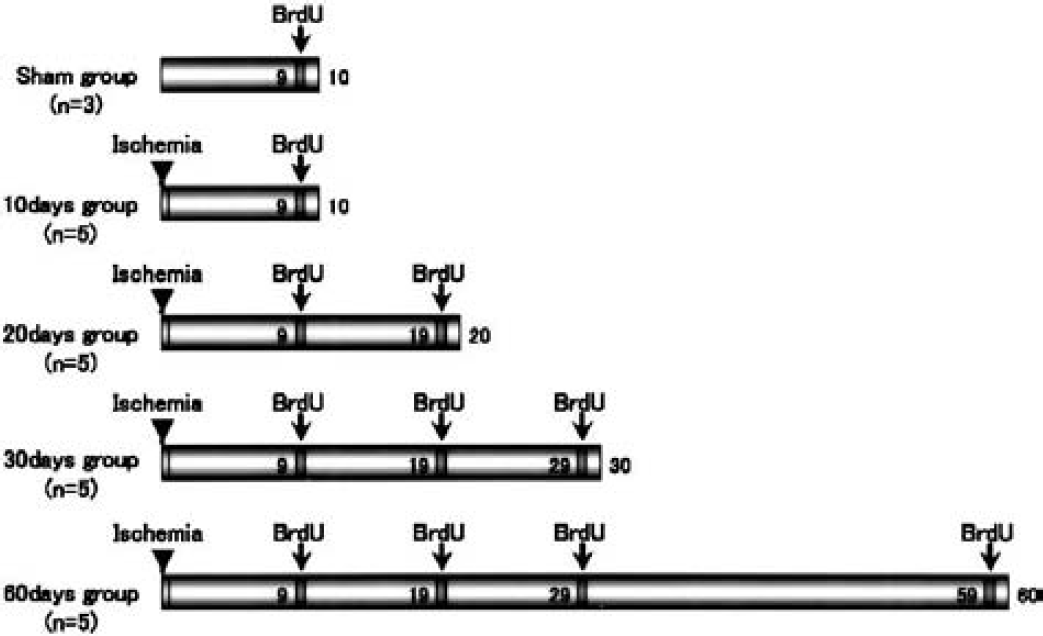
The patterns of bromodeoxyuridine (BrdU) injection. Bromodeoxyuridine was injected at 9 days in sham-operated (n = 3) and 10-d (n = 5) groups, at 9 and 19 days in the 20-d group (n = 5), at 9, 19, and 29 days in the 30-d group (n = 5), and at 9, 19, 29 and 59 days in the 60-d group (n = 5). Bromodeoxyuridine was intraperitoneally injected (50 mg/kg) three times at 8-hour intervals during the day. In each group, gerbils were killed 24 hours after the last BrdU injection.
Tissue preparation and single immunohistochemistry
All gerbils were killed with deep anesthesia by diethyl ether. The brains were quickly removed and frozen in 2-methylbutane chilled by liquid nitrogen. The brain was serially cut in 10-μm-thick coronal sections on a cryostat at −20°C, and the slices were mounted on glass slides coated with polylysine, and then were stored at −80°C until use.
For immunohistochemical detection of BrdU labeling, brain sections were fixed in 4% paraformaldehyde, and pretreated to denature DNA in 50% formamide-2 × saline–sodium citrate buffer at 65°C for 2 hours and then incubated at 37°C for 30 minutes in 2N HCl. Finally, sections were rinsed for 10 minutes at 25°C in 0.1 mol/L boric acid, pH 8.5. Sections were then incubated in 0.3% H2O2 for 30 minutes and blocked with 10% normal horse serum for 30 minutes. Sections were incubated overnight with mouse monoclonal anti-BrdU (1:200; Oncogene, Boston, MA, U.S.A.) diluted in phosphate-buffered saline with 2% horse serum and 0.3% Triton X-100, and were then incubated with biotinylated horse anti-mouse immunoglobulin G (IgG) (1:200; Vector Laboratories, Burlingame, CA, U.S.A.) for 1 hour at room temperature.
For immunohistochemical detection of PSA-NCAM, brain sections were fixed in 4% paraformaldehyde, and were incubated in 0.3% H2O2 for 30 minutes and blocked with 10% normal goat serum for 30 minutes. Sections were then incubated 24 hours at 4°C with mouse monoclonal anti-PSA-NCAM (1:1,000; provided by Dr. T. Seki, Tokyo, Japan) diluted in phosphate-buffered saline with 2% goat serum and 0.3% Triton X-100. Then the sections were incubated in biotinylated goat anti-mouse immunoglobulin M (IgM) (1:200; Vector Laboratories) for 1 hour at room temperature.
Immunoreactivities were developed in horseradish peroxidase/streptavidin/biotin complex solution (Vectastain ABC Kit, Vector Laboratories) for 30 minutes, and were incubated for 2 minutes in a peroxidase reaction solution (0.02% diaminobenzidine, 0.02% H2O2).
Double immunofluorescence
For the immunofluorescence detection of PSA-NCAM and BrdU, sections were first incubated with the mouse monoclonal anti-PSA-NCAM (1:1,000) for 36 hours at 4°C, followed by the rhodamine-labeled goat anti-mouse IgM (1:500, KPL, Gaithersburg, MD, U.S.A.) for 1 hour. After being washed in phosphate-buffered saline, the sections were mounted on glass slides and viewed under Olympus microscopy (BX51; Olympus, Japan) equipped with rhodamine filter set. After obtaining images (1,024 × 1,024 pixels) by Hamamatsu Aquacosmos System (Hamamatsu Photonicus, Hamamatsu, Japan), the sections were pretreated to denature DNA (as previously described) and blocked with 10% normal horse serum for 30 minutes. Sections were incubated with mouse monoclonal anti-BrdU (1:200) for 12 hours at 4°C followed by the FITC-labeled horse anti-mouse IgG (1:500; Vector Laboratories) for 1 hour. Sections were examined under microscopy equipped with an FITC filter set. The images for BrdU labeling were merged with those for PSA-NCAM.
For the double immunofluorescence detection of BrdU and NeuN, sections were pretreated to denature DNA and were first incubated with the sheep polyclonal anti-BrdU (1:1,000; Bio-design, Saco, MA, U.S.A.) and mouse monoclonal anti-NeuN (1:500; Chemicon, Temecula, CA, U.S.A.) for 12 hours at 4°C, followed by the FITC-labeled donkey anti-sheep IgG (1:500; Molecular Probes, Eugene, OR, U.S.A.) and the rhodamine-labeled horse anti-mouse IgG (1:500, Chemicon) for 1 hour. For the double immunofluorescence detection of PSA-NCAM and NeuN, sections were first incubated with the mouse monoclonal anti–PSA-NCAM (1:1,000) and mouse monoclonal anti-NeuN (1:500) for 36 hours at 4°C, followed by the FITC-labeled goat anti-mouse IgM (1:500, Sigma) and the rhodamine-labeled horse anti-mouse IgG (1:500, Chemicon) for 1 hour.
For the double immunofluorescence detection of BrdU and GFAP, sections were pretreated to denature DNA and were first incubated with the mouse monoclonal anti-BrdU (1:200) and goat polyclonal anti-GFAP (1:400; Santa Cruz Biotechnology, Santa Cruz, CA, U.S.A.) for 12 hours at 4°C, followed by the FITC-labeled horse anti-mouse IgG (1:500) and Alexa Fluor 546-labeled donkey anti-goat IgG (1:1,000; Molecular Probes) for 1 hour. For the double immunofluorescence detection of PSA-NCAM and NeuN, sections were first incubated with the mouse monoclonal anti-PSA-NCAM (1:1,000) and goat polyclonal anti-GFAP (1:400) for 36 hours at 4°C, followed by the FITC-labeled rabbit anti-mouse IgM (1:500, Rockland, Gilbertsville, PA, U.S.A.) and Alexa Fluor 546-labeled donkey anti-goat IgG (1:1,000) for 1 hour.
These sections were scanned with confocal microscope equipped with argon and HeNe1 laser (LSM-510; Zeiss, Jena, Germany). Sets of fluorescent images were acquired sequentially for the red and green channels to prevent crossover of signals from green to red or red to green channels.
Cell counting
With single immunohistochemical staining, the number of BrdU-labeled, PSA-NCAM-positive and NeuN-positive cells in the SGZ and GCL was counted in five coronal hippocampal sections (10 μm, spaced 50 μm apart) per animal in the sham-operated (n = 3), and 10, 20, 30, and 60 days of ischemic groups (each group, n = 5). With double immunofluorescence staining, the number of double positive cells for BrdU + PSA-NCAM, BrdU + NeuN, and PSA-NCAM + NeuN was also counted. The area of the DG that included the SGZ and GCL was examined on each section using a computer-based imaging system (UTHSCSA Image Tool version 2.0 Alpha 3, Microsoft, Redmond, WA, U.S.A.). The cell numbers were presented as mean ± SD (per mm2) within the investigated region. On BrdU-labeled cell numbers, statistical analyses were performed only between the sham-operated group and the 10-d group because of the same BrdU administration protocol. On the PSA-NCAM–positive cell numbers, statistical analyses were performed between the sham-operated group and each ischemic group. They were performed by Student's t-test, with P < 0.05 considered significant.
RESULTS
Change of bromodeoxyuridine-labeled cell
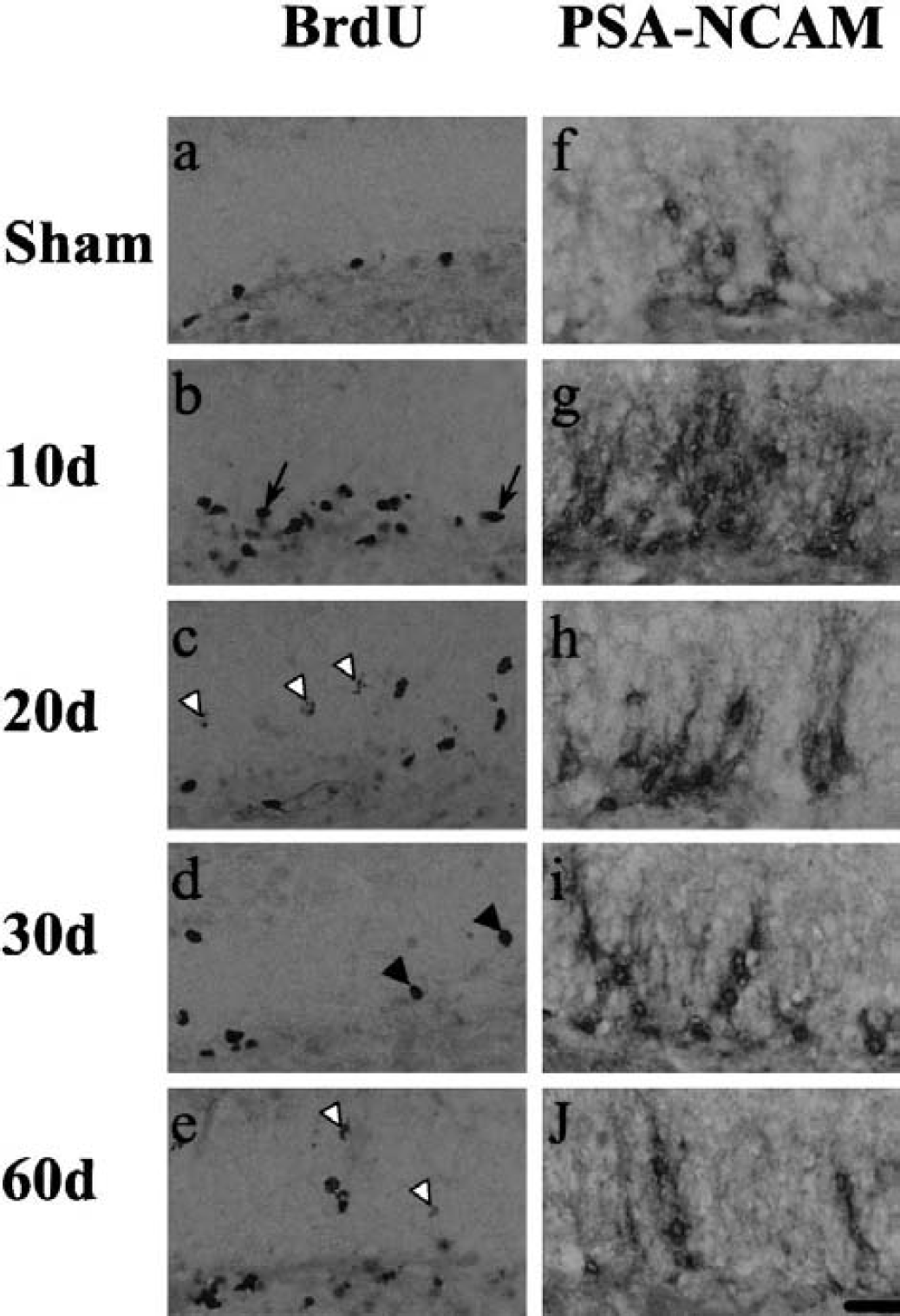
In sham-operated animals, the basal level of BrdU-labeled cells was found exclusively in the SGZ and rarely in the GCL of the DG (Figs. 2a and 3a).
Immunohistochemistry for bromodeoxyuridine (BrdU)
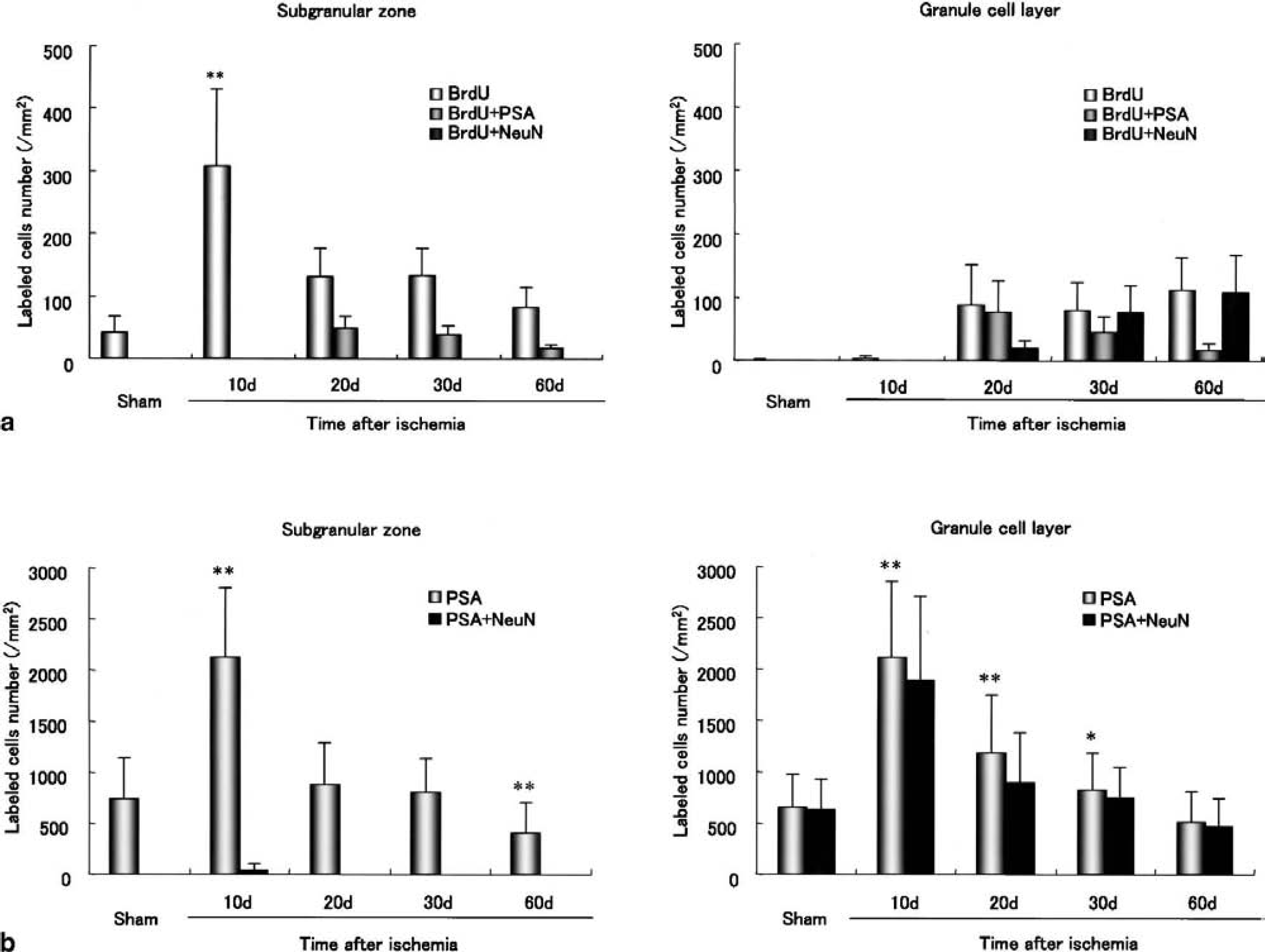
Change of the number of bromodeoxyuridine (BrdU) single-labeled cells and double-labeled cells with PSA-NCAM or NeuN (a), and the number of PSA-NCAM single-labeled and double-labeled with NeuN (b) in the subgranular zone (left side of each panel) and granule cell layer (right side of each panel) of the dentate gyrus after ischemia. Significant differences from the sham at *P < 0.05, **P < 0.01.
The number of BrdU-labeled cells increased after the transient ischemia in the following way. Ten days after ischemia, it dramatically increased (approximately sevenfold) in the SGZ (Figs. 2b; 3a, left), whereas those cells increased only slightly in the GCL (Fig. 3a, right). Twenty and 30 days after ischemia, BrdU-labeled cells were located both in the SGZ and GCL, and the number of those cells dramatically increased in the GCL, and decreased approximately 40% of the 10-d level in the SGZ, although additional BrdU was administered 19 or 29 days after ischemia (Fig. 3a). Sixty days after ischemia, the number decreased in the SGZ (Figs. 2e; 3a, left), but still slightly increased in the GCL (Fig. 3a, right). The total number in the DG (SGZ + GCL) was approximately two thirds of the 10-d level, although additional BrdU was administered 19, 29, and 59 days after ischemia.
The morphology of BrdU-labeled nuclei also markedly changed in the DG after ischemia. With time, the BrdU-labeled nuclei became oval (Fig. 2b, arrows) in the SGZ and round (Fig. 2d, arrowheads) in the GCL. Furthermore, the intensity of BrdU-labeled nuclei became weak in some cells in the GCL 20 days after ischemia or later (Figs. 2c–e, open arrowhead).
Change of PSA-NCAM–positive cell
PSA-NCAM–positive cells were located both in the SGZ and GCL of sham-operated animals and the numbers were similar (Figs. 2f and 3b). Ten days after ischemia, the number of PSA-NCAM–positive cells increased threefold both in the SGZ and the GCL (Figs. 2g and 3b). Twenty and 30 days after ischemia, the number returned to sham-control level in the SGZ (Figs. 2h; 3b, left), but in the GCL was still elevated (at 1.8-fold at 20 days; at 1.3-fold at 30 days) (Figs. 2h and 2i; 3b, right). After that, the number continued to decrease in both zones until 60 days, especially in the SGZ with approximately half that of control (Figs. 2j; 3b, left).
Relation of cell proliferation and migration
In sham-operated animals, BrdU-labeled cells were located only in the SGZ (Figs. 3a and 4a), and PSA-NCAM–positive cells were located both in the SGZ and GCL (Figs. 3b and 4a). However, there was no double-labeled cell for BrdU + PSA-NCAM (Figs. 3a, 3b, 4a). Although BrdU-labeled cells mainly increased in the SGZ (Figs. 3a and 4b) and PSA-NCAM–positive cells increased both in the SGZ and GCL (Figs. 3b and 4b) 10 days after ischemia, no double-labeled cell was detected (Figs. 3 and 4b). The double-labeled cells began to be detected mainly in the GCL and partially in the SGZ 20 (Figs. 3a; 4c, arrowheads, arrow), 30 (Figs. 3a; 4d, arrowheads, arrow), and 60 (Figs. 3a; 4e, arrowhead) days after ischemia. The number of double-labeled cells decreased as the number of single PSA-NCAM–labeled cells decreased in the SGZ and GCL (Figs. 3a and 3b).
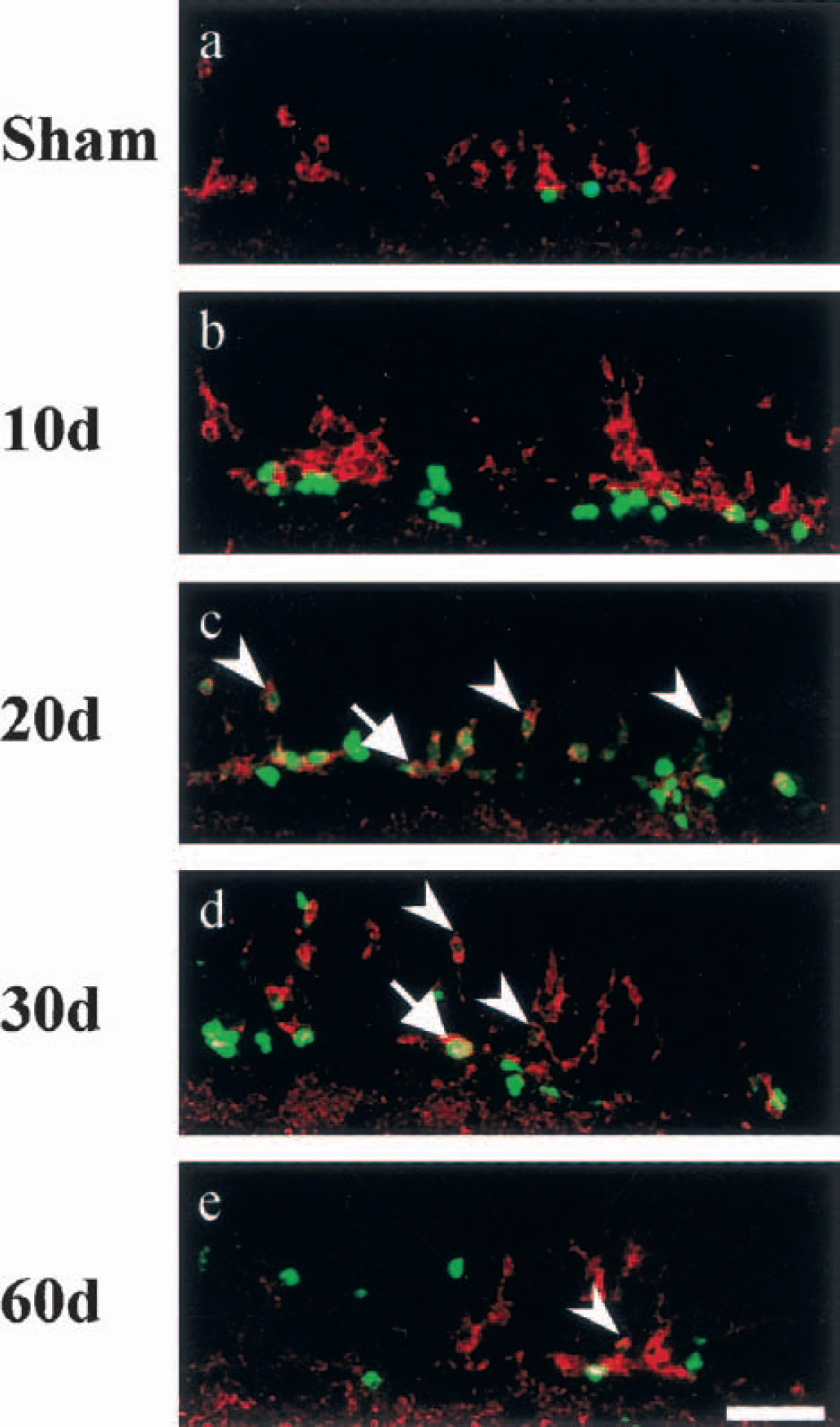
Double immunolabeling for bromodeoxyuridine (BrdU) (green) and PSA-NCAM (red) in the gerbil dentate gyrus of sham-operated
Neuronal feature of proliferated and migrating cells
In sham-operated animals, BrdU-labeled cells were mainly present in the SGZ (Figs. 3a and 5a), but neuronal marker NeuN was expressed only in the granule cells of the GCL (Fig. 5f). Ten days after ischemia, there was no double-labeled cell in the GCL, although the number of BrdU-labeled cells increased in the SGZ (Figs. 3a, 5b, 5l). However, 20 days after ischemia, there were some BrdU-labeled cells in the GCL (Figs. 3a; 5c, arrows) and these cells expressed very weak NeuN signal (Figs. 3a; 5h, 5m, arrowheads). Thirty and 60 days after ischemia, BrdU-labeled cells in the GCL (Figs. 3a; 5d, 5e, arrowheads) expressed NeuN signal stronger (Figs. 5i and 5j, arrowheads), and these merged images (Figs. 5n and 5o, arrowheads) became more yellowish than those of the 20-d group (Figs. 5h and 5m, arrowheads). There was no BrdU-labeled cell expressed NeuN signal in the SGZ after ischemia (Figs. 3a; 5k–o).
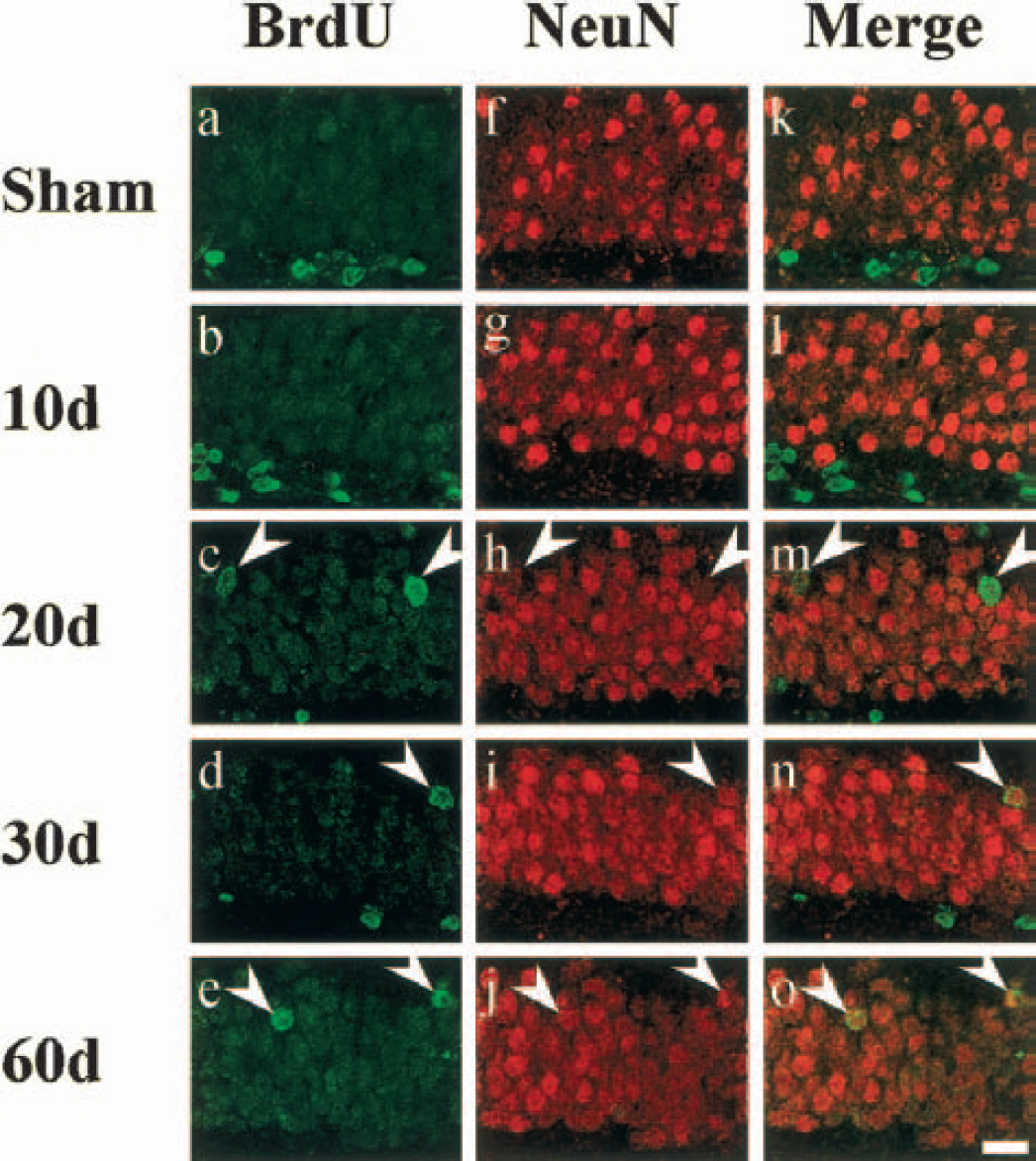
Immunolabeling for bromodeoxyuridine (BrdU) (left, green), NeuN (center, red), and their merged images (right) in gerbil dentate gyrus of sham-control (
As for PSA-NCAM + NeuN staining, all the PSA-NCAM–positive cells in the GCL expressed NeuN in sham-operated animals (Figs. 6a, 6f, 6k). After the transient ischemia, the PSA-NCAM–positive cell numbers increased both in the SGZ and the GCL, and some PSA-NCAM–positive cells in the SGZ and most in the GCL expressed NeuN 10 days after ischemia (Fig. 3b; 6b, 6g, 6l [arrows]); a part of PSA-NCAM–positive cells did not express NeuN (Figs. 6b, 6g, 6l [arrowhead]). Although the PSA-NCAM–positive cell numbers continuously decreased in the GCL from 20 to 60 days (Fig. 3b, right), all such cells expressed NeuN (Figs. 6c–e, 6h–j, 6m–o).
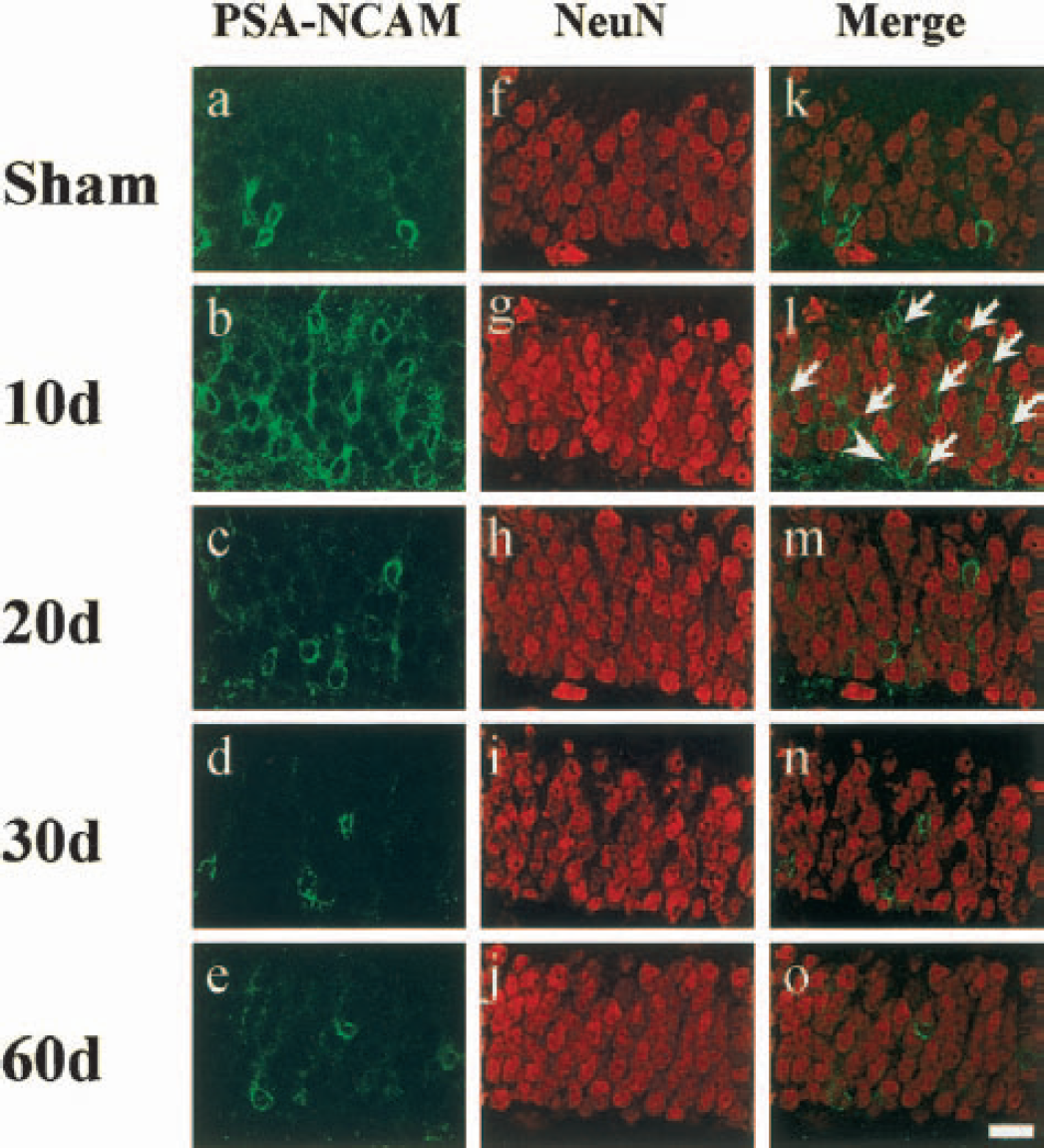
Immunolabeling for PSA-NCAM (left, green), NeuN (center, red), and their merged images (right) in the gerbil dentate gyrus of sham-control
However, GFAP-positive cells labeled by BrdU were not detected in the SGZ and GCL of sham-operated and ischemic animals (Figs. 7a–c, arrows). Double-positive cells expressed with GFAP and PSA-NCAM could not be detected in the DG of sham-operated and ischemic animals, but GFAP-positive processes were partly in contact with PSA-NCAM–positive cell bodies and dendrites in the DG of sham-operated and ischemic animals (Figs. 7f–j, arrowheads).
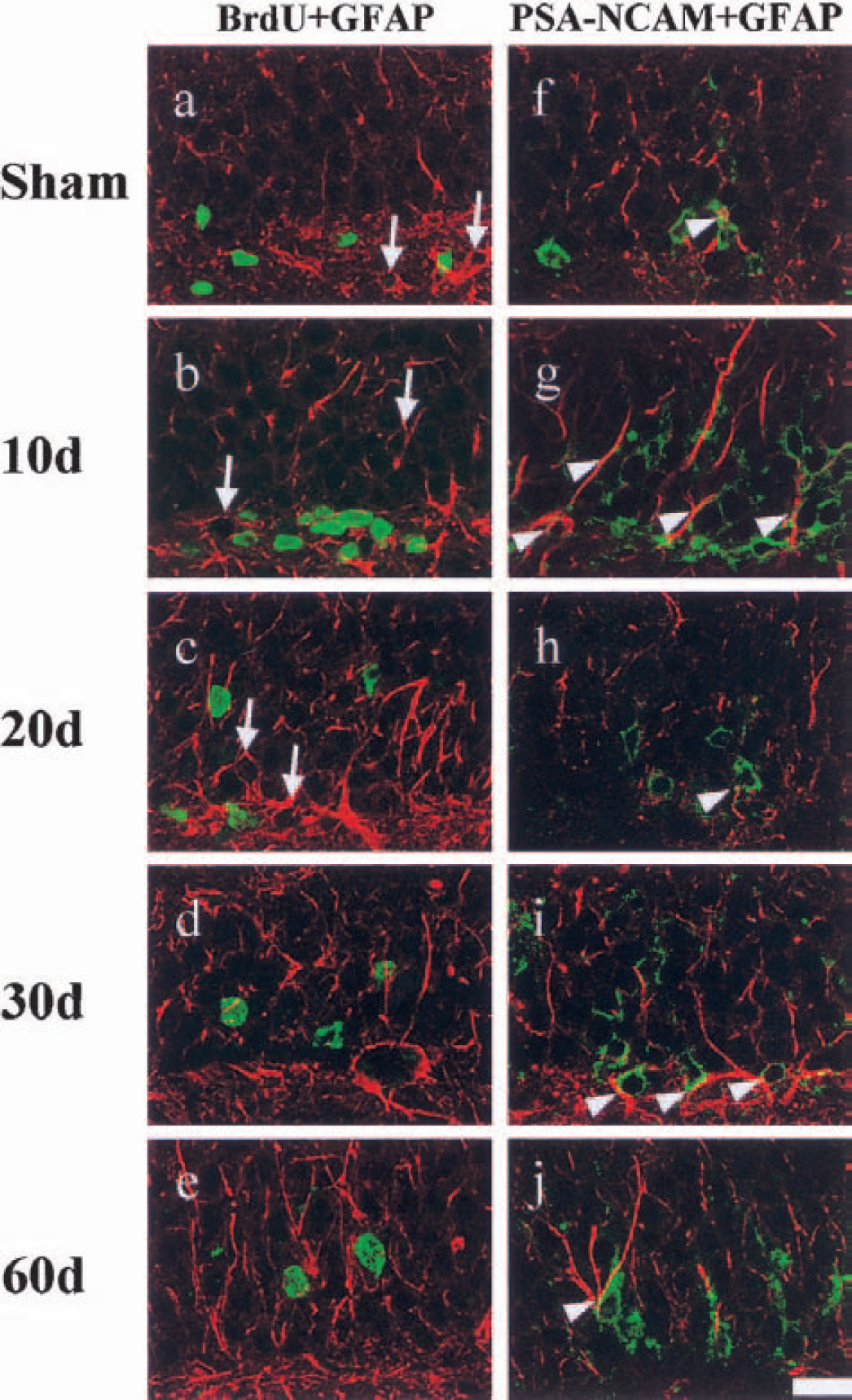
Double immunolabeling for bromodeoxyuridine (BrdU) (green) and GFAP (red)
DISCUSSION
Although some reports investigated postischemic neurogenesis in the DG (Liu et al., 1998; Kee et al., 2001; Jin et al., 2001; Yagita et al., 2001), all of them evaluated only proliferation and differentiation, not migration. In the present study, we demonstrated the three steps of neurogenesis in the gerbil DG after transient global ischemia using BrdU, PSA-NACM, and NeuN as markers for cell proliferation, migration, and differentiation, respectively. This is the first report to examine the detailed relation between these three steps in the DG after 5 minutes of transient global ischemia.
Cell proliferation, the first step of neurogenesis, was observed as a sevenfold increase of BrdU-labeled cells in the SGZ 10 days after the ischemia (Figs. 2b and 3a), which is consistent with our previous report (Iwai et al., 2001). One possible mechanism of this proliferation could be a modulation of NMDA and AMPA/kainate receptors (Bernabeu and Sharp, 2000). NMDA-receptor binding transiently reduced in the rat DG 1 week after transient forebrain ischemia, which then returned to the control level in an additional week (Westerberg et al., 1989; Ogawa et al., 1991). Such a time course readily fits with the BrdU-labeling time course in our present and previous (Iwai et al., 2001) result.
However, various growth factors could also affect stem cell proliferation in the adult hippocampus (Craig et al., 1996; Kuhn et al., 1997). Yoshimura et al. (2001) reported that endogenously synthesized fibroblast growth factor-2 (FGF-2) is essential to maintain proliferation and differentiation of progenitor cells in the adult hippocampus after brain insult. FGF-2 is transiently upregulated in the DG after 10 minutes of transient global ischemia in the rat (Endoh et al., 1994).
To investigate the spatial and chronologic relation of newly proliferated cells between 10 days and 20 days, 20 days and 30 days, and 30 days and 60 days, respectively, we designed four different BrdU administration protocols with five separate animals groups (Fig. 1). Our original expectation of the chronologic change of BrdU-labeled cell was that BrdU-labeled cell numbers would additionally increase. To our surprise, however, BrdU-labeled cell numbers in the DG reached maximum at 10 days and gradually decreased until 60 days after ischemia (Fig. 3a). Our previous report (Iwai et al., 2001) showed that new cell proliferation in the SGZ returned to control levels 20 days after 5 minutes of transient global ischemia. Liu et al. (1998) showed that the number of dividing cells returned to control levels 3 to 5 weeks after ischemia. Therefore, most BrdU-labeled cells detected in the DG 20, 30, and 60 days were labeled 9 days after ischemia and gradually decreased until 60 days. Although a possible explanation of this phenomenon is apoptotic elimination, it has not yet been directly demonstrated (Gage et al., 1998).
Cell migration, the second step of neurogenesis, was investigated using the PSA-NCAM immunostaining method in this study. PSA-NCAM, the polysialylated NCAM isoform, remains expressed in adult neuronal populations showing ongoing neurogenesis, cell migration (Hildebrandt et al., 1998; Hu et al., 1996), axonal outgrowth, nerve branching, and synaptic plasticity (Doherty et al., 1990; Zhang et al., 1992; Tang et al., 1994; Cremer et al., 2000; De Stefano et al., 2001; Fox et al., 2001). In the adult rodent brain, several regulators of PSA-NCAM expression have been reported in relation to neural plasticity. Recent reports showed that PKCδ was induced through activation of the NMDA receptor, and PKCδ negatively regulated neural cell adhesion molecule polysialylation state (Miettinen et al., 1996; Gallagher et al., 2001). In the present study, however, the PSA-NCAM–positive cell numbers reached maximum 10 days after ischemia both in the SGZ and GCL (to a threefold increase; Figs. 2g and 3b). Ten days after ischemia, because most (approximately 90%) PSA-NCAM–positive cells expressed NeuN (Fig. 6l, arrows), the increase of these cell numbers in the GCL predominantly represents neural plasticity.
The present study showed that BrdU-labeled cell migration from the SGZ to GCL began between 10 and 20 days. Bromodeoxyuridine-labeled cells decreased in the SGZ and dramatically increased in the GCL between 10 and 20 days after ischemia (Figs. 2b, 2c, 3a). The double-labeled cells began to be detected mainly in the GCL and partially in the SGZ 20 (Figs. 3a; 4c, arrowheads, arrow), 30 (Figs. 3a; 4d, arrowheads, arrow), and 60 (Figs. 3a; 4e, arrowhead) days after ischemia. These results suggest that the majority of PSA-NCAM–positive cells after 20 days of ischemia represent the migration step of neurogenesis.
Ten days after ischemia, however, some (approximately 10%) of the PSA-NCAM–positive cells did not express NeuN, which represent migration of newly generated cells until 10 days (Fig. 6l, arrowhead). Other molecules, such as TOAD-64 and doublecortin, are expressed in the migrating newborn neuron after brain injury. Dash et al. (2001) reported that double-positive cells with BrdU and TOAD-64 were detected in the GCL 3 days after traumatic brain injury, and Jin et al. (2001) reported that double-positive BrdU and doublecortin were detected in the SGZ 1 week after focal cerebral ischemia. Nacher et al. (2001) reported that almost all doublecortin-positive cells also expressed PSA-NCAM. Further investigations are necessary to completely elucidate how long PSA-NCAM expression takes on BrdU-labeled cells.
NeuN and GFAP immunostaining showed the differentiation to neuronal cell, the final step of neurogenesis. As the most important result in the present study, the double BrdU + PSA-NCAM–positive cells gradually decreased, with concomitant increase of the double BrdU + NeuN-positive cells from 20 to 60 days after ischemia (Figs. 3a, right; 5m–o). These results suggest that the translation period of migration to differentiation steps takes approximately 1 month, and that neurogenesis almost finishes within 2 months after transient global ischemia.
Conversely, BrdU + GFAP-positive cells were not detected in the SGZ and GCL of sham-operated and ischemic animals (Figs. 7a–c), similar to findings by Liu et al. (1998). PSA-NCAM + GFAP-positive cells were also not detected in the SGZ and GCL of sham-operated and ischemic animals. However, GFAP-positive processes were partly in contact with PSA-NCAM–positive cell bodies and dendrites in DG of sham-operated and ischemic animals (Figs. 7f–j), which is contrary to the findings of Fox et al. (2001). However, Fox et al. did not carry out the double staining with neuronal marker. In accordance with Seki and Arai (1999), PSA-NCAM may not be expressed in the glial cells.
In the present study, spatial and chronologic relations of postischemic neurogenesis were demonstrated in the DG of gerbils after 5 minutes of transient global ischemia. These observations may help clarify the mechanism of functional recovery after ischemia with activation of internal neural stem cells.
Footnotes
Acknowledgments:
The authors thank Dr. T. Seki (Tokyo, Japan) for his generous gift of the anti–PSA-NCAM antibody.
