Abstract
Despite the high prevalence, stroke remains incurable due to the limited regeneration capacity in the central nervous system. Neuronal replacement strategies are highly diverse biomedical fields that attempt to replace lost neurons by utilizing exogenous stem cell transplants, biomaterials, and direct neuronal reprogramming. Although these approaches have achieved encouraging outcomes mostly in the rodent stroke model, further preclinical validation in non-human primates (NHP) is still needed prior to clinical trials. In this paper, we briefly review the recent progress of promising neuronal replacement therapy in NHP stroke studies. Moreover, we summarize the key characteristics of the NHP as highly valuable translational tools and discuss (1) NHP species and their advantages in terms of genetics, physiology, neuroanatomy, immunology, and behavior; (2) various methods for establishing NHP focal ischemic models to study the regenerative and plastic changes associated with motor functional recovery; and (3) a comprehensive analysis of experimentally and clinically accessible outcomes and a potential adaptive mechanism. Our review specifically aims to facilitate the selection of the appropriate NHP cortical ischemic models and efficient prognostic evaluation methods in preclinical stroke research design of neuronal replacement strategies.
Keywords
Introduction
Ischemic stroke (IS) is a leading cause of disability with high socioeconomic cost worldwide. 1 The standard acute-phase interventions such as tissue plasminogen activator (tPA) and mechanical thrombectomy dramatically improved the outcomes of IS patients. 2 However, once survivors miss the opportunity of revascularization within 24 hours after the initial stroke, they must rely on spontaneous or rehabilitation-supported functional recovery.3,4 It is reported that almost half of IS patients have lifelong disabilities, 5 and survivors with motor cortex involvement and descending motor fiber damage had shown motor deficits, especially the impairment of hand dexterity, severely affecting their quality of life. 6 To date, neurological deficits in IS patients remain incurable due to the limited regeneration capacity in the central nervous system (CNS). Therefore, new effective treatment strategies with a wider therapeutic window and limited side effects are needed for subacute and chronic IS patients to restore tissue repair and functional recovery.
Neuronal replacement strategies are highly diverse and relatively new biomedical fields that utilize newborn neurons to replace damaged or dying ones. 7 Although this sounds like a daunting challenge, the landmark discovery of lifelong neurogenesis in adult NHP and humans demonstrated that neural stem cells (NSCs) continuously generate newborn neurons and integrate into host neuronal circuitries.8–10 Currently, there are three main neuronal replacement strategies: exogenous stem cell transplants, biomaterials, and direct neuronal reprogramming. 11 These approaches are at different stages of development. The first approach utilizing exogenous cells has proven to achieve encouraging outcomes in rodents, demonstrating that translated cells can survive, differentiate into functional neurons, integrate into host neural circuitry and improve neurological outcomes.12–14 The latter two approaches utilizing endogenous cells also proved that neuronal replacement is feasible and achieves behavioral recovery in rodent models of stroke.15,16 Thus, these neuronal replacement strategies offer promising future treatment options for stroke.
From bench to bedside, most initial stroke studies were conducted in rodents in consideration of their abundant source, low cost, and easy operation. Although impressive results were occasionally obtained in rodent models, these clinical trials ultimately failed when translating directly from rodents to humans without an intermediate step.17,18 Several arguments tried to explain the reason why bench results get “lost in clinical translation.” Specifically, the huge interspecies differences between rodents and humans were considered an important issue. 19 Correspondingly, the Stroke Therapy Academic Industry Roundtable (STAIR) proposed the use of large animals, especially non-human primates (NHP), following lower-order species because of their phylogenetic proximity to humans.20,21 Thus, NHP stroke models are crucial for the safe and effective clinical translation of cell replacement strategies.
Here, we review (1) the recent progress of promising neuronal replacement therapy in NHP stroke studies; (2) NHP species used in IS study and their advantages in terms of genetics, physiology, neuroanatomy, immunology, and behavior; (3) various methods for establishing NHP focal ischemic models targeting the motor cortex; and (4) a long-term motor functional outcome assessment and a potential adaptive mechanism of neural regeneration via a comprehensive analysis of infarction volume, cerebral blood flow (CBF), neurobehavioral assessment, and neural replacement. Taking this approach, we aim to provide useful information for investigators to select the most appropriate NHP models and efficient prognosis evaluation methods for their neural replacement preclinical research.
Neuronal replacement strategies in NHP stroke studies
Ischemia damage several cell elements of the neurovascular unit (NVU) including neurons, astrocytes, endothelial cells, pericytes, oligodendroglia, and microglia.22–24 Neurons were found to be most vulnerable and cause widespread damage in infarction and penumbra.25–28 Neuronal replacement strategies initially aimed to replace the dead neurons with young neurons through tissue engineering or cellular approaches in subacute or chronic stroke.7,11,29 Subsequently, more researchers proposed its bystander effects, including immunomodulation, angiogenesis, glia replacement, and neuroprotection.30–32 On one hand, the transplant substrates or biomaterials efficiently reproduce the physical environment of the brain tissue to provide a scaffold for replacement cells (neurons, astrocytes and oligodendrocytes).33–35 On the other hand, the transplant NSCs or biomaterials release different bioactive molecules and factors to inhibit microglia activation,36,37 promote neurogenesis38,39 and improve angiogenesis.16,40 Thus, neuronal replacement strategies seem a multimechanistic approach through multimodal therapeutic action on multiple cell elements of NVU. 41 Compared to bystander effects, neuronal replacement is more likely to meaningfully restore lost function through the expansion of the available neural substrate.30,32 For this reason, we will next shift our focus back to neuronal replacement and discuss the recent progress of neuronal replacement therapy in NHP stroke studies.
Exogenous stem cell transplants
Exogenous stem cell transplants have been successful at unprecedented levels, and some have reached the stage of clinical trials. 42 The transplanted cell lines used for stroke include embryonic stem cells, mesenchymal stem cells, olfactory ensheathing cells, bone marrow mononuclear cells, multipotent adult progenitor cells and induced pluripotent stem cells (iPSCs).38,43 In analyzing clinical trials, most phase Ι studies have demonstrated safety and functional improvement over baseline.44–50 Despite the positive progress, limited neuronal replacement is still a common challenge. 51 Further preclinical NHP stroke study is required to better understand important factors determining cell transplantation success, such as transplanted cell type, route of administration, treatment time window, and dosing number. Lee et al. 52 transplanted human neural stem cells (hNSCs) into the intact brain of NHP without immunosuppression. They revealed that hNSCs could survive and differentiate into neurons without evidence of tumor formation two years after transplantation. Roitberg et al. 53 further transplanted hNSCs into the NHP brain after one week of ischemic injury. They found that hNSCs can survive up to 105 days and partly undergo neuronal differentiation. While most hNSCs were mainly situated around the injection tracks and did not repopulate the ischemic lesion. Thus, further efforts will still need to improve graft survival and achieve electrophysiological integration into the damaged brain for functional outcomes in NHP preclinical study before the large-scale application of cell transplantation in clinical.
Biomaterials
Biomaterial strategy is transplanting acellular biomaterials into the injury area with the goal of augmenting endogenous neurogenesis. 54 In addition to their function as structure support, biomaterials constantly deliver neurotrophic factors or signaling cues to improve the local microenvironment in favor of regeneration.31,55,56 Biomaterial approaches are demonstrated to enhance and redirect endogenous neuroblast migration from neurogenic niches to specified damaged brain regions and induce the neuroblasts to be differentiated into mature neurons in rodents.34,37,57–60 Presently, there are some initial applications to target the NHP stroke brain with biomaterials. Bjugstad et al. 61 explored the tolerability of polyethylene glycol (PEG)-based hydrogel in the striatum and cerebral cortex of healthy NHP. These biomaterials induced mild inflammation and gliosis, which indicate their potential use for neural tissue regeneration. In a chronic NHP stroke study, Cook et al. 62 injected hyaluronic acid (HA) hydrogel slowly releasing brain-derived neurotrophic factor (BDNF) into the stroke cavity of a macaque monkey. At two weeks after delivery, hydrogel-released BDNF can be detected up to 2 cm from the infarct, a distance relevant to neural repair and recovery in humans. In an acute NHP stroke study, Kawaguchi et al.63,64 demonstrated that intravenous injection of liposome-encapsulated hemoglobin (LEH) could significantly reduce the volume of infarct area and improve behavioral outcomes at 8 days after middle cerebral artery occlusion (MCAO). However, all the above NHP studies did not provide any data related to endogenous neurogenesis due to short-term observation after stroke. Further investigation of survival, maturity, and synaptic integration of redirected neuroblasts is needed in stable stroke models of NHP with long-term survival time after ischemic injury.
Direct neuronal reprogramming
Direct neuronal reprogramming is the forced conversion of local glia into neurons by transduction with various neurogenic fate determinants.65,66 This is an attractive approach to simultaneously replace the lost neuronal population and reduce the astroglial scar following brain injury. 67 NeuroD1-induced neuronal reprogramming has been explored in the focal ischemic injury model in mice. It demonstrated that NeuroD1-mediated reprogramming could regenerate a large number of functional new neurons to restore lost neuronal functions after stroke.68,69 Based on these successful investigations in rodents, Ge et al. 70 further tested adeno-associated virus (AAV) NeuroD1-based neuronal reprogramming in an NHP stroke study. They found that NeuroD1 efficiently converts 90% of the infected astrocytes into neurons in the ischemic cortex of the monkey, and those newly regenerated neurons could survive over 1 year. Moreover, this approach not only significantly increased the neuronal density and synaptic formation, but also reduced the number of reactive microglia and macrophages. 71 Although these results suggest direct neuronal reprogramming in vivo seems an effective therapy to regenerate new neurons in NHP following stroke, further studies are still needed to repeatedly verify the safety and stability of in vivo gene operation.
Presently, clinical implementation of neuronal replacement therapy for tissue repair and functional restoration is distant. However, these strategies offer expanded therapeutic time window expansions from hours to months and provided treatment options for subacute and chronic stroke patients, so neuronal replacement therapy should be actively pursued in the preclinical and translational space. 72 Currently, although the safety of these approaches has been initially demonstrated in NHP, further attention should be given to improving neural survival rate, identifying lesion-specific neuronal phenotypes, monitoring neural activity and testing neuroplasticity functional integration in NHP stroke models. Thus, the establishment of chronic models of NHP allowing long-term imaging and neurological readouts are important to accelerate and improve clinical translation of neuronal replacement strategies for stroke.
NHP species used in stroke study
NHP is classified as a gyrencephalic primate whose brains have a highly folded cortex and a lissencephalic primate whose brains have a smaller number of cortical sulci. 73 They could also be divided into New World monkeys and Old World monkeys in accordance with different habitats. 74 Of them, most Old World monkeys belong to gyrencephalic species. Table 1 summarizes the species, common name, gyrification, 75 habitat, body weight,76,77 and precision grip of the NHP that are commonly used in stroke studies.
NHP species used in ischemic stroke research.
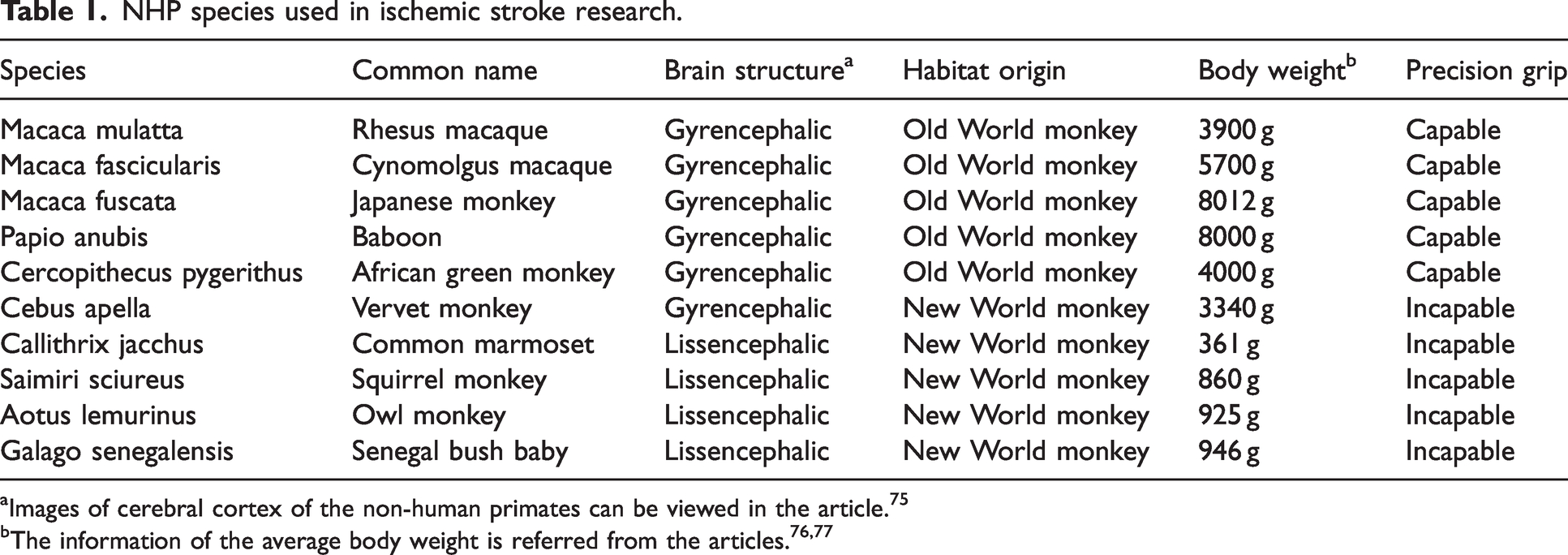
Images of cerebral cortex of the non-human primates can be viewed in the article. 75
The most used lissencephalic NHP species to conduct stroke experiments are the common marmoset and squirrel monkeys. Their small body size and brain volume make them easy to operate. Their relatively flat cortex allows cortical surface imaging. In addition, their sensorimotor and visual representation is on the cortical surface, which provides advantages in precise functional mapping by electrophysiological recording. 78 In addition, they are incapable of precision grip, as characterized by the absence of the ability to hold small objects between the tips of the index finger and the thumb. This disadvantage affects the behavioral assessments after stroke. Moreover, the lissencephalic brain structure is thought to be more rat-like than human-like, which adds an uncertain layer to clinical translation. 79
The most adopted gyrencephalic species are rhesus macaque and cynomolgus macaque. Such NHPs require high costs for purchasing and feeding, high standard surgical and imaging facilities, high ethical considerations, a professional veterinarian for animal anesthesia and post-operative care, and specific staff for the development and maintenance of the stroke model. 80 The COVID-19 pandemic worldwide has further aggravated the quantity shortage and substantial appreciation of the prices of macaques. Despite the above limits in economic and ethical factors, gyrencephalic NHP is the best choice for preclinical stroke study because it can better mimic the physiological symptoms after ischemic stroke.
The gyrencephalic NHP has inherent advantages, and next, we take macaques for example. Genetically, a macaque shares more than 92% of the human genomic sequence. 81 Physiologically, macaques have similar long gestations, postnatal development, and lifespan. 82 Immunologically, macaques share great similarities in leukocyte composition, fibrinolytic protein inflammatory molecules, and hemostatic components to humans. 83 They mimic the cerebrovascular reactions and inflammatory response to stroke in humans comprehensively. Behaviorally, macaques are capable of precision grip and bipedal walking, making them suitable for assessing sensory and motor impairment. Neuroanatomically, macaque brains closely resemble human brains on various criteria (Table 2): (1) In terms of mass, the human brain is ∼3,800 times larger than the mouse brain and ∼17 times larger than the macaque brain. 84 The encephalization quotient is a measure of brain size relative to a taxonomic standard. The encephalization quotient for humans is 7.8, that for macaques is 2.1, and that for rodents is in the 0.4–0.5 range.84,85 The similar brain mass and size of macaques enable the evaluation of relevant clinical neuroimaging. (2) The circle of Willis in macaques is almost the same as that in humans86–89 In addition, the distribution, branch, and diameter of the middle cerebral artery (MCA) of macaques closely resemble those of humans.80,90,91 The similar cerebral vasculature makes it achievable to enter the circle of Willis and reach the MCA endovascularly, which allows for the establishment of an endovascular stroke model of MCAO and the production of the reproducible infraction. (3) Macaques have folded cortex, 92 numerous cortical neurons,93–95 a high percentage of white matter,96–98 and greater myelination 99 compared to rodents. Thus, macaque stroke models can mimic white matter susceptibility to ischemia and the disconnection of brain connectivity in stroke patients most closely to the pathology seen in humans. (4) The similar location and termination patterns of the corticospinal tract (CST) would be beneficial for evaluating the restoration of fine motor skills and voluntary movements after stroke.100,101
The neuroanatomical difference between rodents, macaques, and humans.
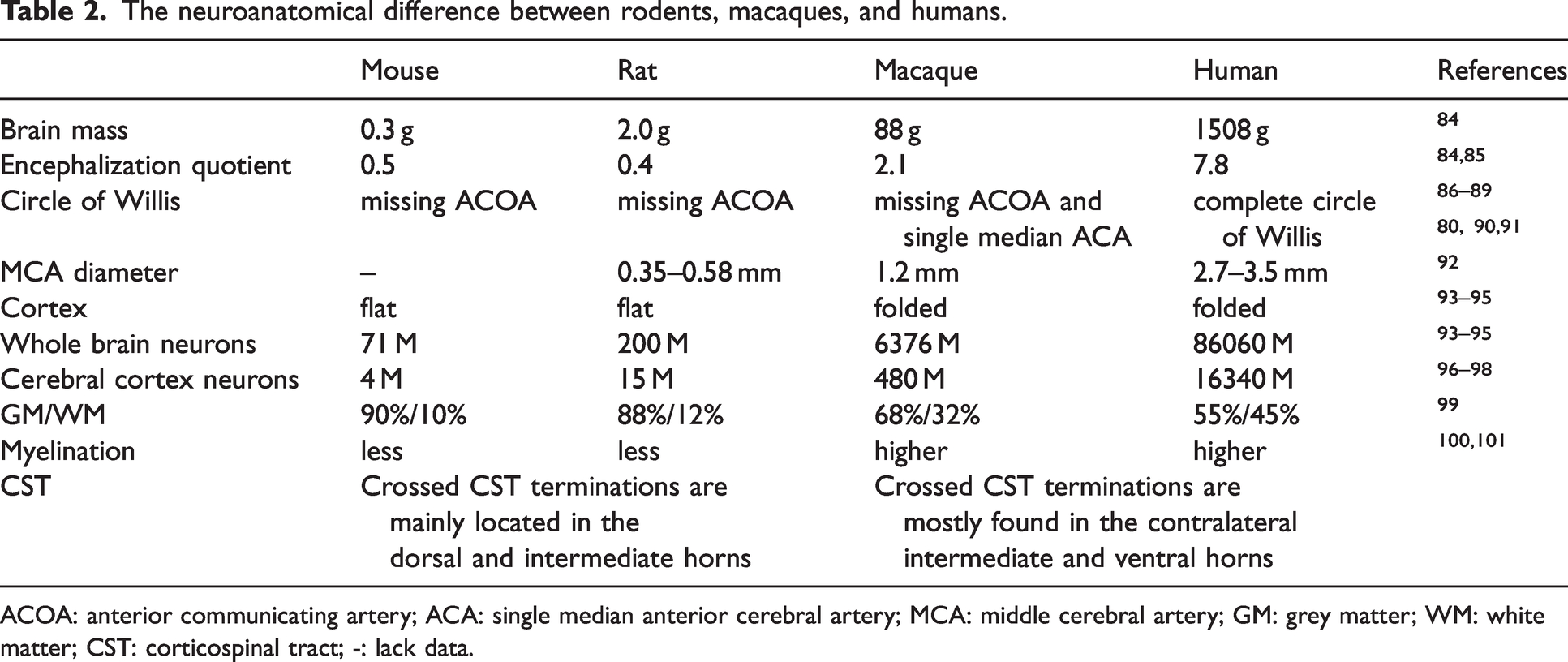
ACOA: anterior communicating artery; ACA: single median anterior cerebral artery; MCA: middle cerebral artery; GM: grey matter; WM: white matter; CST: corticospinal tract; -: lack data.
Although NHP is emerging as a unique stroke model with the above advantages, other animal species are encouraged to study the regeneration and pathophysiology of stroke. Recent studies suggested that a zebrafish is a powerful tool in stroke research because of live imaging in vivo, easy genetic manipulation, and rapid reproduction. 102 In addition, other larger animals such as canines, swine, sheep, and cats have also been reported in stroke studies. 103 These large animals are also attractive models because of similar anatomical and physiological advantages to the human brain, easy access, low cost and accessible ethical acceptance. 104 Indeed, the ethical problem is particularly important because neuronal replacement would require long-term observation in disabled animals. Given the “human-like” deficits and semi-sapience of NHP, other large species may be better alternatives to test many key aspects related to neuronal replacement approaches. NHP primate experimentation, as all in vivo experimental research, should include attendance of professional veterinarians to support and implement proper intra- and postoperative care for experimental subjects. In summary, the use of NHP models should carefully balance their usefulness against logistical, financial and ethical aspects.
Focal cortical ischemia models in NHP
Currently, MCAO models of NHP are primarily used in preclinical thrombolytic therapy and neuroprotectant test. 105 In such models, MCAO extensively injures the cortex and subcortical basal ganglion and is associated with high mortality. 106 Even if NHPs survive, they generally have severe behavioral function deficits and limited functional recovery because of the large-scale brain injury. 107 Thus, the MCAO models of NHP are not ideal for examining the neuronal regeneration and synaptic plasticity that may underlie the recovery following stroke. A relatively smaller focal injury induced in the motor cortex causing a long-term contralateral limb movement disorder is needed to explore adaptive mechanisms that regulate motor recovery in neural regeneration strategies. Ideally, a focal cortical stroke model in NHPs needs to meet three basic criteria: 1) formation of a relatively smaller infarct size in a well-defined motor cortex; 2) persistent notable motor deficits in the contralateral limb, especially dexterous hand function impairments; and 3) a long-term survival period that enables the NHP to enter the chronic phase in which neural repair occurs. Such models are useful to study neural repair and cortical plasticity, thus providing a unique opportunity for developing neural regeneration treatments. Next, we introduce various NHP models of focal ischemic lesions targeting the primary motor cortex (M1) in detail.
Nonvascular stroke model
In nonvascular models, chemicals are injected into the cortex of the M1 area of NHP to selectively destroy neuron cell bodies to mimic ischemic injury in stroke. For gyrencephalic macaque, ibotenic acid as a neurotoxin that works through activation of N-methyl-D-aspartic acid (NMDA) receptors has been applied to establish IS model. Briefly, ibotenic acid (10 µg/µl) was injected into the hand representation of macaque using a Hamilton microsyringe. 1.0 µl of ibotenic acid was injected at 3 mm depth at each injection site, and penetration sites on the cortical surface were separated by 2 mm. 108 The motor cortical lesion induced by this method severely damage reach and grasp ability and cause long-lasting hand dexterity deficits in monkey.109,110 For the lissencephalic marmoset monkey, malonate as a mitochondrial toxin causing neural energy failure and subsequent excitotoxicity is typically utilized to induce focal cortical damage. In Le Friec’s study, stereotaxic injection of 3 M malonate into the primary motor cortex causes long-lasting motor deficits of force and dexterity. 111 In summary, the above nonvascular models successfully induced a focal cortical lesion with anatomical and functional consequences comparable with ischemic stroke, while the pathophysiology obviously differs from that of stroke patients.
Electrocoagulation model
Electrocoagulation of the entire vascular bed of the target cortical tissue via microforceps has been employed to establish stroke models in lissencephalic primates with a flat cortex. The method is simple, that is, the surface vasculature over the target representation area in M1 is permanently occluded using the microforceps connected to bipolar electrocoagulation. 112 The border of the infarct could be visually defined because the ischemic tissue turns blanched, and no reperfusion is detected. This model produces an ischemic injury involving all cortical layers but spares the subcortical white matter. In 2003, Nudo et al. 77 found that the M1 lesions in such a model resulted in paralysis in the contralateral musculature of the distal forelimb and the disruption of the skilled hand use of squirrel monkey. 113 Currently, the electrocoagulation model is widely applied to study cortical plasticity and motor functional recovery post-infraction after stroke in lissencephalic monkeys.114,115 Taken together, surface electrocoagulation is easy operation, and induced reliable infarcts within a restricted functionally defined cortex. However, this method may affect the electrophysiological test and it is not suitable for macaques because their primary motor cortex with monosynaptic projections to motoneurons partly lies deep in the central sulcus. 116
Endothelin-1 (ET-1) injection model
ET-1 is a 21-amino acid peptide with potent vasoconstriction properties. 117 ET-1 injection can constrict strongly segmental vessels and reduce regional CBF within a certain period of time. Then, CBF is gradually restored as the effect fades, resulting in ischemia-reperfusion injury. 118 The indicator of a successful ET-1-induced occlusion modeling is the vasoconstriction and blanching cortex. Compared with the direct intravascular injection of ET-1 to the MCA segment, intracortical microinjection is considered more suitable for neural regenerative study because it offers the ability to specifically target a representation of the primary motor cortex.
Cortical microinjection of ET-1 has been used to induce focal infarction in NHP. Generally, the monkey was mounted on a stereotaxic apparatus. Following exposure to the motor cortex, micro-injection of ET-1 was made according to a combined MRI and histology atlas of the monkey brain in stereotaxic coordinates.119–121 Ge et al. 70 injected ET-1 (2 µg/µl) into the M1 area at six sites based on the stereotaxic atlas of the rhesus monkey brain. 10-µL Hamilton syringe needle was inserted 2–4 mm under the dura and injected five boluses of 500 nl with a distance of 500 µm in each site. They successfully established focal ischemic injury target monkey motor cortex. In Dai et al.’s study, ET-1 was intracortically injected into five sites (5 µg/10 µl/site) of a digit representation with an injection depth of 2.5–3.5 mm under the cortex surface. 122 They found that ET-1-induced ischemic injury impaired the manual dexterity of the monkey within 28 days after injury. 122 Herbert et al. 123 injected ET-1 (400 pmol/µl) into the shoulder/elbow representation in the M1 cortex. For the subjects with 10 injection sites and a 2.5–4.0 mm depth range, the infarcted tissue involved the precentral gyrus and caused mildly impaired skilled reach performance. Thus, these above studies confirmed that ET-1 microinjection allowed the creation of controlled ischemic lesions based on different doses and depths with low mortality and high repeatability. However, ET-1 may interfere with recovery mechanisms because ET-1 has been shown to induce astrocytosis and facilitates axonal sprouting.124,125
Photothrombosis model
Photothrombotic stroke is a model of superficial blood vessel occlusion by using light exposure to illuminate the photosensitizer to form a microvascular thrombus. 126 Rose bengal is the most used photosensitive dye. Following intravenous infusion and circulation in the body, it could initiate chemical reactions when irradiated by a cold light source, further causing endothelial damage, platelet activation, and thrombosis. 127 Ikeda et al. 128 applied a photothrombotic method on the lissencephalic marmoset. Specifically, following an intravenous injection of rose bengal (20 mg/kg), green light (533 nm, with 8 mm diameter) was irradiated on the intact skull-covered sensorimotor cortex for 5 min. They successfully created a cortical infraction causing obvious motor functional impairment. Zhu et al. 129 established a photothrombotic stroke model on gyrencephalic cynomolgus macaques. In surgery, the skull and cerebral dura mater were removed to expose the cortex followed by an intravenous injection of rose bengal (35 mg/kg), then cold light (SCHOTT KL 1500 LCD) was irradiated on the target cortex for 10 min. Later, Yang et al. 130 applied a green light source on both the M3 segment of the MCA and the adjacent gyrus of macaques for 20 min. Both experimental procedures cause stable focal cortical ischemic injury and contralateral forelimb paralysis in macaques. The local blood flow interruption by this method leads to cerebral edema and neural necrosis in the irradiated area, which are similar to the pathological changes in clinical stroke. The most attractive advantage of this method is the simple process and low cost, making it widely used in a primate model. Meanwhile, its disadvantage is the lack of an ischemic penumbra and the absence of collateral circulation.
Clamp model
In addition to damaging the sensorimotor cortex directly in the above methods, occlusion of the cortical branches of MCA also produces a consistent cortical infraction. The key to this method is to block the M3 or distal M2 branches of the MCA supplying cerebral blood to the M1 area exclusively and accurately. In brief, craniotomy is performed on the lateral temporal bone of the skull, and the dura mater is separated from the tissue. Subsequently, M3 in the Sylvian insular cistern is exposed via the standard microsurgical technique. Two aneurysm clips are placed about 3 mm between them, occluding the M3 segment. Finally, the clips are removed after 90 min of occlusion, and reperfusion is achieved. McEntire et al. 131 utilized this method and successfully produced a necrotic infarct localized in the sensorimotor cortex, which resulted in a significant loss of fine motor function characterized by impaired dexterity. The main advantage of this model is precise infarction with a high success rate and visual confirmation of MCAO. Additionally, this method allows subsequent reperfusion of ischemic tissue when the clip is withdrawn. The main disadvantage is the relatively large traumatic craniotomy, which brings pains to affect monkeys psychologically and physiologically. In addition, the mechanical external obstruction of MCA differs from the clinical pathophysiological processes of natural stroke.
Endovascular stroke model
To mimic the thrombotic cerebrovascular occlusion, endovascular models using interventional methods have been established. 132 Common emboli for inducing an endovascular MCAO model in NHPs include glue, 133 microbeads,134,135 microcatheters, 136 balloons,137,138 autologous blood clot, 139 microcoils, 140 and silks. 141 Notably, only thinner intraluminal devices such as microcoils and silks with sufficiently small diameters are able to block the upper trunk of the M2 even M3 segment specifically causing cortical damage. Zhao et al. 142 introduced a microcoil (1 mm × 10 cm) into the distal M2 segment to occlude the artery for 2 hours in rhesus monkeys, and produced the ischemic infarct area around the Sylvian fissure, including the upper part of Brodmann 4 and 6 areas, which correspond to the distal forelimb in the cortex of primates. In addition, delivery of multiple 5 mm segments of 4–0 silk sutures to the M3 branches of MCA also purely caused cortical strokes without deep nuclei involvement.143,144 These endovascular models are reproducible, minimally invasive, minimize pain and distress, spare the deep nuclei, and allow serial behavioral assessments in monkeys. However, this method needs high technological requirements and professional operation neurosurgeons.
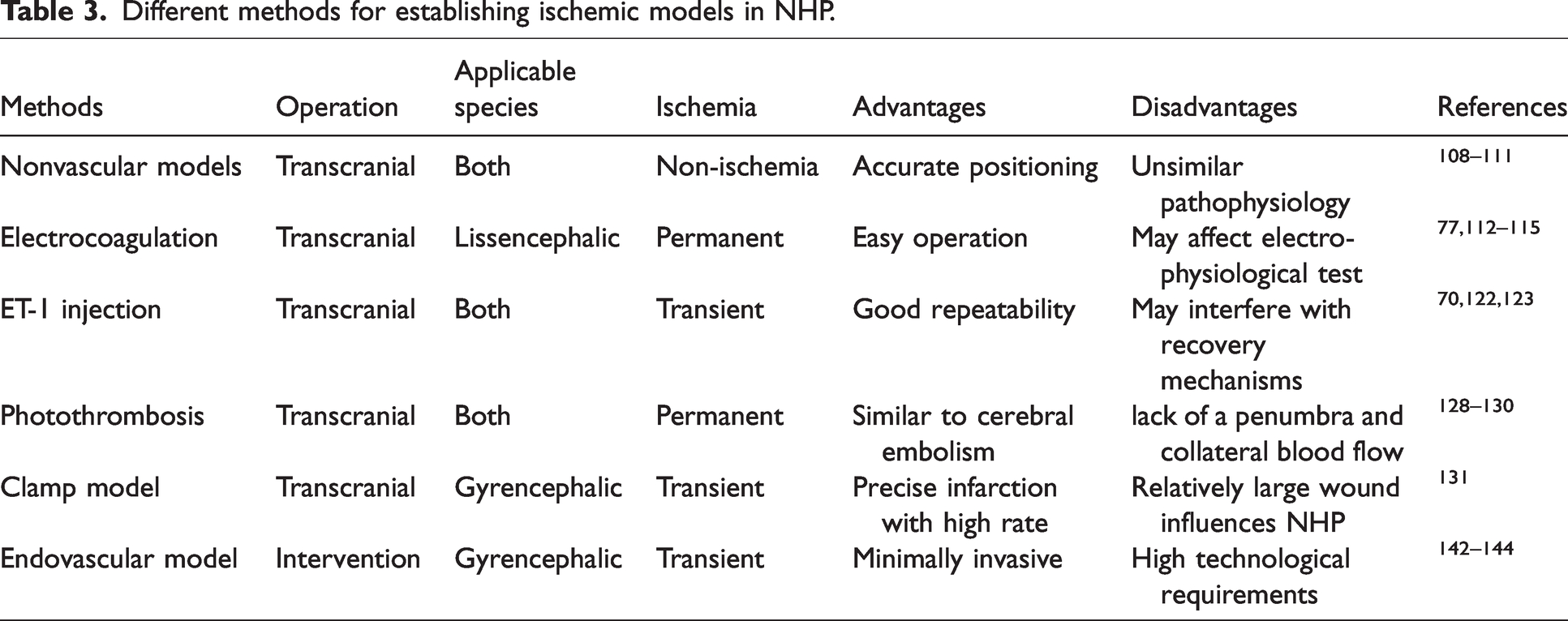
In summary, the above different methods can produce cortical ischemic injury in NHPs. These methods can be divided into transcranial and endovascular approaches in accordance with the different paths into the brain. In addition, these methods establish a permanent ischemic stroke model or an ischemia-reperfusion model on the basis of whether CBF is restored. Notably, the electrocoagulation method is only suitable for lissencephalic NHP, while the endovascular method only applies to large gyrencephalic NHP. We draw a brief schematic of all the mentioned methods (Figure 1) and describe the advantages and disadvantages of each method (Table 3). Researchers should choose the most appropriate model on the basis of the NHP species, experimental condition, and research purpose comprehensively.
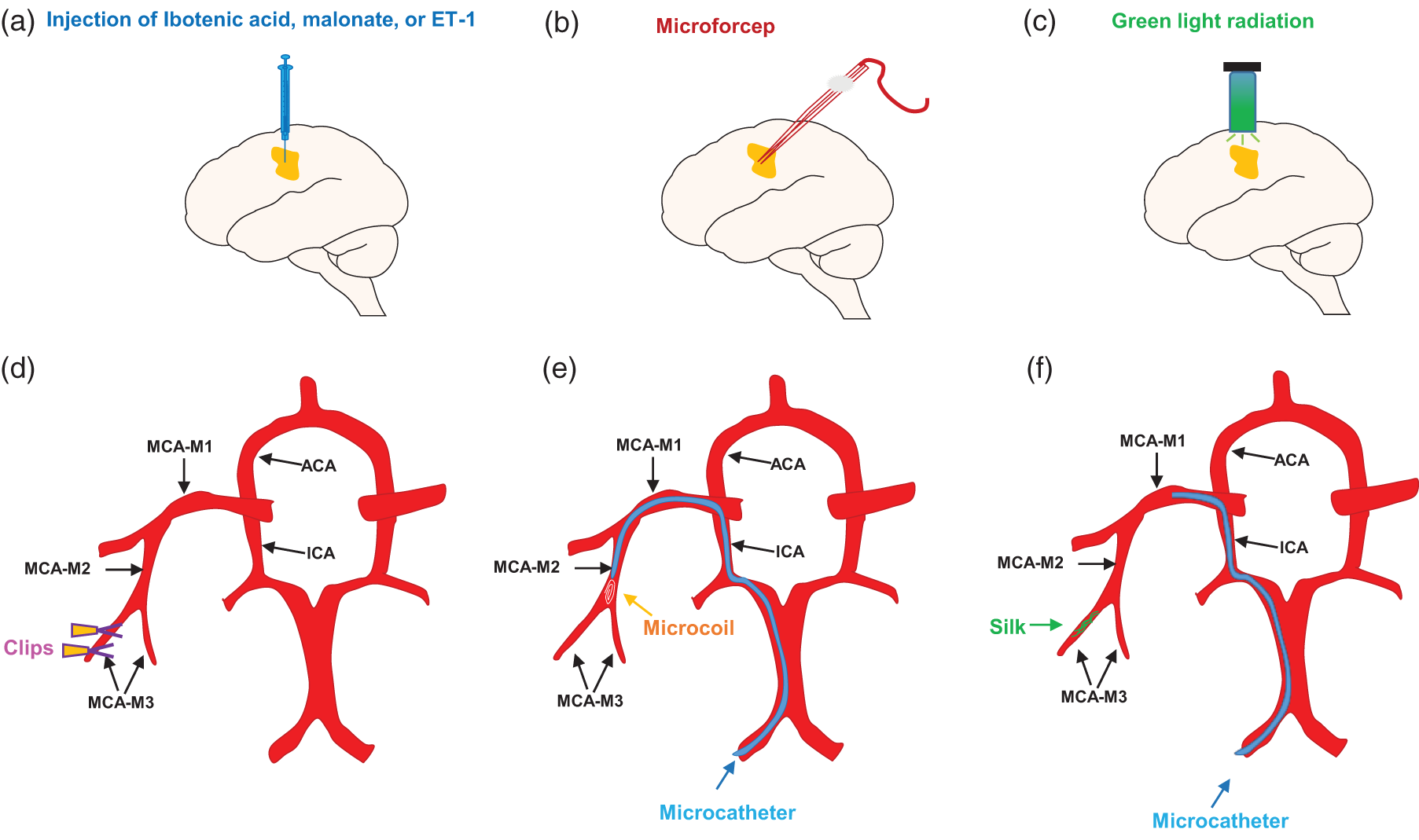
Schematic diagram of the method for establishing an ischemic stroke model targeting the primary motor cortex in NHP. (a) Nonvascular ischemic stroke model and ET-1 injection model. Chemicals (ibotenic acid or malonate) or vasoconstrictor substance (ET-1) are injected into the target cortex by using a microinjection. (b) Electrocoagulation model. Electrocoagulation of the entire vascular bed of the target cortical tissue is conducted using microforceps. (c) Photothrombosis model. After intravenous injection of rose bengal, photoirradiation is performed on the target cortex or the M3 segment of the MCA. (d) Clamp model. Clips are used to clamp the M3 segment of the MCA. (e) Endovascular stroke model. MicroCoils are placed after entering the upper trunk of the M2 segment through the microcatheter and (f) Endovascular stroke model. Silk sutures are placed after entering the M3 segment through a catheter.
Different methods for establishing ischemic models in NHP.
Outcome measures
In the preclinical stroke study of regenerative therapy in NHP, objective measurement of infraction volume, CBF, and neurobehavioral outcomes is an initial step to judge the validity of interventions. In addition, further evidence of the long-term survival of newborn neurons, synaptic integration in host networks, and functional circuitry repair is essential for mechanistic investigation. Through the tests of neuroimaging, histology, electrophysiology, optogenetics, chemical genetics, single-cell sequencing, virus tracing, and behavior examination, we obtain a clinically and experimentally accessible neurological evaluation to improve the clinical translation of neuronal replacement therapy.
Infarction volume
Stroke patients with larger infarcts generally have worse neurological deficits, 145 so infarction volume measurement is essential to predict functional outcomes. At 24 h after ischemic injury in NHP, infarction volume could be detected by pathological and neuroimaging examination. Nissl's staining solution reacts with the Nissl body in neurons, then clearly discerns the necrotic neurons in the infraction of NHP.146,147 Hematoxylin–eosin (H&E) staining could reflect not only neural death but also inflammatory infiltration and glial proliferation after stroke. 148 2, 3, 5-Triphenyltetrazolium chloride (TTC) staining is the most common pathological method for evaluating infarction volume. Nevertheless, TTC staining is generally not applicable to the assessment of infarctions in the chronic stage of stroke.149,150
Imaging evaluation is also important for assessing infarction volume in NNP stroke. Structural magnetic resonance imaging (MRI) visually shows the size of the ischemic infarction and the volume involved. Ramirez-Garcia et al. 151 used MRI to dynamically monitor the infarction size after occlusion of MCA and found that the infarction size correlated with the behavioral sore after stroke in cynomolgus macaques. However, MRI sometimes fails to show the infarct areas in the acute stage (within 4.5 h). By contrast, diffusion weighted imaging (DWI) can reflect the ischemic size and location within minutes after stroke by a quick examination of rapid changes in intracellular and extracellular water contents. Cooke et al. 134 demonstrated that DWI hyperintensities correspond to histological infarctions stained with H&E in cynomolgus macaques. In addition, diffusion tensor imaging (DTI) is used to visualize the conductive fibers in the neurofunctional region. Chin et al. 152 used DTI and found a significant decrease in fractional anisotropy in the internal capsule of a monkey after 7 days of stroke. Collectively, neuroimaging evaluation is an important non-invasive tool to dynamically monitor the infarction volume of NHPs in vivo.
Regional CBF
Imaging evaluation methods are required to examine changes in vascular occlusion and to confirm the success of cerebral ischemia models during surgery. Transcranial Doppler ultrasound (TCD), 153 computed tomography angiography (CTA), 135 digital subtraction angiography (DSA), 154 and magnetic resonance angiography (MRA) 155 arterial spin labeling (ASL) 156 are commonly used to clearly show the interruption or refusion in blood flow during operation in NHP stroke study. To further reflect ischemic changes in brain tissue after the operation, positron emission tomography (PET) is commonly used to monitor CBF, the cerebral metabolic rate of oxygen (CMRO2), oxygen extraction fraction, and local cerebral blood volume. Takamatsu et al. 157 conducted PET studies and detected the neuroprotective effects of FK-506 only in areas with >40% CMRO2 reduction at 3 h after reperfusion in cynomolgus monkeys. Perfusion weighted imaging (PWI) also reliably reflects cerebral perfusion through the measurement of relative CBF, relative cerebral blood volume (CBV), and mean transit time. Yi et al. 158 analyzed CBV/CBF maps, which demonstrated a diffusion–perfusion match after the stroke of rhesus macaques. Taken together, there are various available imaging methods for evaluating vascular occlusion and cerebral infarction in NHP. Although high spatial and temporal resolution, expensive testing costs limit its applications to some degree in NHP studies.
Neurobehavioral assessment
Outcome measurements in NHP stroke studies generally focus on neurological functions, including motor, sensory, and cognitive performance. Correspondingly, a series of NHP stroke scales are used to evaluate neurobehavioral outcomes. 106 However, these neurological scales lack a unified standard and rely on the expertise of the examiner. To quantify reparative functional outcomes and improve clinical translation, these scales need to be supplemented by behavioral assessments. Thus, hill and valley, two-tube, and six-well tasks are required to test the limb mobility and space exploration ability of NHP. 159 Additionally, the food reward retrieval tests, such as Klüver board, Brinkman board, and vertical slot tasks are used to evaluate the digit grasping ability.122,140
The dexterous hand functional test is especially important in cortical ischemic injury targeting M1. In the acute stage of stroke, NHP animals spent a longer time retrieving food and showed decreased successful retrievals. However, spontaneous recovery of manual dexterity improved the scores in the behavioral tasks and even recovered to preoperative levels in the chronic stage. 160 In this regard, researchers generally recorded behavioral tasks using a digital video camera for kinematic analyses by calculating detailed indexes, such as the flexion of the index finger, coordinates of the tip of the thumb, joint angle, operation time, and grasping forms. 161 Murata et al. 108 analyzed digit movements frame by frame and found that the behavioral indexes of manual dexterity remained lower until months after M1 lesions. The damaged monkeys frequently used alternate grip strategies to grasp a small object with the affected hand. Thus, comprehensive analysis of stroke scales and behavioral test especially fine manual performance is helpful for researchers to track the functional recovery after stroke after NHP receive neural replacement therapy.
Neural regeneration and repair
Indeed, any neural replacement strategies need to demonstrate that the behavioral recovery is due to the substitution of dying ischemic neurons by newborn neurons, that is providing a causative link between neuronal replacement and behavior improvement. Thus, the newborn neural long-survival, subtype, activity, synaptic formation and functional connectivity with the host are needed to study. First, stroke-induced activation of NSCs and proliferation of neural progenitor cells (IPC) should be detected using mitotic indicators such as 5-bromodeoxyuridine (BrdU) by immunochemistry.9,162 Second, newborn neurons should survive and fully mature into the neuronal subtype lost with the pathology and restructure the site-specific phenotypes and tissue architecture. Single-cell transcriptomics is an advanced technique for identifying neurogenic-related cell types and differentiation trajectories.8,10 Third, electrophysiological recording,163–165 calcium imaging,166,167 and functional magnetic resonance imaging (fMRI) 168 allow monitoring of adult-born neuronal activity. Fourth, electron microscopy 169 and quantitative analysis of synaptophysin 170 are needed to identify synaptogenesis. Monosynaptic virus tracing and MED64 system are necessary to confirm synaptic contacts between host cells and newborn neurons.171,172 Fifth, newborn neurons should functionally integrate into the host brain circuitry. Transcranial magnetic stimulation could noninvasively test the cortical motor function by recording the motor evoked potential (MEP) following the magnetic stimulus delivered over the primary motor cortex. 173 Finally, optogenetic and chemogenetic technology that selectively silences or activates newborn neurons allows for assessing the contribution of regenerative neurons to functional recovery.174–176 We draw a brief schematic of all the mentioned methods applied in the neural replacement study (Figure 2) and summarize the advantages and disadvantages of each technological approach (Table 4).
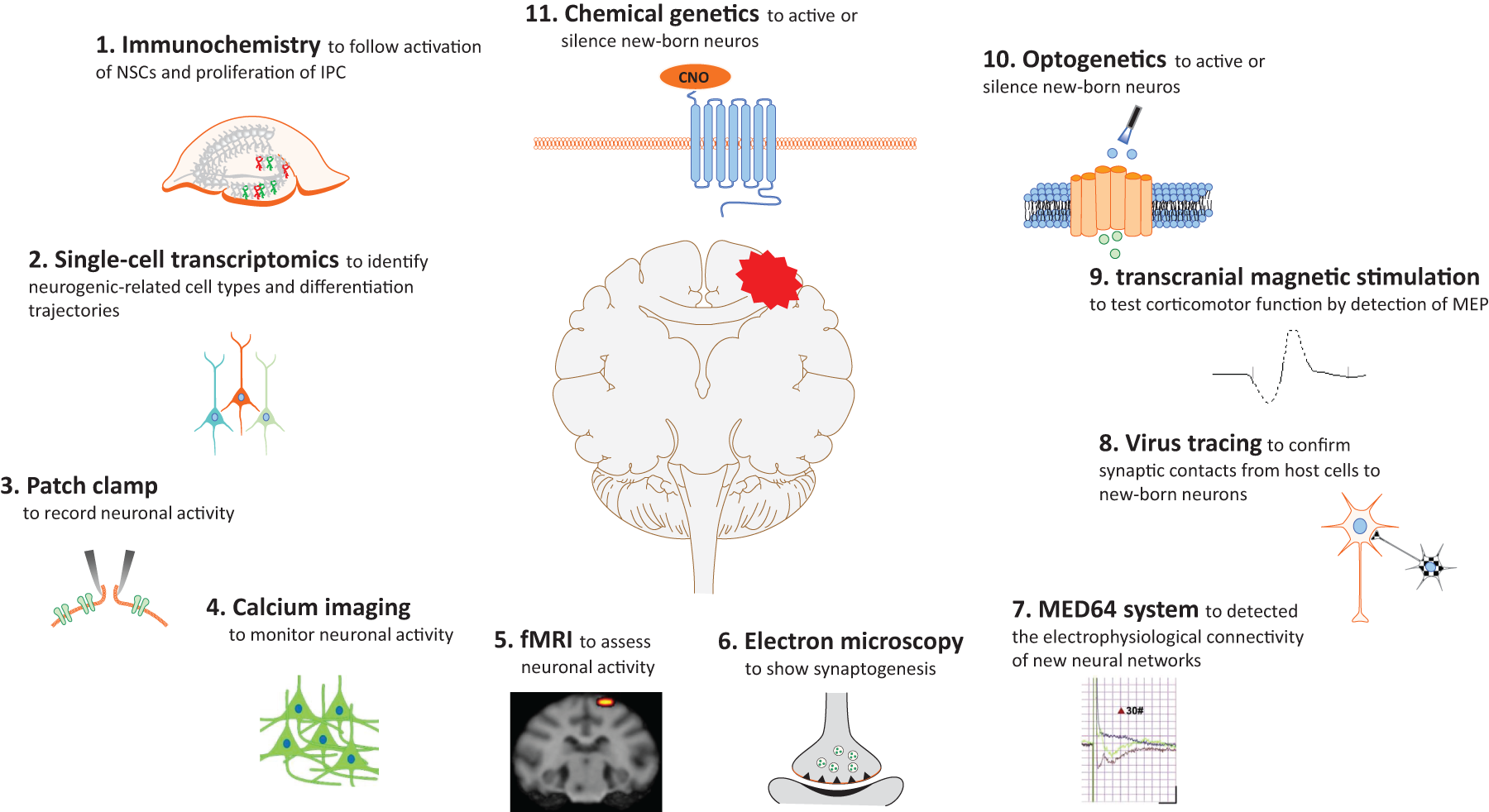
Schematic summary of the technological approaches used to study neural regeneration in neuronal replacement strategies. Combination of “traditional” techniques, such as immunochemistry, electron microscopy, clinical imaging, and electrophysiological recording, and new technological tools, such as single-cell transcriptomics, virus tracing, calcium imaging, photogenetics, and chemical genetics, are needed to study neural subtype, activity, and functional connectivity with the host. (Element 5 reprinted from figure 5 in “NT3-chitosan enables de novo regeneration and functional recovery in monkeys after spinal cord injury” by Jiasheng Rao et al.).
The methods, applications, advantages and disadvantages of the technological approaches used to study neural replacement.

NSCs: neural stem cells; IPCs: neural progenitor cells; NHP: non-human primate; RMP: resting membrane potential; Rinput: membrane input resistance; Cm: membrane capacitance; AP: action potential; sEPSC: spontaneous excitatory postsynaptic currents; mEPSCs: miniature excitatory postsynaptic currents; LPP: lateral perforant path; fMRI: functional magnetic resonance imaging; BOLD: blood oxygenation level-dependent; fEPSPs: field excitatory post-synaptic potential; BDA: biotinylated dextran amine; MEP: motorevoked potential; DREADDs: designer receptors exclusively activated by designer drugs; GPCRs: G-protein-coupled receptors; CNO: clozapine-N-oxid.
Conclusions
For designing NHP preclinical stroke studies of neuronal replacement strategies, we draw a program flowchart to address three major questions: What NHP species should be selected? Which focal ischemic model should be established? How to measure functional recovery and the underlying mechanism (Figure 3)? NHP is a link between basic research and clinical application because NHP closely resembles humans. The NHP stroke models that damage the cortex targeting M1 are more suitable for the study of neurological deficits caused by long-term cerebral infarction. The researchers should choose the most appropriate NHP stroke models by analyzing the advantages and disadvantages of each model.
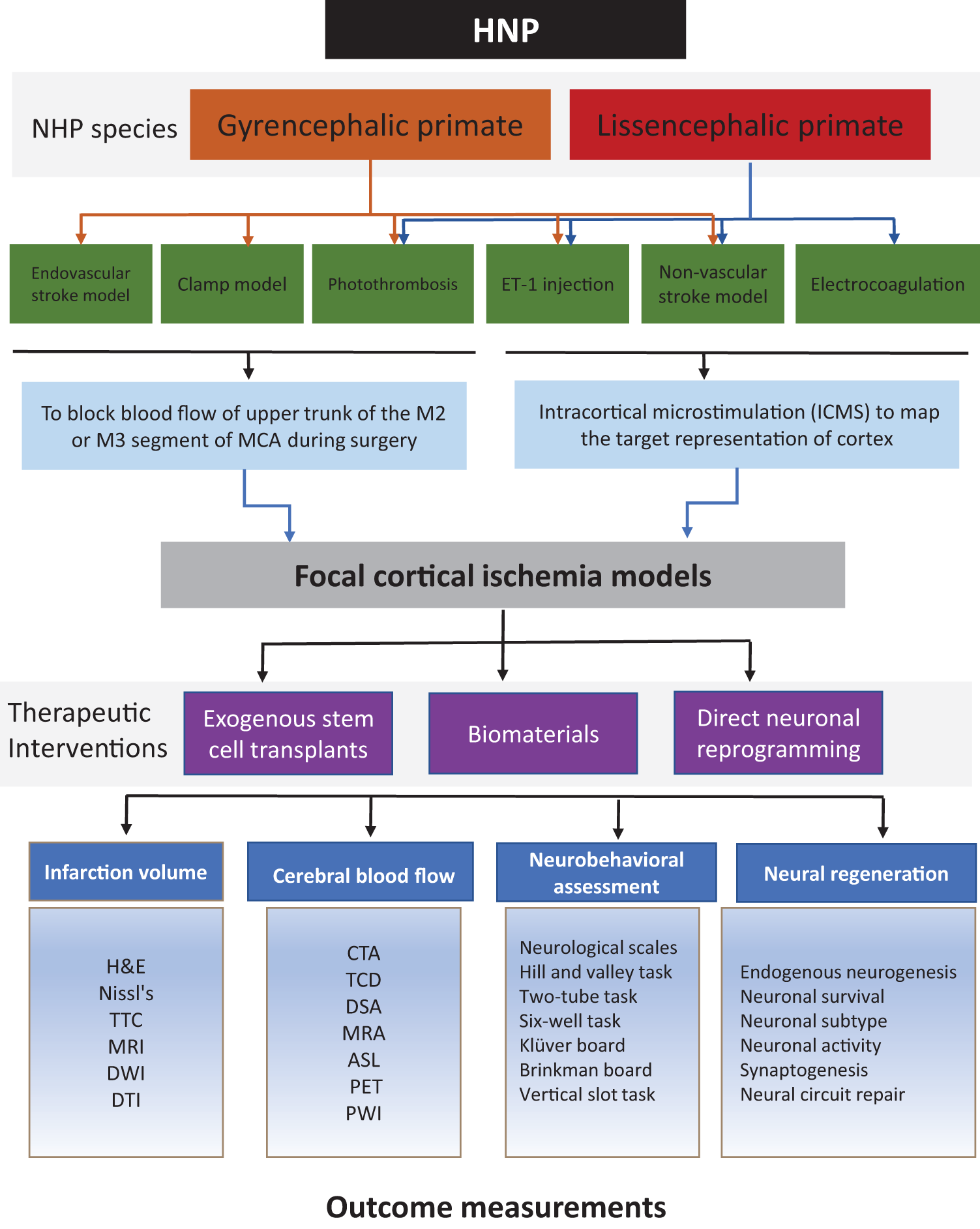
Flowchart describing the most relevant aspects to consider when designing an NHP stroke model in the preclinical study of neuronal replacement strategies. NHP species, stroke model, and outcome measurements are the three major aspects needed to address.
Stroke study in NHP pose significant challenges, including specialized infrastructure, strict ethical considerations, high imaging cost, strict tissue fixation procedures, complex neurobehavioral assessment, and limited application of NHP specific tracer and antibodies. In our view, NHP stroke studies of neuronal replacement should only be undertaken for promising therapies that have been fully tested in rodent species and other lower species. To better enhance the predictive power of NHP in preclinical stroke study, the multi-center preclinical trials by collaborative groups, and more funding from industry support or publicly programs will likely be required. Work of the past decade fueled by technical advances, such as single-cell sequencing, electrophysiology, virus tracing, and photogenetics, would further amplify our knowledge of neuronal circuitry and diversity. In addition, behavioral evaluation and chemical genetic analysis are essential to better quantify a high level of specificity when newborn neurons participate in behavioral recovery following stroke.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (Grants 31730030, 31971279, 82272171, 82271403, 82201542, 81941011, 31771053 and 31900749), Beijing Natural Science Foundation (Grant 7222004), and The Key Research and Development Program of Guangzhou (Grant 202206060003).
Acknowledgements
We would like to thank Jiasheng Rao (School of Engineering Medicine, Beihang University, China), and the anonymous reviewers for providing useful comments on the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
