Abstract
The common marmoset (Callithrix jacchus), a New World monkey, has recently been used as a model of focal cerebral ischaemia. Here, we sought to develop a stroke model in this species using an intraluminal approach to occlude the middle cerebral artery (MCA). This technically simple procedure allows both transient and permanent ischaemia with minimal morbidity. Ten common marmosets underwent either transient (3 h) or permanent ischaemia by the insertion of a nylon filament through the external carotid artery up to the origin of the MCA. Cerebral blood flow (CBF) was monitored by the laser-Doppler flowmetry technique. Sensorimotor functions were regularly evaluated, and histologic, immunohistochemical, and magnetic resonance imaging analyses were performed 8 days after the occlusion. The surgical procedure was achieved straightforwardly without postoperative mortality or cerebral haemorrhage. All animals displayed a consistent decrease in CBF that remained stable over 3 h. Infarction affected both cortical and subcortical structures. Although not statistically significant, the volume of infarction was smaller in marmosets subjected to transient ischaemia compared to those permanently occluded (237±139 and 358±118 mm3, respectively). In all the behavioural tests used, reperfused marmosets exhibited fewer neurologic and functional impairments compared to permanently occluded ones. We show the feasibility of the induction of permanent or transient focal cerebral ischaemia in the marmoset using an intraluminal approach with minimal invasion. This model could be suitable as an advanced screening for potential stroke therapies in which behavioural, imaging, and histologic analyses can be compared.
Introduction
Non-human primates might be considered the species of choice to study the pathophysiology and the treatment of cerebral ischaemia because of their similarities with the human cerebrovascular system, brain metabolism, grey to white matter ratio (Zhang and Sejnowski, 2000), and rich behavioural repertoire that can be used for subtle functional recovery analyses (Fukuda and del Zoppo, 2003). Furthermore, the pharmacokinetics and pharmacodynamics in primates are often radically different from those in rodents. Although landmark studies with respect to the pathophysiology of stroke have been performed in large non-human primates such as baboons and macaques (Astrup et al, 1977), these species have been relatively abandoned over the last two decades. The reasons for this are mainly due to the high cost and the absence of technically simple stroke models in these species. A number of recent reports have been published to underline the necessity of the use of not only rodents but also other animal species and, especially, non-human primates, in the study of the pathophysiology and of the treatment of cerebral ischaemia (STAIR, 1999; Iadecola et al, 2006). Recently, the common marmoset (Callithrix jacchus) has been used in modelling cerebral ischaemia (Marshall and Ridley, 1996; Virley et al, 2004). Marmosets, weighing 250 to 400 g at adult age, have a brain size approximately four times that of the rat. They breed readily in standard animal facilities and are relatively easy to handle, which is advantageous for behavioural testing and postoperative care management (Mansfield, 2003). Moreover, thanks to the relatively small body size of the marmoset, studies made on these animals can be performed by only one qualified person and can use the infrastructure and the material commonly used in rat investigations. Thus, once breeding is established, the cost of an ischaemia experiment on this species is approximately twice compared to that performed on rats. Marshall and Ridley (1996) described a model of stroke by electrocoagulation of the middle cerebral artery (MCA) in the marmoset. However, this approach has several disadvantages: impossibility to achieve reperfusion, necessity of a large craniotomy, dissection of the dura mater, and displacement of the frontal lobe to reach the proximal portion of the MCA. Recently, Virley et al (2004) reported a model of temporary ischaemia in the marmoset after the application of the vasoconstrictor agent endothelin-1 on the distal segments of the MCA. In this model, which also requires craniotomy, the duration of the occlusion cannot be strictly controlled and, accordingly, provokes an increased interindividual variability of the extent of the lesion.
The intraarterial suture occlusion of the MCA, through the internal carotid artery (ICA), is the most widely used model of stroke in rodents (Longa et al, 1989). This approach, also termed the thread model, is technically simple in rodents, and does not require craniotomy and allows one to study permanent or transient ischaemia. In this study, we sought to adapt this surgical approach to the marmoset.
Materials and methods
All procedures were performed according to the appropriate European Directives and approved by the Regional Ethics Committee. Six male and four female laboratory-bred marmosets (C. jacchus) were used (weighing 240 to 330 g). Two weeks before the surgery, animals were housed in individual cages in a colony room maintained in a 12-h light/dark cycle.
Anaesthesia
Anaesthesia was induced with isoflurane (5%) and maintained during the entire experiment with isoflurane (1% to 1.5%) in an O2/N2O mixture (30%/70%). The animals were orally intubated and mechanically ventilated (Harvard Apparatus, South Natick, MA, USA). At the outset of the surgery, mean arterial CO2 tension was 38.5±8.6 mm Hg, arterial pH was 7.48±0.06, and arterial oxygen tension was 174±38 mm Hg. These parameters did not significantly vary throughout the experiment. Rectal temperature was kept close to 37.5°C with a heating pad. The tail artery and vein were cannulated for arterial pressure monitoring (Stoelting, Wooddale, IL, USA), blood sampling for gases and pH analyses (Ciba Corning, Halsted, UK), and for atracurium (0.5 mg/kg; and 0.75 mg/kg h; Tracrium, Faulding, Royal Leamington, UK) and saline (2 × 0.5 mL) administration, respectively.
Middle Cerebral Artery Occlusion
Focal cerebral ischaemia was induced by MCA occlusion (MCAo) based on the intraluminal filament approach previously described in rodents (Longa et al, 1989). Under aseptic conditions, a midline neck incision was made, and the right common carotid artery, external carotid artery (ECA), and extracranial branch of the ICA were carefully exposed, and the ECA was coagulated distally. Then, the head of the animal was secured in a rotating (360°C) stereotaxic frame (Kopf Instruments, Tujunga, CA, USA); an incision of the scalp was made, and the right temporal muscle was reflected towards its origin. A laser-Doppler flowmetry probe (FlooLab; Moor Instruments, Millwey, UK) was positioned on the parietal bone, thinned with saline-cooled dental drill, to measure cerebral blood flow (CBF). Once the CBF values were stable, the stereotaxic frame was rotated 180°C to show the previously dissected ventral side. Under the operating microscope, the extremity of a nylon thread (0.18 mm in diameter) was coated by a thermofusible adhesive (melting temperature 70°C) by passing it through a calibrated hole made in a heated piece of metal. The thread, with a distal cylinder of 3 mm length and 0.54 mm diameter, was inserted into the ECA and gently advanced (approximately 27 mm from the bifurcation between ICA and ECA) up to the origin of the MCA until the laser-Doppler flowmetry signal decreased. Reperfusion was achieved simply by gently withdrawing the filament. In temporary occluded marmosets (n=4, 3 males and 1 female), CBF was continuously monitored during the occlusion period and up to 30 mins after reperfusion. In those subjected to permanent ischaemia (n=6, 3 males and 3 females), CBF was also continuously monitored during the first 200 mins after the occlusion. After suturing the skin, each animal received an injection of an analgesic (tolfenamic acid, 4 mg/kg, intramuscularly, Tolfedine 4%, Vétoquinol, Lure, France) and an antibiotic (cefamandole 15 mg/kg, intramuscularly; Kefandol, France, for the next 3 days). After recovery from anaesthesia, the marmosets were returned to their cages and given access to water and soft food, and then observed for 1 to 2 h and daily. Usually, the day after occlusion, the marmosets were able to move and to feed without assistance.
Behavioural Assessment
Behavioural testing was performed on eight marmosets, subjected to either transient (n=4) or permanent (n=4) MCAo. Before surgery, marmosets were trained in behavioural tests to habituate them and to determine preoperative scores. The marmosets were then retested daily from day 1 (D1, first day after surgery) to D7 after ischaemia. Behavioural testing was performed in the home cage in the undernoted order.
Overt Abnormal Signs
The global status of the marmoset was assessed according to the following criteria: motility, circling behaviour, movement's accuracy, presence of miosis, and fur's aspect.
Neurologic Scoring
Neurologic status was evaluated according to Marshall and Ridley (1996). Briefly, the absence (score=2), scarce occurrence (score=1), or presence (score=0) of the following abnormal movements and postures was observed: forelimbs and hindlimbs slipping or dangling under the perch, at rest or during movement, hand crossing the chest, head tilting, and reaction to a visual stimulus.
Response to Tactile Stimulation
To assess somatosensory functions (Virley et al, 2004), the marmoset was coaxed to move around the cage while the experimenter, without intruding into its visual field, stimulates with a paintbrush each of its forelimbs, hindlimbs, and ears. Scoring was performed as follows: clear response=2, moderate response=1, or no response=0. Two trials for each stimulus were randomly performed and scores were averaged to give a maximum score of 6 for each body side.
Grip Test
Grip strength of each forepaw and hindpaw was assessed by encouraging the marmoset, held around the torso, to grip the shaft of a paintbrush while the experimenter pulled it away (Marshall and Ridley, 1996; Virley et al, 2004). Scoring was as follows: strong grip and resistance to the pull=2, moderate grip without resistance=1, and no grip=0. The procedure was repeated twice.
Hill and Valley Staircase Tests
These tests measured the skilled independent use of the forelimb (Annett et al, 1992b; Marshall and Ridley, 1996). Marmosets were required to reach and grasp food rewards (small pieces of apple) placed on five steps of two stairs located behind a Plexiglas screen attached to the front of the cage. In the hill version, there were two laterally positioned vertical slots, so that the animal used its right arm to retrieve the food pellets situated on the right stair, and vice versa. In the valley version, there was only one centrally positioned slot and the animal used its left arm to reach the right stair, and vice versa. In each version, the maximal time was 300 secs and the procedure was repeated twice, separated by 15 mins. The mean of cumulated time to retrieve the rewards was then calculated, with a maximum score of 1,500 secs (5 steps × 300 secs).
Adhesive Removal Test
This test, which requires sensory and motor capacities, was originally devised by Schallert et al (1983) in the rat and further adapted for the marmoset (Annett et al, 1992a; Marshall and Ridley, 1996). Briefly, the paradigm measures the marmoset's ability to detect and remove adhesive tapes of equal size (2 cm2 × 4.5 cm2) placed bilaterally around the hindpaws. Adhesives were pasted with an equal pressure, and the order of placing (right or left) was changed between each trial and each animal. The time to contact each paw (defined by a bite or a scratch) and the latency required to remove the adhesives were measured within a maximum of 10 mins. The procedure, performed in the home cage, was repeated twice with an intertrial interval of 15 mins. The animals were trained to the task and a preoperative value was collected the day before surgery (D1) for each animal. Likewise, individual postoperative data were averaged in two blocks: block 1 (mean of D1, D2, and D3) and block 2 (mean of D6 and D7).
Magnetic Resonance Imaging Studies
Eight days after induction of ischaemia, six marmosets underwent T2-weighted imaging and angiography with a 7 T MRI (Pharmascan; Bruker Biospin, Ettlingen, Germany) using a transmit/receive 60 mm resonator. A fast low angle shot (FLASH) scout scan was used for localization. T2-weighted MRI images (20 coronal slices; 1.5 mm thick) were performed with a fast spin-echo sequence (rapid acquisition with relaxation enhancement (RARE) with a factor of 8; repetition time (TR)=5,000 ms; echo time (TE)=60 ms; number of experiments (NEX)=4; field of view=50 mm × 50 mm; matrix=256 × 192). For magnetic resonance angiography, a high-resolution, time-of-flight FLASH sequence was used (TR=10 ms; TE=1.8 ms; NEX=2; field of view=50 mm × 50 mm × 33 mm; matrix=256 × 192 × 192).
Histology, Histochemistry, and Immunohistochemistry
The marmosets were deeply anaesthetized and transcardially perfused with a solution of saline. The brains were removed, frozen, and cut in the coronal plane in 20-μm-thick sections using a cryostat (Leica CM3050, Nussloch, Germany). One section in every 40 was stained by thionin to visualize the infarcted area. For immunohistochemistry, the brains of three marmosets (two transient and one permanent) were fixed in situ by transcardiac perfusion with a solution of 4% paraformaldehyde acid, and thereafter cut in the coronal plane at 40 μm with a freezing microtome (Leica SM 2400, France). Adjacent sections were incubated with either glial fibrillary acidic protein (GFAP) (rabbit anti-GFAP 1:5,000; Sigma, St Quentin Fallavier, France), or NeuN antibodies (mouse anti-NeuN 1:2,000; Chemicon, Chandlers Ford, Hampshire, UK). Secondary peroxidase anti-rabbit (Sigma) and biotinilated anti-mouse antibody (Sigma) were used, respectively. To examine the occurrence of intracerebral haemorrhage, Perls’ staining technique has been used (Meguro et al, 2007). Four brain sections of each animal were incubated in ferrocyanid solution (2%) and hydrochloric acid (2%) v/v for 30 mins at room temperature. The sections were then rinsed several times with tap water and allowed to dry for 5 h at room temperature.
For the measurement of infarct volume, 20 to 30 equidistant coronal slices covering the entire lesion were used, and the infarcted surface was measured on each slice through the use of ImageJ software (Abramoff et al, 2004). The total histologic volume of infarction was calculated by integration of the areas over the analyzed sections and the distance between them.
Statistics
Data are presented as mean±s.d. The following tests were used when appropriate: Student's t-test and two-way analyses of variance with repeated measures, followed by post hoc Fisher's protected least significant difference (PLSD) test. A univariate t-test was used to compare the behavioural performances to a reference value. A P<0.05 was considered significant (Statview software).
Results
Induction of Ischaemia
Magnetic resonance imaging-derived angiography shows that the ICA presents a distinct carotid siphon before the emergence of the MCA (Figure 1A). Despite this curvature, the filament, with a distal cylinder of 3 mm length and 0.54 mm diameter, was introduced through the ECA at the origin of the MCA without resistance. An abrupt decrease in CBF, proof of the efficiency of MCAo, was observed when the filament was introduced approximately 27 mm from the bifurcation between ICA and ECA. In permanently occluded marmosets, the adequate position of the embolus occluding the origin of the MCA was also documented by visual inspection after brain removal after euthanasia and by magnetic resonance imaging-derived angiography showing the disappearance of the signal of the MCA (Figure 1B). In transiently occluded animals, the MCA was visible on angio-MRI, which attests to complete reperfusion (Figure 1C). The occlusion of the MCA induced a similar CBF decrease in permanently and temporarily occluded marmosets. During the 3 h after the occlusion, no difference in decreased CBF was observed between the two groups (two-way analysis of variance (ANOVA), P=0.08) (Table 1). In transiently occluded marmosets, withdrawal of the filament resulted in a hyperperfusion that lasted at least 30 mins after reperfusion (Table 1). During the whole period of experiment, arterial pressure, heart rate, and rectal temperature were similar in the two groups and remained stable (Table 1). No postoperative mortality was observed in our experiments.
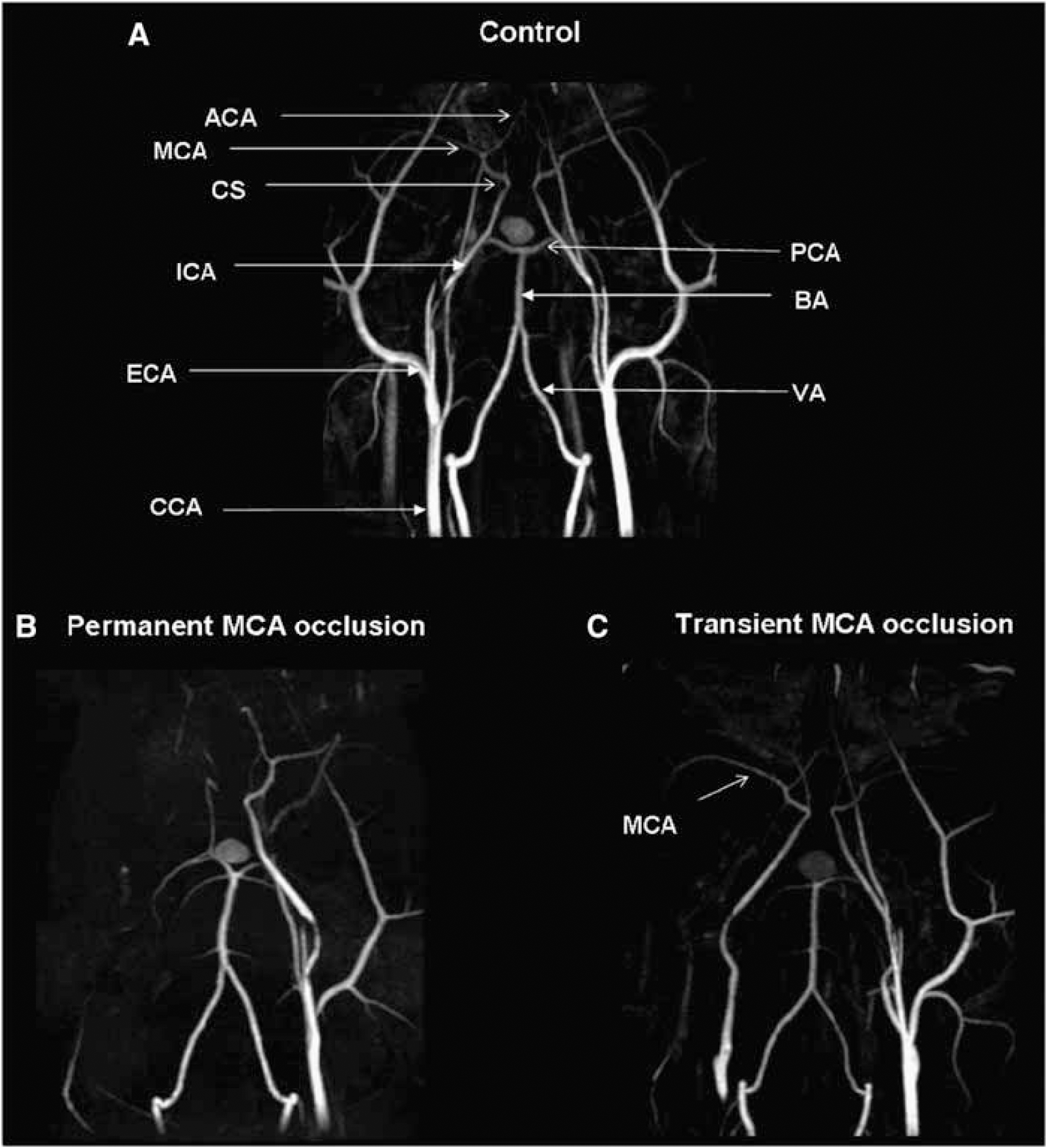
Representative images of magnetic resonance angiography performed 8 days after MCAo in permanently and transiently occluded marmosets in comparison to a nonoperated animal. Maximum intensity projection maps of time-of-flight magnetic resonance angiograms are presented in the horizontal plane. ACA, anterior cerebral artery; BA, basilar artery; VA, vertebral artery; CCA, common carotid artery; CS, carotid siphon; ECA, external carotid artery; ICA, internal carotid artery; MCA, middle cerebral artery; PCA, posterior cerebral artery.
Physiologic parameters in permanently and transiently occluded marmosets
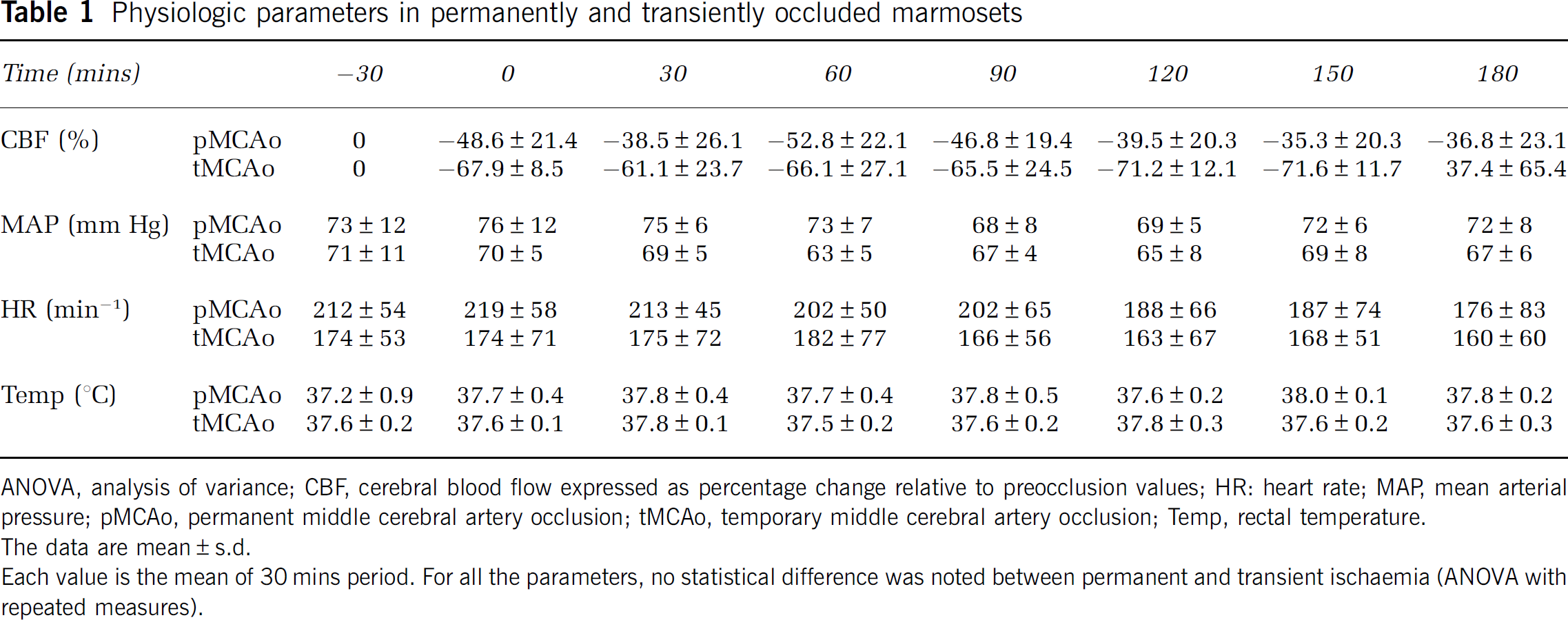
ANOVA, analysis of variance; CBF, cerebral blood flow expressed as percentage change relative to preocclusion values; HR: heart rate; MAP, mean arterial pressure; pMCAo, permanent middle cerebral artery occlusion; tMCAo, temporary middle cerebral artery occlusion; Temp, rectal temperature.
The data are mean±s.d.
Each value is the mean of 30 mins period. For all the parameters, no statistical difference was noted between permanent and transient ischaemia (ANOVA with repeated measures).
Histology and Magnetic Resonance Imaging
Eight days after the MCAo, histologic analyses revealed a well-delimited infarction in cortical and subcortical ipsilateral regions. In all animals, ischaemic damage was observed in the caudate, the putamen, the internal capsule, and also in the cortical areas surrounding the lateral sulcus (Figure 2A). The volume of hemispheric infarction was 358.1±117.8 and 237.1±139.2 mm3 in permanently and transiently occluded marmosets, respectively. The difference between the two groups did not reach statistical significance (Student's t-test P=0.18). In both groups, neither intracerebral swelling nor overt haemorrhage was observed. Perls’ staining, performed on brain sections of all the animals, confirmed the absence of intracerebral haemorrhage (Figure 2B).
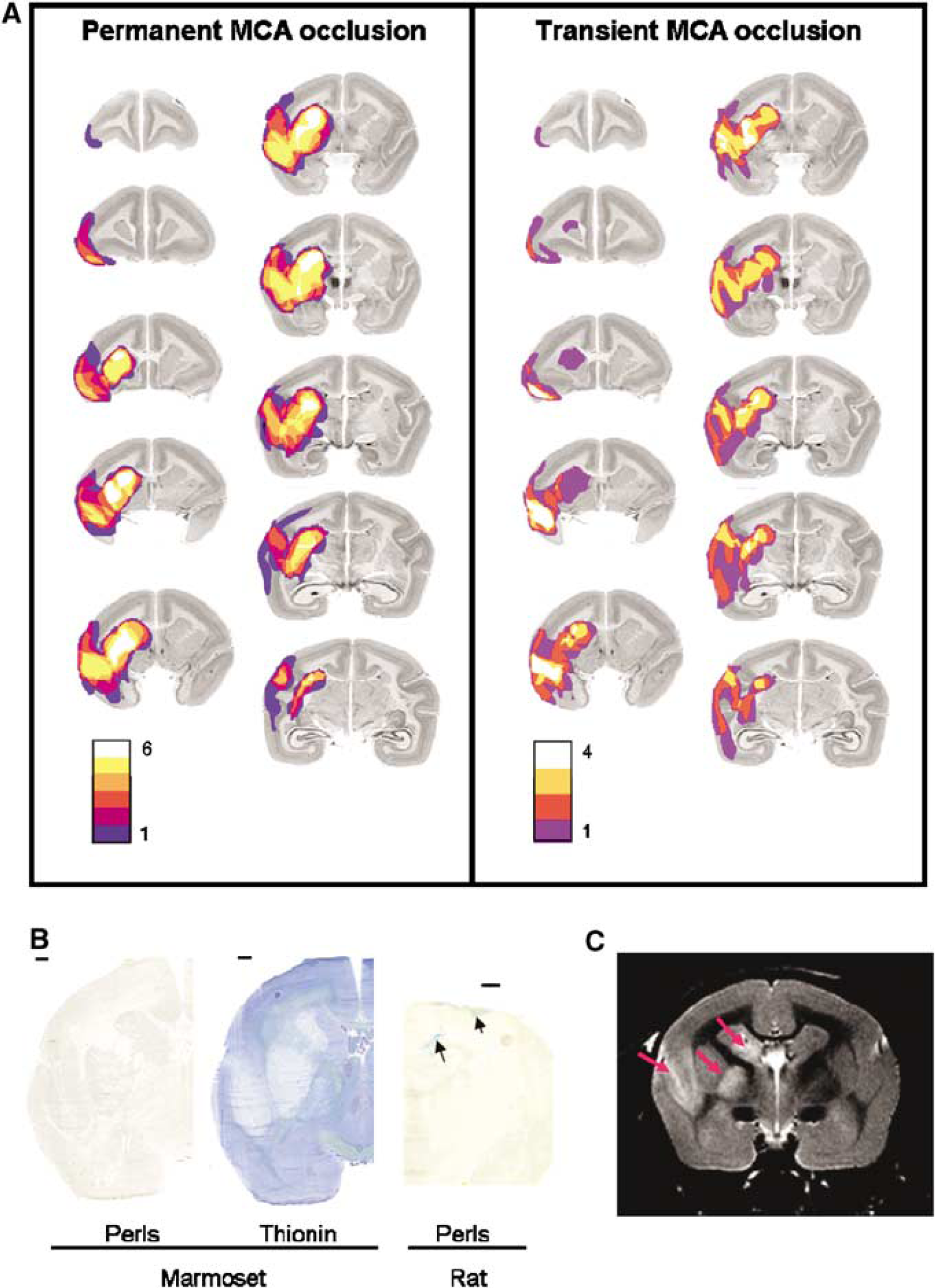
(
In animals that underwent MRI examinations (n=6), T2 images also showed an infarction located in extended cortical and subcortical structures (Figure 2C).
Immunohistochemical techniques based on the NeuN antibody, a marker of intact neuronal nuclei, confirmed neuronal loss within the lesion, which was demarcated by weak thionin staining (Figure 3). There was no apparent selective cellular loss outside the lesion 8 days after occlusion of the MCA. GFAP immunostaining revealed a marked astroglial reaction in the affected cortical and subcortical regions (Figure 3).
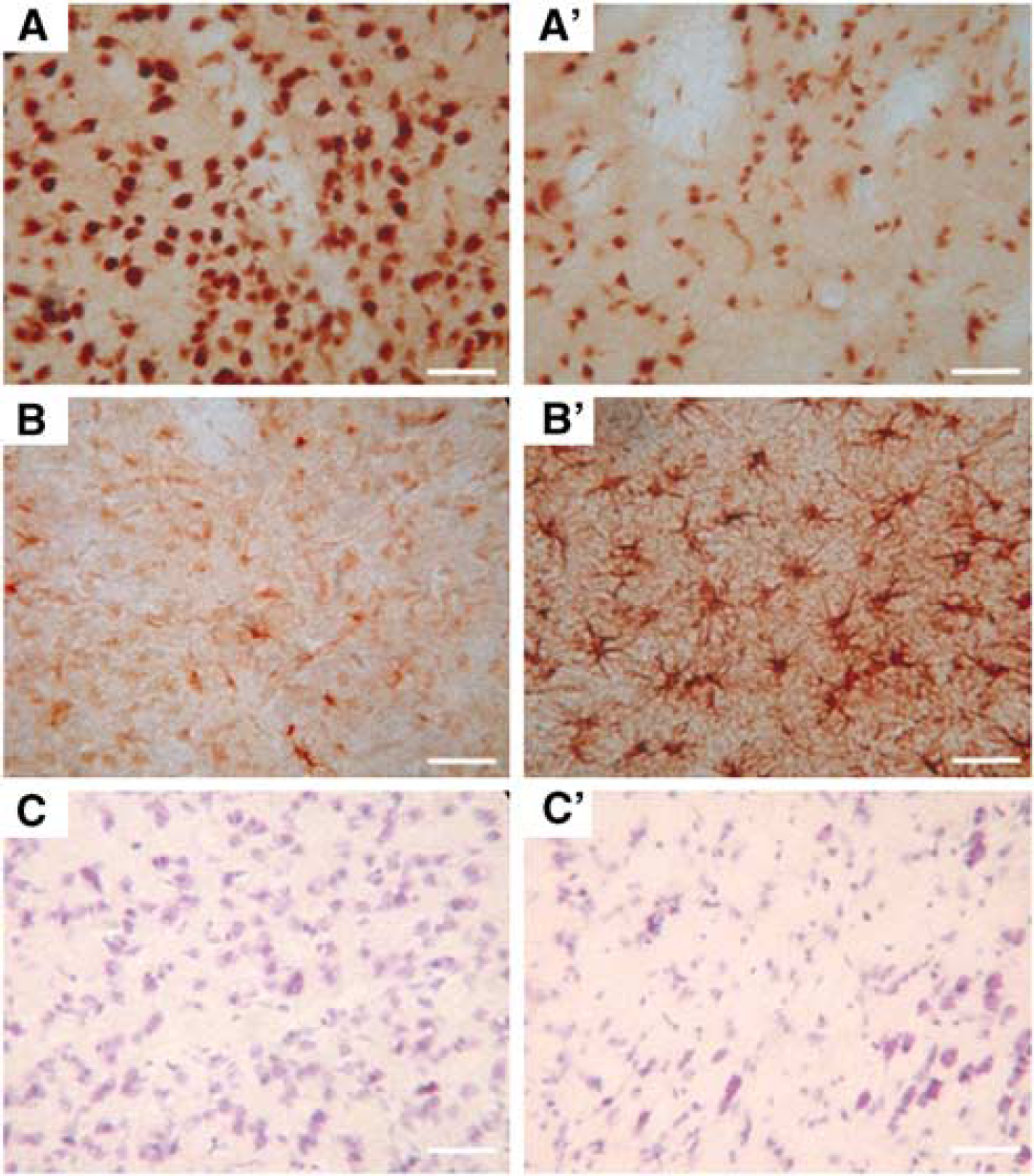
Representative photomicrographs illustrating both neuronal loss and the astroglial reaction in the putamen after 8 days recovery after 3-h temporary MCAo in the marmoset. (
Behavioural Results
Whatever the behavioural test considered, there was no significant difference between groups or between left and right performances during the presurgical period.
Postoperative General Status
After surgery, all marmosets showed a decrease in general activity, which was slight and transient (2 days) in reperfused, and noticeable and longer (until D7) in permanently occluded animals. We also noted an ipsilateral miosis until the end of the experiment in animals subjected to permanent, but not to transient, MCAo.
After surgery, reperfused marmosets only showed a slight and transient (during 2 days) decrease in general activity. In contrast, marmosets subjected to permanent MCAo showed a long-lasting decrease in overall activity. However, after 4 days, they progressively displayed a marked improvement, although they were still clumsy at D7. We also noted an ipsilateral miosis, which lasted until the end of the experiment in animals subjected to permanent, but not to transient, MCAo. A ptosis was occasionally observed during the first 2 days in both groups. In some animals, we observed a transient body tremor and frequent eyelid blinking. Moreover, permanently occluded animals displayed a circling behaviour during the first 3 days, which often appeared during crises that seemed triggered by an emotional stress (changing the apparatus between tests, for instance).
Neurologic Score
Whereas transiently occluded marmosets exhibited no significant deficit in the contralateral side (score=19.5±2.6), those subjected to permanent MCAo displayed a long-lasting deficit until D7 (score=8.8±2.2 at D1 and 15.5±5.0 at D7, univariate t-test, P<0.0007 and P<0.05 for D1 and D7, respectively).
Response to Tactile Stimulation
After surgery no deficit was noted in either ischaemic group on the ipsilateral side. However, on the contralateral side, both groups showed a deficit (two-way ANOVA with repeated measures, group effect: P<0.0001, time effect: P<0.002, no group × time interaction: P=0.58). Marmosets subjected to transient MCAo were only slightly impaired 1 day after the intervention (univariate t-test, P<0.05), and showed a rapid spontaneous recovery (time effect, P<0.02). Conversely, permanently occluded animals displayed a long-lasting deficit, which persisted until the end of the experiment (univariate t-test, P<0.003 at D7; no time effect P=0.11).
Grip Test
On the ipsilateral side, no significant deficit was observed in either group, with a score close to maximum until the end of the experiment. On the contralateral side, temporary occluded animals did not show any impairment, whereas those subjected to permanent occlusion exhibited a significant deficit when compared to the maximal score (score at D1: 1.3±0.7; univariate t-test, P<0.0001). This deficit was however transient and disappeared at D6 (univariate t-test, P=0.13).
Hill and Valley Staircase Tests
After transient or permanent ischaemia, the marmosets spent more time retrieving the pellets at the hill staircase (two-way ANOVA with repeated measures; time effect: P<0.0001 for both sides). This bilateral deficit was not different between the two groups (no group effect: P=0.15 and P=0.12 and no group × time interaction: P=0.67 and P=0.32 for ipsi- and contralateral side, respectively), and was still present at D7 on the contralateral side (P<0.003). On the ipsilateral side, this deficit disappeared at D6 (P=0.18) (Figures 4A and 4B).
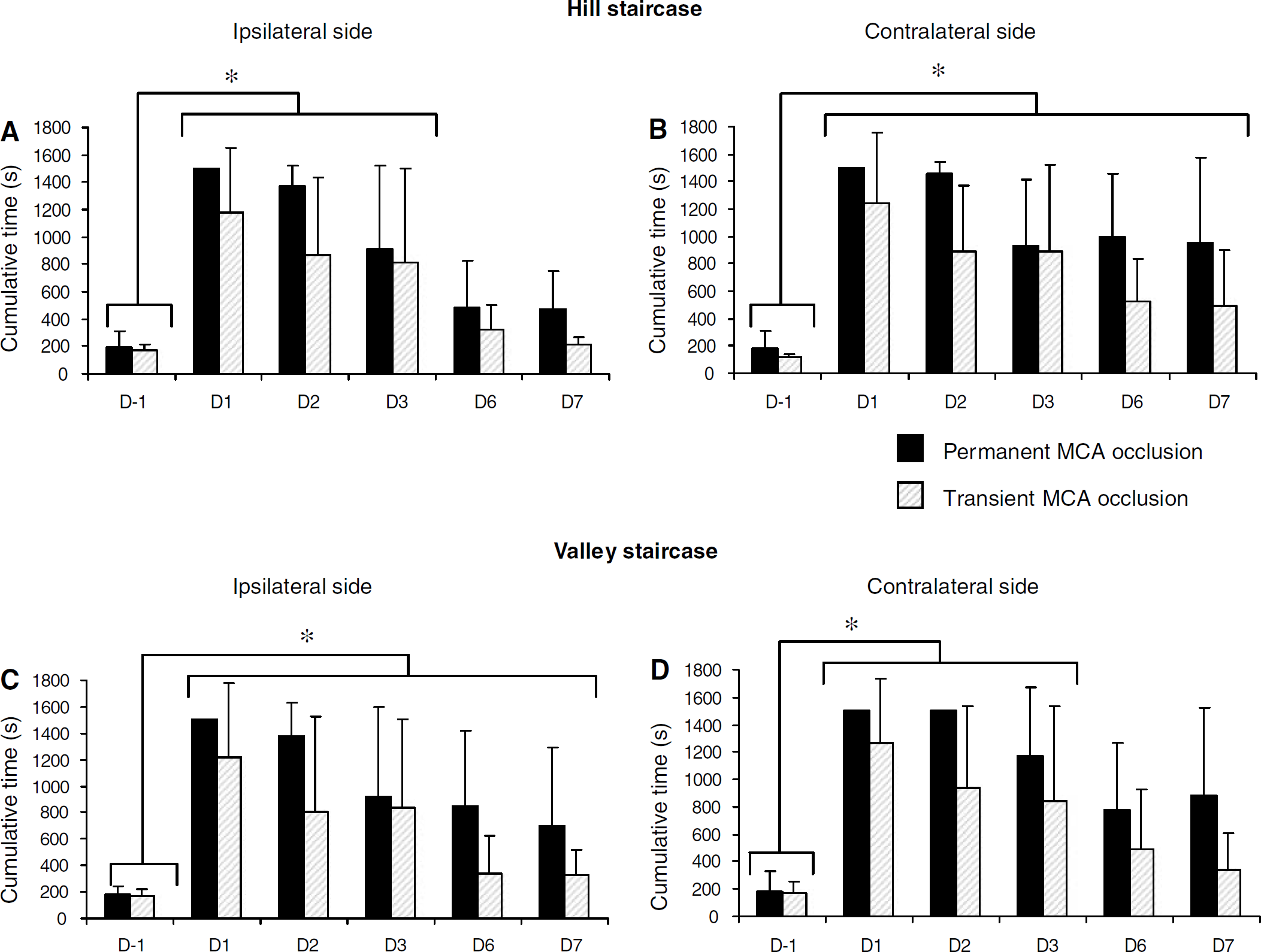
Forelimb reaching performances in the staircase tests after permanent and transient MCAo as a function of time (D0=day of ischaemia). The data are expressed as the mean cumulative time (±s.d.) to retrieve all pellets. (
At the valley staircase, retrieving the rewards with the ipsilateral forelimb was significantly affected until D3, whatever the group considered (Figure 4D) (two-way ANOVA with repeated measures, no group effect: P=0.14; time effect P<0.0001; and no group × time interaction: P=0.72). Regarding the contralateral forelimb, the two groups were similarly impaired (two-way ANOVA with repeated measures, no group effect: P=0.09; a time effect: P<0.0001; and no group × time interaction: P=0.74). Even if an improvement was observed, the deficit on the contralateral forelimb was still present at D7 (P<0.04 at D7) (Figure 4C).
Adhesive Removal Test
Regarding the time to contact the adhesives, the two groups were differently affected by ischaemia (two-way ANOVA; group effect: P<0.05 and P<0.04; time effect: P<0.0005 and P<0.0005; group × time interaction: P<0.03 and P<0.0005 for ipsi- and contralateral sides, respectively) (Figure 5A). On the ipsilateral side, the deficit observed at block 1 fully disappeared at block 2, whatever the duration of ischaemia. This somatosensory deficit was more severe in permanently occluded animals compared to transiently occluded animals (block 1, one-way ANOVA group effect: P<0.03). On the contralateral side, a transient and persistent impairment was observed in temporary and permanently occluded marmosets, respectively (Figure 5A).
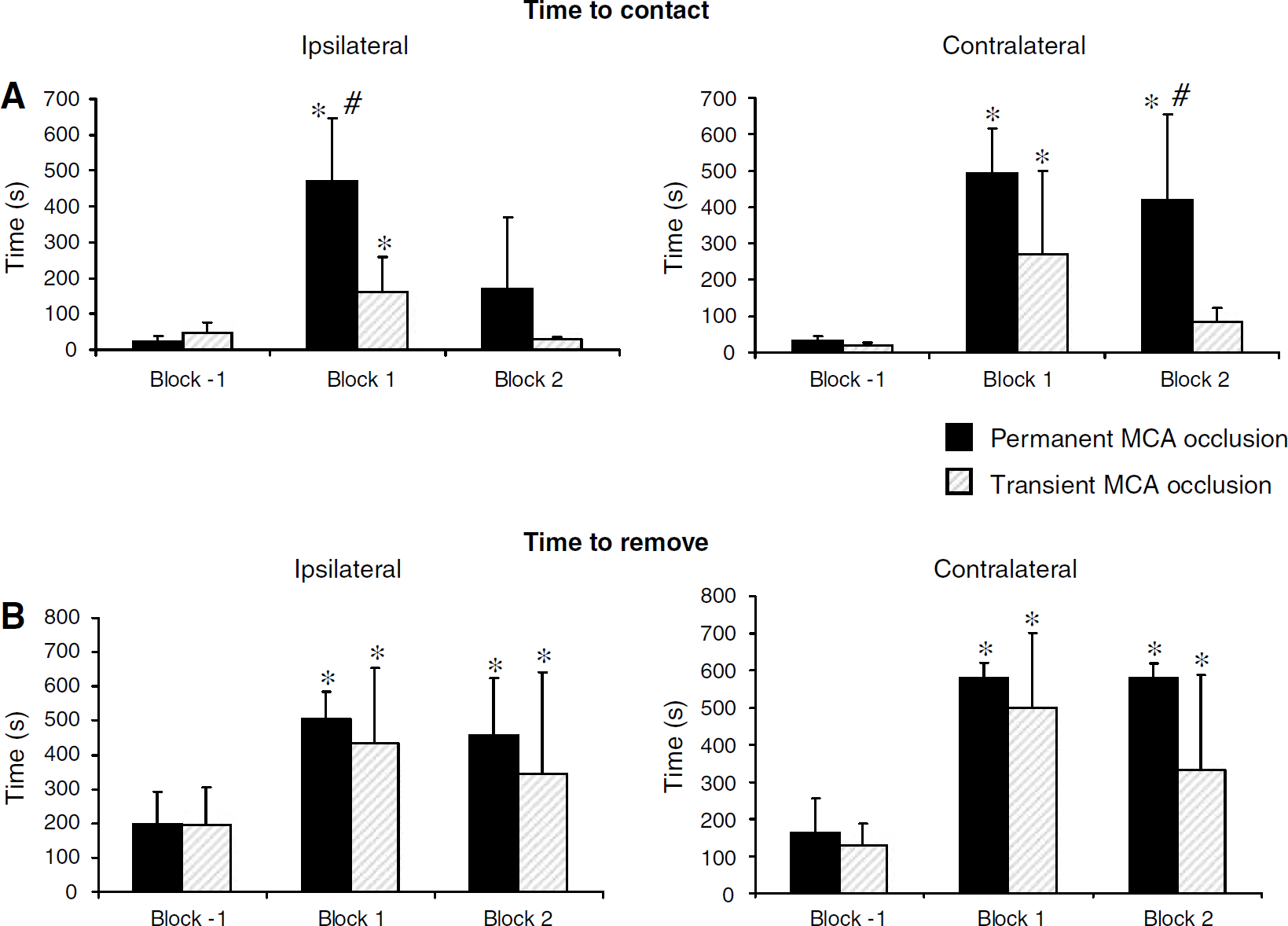
Mean time (±s.d.) to contact (
With respect to the time to remove the adhesives (Figure 5B), a bilateral sensorimotor deficit was observed in both groups up to block 2 (two-way ANOVA with repeated measures; no group effect: P=0.06 and P=0.34; time effect: P<0.0005 and P<0.04; and no group × time interaction: P=0.35 and P=0.85 for ipsi- and contralateral sides, respectively).
Discussion
This study shows the feasibility to induce focal cerebral ischaemia by the intraluminal approach in the non-human primate (the common marmoset). This method has several advantages: (1) the strict control of ischaemia durations; (2) the absence of craniotomy, and thus no ‘artificial’ alterations of intracranial pressure and brain temperature are caused; (3) the simplicity of the surgical procedure, which resembles to that widely performed in rodents in many laboratories; (4) the comparative noninvasiveness, as severe disability and mortality was not observed in the postoperative period. Overall, this paradigm allows one to perform behavioural analyses in conjunction with pathophysiological and neuropathological changes in the acute and chronic stages of the infarct evolution.
The marmosets have been recently used in modelling focal stroke (Marshall and Ridley, 1996; Virley et al, 2004). However, the approaches employed involved sophisticated surgical procedures that required an extensive craniotomy and did not permit to achieve reperfusion with strictly controlled duration of ischaemia. In an attempt to develop a model of cerebral ischaemia with reperfusion by an intravascular method, Virley et al (2004) examined the gross cerebrovascular anatomy of the marmoset, through the use of Araldite corrosion cast and ink perfusion techniques, and showed that its principal features are similar to those previously described for the yellow-handed marmoset and human. The ACA and MCA formed the terminal branches of the ICA; the MCA was the largest branch of the ICA; the lenticulostriate arteries were observed at the proximal MCA portion (M1), which extends upwards to the M2 portion. Some aspects of this anatomy were also observed in our angiography-derived MRI examinations (Figure 1A). Based only on this anatomic analysis of the vasculature, the authors concluded that an extracranial approach to induce temporary MCAo would not be possible due to the presence of the carotid siphon (Virley et al, 2004). However, given that the intravascular approach to occlude the MCA—through the insertion of inflatable catheter from the femoral artery up to the origin of the MCA—has been achieved in the baboon (Hamberg et al, 2002), a species that also has a tortuous ICA, we sought to establish a simple model of transient ischaemia in the marmoset using the intraluminal thread technique. During the entire surgical procedure, the marmosets were anaesthetized with isoflurane, and physiologic parameters remained stable within physiologic limits in both groups. All of the marmosets quickly recovered from anaesthesia and were able to move and to feed without assistance when returned to their cages. In preliminary studies, we tested nylon filaments with distal cylinders of different diameters and found that the introduction of one having a distal cylinder of 3 mm length and 0.54 mm diameter was carried out without any resistance. This approach resulted in an abrupt and persistent reduction of CBF. The diameter of this embolus is concordant with those of the ICA and the MCA reported by Virley et al (2004). In reperfused marmosets, the withdrawal of the embolus was also accomplished without resistance or obvious haemorrhage.
The CBF reduction was observed when the filament was introduced approximately 27 mm from the bifurcation of the ICA and ECA. In permanently occluded animals, the adequacy of the position of the embolus that occluded the origin of the MCA was confirmed by visual inspection of the circle of Willis after post-mortem removal of the brain. The filament removed after euthanasia had an ‘S’ shape, suggesting that it fit to the sinuous form of the ICA. Moreover, the magnetic resonance imaging-derived angiography showed the disappearance of the signal from the MCA confirming the permanent interruption of flow in this artery (Figure 1B). By contrast, a reperfused MCA in transiently occluded marmosets was clearly seen (Figure 1C).
The MCAo resulted in an immediate and stable reduction in CBF during the period of occlusion, which was not different between permanently and transiently occluded animals. In the latter group, hyperperfusion was seen after the removal of the suture, indicating an efficient reperfusion. Although the number of marmosets used in our study is relatively small, the MCAo resulted in a reproducible infarction that affected both cortical and subcortical white matter as well as the basal ganglia (Figure 2A). This topology is of interest as the occlusion of the proximal segment of the MCA in larger primates (e.g., baboons) results mainly in internal capsular and basal ganglial damage (Tagaya et al, 1997; Young et al, 1997). The volume of hemispheric infarction measured in our study is comparable to that previously reported in marmosets subjected to extraluminal MCAo (Marshall et al, 2003). Although the mean volume of infarction was smaller in animals subjected to temporary occlusion compared to those that underwent permanent ischaemia, this difference did not reach statistical significance. This can be ascribed to the small number of marmosets included in each group. In all animals analyzed, no overt brain haemorrhage was observed. The absence of haemorrhage was additionally confirmed in some animals through the use of post-mortem Perls’ staining technique.
Eight days after the MCAo, T2-weighted MRI also revealed a lesion that encompassed the cortex around the lateral sulcus, subcortical white matter, and the basal ganglia (Figure 2C). These imaging studies, although performed only on six animals, show the feasibility of imaging the ischaemic lesion with MRI with valuable temporal and spatial information in the same animal. This technology may be of value to assess the potential neuroprotective treatments in this non-human primate model. The immunohistochemical analyses based on the GFAP antibody revealed a large zone of astrocyte reaction affecting both the cortical and subcortical zones. The use of the neuronal marker (NeuN antibody) confirmed neuronal loss within the lesion, but there was no evidence for selective neuronal loss in areas remote to the lesion. Altogether, our approach shows the feasibility of performing multimodal investigations in the marmoset subjected to focal ischaemia (i.e., MRI, histology, immunohistochemistry). Nonetheless, future studies need to be undertaken to address the spatiotemporal evolution of the lesion, the impact of the suture on the endothelium and its role in the development of the lesion, as well as the influence of gender on the extent of ischaemic brain damage. Indeed, all the available studies have used both male and female marmosets, and no comparison of the ischaemic lesion size between the two sexes has been made. In our own studies, and in accordance with those of Virley et al (2004), no difference in behavioural deficits between males and females was observed. Nonetheless, given the small number of animals used, no statistical analysis has been performed.
Despite the brain lesion and neck surgery, all the animals included in our study survived until euthanasia at 8 days after the occlusion. This fact allowed us to perform advanced behavioural analyses to assess the evolution of various functional deficits. Permanent and transient cerebral ischaemia altered the global neurologic status of the marmoset. However, the animals subjected to permanent MCAo exhibited marked and persistent neurologic impairments in the contralateral side compared to the animals that underwent transient ischaemia. Although the neurologic scoring is investigator-dependent, our observations in the permanent MCAo group are in agreement with those of Marshall and Ridley (1996). The neurologic score was also decreased by transient ischaemia in the endothelin model of Virley et al (2004). Nonetheless, in this latter study, the neurologic evaluation was restricted to the first day after the ischaemia. Somatosensory functions were impaired on the contralateral side by both durations of ischaemia, although transiently in the reperfused animals and durably (at least until the end of the first week after surgery) in the permanently occluded marmosets. Such a pronounced and long-lasting neglect of contralateral tactile stimuli induced by ischaemia is in accordance with the results of Marshall and Ridley (1996). Grip strength was strongly, but transiently, reduced in the marmosets subjected to permanent MCAo, whereas almost no deficit was observed in temporary occluded animals. This latter finding contrasts with the study of Virley et al (2004), which reported a decreased grip strength in marmosets subjected to transient ischaemia. But, as for neurologic scoring, the grip test is a rather subjective procedure and hardly comparable from one study to another. The use of an automated method of objective grip strength measurement, based on an apparatus adapted to the marmoset would be most useful (Iwanami et al, 2005). In the adhesive removal test, the sensorimotor capacities of both ischaemic groups were equally impaired in both sides, irrespective of the time point considered. The animals presented a partial recovery at the end of the study. Lastly, during a more demanding task, such as the hill staircase, no difference was observed between the two groups. Both displayed a bilateral hemiparesis, although less severe and transient ipsilaterally. These results contrast with those of Marshall and Ridley (1996), who reported only a unilateral motor deficit at both the adhesive removal and hill staircase tests in the extravascular model of cerebral ischaemia. However, as it has already been suggested in the rat by Marston et al (1995), the bilateral nature of the deficit could be due to a dysfunction of interhemispheric communication, and therefore dependent on the model of ischaemia and the time of analysis of the functional deficit. Indeed, in contrast to the study of Marshall and Ridley (1996), our analyses were performed after the acute period of ischaemia. We cannot infer from our data whether a hemineglect syndrome was associated with the motor impairment induced by ischaemia, as already described after permanent ischaemia induced by the extravascular approach (Marshall and Ridley, 1996).
In conclusion, our study shows the feasibility to induce a focal cerebral ischaemia in the non-human primate through the use of the intraluminal approach, highly relevant for stroke studies. Importantly, this model allows the induction of permanent or transient ischaemia with a strictly controlled occlusion duration. Even if further experiments are necessary to evaluate the long-term (i.e., several weeks) functional recovery after ischaemia, we show that the intraluminal model of cerebral ischaemia is, firstly, suitable to undertake behavioural studies and, secondly, that it induces functional impairments that are close to the neurologic and somatosensory symptoms observed in stroke patients. Since the ultimate goal of any therapy is recovery, or at least, improvement of patients’ functions, our approach may be critical in this regard.
Footnotes
Acknowledgements
We thank D Divoux and L Chazalviel for technical assistance.
