Abstract
Cerebral ischemia stimulates endogenous neurogenesis within the subventricular zone and the hippocampal dentate gyrus of the adult rodent brain. However, such newly generated cells soon die after cerebral ischemia. To enhance postischemic survival of neural precursor cells (NPC) and long-lasting neural regeneration, we applied the antiapoptotic chaperone heat shock protein 70 (Hsp70) fused to a cell-penetrating peptide derived from the HIV TAT to ensure delivery across the blood-brain barrier and the cell membrane. After transient focal cerebral ischemia in mice, TAT-Hsp70 was intravenously injected concomitant with reperfusion and additionally on day 14 after stroke. TAT-Hsp70 treatment resulted in smaller infarct size (27.1 ± 9.0 versus 109.0 ± 14.0 and 88.5 ± 26.0 mm3 in controls) and in functional improvement as assessed by the rota rod, tight rope, and water maze tests when compared with saline- and TAT-hemagglutinin-treated controls. In addition, postischemic survival of endogenous doublecortin (Dcx)-positive NPC was improved within the lesioned striatum of TAT-Hsp70-treated animals for up to 4 weeks after stroke without changing overall cell proliferation of BrdU+ cells. Thus, TAT-Hsp70 treatment after stroke may be a promising tool to act neuroprotective and improve postischemic functional outcome, and also to increase survival of endogenous NPC after stroke.
Introduction
In the adult mammalian brain, limited endogenous neurogenesis is observed in the subgranular zone of the hippocampal dentate gyrus and in the subventricular zone (SVZ) (Gross, 2000; Taupin and Gage, 2002). Here, astrocyte-like neural stem cells function as primary neural precursor cells (NPC) that eventually give rise to neuroblasts, which migrate through the rostral migratory stream toward the olfactory bulb and differentiate into mature neurons (Alvarez-Buylla and Garcia-Verdugo, 2002; Doetsch et al, 1999).
Various pathologic conditions, such as cerebral ischemia, are able to stimulate endogenous neurogenesis resulting in neuronal differentiation of proliferating 5-bromo-2′-deoxyuridine-5′-monophosphate (BrdU)-positive cells (Arvidsson et al, 2002; Li et al, 2002; Liu et al, 1998; Zhang et al, 2001). Transient focal cerebral ischemia increased the number of SVZ-derived BrdU+ cells followed by lesion-directed migration and subsequent coexpression of early and mature neuronal markers (Arvidsson et al, 2002; Yamashita et al, 2006).
Although a postischemic increase of SVZ-derived BrdU+ cells and their subsequent neuronal differentiation has recently been shown by several groups, a proof of functional relevance is still missing (Arvidsson et al, 2001; Li et al, 2002; Zhang et al, 2001). Because the majority of these cells die within several weeks after stroke, the extent of postischemic neuronal replacement by newborn cells seems to be limited (Haas et al, 2005; Parent, 2003). Different neuroprotective strategies, such as growth factor application, temporarily lead to an increase in postischemic generation of NPC, but did not result in sustained cell survival after stroke (Wada et al, 2003; Wang et al, 2004).
Proteins of the heat shock protein 70 (Hsp70) family may serve as a valuable tool to enhance postischemic cell survival of NPC. Postischemic neuroprotection by these proteins is attributed to chaperone activity as well as to inhibition of necrosis, alteration of intrinsic and extrinsic apoptotic pathways, such as the inhibition of cytochrome c release, and modulation of inflammation (Giffard et al, 2008). However, blood-brain barrier transition and cellular entry of Hsp70-like proteins is very limited. So far, Hsp70-mediated neuroprotection after stroke has only been shown by protein overexpression in transgenic mice, by transfection or viral-vector-mediated overexpression (Badin et al, 2006; Hoehn et al, 2001; Rajdev et al, 2000; Zheng et al, 2008). For effective blood-brain barrier transition and cellular entry after systemic injection in experimental stroke, Hsp70 was fused to the basic domain derived from the human immunodeficiency virus TAT protein (Nagel et al, 2008a, b ; Wheeler et al, 2003). In the study described here, TAT-Hsp70-mediated effects on infarct volume and functional outcome, as well as on lesion-specific cell proliferation and survival of NPC, were analyzed after systemic application in transient focal cerebral ischemia in mice.
Materials and methods
Expression and Purification of Hsp70 and Tat Fusion Proteins
To create an expression vector for the TAT-Hsp70 fusion protein, rat hsp70.1 cDNA was cloned into the pTAT-hemagglutinin (HA) expression vector (kindly provided by SF Dowdy, University of California in San Diego, CA, USA) as described by Nagel et al (2008a). pTAT-HA was derived from pRSET B (Invitrogen GmbH, Karlsruhe, Germany). TAT-Hsp70 and TAT-HA were expressed in Escherichia coli strain BL21 (DE3)pLysS (Novagen, San Diego, USA) and isolated in their native conformations in a 10 mmol/L Tris (pH 10), 20% glycerol, 274 mmol/L NaCl, 0.1% Pluronic, 0.02% Tween 80 buffer as described previously (Nagel et al, 2008b). Anti-HA antibodies were purchased from Covance Inc, San Diego, USA. TAT-Hsp70 was dialyzed against five changes of a 250-fold buffer solution (274 mmol/L NaCl, 10 mmol/L Tris, pH 10) before application. Protein purity was assessed by Coomassie dye (Coomassie Brilliant blue; Merck, Darmstadt, Germany)-stained SDS gels. The protein concentration was quantified by comparison with protein standards, loaded on Coomassie-stained polyacrylamide gels.
Experimental Groups
All experimental procedures were performed according to the EU guidelines for the care and use of laboratory animals and approved by local authorities. For all experiments adult male C57Bl/6N (Charles River, Sulzfeld, Germany) mice weighing 22 to 27 g were used and assigned to seven groups consisting of 7 to 8 animals each. Animals were killed on day 4 (group I to III) and on day 28 (IV to VII) after induction of transient focal cerebral ischemia. Starting with reperfusion, saline (I), 1 nmol of TAT-HA (negative control; II, IV, and VI) or 1 nmol of TAT-Hsp70 (III, V, and VII) was injected intravenously in a volume of 100 μL. Animals of groups VI and VII received a second injection on day 14. All animals received intraperitoneal BrdU injections (Sigma, Taufkirchen, Germany) on days 0 to 3 (I to III) or 8 to 18 (IV to VII; daily dose: 50 mg/kg bodyweight). Mortality rate was 0% for group I to III, 17.6% for group IV to V, and 11.2% for group VI to VII.
Induction of Focal Cerebral Ischemia
Cerebral ischemia was induced by transient occlusion of the middle cerebral artery (MCA) using the intraluminal filament technique as described previously (Weise et al, 2006). Briefly, animals were anesthetized (0.8% to 1.5% isofluran, 30.0% O2, remainder N2O) and rectal temperature was maintained at 36.5°C to 37.0°C using a feedback-controlled heating system. The laser-Doppler flow was recorded using a flexible fiber optic probe (Perimed, Stockholm, Sweden), which was attached to the skull overlying the core region of the MCA territory to assess cerebral blood flow (CBF). After a midline neck incision the left common and external carotid arteries were isolated and ligated. After placing a microvascular clip (Aesculap, Tuttlingen, Germany) on the internal carotid artery, a 7–0 silicon-coated nylon monofilament (Doccol, Redlands, USA) was introduced through an incision into the distal part of the common carotid artery and, after removing the clip, advanced 9 mm distal from the carotid bifurcation for MCA occlusion. After 45 mins, the thread was removed to allow reperfusion of the MCA. The laser-Doppler flow recordings continued for > 15 mins after thread removal to monitor appropriate reperfusion (> 80% of initial CBF).
Immunohistochemistry
Animals received intraperitoneal injection of chloral hydrate (420 mg/kg body weight) and were transcardially perfused with 4% paraformaldehyde at day 4 or 28 after ischemia. Brains were removed, postfixed in paraformaldehyde, and paraffin embedded before 2 ím coronal sections were prepared. Sections were deparaffmized, boiled in 0.2% citrate buffer, and incubated with blocking solution (0.3% Triton X-100; 10% normal goat serum in phosphate-buffered saline; 1 h, room temperature (RT)). For analysis of BrdU+ cells, we exposed sections to blocking solution and subsequently stained with a rat monoclonal anti-BrdU antibody (1:50, 18 h, 4°C; Serotec, Düsseldorf, Germany) and a goat anti-rat Alexa 594 secondary antibody (1:500, 1 h; Molecular Probes, Karlsruhe, Germany). To analyze the differentiation of BrdU+ cells, we incubated sections with a mouse monoclonal anti-BrdU antibody (1:500; Roche, Grenzach, Germany) and a goat polyclonal anti-doublecortin antibody (1:50; Santa Cruz Biotechnology, Heidelberg, Germany), a rat polyclonal anti-GFAP antibody (1:500; Zymed, Karlsruhe, Germany), or a rabbit polyclonal anti-NG2 antibody (1:250; Chemicon, Watford, UK) for 18 h at 4°C. For double staining against BrdU and NeuN, CNPase, or Nestin, a rat monoclonal anti-BrdU antibody (see above) and a mouse monoclonal anti-NeuN antibody (1:200, 18 h, 4°C; Chemicon, Watford, UK), a mouse monoclonal anti-CNPase antibody (1:400; Chemicon, Schwalbach, Germany), or a mouse monoclonal anti-Nestin antibody (1:500; Chemicon, Schwalbach, Germany) were used. After washing, the sections were incubated for 1 h at RT with secondary antibodies. The following secondary antibodies were used: goat anti-mouse Cy-3 (1:400; Dianova, Hamburg, Germany) or goat anti-rat Alexa 594 (1:400; Dianova, Hamburg, Germany) for BrdU staining, goat anti-rat Alexa 488 (1:250; Molecular Probes) or donkey antigoat Alexa 488 (1:250; Molecular Probes) for glial fibrillary acidic protein (GFAP) or doublecortin (Dcx) staining, goat anti-mouse Cy-3 (1:100; Jackson Immuno-Research, Newmarket, Suffolk, UK) for CNPase staining, goat antirabbit Alexa 488 (1:250; Molecular Probes) for NG2 staining, and goat anti-mouse Alexa 488 (Molecular Probes, Karlsruhe, Germany) for NeuN (1:400) and Nestin (1:250) staining. Terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick-end labeling (TUNEL) staining was performed incubating sections with proteinase K (7 mins, 37°C), followed by exposure to the terminal deoxynucleotidyl transferase enzyme reaction according to the manufacturer's manual (Roche). After several washing steps and exposure to TUNEL blocking solution (20mins, RT), sections were stained with a streptavidin-Alexa594-conjugated secondary antibody (2h, RT; Molecular Probes, Karlsruhe, Germany). For analysis of TAT-Hsp70-transduced cells 24 h after ischemia, deparaffinized sections were stained with a mouse monoclonal antibody (1:5, 2h, 37°C; Covance, Munich, Germany) directed against the HA sequence of TAT-Hsp70 followed by incubation with a goat anti-mouse Cy-3-coupled secondary antibody (1:400, 1 h, RT; Dianova, Hamburg, Germany). The number of HA+ cells was counted both in ischemic and in nonischemic hemispheres defining regions of interest within the basal ganglia (BG) and subsequent calculation of HA+ cells per square millimeter.
Quantitative Analysis of Infarct Volume and Postischemic Cell Proliferation
For infarct size determination animals were killed at days 4 and 28, respectively. Brains were removed and cut into four slices of 2 mm each. Slices were stained with 2,3,5-triphenyltetrazolium chloride (2%), and computer-based analysis of infarct volumes was performed using the software ImageJ by subtracting the area of the nonlesioned ipsilateral hemisphere from that of the contralateral side. Infarct volume sizes were calculated by integration of the lesioned areas. Quantitative analyses were performed defining regions of interest within the SVZ and the BG. Stereotactic coordinates for quantitative analysis in the SVZ were 0.14 mm anterior, 2 to 3 mm ventral, and 1 to 1.25 mm lateral from bregma. Cell count analysis within the BG was performed at 0.14 mm anterior, 2.5 to 3.25 mm ventral, and 1.5 to 2.25 mm lateral from bregma. Given the different densities of labeled cells in the structures, we performed cell counts in areas of 0.25 mm2 (BrdU staining) or 0.15 mm2 (TUNEL staining). Subsequently, the number of cells counted was recalculated for an area of 1 mm2 (means ± s.e.m.). Two to three sections per animal and regions of interest were used. Cell death (TUNEL) was assessed within ischemic BG. The number of proliferating cells (BrdU) was determined within the BG and the SVZ.
Quantitative Analysis of Neuronal Dcx+ Precursor Cells
Because Dcx+ cells tend to form chain-like aggregates within ischemic BG, quantification was performed analyzing complete ischemic BG (0.14 mm anterior, 2.0 to 4.5 mm ventral, 1.0 to 3.0 mm lateral from bregma) and the SVZ (see above) in two sections per animal at day 28 after ischemia. Entire BG and SVZ were photographed and Dcx+ cells were quantified using Image Analysis software (AxioVision, Zeiss, Jena, Germany). Numbers of Dcx+ cells are given as cells per section analyzed (means ± s.e.m.).
Behavioral Analysis
To study TAT-Hsp70-mediated effects on motor coordination and spatial learning strategies, we tested the animals on the rota rod, the tight rope, and in the water maze test. For assessment of motor coordination (rota rod and tight rope tests), animals were trained 1 day before experimental cerebral ischemia; testing days were on day 4, 7, 10, 14, 21, and 28 after ischemia. The rota rod test was performed using an accelerating treadmill for mice (TSE Systems, Bad Homburg, Germany; 3 cm diameter). Mice were placed on the rotating drum with an accelerating speed of 4g to 40g Maximum speed was achieved after 260 secs, and maximum testing time was 300 secs. The time until animals dropped was registered and analyzed for all animals and time points tested. Using the tight rope test, we placed animals on a 60 cm long tight rope grasping the string with their forepaws. Healthy animals grasp the string with four legs and tail and move to reach a side pole, whereas mice having ischemia-induced motor deficits cannot lift their hind legs and eventually fall off onto the cage bedding placed underneath. All animals were tested twice per time point. Maximum test time was 60 secs and results were scored from 0 (min) to 20 (max) according to a validated score described earlier depending on the time animals spent on the rope and whether they reached the platform (Gerber et al, 2001). Scores were used for statistical analysis.
The water maze test was performed using a swimming pool (diameter: 100 cm; height: 30 cm) filled to a depth of 15 cm with water kept at RT (20°C). A transparent plexiglass platform (11 × 11 cm) was submerged with the top located 1 cm below the surface. Animals of groups IV to VII were tested on days 26, 27, and 28 after ischemia. A full experiment consisted of 20 trials; 4 trials performed in the morning of days 26, 27, and 28 and 4 trials in the afternoon on days 26 and 27 (maximum of 90 secs per trial, intertrial interval: 1 min). For these experiments the platform was always located in the center of the same quadrant and animals were always put into the same of one of the remaining quadrants. If an animal did not reach the platform within 90 secs, it was guided to the platform and allowed to stay there for 30 secs. For assessment of new learning strategies (task switch) the platform was set into another quadrant, animals were put into the same quadrant as before, and four trials were performed (see above) in the morning of day 28. The time needed to reach the platform was statistically analyzed using a computer-based system (TSE Systems).
Statistics
All data are given as mean ± s.e.m. For comparison between two groups the two-sided Student's t-test was used. For comparison between more than two groups one-way analysis of variance followed by the Tukey's post hoc test was performed. A P-value < 0.05 was considered statistically significant.
Results
TAT-Mediated Cerebral Tissue Transduction
Intravenous injection of TAT-Hsp70 resulted in high numbers of HA+ cells within the ischemic and nonischemic hemispheres of treated animals, suggesting TAT-Hsp70 to permeate sufficiently both the blood-brain barrier and the cell membranes (Figure 1). No significant differences in the number of HA+ cells in ischemic and nonischemic tissues were observed (673.0 ± 76.0 versus 769.0 ± 99.0 per mm2).
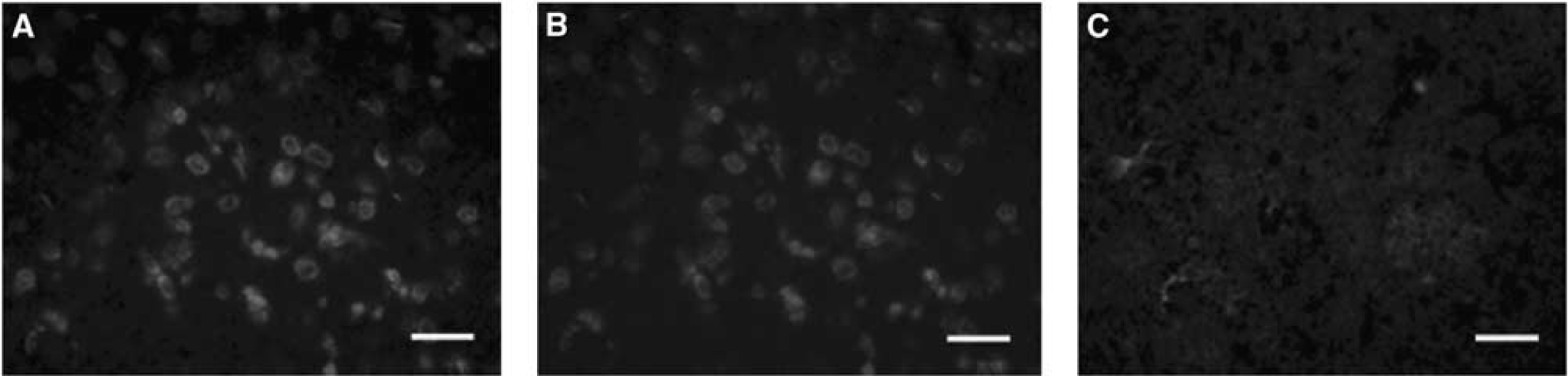
TAT-Hsp70-mediated tissue transduction. Intrastriatal detection of the hemagglutinin (HA) sequence of TAT-Hsp70 24 h after cerebral ischemia and systemic administration of TAT-Hsp70 (
TAT-Hsp70 Reduces Cerebral Infarct Volume and Neural Cell Injury
Quantitative analysis of 2,3,5-triphenyltetrazolium chloride-stained tissues (Figure 2A) on day 4 after experimental stroke showed significant infarct size reduction (P<0.05) in TAT-Hsp70-treated animals (27.1 ± 9.0mm3) when compared with controls either treated with saline (109.0 ± 14.0 mm3) or TAT-HA (88.5 ± 26.0 mm3). As observed on day 4, after a 28-day survival period infarct volumes in TAT-Hsp70-treated animals were significantly smaller (P<0.05) when compared with TAT-HA- and saline-treated controls (42.0 ± 17.0 versus 96.7 ± 20.0 mm3). A second systemic injection of TAT-Hsp70 on day 14 after cerebral ischemia did not lead to a further reduction of infarct volumes compared with TAT-HA and saline control animals (55.7 ± 21.0 versus 106.5 ± 6.0 and 96.0 ± 9.0 mm3). Along with TAT-Hsp70-mediated reduction of infarct size administration of TAT-Hsp70 also significantly decreased the number of TUNEL+ cells in ischemic BG and the periinfarct zone compared with control animals (Figure 2B). These observations did not coincide with changes in CBF as animals of all treatment paradigms showed a ∼100% reperfusion rate within the first 15 mins of reperfusion (Figure 3). Because of a lack of difference in both infarct sizes and numbers of TUNEL+ cells of TAT-HA- and saline-treated animals, only TAT-HA-treated mice were used as (negative) controls in further experiments.
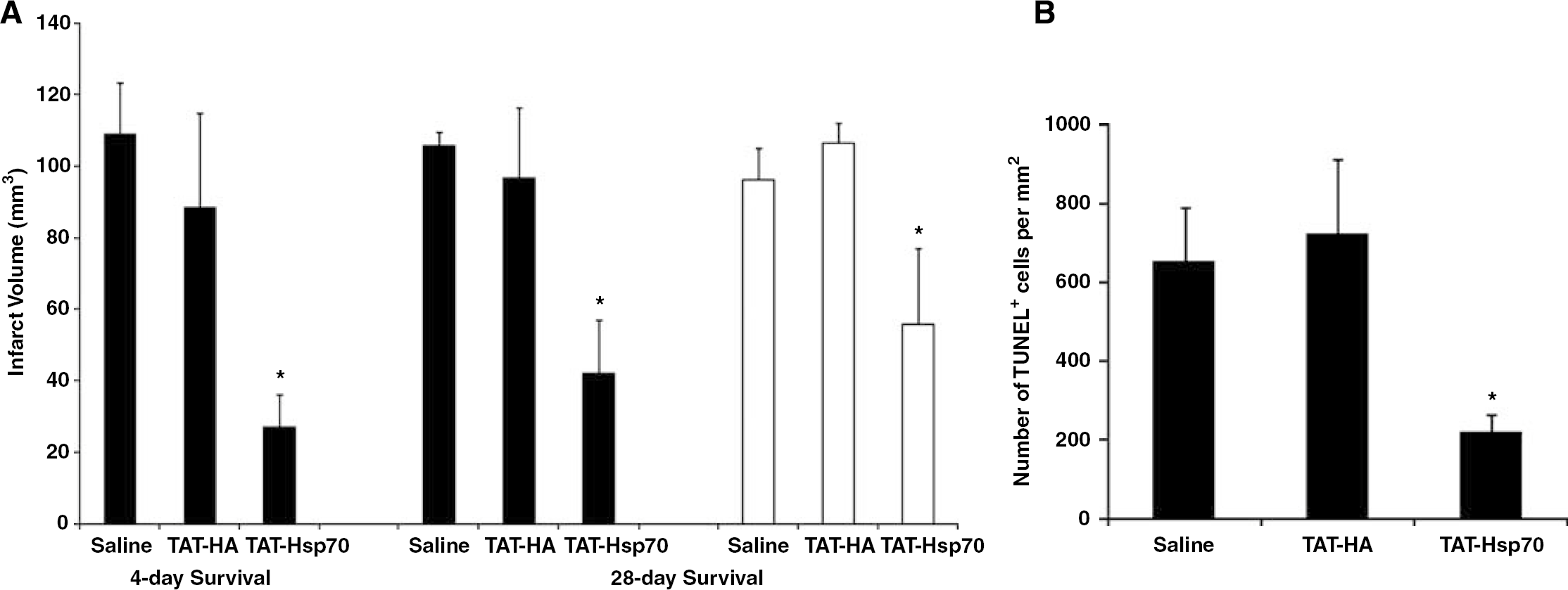
TAT-Hsp70-mediated reduction of infarct volume and neuroprotection. (
Recordings of cerebral blood flow (CBF). Laser-Doppler flow was continuously recorded during induction of focal cerebral ischemia and subsequent intravenous drug injection until at least 15 mins after the beginning of reperfusion. (
Systemic Application of TAT-Hsp70 Reduces Functional Deficits After Cerebral Ischemia
Analysis of postischemic motor coordination deficits was performed by means of the rota rod and the tight rope tests on days 4, 7, 10, 14, 21, and 28 after cerebral ischemia. Although all animals showed better test performances over time in both the rota rod and the tight rope tests, TAT-Hsp70-treated animals always performed significantly (P<0.05) better than controls (Figures 4A and 4B). However, there was no significant difference between animals treated with one or two injections of TAT-Hsp70.
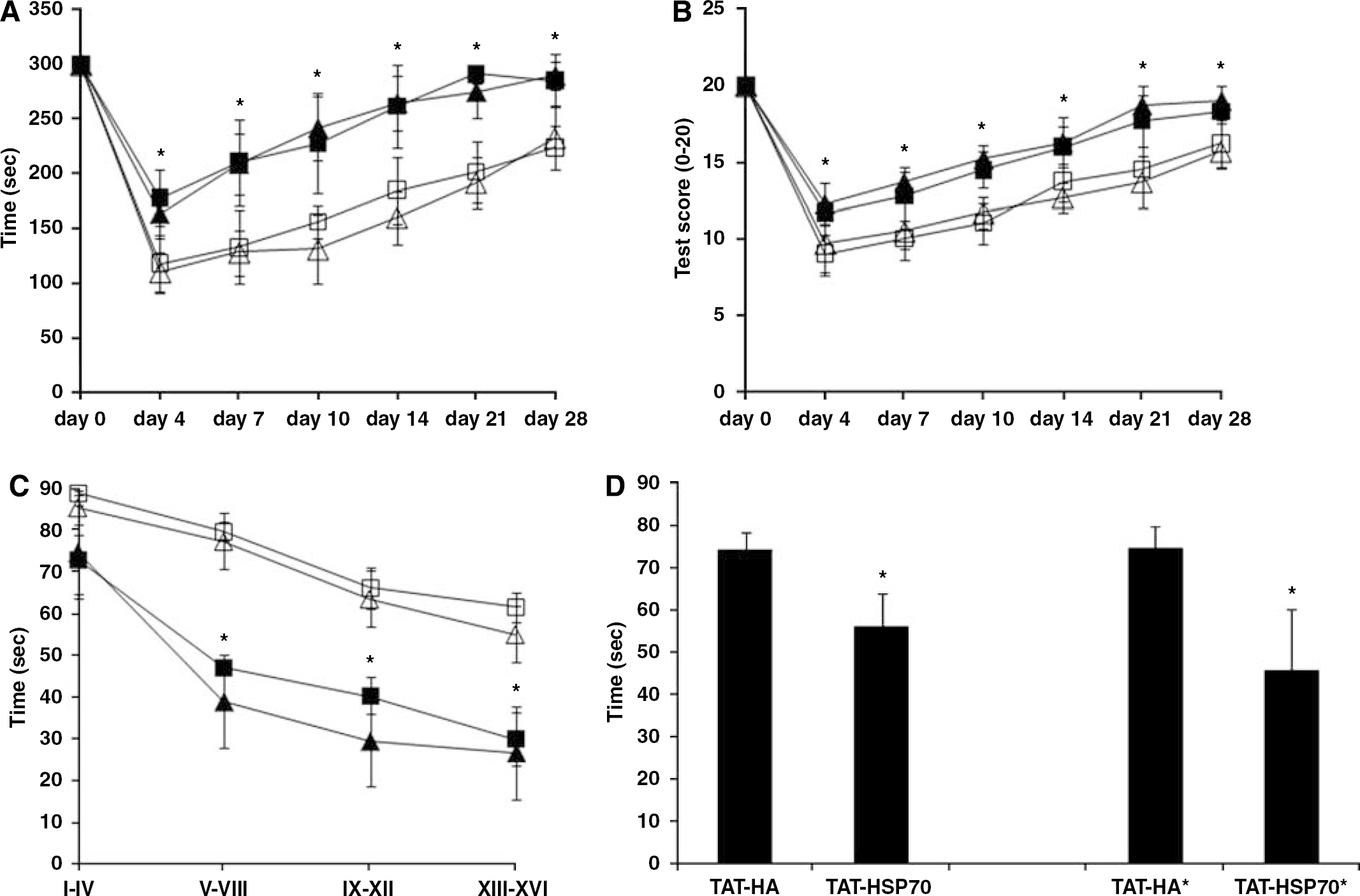
TAT-Hsp70 treatment decreases postischemic loss of motor function and improves spatial learning and memory capabilities. Rota rod (
For the modified water maze test the time necessary to reach the platform constantly decreased with the number of trials in both the control and the treatment groups suggesting that animals from all four groups were able to learn and memorize (Figure 4C). However, TAT-Hsp70-treated animals from both groups (i.e., single and double injection) were significantly (P<0.05) faster in reaching the platform than control animals. Although a change in platform position (task switch) initially reincreased the time needed to reach the platform in all animal groups, TAT-Hsp70-treated animals were significantly faster (P<0.05) in reaching the platform than TAT-HA-treated animals (Figure 4D).
TAT-Hsp70-Mediated Neuroprotection Does not Alter Postischemic Cell Proliferation
Endogenous postischemic cell proliferation was analyzed using BrdU staining. TAT-Hsp70 treatment did not affect the number of BrdU+ proliferating cells in both the left/ipsilateral SVZ and the (ischemic) BG, neither on day 4 (data not shown) nor on day 28, regardless of the number of TAT-Hsp70 injections (Figure 5). This suggests that TAT-Hsp70 is neuroprotective (Figure 2) rather than affecting ischemic cell proliferation.
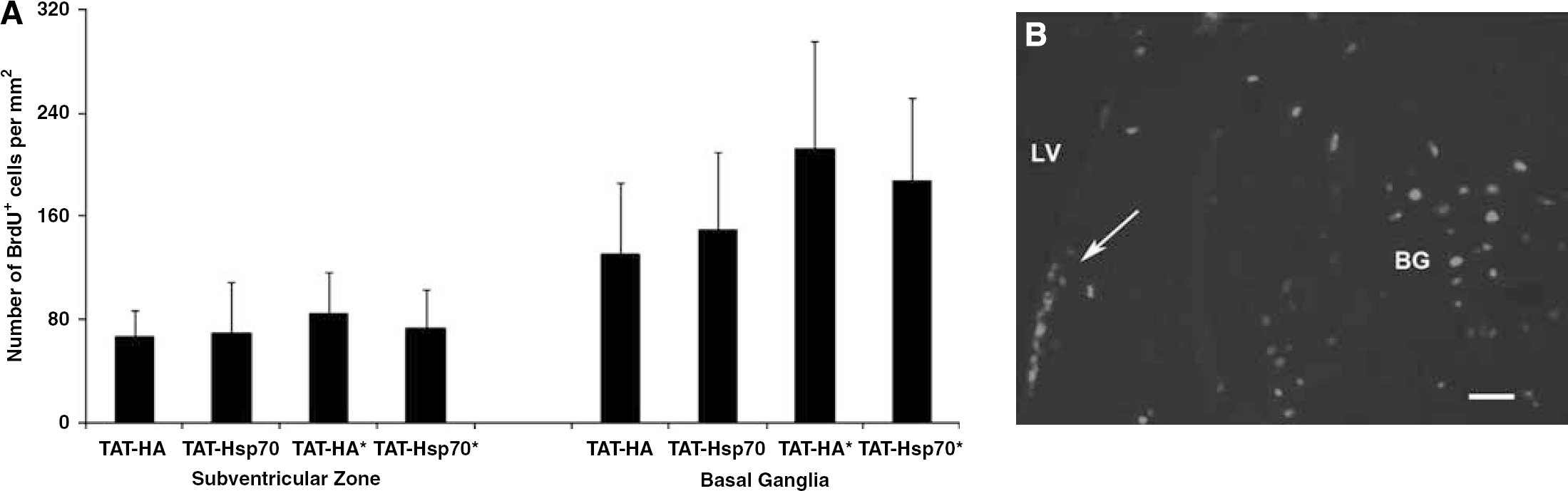
TAT-Hsp70 treatment does not affect postischemic cell proliferation. (
Increased Number of Neural Precursor Cells Within the Lesion Site After TAT-Hsp70 Treatment
Repeated injections of TAT-Hsp70, that is, injections on days 0 and 14, yielded a significantly (P<0.05) higher number of Dcx+ NPC than in controls within the ischemic BG at day 28 after cerebral ischemia (Figure 6A). Doublecortin-positive cells in TAT-Hsp70-treated animals were typically organized in clusters forming chain-like aggregates (Figures 6B and 6C). On the contrary, single injection of TAT-Hsp70 at day 0, at the beginning of the reperfusion, did not alter the number of Dcx+ cells significantly (Figure 6A). The number of Dcx+ NPC within the ipsilateral SVZ after TAT-Hsp70 treatment (days 0 and 14) was 37.0 ± 8.3 and therefore not significantly different from TAT-HA controls (34.0 ± 11.3). Doublecortin-positive cells were always restricted to the ischemic hemisphere (except for the contralateral SVZ) and could not be detected in the nonischemic BG (data not shown). We did not observe any Dcx+ cells within the lesion site on day 4 after cerebral ischemia in any experimental group (data not shown). These data suggest TAT-Hsp70-mediated improved survival of NPC within the lesion site after experimental ischemia.
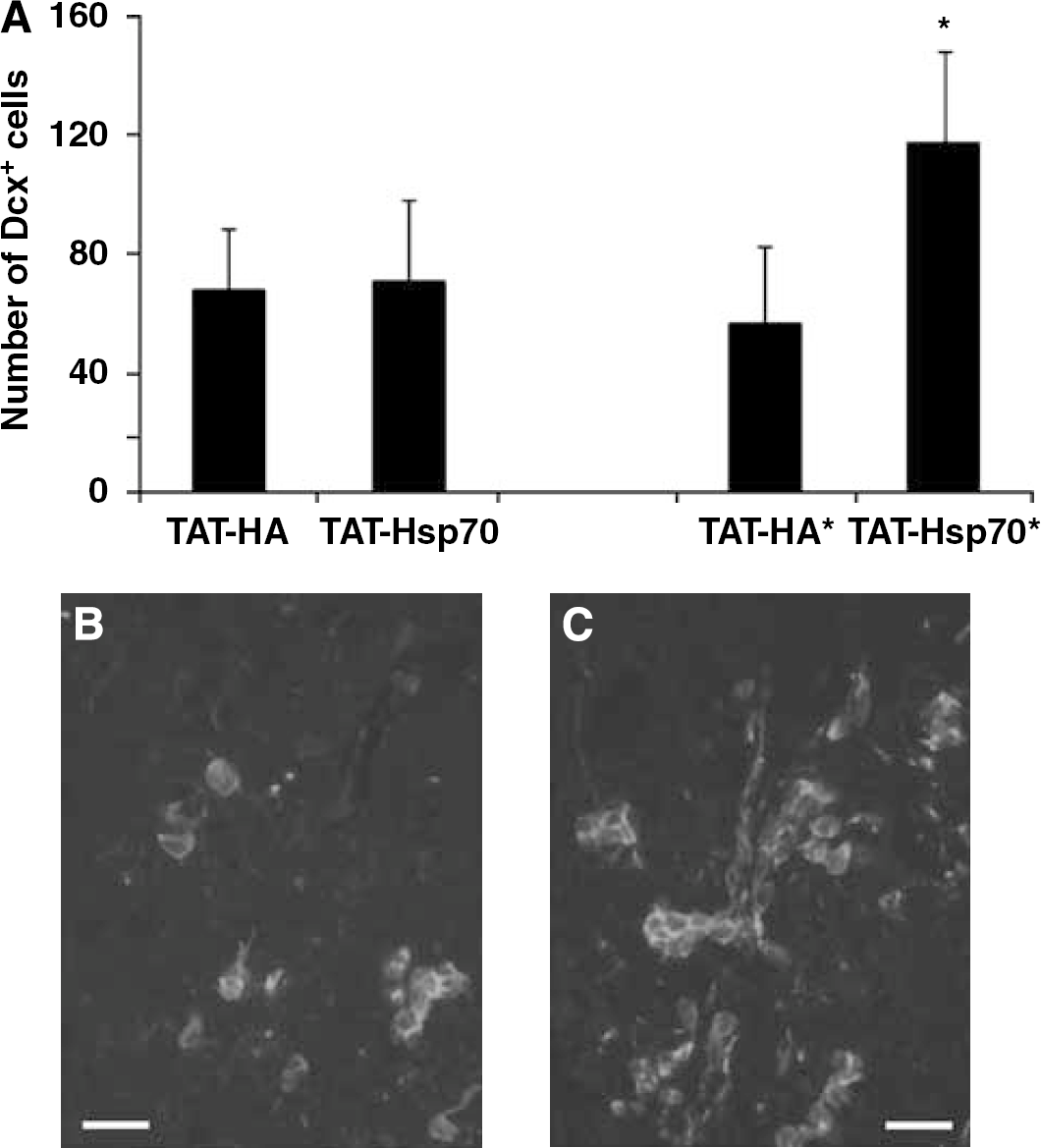
Increased survival of Dcx+ neuronal precursor cells in postischemic basal ganglia after TAT-Hsp70 treatment. (
Postischemic Differentiation of BrdU+ Proliferating Cells
Although the number of BrdU+ proliferating cells after cerebral ischemia was not changed by TAT-Hsp70 (Figure 5A), TAT-Hsp70 treatment yielded a significantly higher number of BrdU+ /Dcx+ cells at day 28 after ischemia (Figures 7A and 7D). Repeated application of TAT-Hsp70 (days 0 and 14) resulted in 11.1% ± 5% BrdU+ /Dcx+ cells compared with 2.6% ± 1.2% cells in TAT-HA-treated animals within the lesion site at day 28 after cerebral ischemia. However, no colocalization of BrdU+ cells with the mature neuronal marker NeuN was observed (data not shown). Regarding astrocytic differentiation of BrdU+ cells 28 days after cerebral ischemia, no significant difference in the relative number of BrdU+/GFAP+ cells after repeated injection of TAT-Hsp70 (7.9% ± 4.1%) was observed as compared with TAT-HA-treated animals (5.9% ± 3.5%; Figures 7B and 7E). No colocalization with the oligodendritic markers NG2 and CNPase was observed, suggesting a lack of relevant oligodendritic differentiation of newly generated BrdU+ proliferating cells (data not shown). Staining against the neural marker Nestin only revealed a few scattered cells within the ischemic BG showing no significant difference between the treatment groups (Figures 7C and 7F).
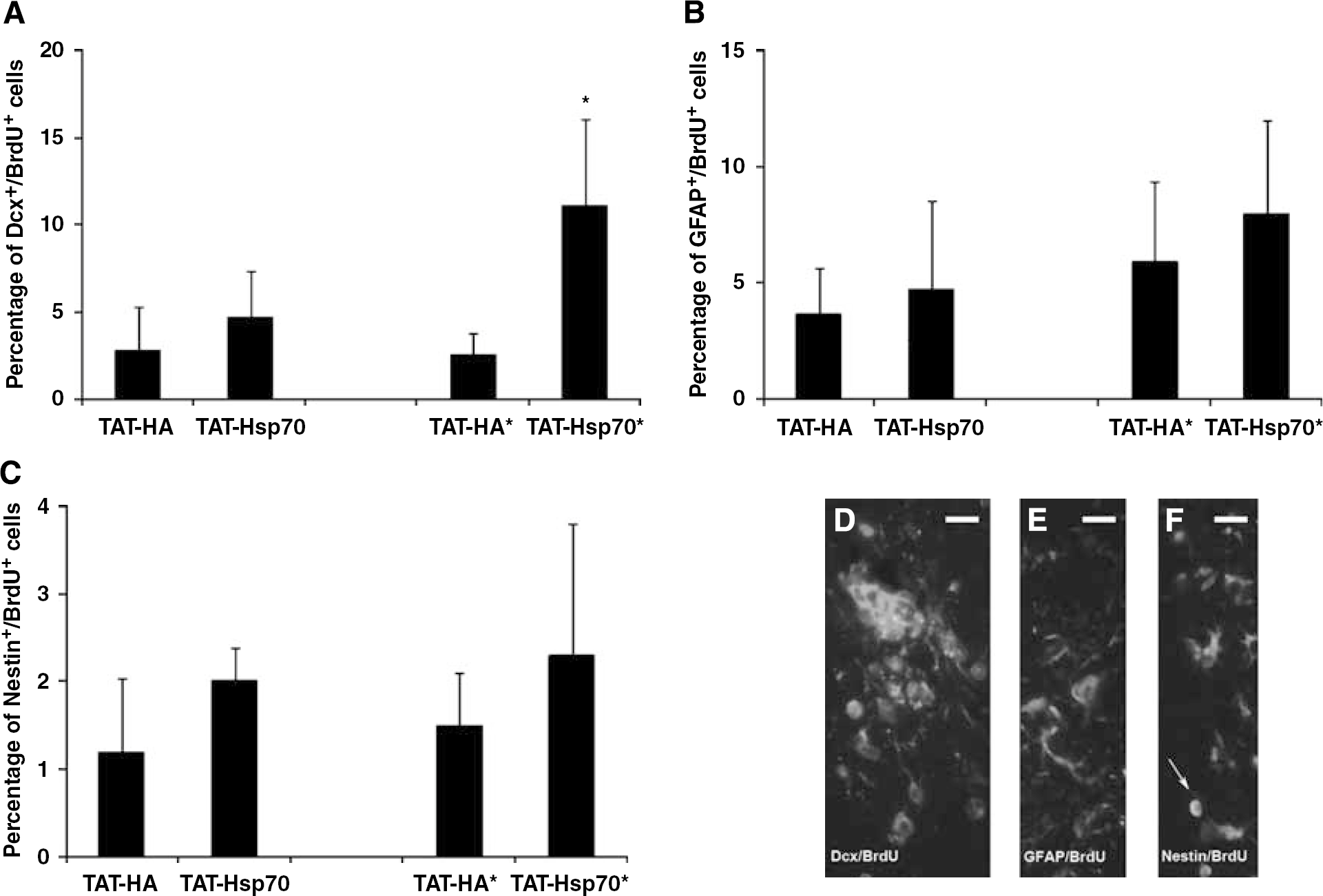
TAT-Hsp70 treatment increases the number of Dcx+/BrdU+ cells in postischemic basal ganglia. Quantitative analysis of BrdU+/Dcx+ cells (
Discussion
In this study, TAT-Hsp70-mediated effects on infarct volume, functional outcome, cell proliferation, and survival of endogenous NPC after transient focal cerebral ischemia in mice were analyzed. In line with Nagel et al (2008a), who studied TAT-Hsp70-mediated effects in an in vivo model of Parkinson's disease, the transduction rate after systemic application of TAT-Hsp70 was high in both ischemic and nonischemic hemispheres (Figure 1). So far, Hsp70-mediated postischemic neuroprotection has only been described after protein overexpression within 2 days after the onset of cerebral ischemia (Badin et al, 2006; Hoehn et al, 2001; Sun et al, 2006; Zheng et al, 2008). Therefore, we chose a different ‘therapeutic’ approach by systemic application of TAT-Hsp70 during reperfusion (and on day 14) and analyzed infarct volumes on days 4 and 28 after experimental stroke. When compared with controls infarct size in TAT-Hsp70-treated animals (single as well as repeated administration) was significantly reduced at both time points (Figure 2A). In line with TAT-Hsp70-mediated reduction of infarct volume, fusion protein treatment also yielded a direct neuroprotective effect shown by a significantly reduced number of TUNEL+ cells (Figure 2B). These effects, however, were independent of CBF values as animals in all treatment groups showed an ∼100% reperfusion rate within the first 15 mins after thread removal (Figure 3).
Although overexpression of Hsp70 with subsequent short-term neuroprotection after experimental stroke has been shown before (see above), its functional relevance has not been studied sufficiently yet, and postischemic analysis of functional deficits is limited because of simplified stroke scales (Hoehn et al, 2001; Sun et al, 2006; Zheng et al, 2008). Thus, we studied postischemic functional deficits in motor coordination using the rota rod and the tight rope tests, both of which have proved to be sensitive in detecting sensorimotor functional deficits after experimental stroke in mice (Gerlai et al, 2000). Both groups of animals receiving either a single or a repeated injection of TAT-Hsp70 performed significantly better in the two tests when compared with controls (Figures 4A and 4B). Although animals from all experimental groups showed better test performance over time, at the time points analyzed, TAT-Hsp70-treated animals significantly scored better, which was also associated with smaller infarct size assessed on days 4 and 28. The improvement in motor function over time in all animals, as shown by better test results with increasing survival time, can probably be attributed to the high rate of spontaneous recovery of rodents after cerebral ischemia (Nygren and Wieloch, 2005; Winter et al, 2004). Thus, reduced loss of motor coordination deficits in TAT-Hsp70-treated animals may be associated with TAT-Hsp70-mediated neuroprotection.
As cerebral ischemia does not only affect motor coordination but also learning and memory capabilities of rodents (Bendel et al, 2005), we additionally exposed the animals to a modified water maze test. All animals receiving TAT-Hsp70 treatment, both single or repeated administration, reached the platform significantly faster when tested on days 26 and 27 after ischemia, and they achieved better test scores in the task switch on day 28 (Figures 4C and 4D) when compared with TAT-HA-treated controls. Most likely, this is because of TAT-Hsp70-mediated neuroprotection on days 4 and 28. However, one has to keep in mind that complex cognitive functions such as learning and memory do not simply depend on infarct size alone, but also on neuronal circuits between different structures like the hippocampus or the mammillary bodies, which are usually not affected by a 45 mins occlusion of the MCA (Vann and Aggleton, 2004). Therefore, the precise neuroanatomic basis for a better water maze test performance of TAT-Hsp70-treated animals remains elusive and further studies with emphasis on analysis of TAT-Hsp70-mediated effects on hippocampal neurons are needed.
Consistent with other reports (Arvidsson et al, 2002; Jin et al, 2001), focal cerebral ischemia induced endogenous cell proliferation and increased the number of NPC, labeled by BrdU and Dcx. Postischemic survival of Dcx+ NPC within the lesion site of animals repeatedly treated with TAT-Hsp70 after ischemia was significantly higher when compared with TAT-HA-treated controls (Figure 6). Thus, systemic application of TAT-Hsp70 may be an interesting ‘therapeutic’ approach to improve limited survival of NPC (Haas et al, 2005; Parent, 2003). However, single injection of TAT-Hsp70 at the beginning of the reperfusion did not alter the number of Dcx+ cells in the lesion site. The latter result might be because of a ‘delayed’ increase of endogenous neurogenesis after stroke; we did not observe any Dcx+ cells within the site of lesion at day 4 after induction of cerebral ischemia (own unpublished observation). Therefore, Dcx+ cells analyzed on day 28 within the ischemic BG must have been generated beyond day 4. We hypothesize that these late-born cells may therefore profit from a second antiapoptotic treatment on day 14 after stroke. In line with these results, animals receiving repeated injections of TAT-Hsp70 showed a higher percentage of BrdU+ cells expressing Dcx (Figures 7A and 7D); the cause of which is rather a TAT-Hsp70-mediated enhanced survival of NPC within the lesion, than an increased neuronal differentiation of BrdU+ proliferating cells. As a matter of fact, TAT-Hsp70 treatment did not alter the survival of Dcx+ NPC within the SVZ after cerebral ischemia (own unpublished observation). According to this, we did not observe any colocalization of BrdU+ cells with the neuronal marker NeuN in the lesion site at 4 weeks after cerebral ischemia (unpublished observation), arguing against a TAT-Hsp70-mediated enhanced neuronal differentiation. However, survival periods beyond 4 weeks after stroke are necessary to assess whether TAT-Hsp70-mediated increased survival of NPC eventually leads to mature neurons within the lesion site.
In contrast to a higher percentage of BrdU+/Dcx+ cells in TAT-Hsp70-treated animals, TAT-Hsp70 did not affect the total number of proliferating BrdU+ cells after stroke (Figure 5), which contradicts a TAT-Hsp70-mediated effect on postischemic cell proliferation. Nevertheless, the overall number of BrdU+ cells may not be equal to the number of proliferating NPC within the lesion site, as the thymidine analogue BrdU is integrated during cell proliferation as well as during DNA repair (Taupin, 2007). In comparison to TAT-Hsp70-mediated neuroprotection, enhanced cell injury in controls is associated with increased DNA repair, resulting in higher BrdU uptake, thereby masking a probable difference in the number of ‘genuinely’ proliferating cells between both groups.
In summary, systemic TAT-Hsp70 treatment after focal cerebral ischemia is neuroprotective resulting in reduced infarct size and less motor coordination and cognitive deficits. In addition, the survival of newly generated Dcx+ NPC within the lesion site is enhanced after TAT-Hsp70 treatment, indicating that this treatment approach may simultaneously affect different pathophysiological cascades and thereby provide an interesting versatile tool for the development of new neuroprotective therapies in stroke.
Footnotes
Acknowledgements
We thank Barbara Müller and Birgit Dietz for excellent technical assistance.
The authors declare that they have no competing financial interest.
