Abstract
Abnormal cerebral energy metabolism owing to dysfunction of mitochondrial electron transport has been implicated in the pathogenesis of Parkinson's disease (PD). However,
Keywords
Introduction
Parkinson's disease (PD) is a degenerative neurologic disease, which is manifested by tremor, rigidity, bradykinesia, and postural instability (Calne et al, 1992). Although PD is characterized neuropathologically by alpha-synuclein-immunopositive Lewy bodies in the substantia nigra and other brainstem structures, there is an increasing recognition that PD is a diffuse brain disease involving both cortical and subcortical structures (Braak et al, 2003). Dysfunction of mitochondrial oxidative metabolism has been implicated in the pathogenesis of PD, through a variety of mechanisms including reduced ATP production and generation of free radicals (Abou-Sleiman et al, 2006). Diminished activity in complex I of the mitochondrial electron transport system (ETS) in post-mortem brain tissue has been reported in the cortex and substantia nigra as well as in platelets (Krige et al, 1992; Schapira, 1994; Keeney et al, 2006).
Dysfunction of the mitochondrial ETS will reduce the cerebral metabolic rate of oxygen (CMRO2). However, because the brain regulates metabolism to match energy demand, both CMRO2 and the cerebral metabolic rate of glucose (CMRglc) are reduced under any condition with diminished energy demand, such as barbiturate anesthesia (Astrup et al, 1981). Specific defects in mitochondrial ETS decrease CMRO2 proportionately more than CMRglc (fewer moles of oxygen consumed per mole of glucose metabolized), thereby producing a reduction in the CMRO2/CMRglc ratio below the normal value of 5.6 (Brierley et al, 1977; Frackowiak et al, 1988). This is in distinction to the situation during physiologic brain activation in which the CMRO2/CMRglc ratio is also below normal, but CMRO2 is not reduced (Dienel et al, 2002). Therefore,
Materials and methods
Participants
Inclusion criteria were as follows:
Clinically definite PD:
Onset after the age of 30 years. Three of the following: rest tremor, rigidity, bradykinesia, or postural instability; or two of these features with one of the first three displaying asymmetry (Calne et al, 1992). Three or more of the following: unilateral onset; persistent asymmetry; rest tremor; or progression (United Kingdom Parkinson Disease Society Brain Bank criteria) (Hughes et al, 1992). Asymmetric resting tremor (Rajput et al, 1991). Symptomatic for less than 4 years. Never taken any anti-Parkinsonian medication.
Exclusion criteria were as follows:
Any of the following: a history of stroke; history of head injury; history of definite encephalitis, oculogyric crisis; previous dopamine receptor antagonist treatment; sustained remission; strictly unilateral features after 3 years; supranuclear gaze palsy; cerebellar signs; early severe autonomic involvement; early severe dementia (within first year of onset) with disturbances of memory, language, and praxis; extensor plantar reflex; any defect on brain imaging; or 1-methyl 4-phenyl 1,2,3,6-tetrahydropyridine (MPTP) exposure. Major neurologic or psychiatric disease other than PD or clinically significant lesions on brain imaging that was performed before enrollment in the study. Regular treatment or exposure in the last 6 months to flunarizine, cinnarizine, reserpine, amphetamines, monoamine oxidase (MAO) inhibitors, or other medications that might interfere with mitochondrial metabolism. Currently taking chloramphenicol or valproic acid. Ever having taken dopaminergic medications for any reason. Anticholinergics, amantadine, CoQ10, selegiline, and vitamins E and C must be discontinued for 30 days before entry into the study. Diabetes mellitus treated by medications. Pregnancy.
All underwent clinical neurologic evaluation by movement disorder specialists and were assigned a duration of symptoms and a clinically more involved side of the body based on this examination. They were all followed clinically for at least 3 years to determine whether each still met the above clinical criteria for idiopathic PD. This determination was made by an investigator blind to the PET data.
Inclusion criteria were as follows:
Disease free by subject's own history, including no history of migraine, childhood febrile seizures, or head trauma with loss of consciousness. Taking no medication by subject's own history. No signs or symptoms of neurologic disease other than mild distal sensory loss in the legs consistent with age. No pathologic lesions on magnetic resonance (MR) scan done for this study (see below). Mild atrophy and punctate asymptomatic white matter abnormalities were not considered pathologic.
Exclusion criteria were the same as for the participants with PD.
Normal controls were recruited as part of a larger study including patients with Huntington's Disease and then retrospectively age matched to the patients with PD without reference to PET measurements (Powers et al, 2007).
Image Acquisition
T1-weighted MR images were acquired with a Siemens Sonata 1.5 T scanner (Siemens Medical Solutions USA Inc., Malvern, PA, USA). A midsagittal scout spin-echo sequence was used to position the subject and then a three-dimensional magnetization prepared rapid acquisition gradient echo (MPRAGE) sequence was acquired (TR (repetition time)/TE (echo time)/TI (inversion time) = 1,900/3.93/1,100 ms, FA (flip angle) = 8°, 7:07 mins, 128 times 256 times 256 matrix, 1.25 times 1 × 1 mm voxels). In 10 patients and 11 controls, high-resolution MR images were acquired with a Siemens Allegra 3T scanner (Siemens Medical Solutions USA Inc.) to permit the identification of the substantia nigra. A two-dimensional turbo spin-echo sequence (TR/TE = 5,540/99 ms, FA = 180°, 384 times 512 matrix, 0.5 times 0.5 times 2 mm voxels acquired as 21 interleaved, contiguous planes) was coregistered to an MPRAGE image, which, in turn, was coregistered to the MPRAGE image from the 1.5 T scanner.
Positron emission tomography images were obtained in the two-dimensional acquisition mode with a Siemens/CTI ECAT EXACT HR 47 PET scanner (Siemens Medical Solutions USA Inc.) with participants lying supine in a quiet dark room. Cerebral blood flow, cerebral blood volume, CMRO2, and oxygen extraction fraction were measured as previously described (Raichle et al, 1983; Mintun et al, 1984; Martin et al, 1987; Powers et al, 2007). Arterial blood samples for the measurement of pCO2 and oxygen content were collected and analyzed (Instrumentation Laboratory, Lexington, MA, USA). In one PD participant and one control, pCO2 measurements were not done because of a broken machine. Dynamic emission PET scans were obtained after slow intravenous injection over 10 to 20 secs of 10 mCi of 18F-fluorodeoxyglucose (18FDG) for the measurement of CMRglc (Powers et al, 2007). All PET emission scans were reconstructed with filtered back projection using the individual attenuation measurements and scatter correction with a ramp filter cutoff at the Nyquist frequency producing images with a resolution of 4.3 mm full width at half maximum.
Image Analysis
The 1.5 T MR image was segmented into the brain and cerebrospinal fluid based on voxel intensities. Manual editing was required to remove external tissue, where there was insufficient cerebrospinal fluid to separate the brain (near sinuses, temporal lobes, eyes, and brainstem), and a single erosion followed by conditional dilation completed the tissue segmentation. The MR image was then edited manually to generate a region of interest encompassing both cerebral hemispheres by removing the cerebellum and brainstem along a plane connecting the posterior commissure and the most inferior point of the interpeduncular fossa.
The substantia nigra was identified on the 3 T turbo spin-echo sequence (Foster et al, 2007). This region included both the pars compacta and the pars reticulata. Although the pars compacta is the primary site of pathology in PD, the spatial resolution of PET limits the small size of the region from which metabolic measures can be made and components of the pars compacta extend finger-like projections into pars reticulata making anatomic separation of the two not feasible on magnetic resonance imaging (Damier et al, 1999).
The putamen was identified on the 1.5 T MPRAGE sequences. Medially and anteriorly, the putamen was defined on the coronal view. Where separation from the caudate or nucleus accumbens by white matter was not clear, an arbitrary straight line was drawn laterally and inferiorly following the direction of the internal capsule on this section. This rule does not correspond perfectly to anatomic putamen, but represents a necessary compromise, because histologic detail is required to distinguish the putamen from caudate or nucleus accumbens accurately. Laterally, the claustrum was excluded whenever the external capsule could be partially or completely visualized. The tail of the caudate was excluded by reference to sagittal views.
The globus pallidus was identified on 1.5 T transverse MPRAGE images by its lateral border with the putamen and its medial border with the internal capsule. The highest and lowest MR images that contained visible pallidum nestled against the putamen defined its superior and inferior borders.
The volume of the substantia nigra, putamen, and globus pallidus for each participant was determined from the number of voxels within each structure and the voxel volume size.
The original segmented MR image plus the three 15O PET images were coregistered to a composite from 40 to 60 mins 18FDG PET image using Automated Image Registration software (AIR, Roger Woods, University of California, Los Angeles, CA, USA) (Woods et al, 1993). Mean counts corrected for partial volume effects owing to nonbrain structures including cerebrospinal fluid were generated for the bihemispheric, substantia nigra, putamen, and globus pallidus regions of interest for each of the three 15O PET images and for the dynamic 18FDG PET images (Videen et al, 1999; Powers et al, 2007). Corrected mean counts were converted to quantitative cerebral blood flow, oxygen extraction fraction, or CMRO2 (Videen et al, 1987).
For the measurement of bihemispheric CMRglc, a modified Marquardt parameter estimation routine was used to derive rate constants for each participant using the partial volume corrected dynamic bihemispheric 18FDG PET counts and arterial whole blood time-radioactivity curves (Powers et al, 2007). For the calculation of the regional substantia nigra, putamen, and globus pallidus CMRglc, we used a single scan strategy using mean 18FDG counts integrated from 40 to 60 mins postinjection and a single set of rate constants (Phelps et al, 1979). We compared the 4 rate constants derived from bihemispheric parameter estimation in the 12 participants with PD and the 12 age-matched normal controls (see below) and found no difference (all
Statistical Analysis
The null hypothesis of no difference in cerebral oxygen metabolism in patients with early PD was tested by the comparison of bihemispheric CMRO2 between participants with PD and age-matched normal controls by two-sided unpaired
This protocol received prior approval by the Washington University Human Studies Committee (IRB). Written informed consent was obtained from all participants.
Results
Participants
Twenty-two subjects with PD were initially enrolled. Complete studies were carried out in 12 subjects. Eight subjects did not have complete PET and MR studies because of technical problems. Two were determined not to have PD by the lack of persisting asymmetry and no motor response to high doses of
Cerebral Mitochondrial Metabolism
The primary analysis showed a statistically significant 24% increase in bihemispheric CMRO2 in PD (
Bilateral cerebral and substantia nigra metabolism in early PD
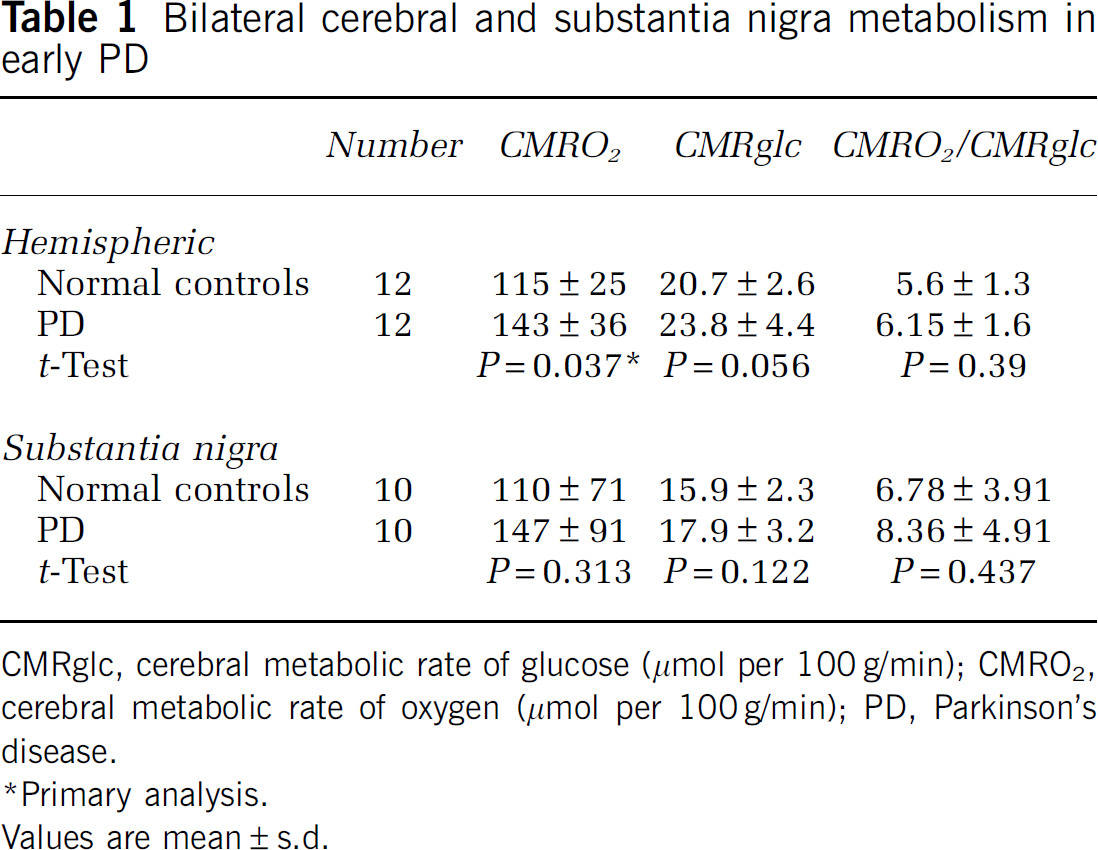
CMRglc, cerebral metabolic rate of glucose (μmol per 100 g/min); CMRO2, cerebral metabolic rate of oxygen (μmol per 100 g/min); PD, Parkinson's disease.
Primary analysis.
Values are mean ± s.d.
Similar results, although with more measurement imprecision as expected, were found in the substantia nigra. Examination of the confidence intervals for the differences between the two groups shows that there is less than a 20% chance that substantia nigra CMRO2 in PD is lower by more than 10%, that CMRglc is lower by more than 16%, and that there is only a 10% chance that CMRglc/CMRO2 is reduced by any amount.
Measurements from the putamen and globus pallidus also showed increases in regional CMRO2 and CMRglc (Table 2). Analysis of regional/bihemispheric ratios showed no difference between controls and participants with PD indicating that the increases in regional metabolism were primarily a reflection of overall bihemispheric changes There were no differences in CMRO2 or CMRglc between the structures ipsilateral and contralateral to the side of the body with the greatest signs (data not shown).
Bilateral basal ganglia metabolism in early PD
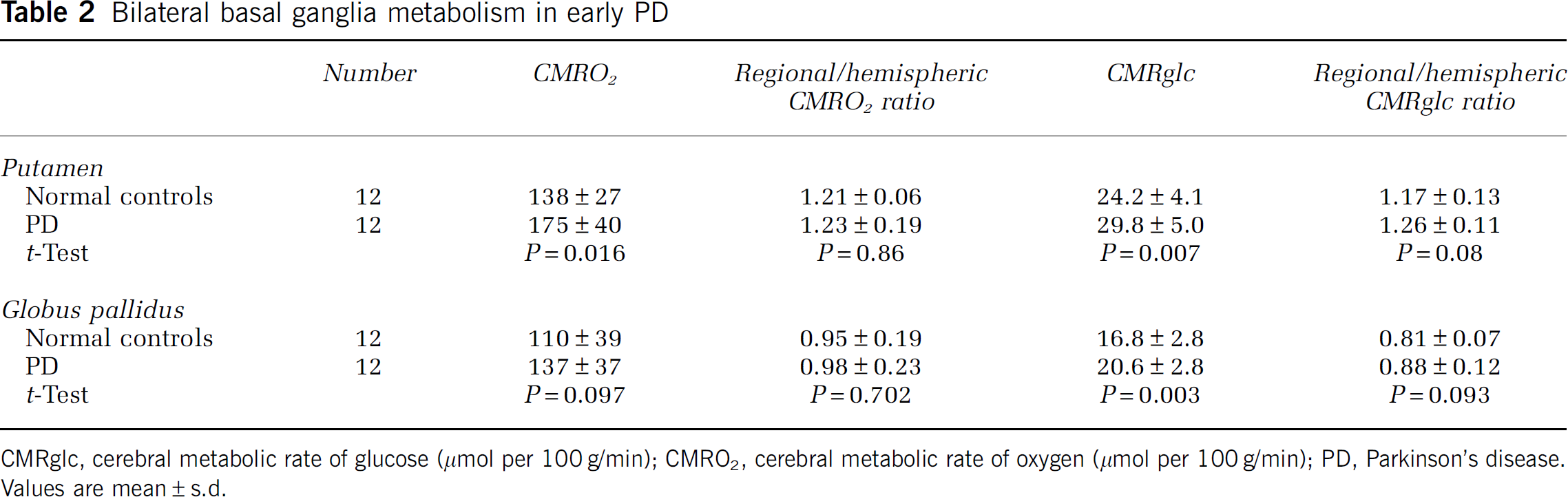
CMRglc, cerebral metabolic rate of glucose (μmol per 100 g/min); CMRO2, cerebral metabolic rate of oxygen (μmol per 100 g/min); PD, Parkinson's disease. Values are mean ± s.d.
Systemic physiologic measurements were comparable between the participants with PD and the controls: pCO2 36 ± 4 versus 37 ± 3 mm Hg (
There were no differences in volume between the normal controls and the participants for the substantia nigra (
Discussion
The CMRO2 method that we have used has been validated for quantitative accuracy in nonhuman primates across a wide range of CMRO2 (Mintun et al, 1984; Altman et al, 1991). Included in these validation studies were measurements during intraaortic sodium cyanide infusion, specifically showing the ability of this technique to accurately measure a reduction in CMRO2 under conditions of reduced mitochondrial ETS activity (Altman et al, 1991).
We corrected for artifactual reductions in PET measurements owing to partial volume effects from increases in ventricular, cisternal, and sulcal cerebrospinal fluid volume owing to atrophy. The method does not correct for partial volume effects on the globus pallidus and putamen from surrounding white matter, which will worsen with atrophy of these structures (Videen et al, 1988, 1999). These white matter effects cause lower values for CMRO2 and CMRglc when the globus pallidus and putamen are smaller. However, we found no difference in the volume of these structures between normal controls and participants with PD, hence this cannot be the explanation for the increased metabolism in PD.
The hypothesis that patients with early PD have a defect in cerebral mitochondrial electron transport was tested by the comparison of bihemispheric CMRO2 between participants with PD and age-matched normal controls. This primary analysis showed a statistically significant 24% increase in bihemispheric CMRO2 in PD. Explanatory analyses showed that CMRO2/CMRglc was not reduced. Similar results, although with more measurement imprecision as expected, were found in the substantia nigra. Increased CMRO2 with a normal CMRO2/CMRglc is inconsistent with a defect in mitochondrial oxidative phosphorylation owing to reduced activity of the mitochondrial ETS (Brierley et al, 1977; Frackowiak et al, 1988). Although a finding of normal CMRO2 in PD would not exclude the possibility of dysfunction of mitochondrial ETS because complex I, III, and IV activity can be substantially reduced before there is a reduction in CMRO2, dysfunction of the ETS cannot be the explanation for increased CMRO2 in PD (Davey et al, 1998). Because PD symptoms were already manifest in these 12 patients, we can exclude deficient energy production owing to a reduced activity of the mitochondrial ETS as a pathogenic mechanism of their disease. Thus, although defects in mitochondrial ETS may be present in some patients with PD, the absence of such defects in these 12 patients with early PD indicates that they cannot be essential to the pathogenesis of neuronal death in early PD.
Previous studies of CMRglc in PD have yielded mixed results. In five studies of global CMRglc, four have reported reductions of approximately 20% and one reported no significant difference compared with age-matched controls (Kuhl et al, 1984; Leenders et al, 1985; Eidelberg et al, 1993, 1994; Piert et al, 1996). In one of these studies, reductions in global CMRglc were seen only after
Basal ganglia metabolism in PD has been reported to be increased, decreased, or unchanged (Kuhl et al, 1984; Rougemont et al, 1984; Martin et al, 1984; Wolfson et al, 1985; Leenders et al, 1985; Mohr et al, 1992; Eidelberg et al, 1993, 1994, 1995; Piert et al, 1996). In these studies, findings are dependent on whether analysis is performed using absolute values, relative values, or more sophisticated image analysis techniques, such as statistical parametric mapping or scaled subprofile modeling (Eidelberg et al, 1993, 1995; Piert et al, 1996). Increased metabolism in basal ganglia structures has been ascribed to the loss of dopaminergic inhibitory pathways (Martin et al, 1984; Wolfson et al, 1985; Eidelberg et al, 1993). After unilateral 6-hydroxydopamine lesions of the substantia nigra in rats, glucose hypermetabolism is focally restricted to the ipsilateral globus pallidus and is transient (Wooten and Collins, 1981). We found no evidence that the increases in basal ganglia CMRO2 and CMRglc were focal or related to the more clinically involved side, but we cannot exclude this possibility due to the small sample size and low power. Nevertheless, loss of inhibitory dopaminergic input seems an unlikely explanation for the general increase in hemispheric CMRO2 that we measured. Alternatively, this general increase in CMRO2 could be due not to an increased metabolic demand but to an uncoupling of ATP production from oxidation in the terminal stage of oxidative phosphorylation. Uncoupling (dysfunction of Complex V ATP synthase) produces an increase in both CMRO2 and CMRglc similar to what we observed (Patel and Brewer, 2003; Tretter and Adam-Vizi, 2007). Whether uncoupling of oxidative phosphorylation occurs in early PD and whether it is important in the pathogenesis of PD will require further study.
Footnotes
Acknowledgements
We thank Lennis Lich, John Hood, Lori McGee-Minnich, and the Washington University Cyclotron Staff for their assistance.
The authors state no conflict of interest.
