Abstract
Perinatal hypoxic-ischaemic encephalopathy (HIE) is the leading cause of irreversible brain damage resulting in serious neurological dysfunction among neonates. We evaluated the feasibility of positron emission tomography (PET) methodology with 15O-labelled gases without intravenous or tracheal cannulation for assessing temporal changes in cerebral blood flow (
Keywords
Introduction
Perinatal asphyxia resulting in hypoxic-ischaemic encephalopathy (HIE) is a major cause of premature mortality worldwide, and 25% of survivors suffer from long-term neurological and neurodevelopmental impairments such as cerebral palsy. 1 The incident occurs in approximately 1.5 per 1000 live births in developed countries. 2 The pathophysiology starts with primary energy failure during the hypoxic-ischaemic (HI) event, involving oxidative metabolism failure, cytotoxic oedema and excitotoxin accumulation. The subsequent secondary energy failure results in glutaminergic excitotoxicity, apoptosis and neuroinflammation. 3 The only clinically feasible therapy for HIE neonates is hypothermia starting within 6 h post-incident, but not more than 50% of treated neonates achieve an improved outcome.4 –6 The predominantly employed Rice–Vannucci neonatal rodent HIE model7,8 has provided extensive advances toward developing promising therapies that could enhance the neuroprotective effect of hypothermia.
Given the large variation in brain damage severity in this animal model,
9
in vivo follow-up observations on an individual basis are vital for consistent progress monitoring. This interindividual variability is also the case clinically in patients and thus reliable prognostic indicators are highly desired, particularly in the acute phase. In neonates, cerebral oxidative metabolism exhibits a distinct elevation in the early maturational process.10,11 Oxygen supply derangement to the brain at this stage accordingly causes devastating outcomes including reperfusion-related injury after the initial decrease in cerebral blood flow (
Studies have utilized the blood oxygenation level-dependent (BOLD) technique14,15 to quantitatively assess blood oxygenation level in the superior sagittal sinus region in humans using magnetic resonance imaging, from which the oxygen extraction fraction (
Positron emission tomography (PET) with 15O-labelled gases has been the gold standard tool for decades for quantitative measurements of
This study depicted temporal changes in
Materials and methods
Study design
An integrated 15O-PET system was developed enabling quantitative
Animal subjects and ethical statement
For the first experiment that determined the
Age, body weight and number of animal subjects on each PET imaging day in the second experiment. Numbers in parentheses denote values after exclusion from the image data analysis.
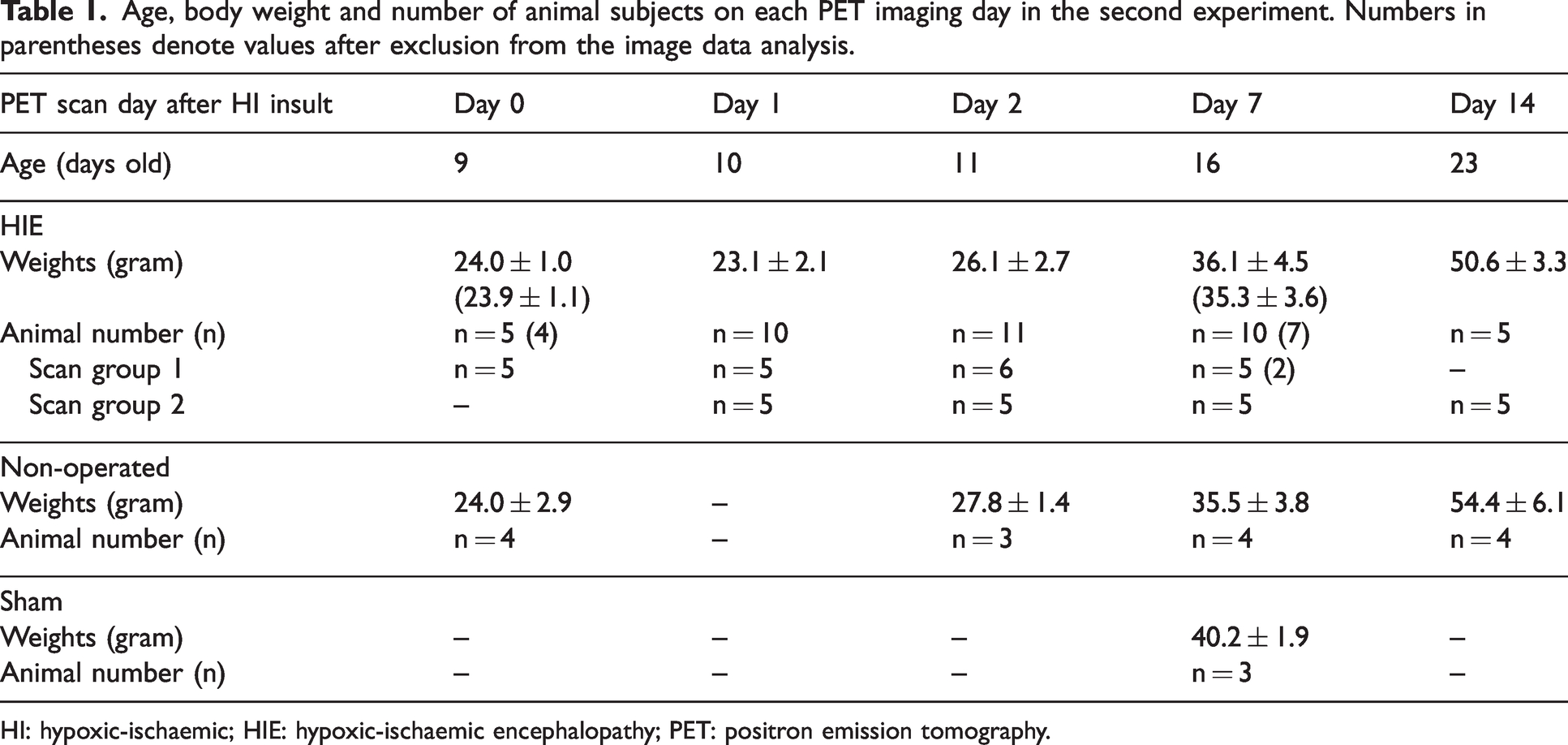
HI: hypoxic-ischaemic; HIE: hypoxic-ischaemic encephalopathy; PET: positron emission tomography.
All animals were group housed under standard conditions (12-h light/dark cycle, temperature 21 ± 3 °C, humidity 55 ± 15%) with ad libitum access to RM3(E) rodent diet (SDS, UK) and tap water at the Central Animal Laboratory, University of Turku, Finland (UTUCAL). The study was approved by the National Project Authorization Board of Finland (ESAVI/20863/2018) adhering to the 3 R principles in accordance with Finnish National legislation (Act 497/2013, Decree 564/2013) and EU Directive 2010/EU/63 on the protection of animals used for scientific purposes. All reported experiments complied with the ARRIVE guidelines. 26
Integrated 15O-PET imaging system
A comprehensive PET system was assembled to sequentially assess
Radioactive gas production, purification/qualification and inhalation control
15O was produced using the cyclotron by a 14N(d,n)15O nuclear reaction. The target gas was 1.0% O2 in N2 and 1.0% CO2 in N2, producing 15O-O2 and 15O-CO2 gases in the target, respectively. Each gas was transported to the hot cell, where 15O-O2 and 15O-CO2 gases were purified. The 15O-CO gas was synthesized from the target 15O-O2 gas by a charcoal column heated at 950 °C. The radiochemical and chemical purity was assessed each time before inhalation by gas chromatography (Model GC-2014, Shimazu, Kyoto, Japan) with Molecular Sieve and HayeSep Q columns (Figure 1(a)).
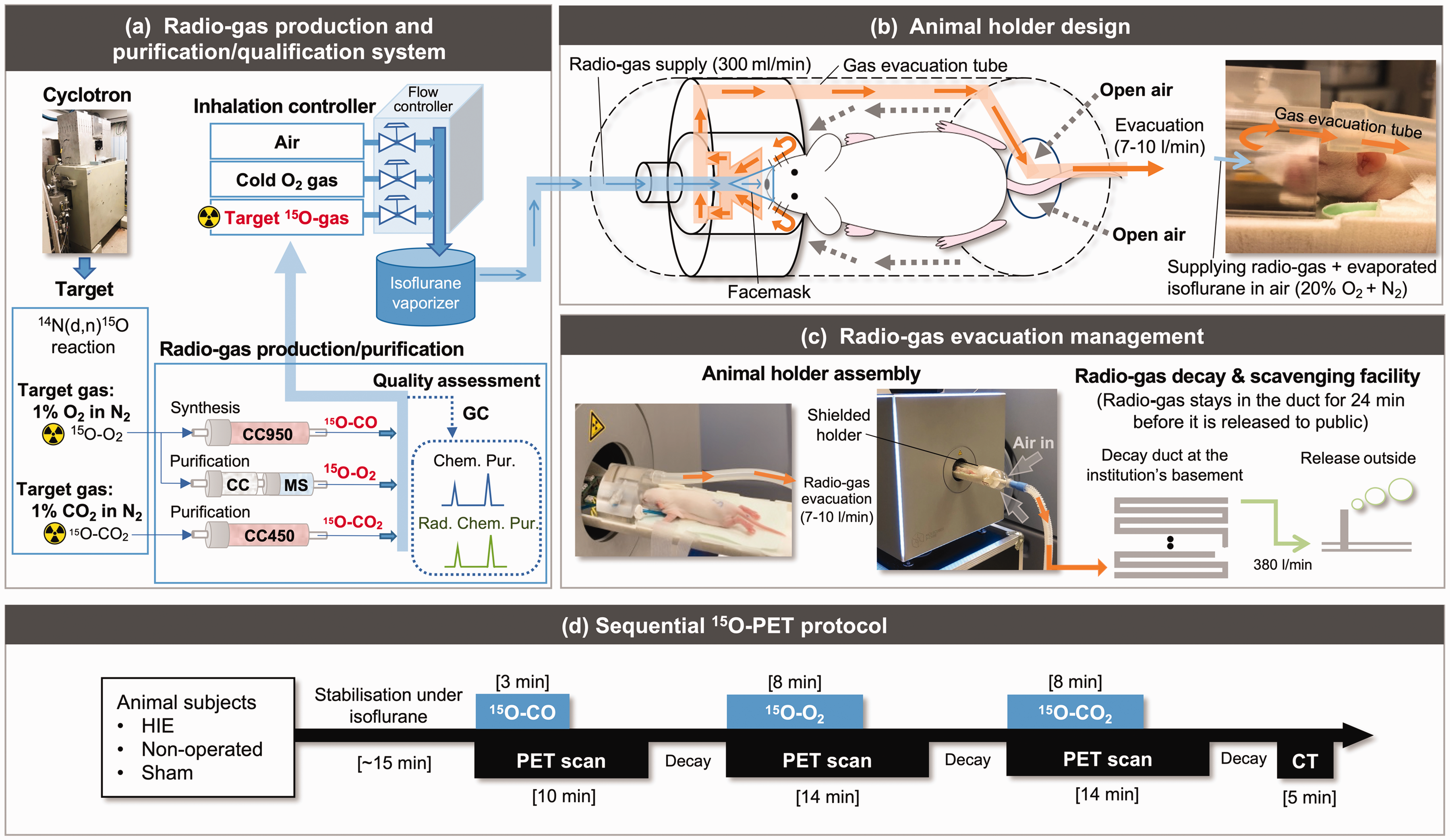
A schematic of the comprehensive PET system newly assembled in this study and consisting of three parts (a–c). (a) Radioactive gas production and purification/qualification system consisting of a cyclotron, target chamber, radioactive gas production/purification unit and inhalation controller. A charcoal column heated at 950 °C (CC950) was utilized for 15O-CO synthesis. For 15O-O2 purification, two columns filled with charcoal at room temperature (CC) and another with MS were used. For the purification of 15O-CO2, a column with charcoal heated at 450 °C (CC450) was used. Gas chromatography (GC) was implemented with two columns with HayeSep Q and Molecular sieve (MS) to assess chemical and radiochemical purity. (b) Animal holder dedicated to 15O-oxygen inhalation PET. A corn-shaped facemask made of a thin fabric sheet is placed over the snout of the animal. Radioactive gases are carried to the facemask for spontaneous inhalation. The animal holder is well sealed and actively scavenged from the front surrounding the radioactive gas supply tube. Fresh air is passively carried from the back into the animal holder. (c) Radioactive gas evacuation system. Approximately 380 l/min of evacuation carries scavenged radioactive gases from the animal holder to the decay ducts installed in the institute basement. Approximately 24 min of transit time in the ducts allows for radioactivity decay by a factor of 4000 before it is released from the chimney to the outside and (d) the 15O-PET/CT protocol consists of three separate PET scans each with 15O-CO, 15O-CO2 and 15O-O2 inhalation conducted in a sequential PET/CT study in the three subject groups of HIE, non-operated and sham. CT: computed tomography; HIE: hypoxic-ischaemic encephalopathy; PET: positron emission tomography; Radio-gas: radioactive gas.
The quality-assured radioactive gases were then mixed with pure oxygen and air so that animals inhaled 20% oxygen during the entire period when animals were in the holder, even while radioactive gases were supplied or when the radioactivity supply rate was changed. The typical flow rate was 300 ml/min and the radioactivity supply rate was 250, 150 and 300 MBq/min, corresponding to 15O-O2, 15O-CO2 and 15O-CO, respectively. Using multiple radioactivity detectors and mass flow controllers reliably controlled these parameters. The inhalation gas was mixed with evaporated isoflurane gas at a given concentration (typically 2–2.5%) before it was transferred to the animal holder.
Dedicated 15O-PET system animal holder
Two sets of animal holder assemblies were constructed to adapt to the HRRT PET scanner (CTI PET Systems, Knoxville, TN, USA) for the first experiment and the β-Cube PET scanner (Molecubes, Ghent, Belgium) for the second experiment. Both holders were sealed to prevent radioactive gas leakage. A corn-shaped facemask made of a fabric sheet (100 μm thickness) gently covered the snout to which the radioactive gases were supplied at a flow rate of 300 ml/min. The air inside the animal holder was evacuated at 7–10 l/min from the front through small holes surrounding the supply tube inside the animal holder. Fresh air was then passively carried to the animal holder from the tail to the front (Figure 1(b)).
Radioactive gas evacuation system
The scavenged radioactive gas from the animal holder was carried to 28 decay ducts (each 9.5 m long and 150 mm diameter, total volume of 4649 l) implemented in the facility building, allowing sufficient radioactivity decay before releasing it from the facility (Figure 1(c)).
Animal preparation and experimental procedures
First experiment (assessment of metabolized 15O-H2O)
Each animal assigned for the first experiment was anaesthetized with isoflurane (1.5–3%), and the left common carotid artery was cannulated with a polyethylene tube (SP-31, outer diameter: 0.8 mm, inner diameter: 0.5 mm, Natsume Seisakusho Co., Ltd, Tokyo, Japan). The cannula was secured by tight ligation around the cannulated vessel with 5-0 silk sutures (Ethicon, NJ, USA) and flushed with heparinized 0.9% NaCl to prevent blood clotting.
The animal was then moved to the animal holder and placed in the HRRT PET scanner. After ensuring the stabilisation of the animal under isoflurane anaesthesia, a 6 min attenuation correction scan was conducted. Next, a 6 min dynamic PET scan with a 5 s frame duration was started 15 s before initiation of 15O-O2 inhalation. Arterial blood samples (0.1 ml each) were manually obtained 4–5 times at 30 s intervals after 30 s of 15O-O2 inhalation from the catheter placed in the carotid artery. The blood samples were immediately divided into two samples. One sample was quickly centrifuged and the plasma radioactivity concentration was measured using a BeWell-Q3 well counter (Molecular Imaging Labo, Suita City, Japan), and the other sample was used to determine the whole blood radioactivity concentration.
Second experiment (sequential 15O-PET imaging)
HIE was induced in 9 d/o rats as described previously.7,8 Briefly, under isoflurane anaesthesia (1.5–3%), the left common carotid artery was exposed, ligated (6-0 silk sutures: Ethicon, NJ, USA) and then completely cut with electrocautery. After resting with their dam for 60 min, pups were exposed to 120 min of hypoxia in a chamber containing 8% oxygen and 92% nitrogen (P360 ProOx, BioSpherix, NY, USA) at 36–37 °C rectal temperature. Pups were then returned to their dam again for recovery for over 60 min and kept in standard housing condition after sequential imaging studies. Sham pups underwent a similar surgical procedure without carotid artery ligation and hypoxic insult. Non-operated pups experienced no interventions.
PET and computed tomography (CT) scans were sequentially performed on eleven HIE, four non-operated and three sham pups using β-Cube and X-Cube small animal scanners (Molecubes). During all scans, the animal was placed on a heating pad to maintain its body temperature, and respiration rate was continuously monitored using a sensor pad placed underneath the animal’s chest.
After ensuring the stabilisation of the animal under isoflurane anaesthesia, a series of list-mode PET scans were acquired (Figure 1(d)). Two 14 min scans were started at the time of initiating continuous inhalation of both 15O-O2 and 15O-CO2 over 8 min. A separate 10 min scan was acquired from the time of initiating continuous inhalation of 15O-CO over 3 min on one HIE pup on days 1, 7 and 14 before the 15O-O2 and 15O-CO2 scans to assess cerebral blood flow (
These sequential scans were repeated four times, first within 6 h (day 0) and then at 24 h (day 1), 48 h (day 2) and 7 days (day 7) post-insult for HIE pups in scan group 1, and on days 1, 2, 7 and 14 post-insult for HIE pups in scan group 2. Age-matched non-operated pups were sequentially scanned four times at ages equivalent to those of HIE pups on days 0, 2, 7 and 14. Sham pups were scanned only on day 7 after the surgical intervention. The PET imaging time course with postoperative days and ages in the three subject groups is presented in Table 1. Image data analysis excluded animals that indicated poor radioactivity inhalation dose in the body during scanning or inconstant body position between 15O-O2 and 15O-CO2 scans.
Data processing
In the first experiment, PET images were reconstructed to generate dynamic images with 12 × 5 s, 8 × 15 s and 6 × 30 s frames for 6 min using the 3D OSEM method with depth-of-interaction compensation. 27 Whole blood time-activity curves were obtained from the left ventricular (LV) region in the reconstructed images.
Well-counter counts for the plasma and whole blood samples were normalized by acquisition time duration(s) and weights for each sample, followed by correction for radioactivity decay to the PET initiation time. They were further corrected for the cross-calibration factor determined using a 5 cm diameter and 10 cm long cylindrical phantom so that the well counter-based activity concentration for whole blood was equivalent to that of the LV values in the PET image, as we previously reported. 28 Plasma counting rates were corrected for the plasma/whole blood radioactivity concentration ratio for 15O-H2O. 29 The arterial blood concentration of 15O-O2 [Bq/ml] was then obtained by subtracting the 15O-H2O concentrations from the whole blood radioactivity concentrations for each sample.
In the second experiment, PET images were reconstructed to generate dynamic images with a 1 min frame duration for the entire period of 14 min using 3D OSEM with depth-of-interaction compensation for 15O-O2 and 15O-CO2 and 10 min for 15O-CO scans. Radioactivity decay correction was set uncorrected in the second experiment to avoid degradation of the pixel count precision in decay-corrected images for short-lived 15O radioisotopes, which occurs specifically in the β-Cube scanner.
Data analysis
Determination of the production rate of metabolized 15O-H2O in arterial blood
For the first experiment, a volume of interest (VOI) was selected on the LV cavity region in the dynamic PET images obtained after 30 s continuous inhalation of 15O-O2 using Carimas 2.1 (Turku PET Centre, Turku, Finland). The total blood arterial input function (AIF),
The following model formulation was employed to estimate the metabolized 15O-H2O curve, 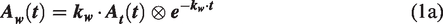

Calculation of parametric values
Parametric values of
For the 15O-O2 inhalation PET data:
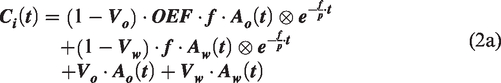
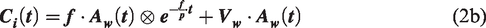


The AIF for 15O-O2 (
The 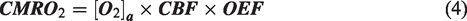
Time course of parametric values
Time courses of absolute
Histological analysis
After the last PET scan on day 14, HIE and non-operated pups (n = 5 and n = 4, respectively) were transcardially fixed with 4% paraformaldehyde under deep anaesthesia with 4% isoflurane. The brains were removed, processed and embedded in paraffin sections. Coronal slices were obtained in 2 mm intervals from the frontal pole using a microtome (Leica RM2265, Nussloch, Germany) and stained with haematoxylin-eosin (H&E) for morphology. Microglial activation was examined using Iba1. Antigen retrieval was performed in citrate buffer (pH 6.0, BioSite BSC-OKHURL) for 20 min using a pressure cooker (Decloaking chamber, Biocare Medical NxGen). Sections were incubated with endogenous enzyme block (hydrogen peroxide) and pre-protein block (BrightDiluent, normal antibody diluent, WellMed BD09-125) to avoid non-specific binding. A primary antibody of Iba1 (1:2000, 019-19741, Wako Ltd., Japan) was applied for 60 min, followed by secondary antibody (BrightVision, 1 step detection system goat anti-rabbit HRP, WellMed DPVR110HRP) for 30 min and detection reagents (BrightDAB, WellMed BS04-110) for 10 min at room temperature.
The area (mm2) of each hemisphere was measured on H&E-stained sections using CaseViewer 2.4 (3DHISTECH Ltd., Budapest, Hungary). The hemispheric volume of each brain was calculated by summing the hemispheric area of each brain slice and multiplying the sum of the section thickness. The mean ipsi-/contralateral hemispheric-volume ratios were compared between HIE and non-operated groups. Additionally, the ipsi-/contralateral hemispheric-volume ratio in each HIE animal was compared with the ipsi-/contralateral ratios of
Statistics
Results are presented as mean ± SD. The student’s paired or unpaired t-test was applied to compare two variables as appropriate. Pearson's analysis was used to test for correlation with GraphPad Prism 9 (GraphPad Software, San Diego, CA, USA). P-values <0.05 were considered statistically significant. Image data analysis was blinded to avoid any subjectivity.
Results
Five of the thirteen rats that were assigned to the first experiment were excluded from the data analysis because of unsuccessful arterial blood sampling or extremely low 15O-O2 inhalation dose during the PET scan. Otherwise, all well counter-based whole-blood radioactivity concentrations agreed with the PET image-derived LV TAC in all cases and were further validated by an earlier study using the same experimental setup.
28
Fitting the predicted metabolized 15O-water TAC to the manually separated 15O-water TAC resulted in average
The whole-body distribution of 15O-gases in a rat scanned on days 1, 7 and 14 post-HI insult is shown in Figure 2 as sagittal PET/CT images accumulated for a period of 0–10 min, 0–5 min and 0–8 min after initiation of 15O-CO, 15O-O2 and 15O-CO2 gas inhalation, respectively. Radioactivity was visible on the surface of the snout inside the facemask (arrow a), in the gas supply tube (arrow b) and on the inner surface of the animal holder (arrow c). These radioactivity spots were negligibly small and did not significantly impact image data analysis. Radioactivity in the brain relative to the heart was lower with 15O-O2 than that with 15O-CO2. Radioactivity in the lung region was greater in 15O-O2 images than 15O-CO2 images.
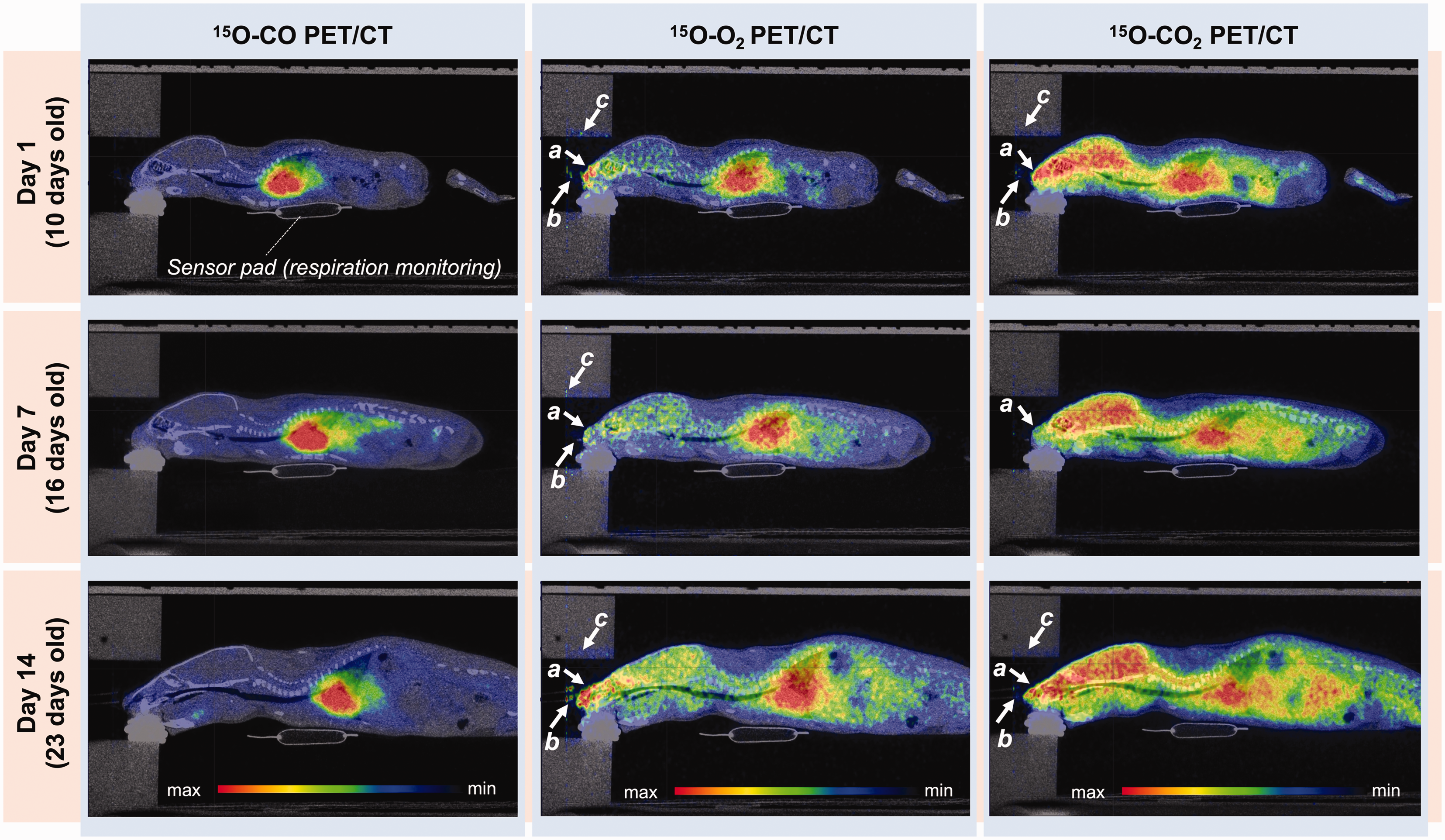
Representative sagittal whole-body PET/CT images of a HIE rat placed in the animal holder on days 1, 7 and 14 post-HI insult. Summed images over 0–10 min after 15O-CO, 0–5 min after 15O-O2 and 0–8 min after 15O-CO2 inhalation were displayed. White arrows (a–c) indicate the effects of radioactivity accumulated in the snout (a), emitted from the radioactive gas supply tube (b) and on the inner surface of the animal holder (c). CT: computed tomography; HIE: hypoxic-ischaemic encephalopathy; PET: positron emission tomography.
Figure 3 shows 15O-gas accumulation in a HIE brain scanned on day 14 (Figure 3(a) to (c)) and LV TAC and tTACs (Figure 3(d) to (f)) for the selected VOIs defined in the 15O-CO image (Figure 3(a)). The decreased radioactivity in the ipsilateral hemisphere is clearly visible in both 15O-O2 and 15O-CO2 PET images (white arrows), while 15O-CO images showed no interhemispheric difference at any time point after HI insult. LV radioactivity concentrations were nearly equal between the 15O-O2 and 15O-CO2 inhalation scans although the supply rate of 15O-CO2 radioactivity was lower (200 MBq/ml) than that of 15O-O2 (500 MBq/ml). The peak values of TACs in both the ipsi- and contralateral brain regions were lower during 15O-O2 inhalation compared with 15O-CO2, as seen in Figure 3(b) and (c), respectively.
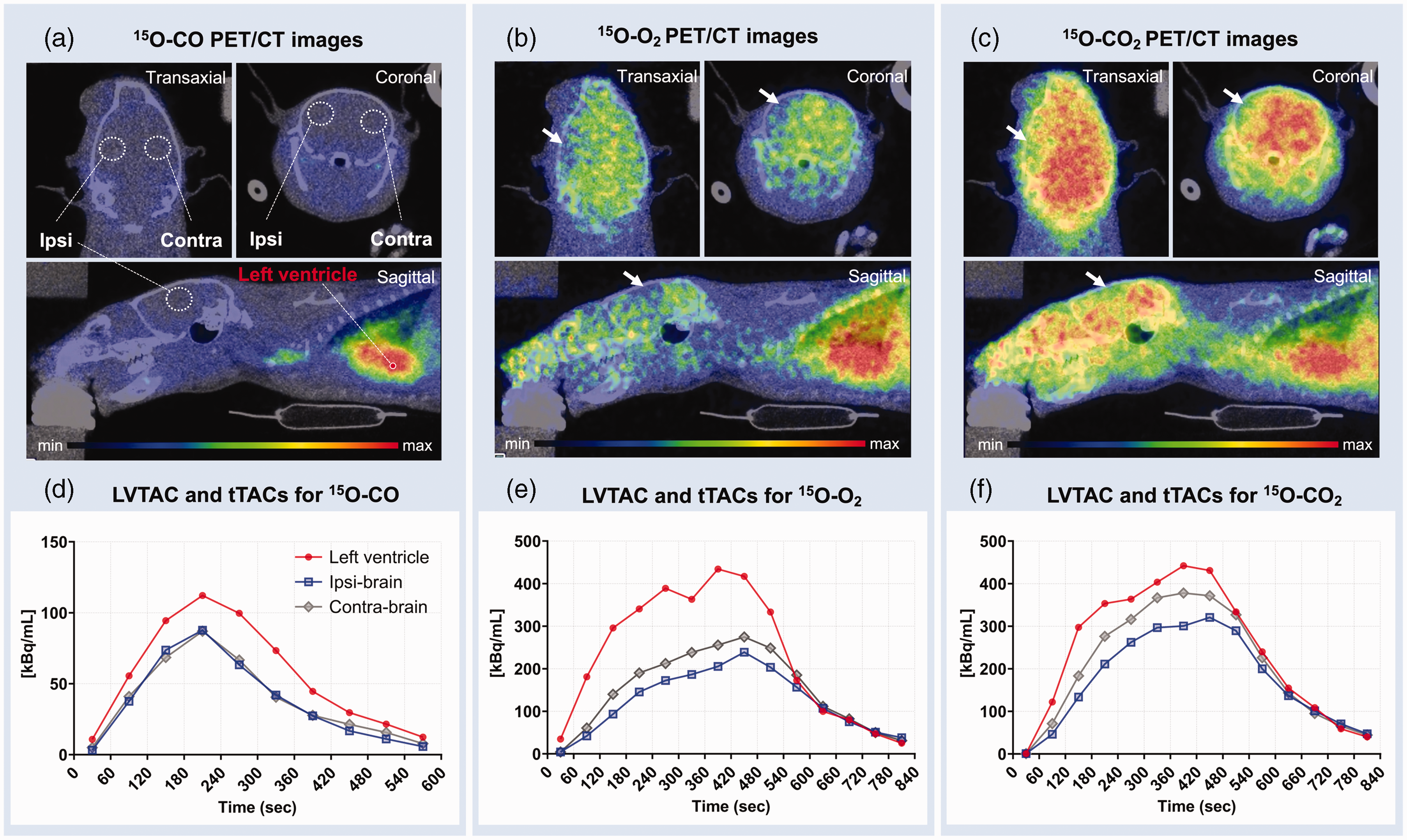
Representative transaxial, coronal and sagittal PET/CT images of the brain of a HIE rat on day 14 post-insult. PET images are summed over 0–10 min after 15O-CO (a), 0–5 min after 15O-O2 (b) and 0–8 min after 15O-CO2 (c) inhalation. The lesion is indicated by the white arrows in the left hemisphere in both 15O-O2 (b) and 15O-CO2 images (c). VOI locations in the LV, the lesion in the ligated-side (ipsilateral) hemisphere and its symmetrical position (contralateral) are also indicated on the 15O-CO images (a). The measured tTACs in ipsilateral (square) and contralateral (diamond) brain regions, and LV TAC (circle) from the same animal successively administered 15O-CO (d), 15O-O2 (e) and 15O-CO2 (f) are also presented. Note that LV TAC for 15O-CO is presented as one-tenth value (d). CT: computed tomography; HIE: hypoxic-ischaemic encephalopathy; LV, left ventricle; PET: positron emission tomography; TAC: time-activity curve; tTAC: tissue TAC; VOI: volume of interest.
A representative result from the NLLSF analysis for fitting
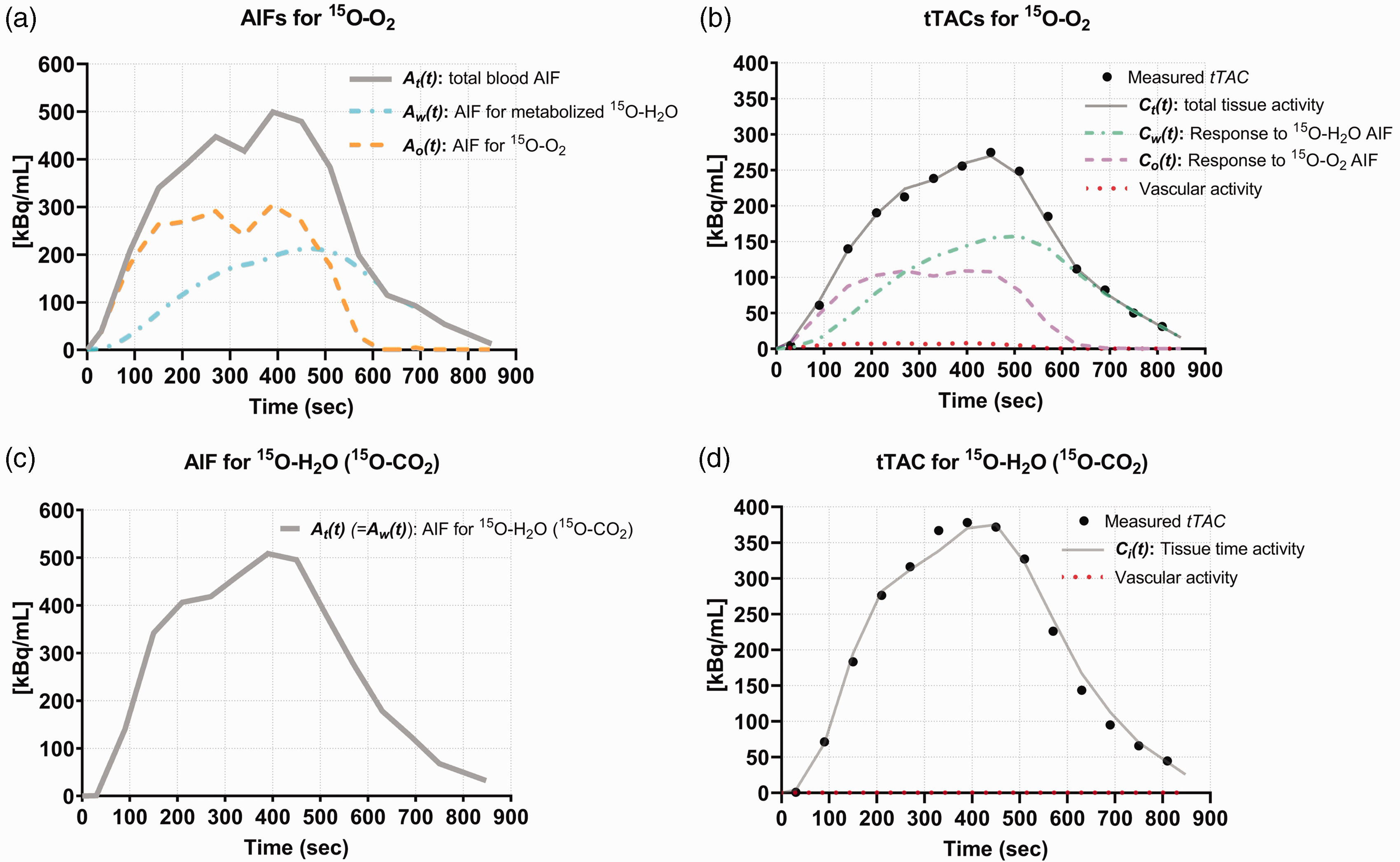
A typical example of AIFs (a) and the results from simultaneous fitting to tTACs (b) for 15O-O2, AIFs (c) and results from simultaneous fitting to 15O-CO2 tTACs (d) obtained from a HIE rat on day 14 after the insult (same case shown in Figure 3). (a) Three input functions are given for the 15O-O2 fitting analysis, i.e. the total blood AIF (
The temporal profile of mean quantitative hemispheric
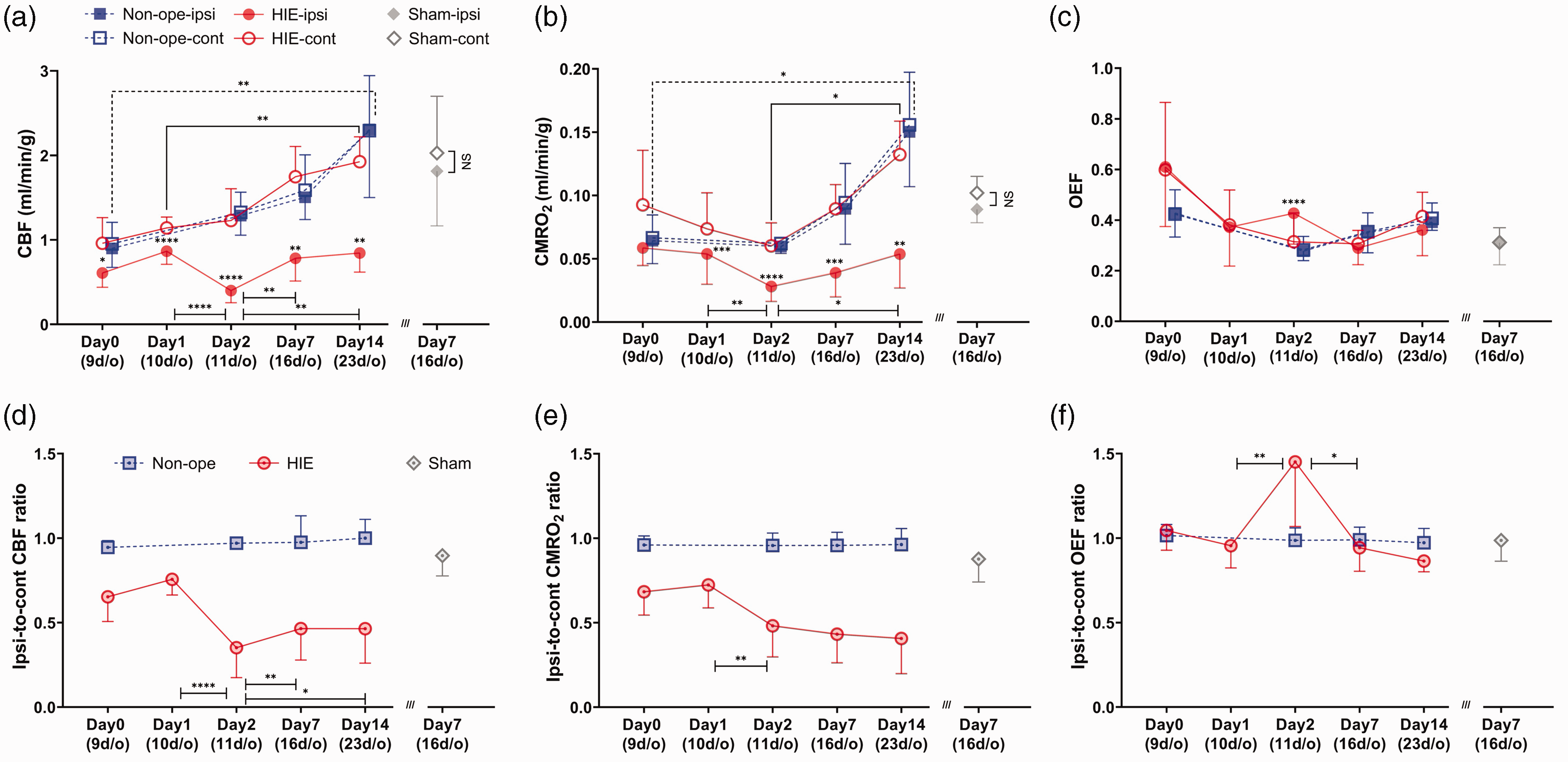
The mean temporal values of (a)
A comparison of 15O-O2 PET/CT images and histology results from a HIE rat on day 14 and an age-matched non-operated rat is shown in Figure 6. Infarct regions measured by H&E staining in the HIE brain coincided with areas showing extremely decreased accumulation of 15O-O2 in PET images (Figure 6(a)). Additionally, increased microglial activation was displayed by Iba1 outside the margins of the infarct cores, where small reductions in 15O-O2
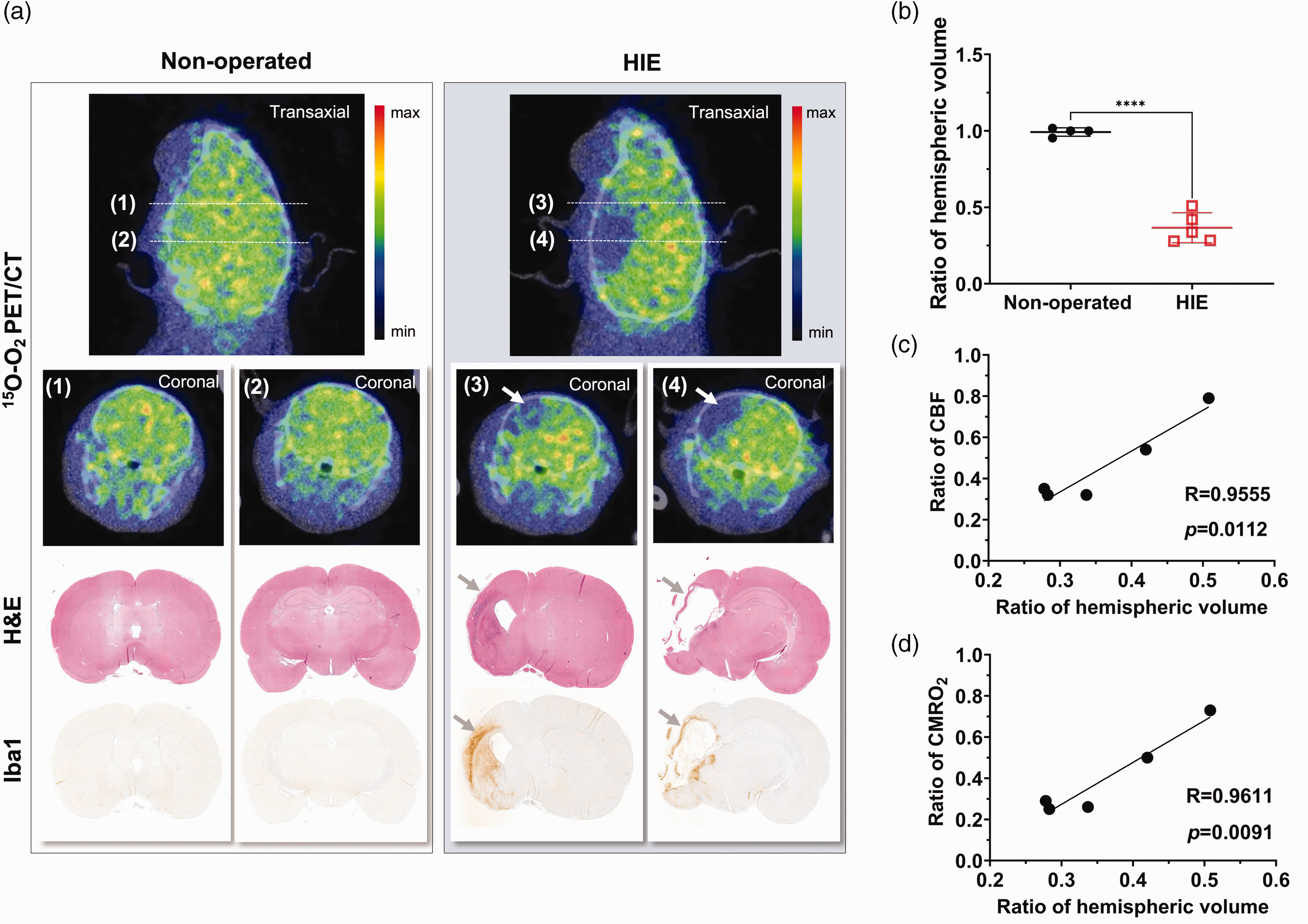
(a) Representative PET/CT images of the transaxial view and each coronal slice at the striatum (1) (3) and hippocampus level (2) (4) of the brains of a HIE pup on day 14 and an age-matched non-operated pup are shown as summed images over 0–5 min after 15O-O2 inhalation. Coronal images stained with H&E and Iba1 of the same animals at equivalent slicing levels are also displayed. Note that the area of the histological tissue loss in H&E images as indicated by grey arrows in the HIE animal is consistent with the defect region in 15O-O2 PET images as indicated by white arrows. Increased microglial activation is also visible around the infarct tissues in Iba1 images as indicated by grey arrows. (b) Ratios of the ipsilateral to contralateral hemispheric volume are presented as mean ± SD, displaying a significantly decreased mean ratio in HIE (****p < 0.0001) compared to non-operated pups (n = 5 and n = 4, respectively). (c, d) The ipsi-/contralateral ratio of
Discussion
Overview of the findings
The comprehensive 15O-oxygen PET system for small animals developed in this study provided quantitative
The technique detected time-dependent changes in
We confirmed that both LV TAC for 15O-oxygen scan in the first experiment agreed with those obtained by the well counter, which is consistent with an earlier finding 28 despite the smaller average animal size in this study. This may be attributed to the contribution of the myocardium surrounding the LV chamber, which has a high flow value, resulting in a similar tTAC in the myocardial wall as the LV blood TAC. We also expect a similar phenomenon in the second experiment, in which a high-resolution β-Cube scanner was employed. However, this remains a hypothesis requiring further verification.
In HIE animals,
Histological tissue losses measured after the last PET scan showed spatial agreement with severely decreased
Methodological advantages
15O-O2 gas is the sole tracer that can trace the kinetics of oxygen molecules and oxygen metabolism. It was shown in both the first and second experiments that 15O-O2 is metabolized quickly in neonatal rats so that the metabolized 15O-H2O replaces 15O-O2 in arterial blood shortly after starting 15O-O2 inhalation, as typically seen in Figure 4(a). Since the 15O-H2O extraction rate to the brain in the capillary bed is over two-fold higher than that of 15O-O2, the contribution of metabolized 15O-H2O to the brain (the second term in equation (2a)) exceeds the level of that of the original 15O-O2 during the continuous inhalation of 15O-O2 (Figure 4(b)), at 3–5 min after 15O-O2 inhalation initiation. To compensate for the contribution of metabolized 15O-H2O, the additional administration of 15O-CO2 (equivalent to intravenous 15O-H2O) was applied as many PET methods that utilized 15O-O2 inhalation protocol.
Simultaneous NLLSF is considered to be a reliable approach for estimating both
Limitations
There are several limitations to this study. Firstly, the animal model used exhibits hemispheric HI injury generated by unilateral ligation of the common carotid artery with subsequent global hypoxic insult. Considering the impact of interhemispheric collateral circulation, the use of ipsi-/contralateral ratios of
A standardized
A
We applied the average
Future implications
The present 15O-oxygen PET system can be further improved if the entire examination duration is shortened. A PET scanner with better counting rate performance is essential for employing a single PET scan protocol during the dual inhalation of 15O-O2 followed by 15O-CO2 with a short interval, as demonstrated previously.46,47 This protocol can reduce the total anaesthetisation time and minimize possible physiological changes during PET scans. The intrinsic vascular radioactivity correction could also be compensated without the need for an additional 15O-CO scan. Continued research in this study would be of great value to evaluate possible pharmacological interventions and the early prognostic value of 15O-PET for neonatal HIE with a larger sample size. Such technical innovations would also facilitate the broader use of 15O-PET in preclinical and clinical applications, including in neonatal patients.
It is also important to establish a technique that can determine the appropriate
Conclusions
We demonstrated that the present 15O-gas inhalation PET system quantitatively assesses temporal changes in
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Turku University Foundation and Instrumentarium Science Foundation, Finland and JSPS KAKENHI [21KK0183] and Cardiovascular Research Foundation, Japan.
Acknowledgements
The authors gratefully acknowledge the contribution of Ms Heidi Partanen for radioactive gas quality control and Mr Aake Honkaniemi for PET scanner operation (Turku PET Centre). We also thank the Histology Core Facility of the Institute of Biomedicine, University of Turku for technical support in histological analysis and UTUCAL for assistance in animal experiments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
SS, VP, EY, TG and HI designed the experiments, participated in the interpretation of data and were responsible for writing the manuscript. Material preparation and data collection were performed by SS, JI, TT, AH, EK and SJ. NK and HI were responsible for the kinetic calculation processing. MT and SK reviewed the manuscript. All authors have read and approved the final manuscript.
