Abstract
Dynamic cerebral autoregulation (dCA) is a key mechanism that regulates cerebral blood flow (CBF) in response to transient changes in blood pressure (BP). Impairment of dCA could increase vulnerability to hypertensive vascular damage, but also to BP lowering effects of antihypertensive treatment. The literature remains conflicted on whether dCA is altered in Alzheimer’s disease (AD) and mild cognitive impairment (MCI). We summarized available data on dCA in AD and MCI, by searching PubMed, Embase, PsycINFO and Web of Science databases (inception-January 2022). Eight studies (total n = 443) were included in the qualitative synthesis of which seven were eligible for meta-analysis. All studies used Transcranial Doppler (TCD) ultrasonography and transfer function analysis or the autoregulatory index to assess dCA during spontaneous or induced BP fluctuations. Meta-analysis indicated no significant difference between AD, MCI and healthy controls in dCA parameters for spontaneous fluctuations. For induced fluctuations, the available data were limited, but indicative of at least preserved and possibly better autoregulatory functioning in AD and MCI compared to controls. In summary, current evidence does not suggest poorer dCA efficiency in AD or MCI. Further work is needed to investigate dCA in dementia with induced fluctuations controlling for changes in end-tidal carbon dioxide.
Keywords
Introduction
With the population ageing, the worldwide prevalence of Alzheimer’s disease (AD) and other forms of dementia is expected to almost triple in the next thirty years. 1 Inevitably, this has significant implications for the individual, society, and economy, particularly in developing countries where anticipated growth is greatest. 2 Currently, no treatments are available to cure the disease or effectively prevent its progression. Although age is the strongest established risk factor for dementia, not everyone develops the disease as they get older. Studies show that genetics, sex, lifestyle, educational level, and comorbidity (including hypertension, atherosclerosis, type 2 diabetes, and cardiac disease) can influence the risk of cognitive decline and dementia.3,4
While many of these risk factors are related to vascular health, the aetiologies of vascular (VaD) versus neurodegenerative dementias like AD have long been viewed as separate. However, mounting evidence indicates the presence of vascular involvement in AD pathology, contributing to both the onset and progression of the disease.5,6 Studies show that cerebral microvascular dysfunction, through chronic hypoperfusion and oxidative stress, contributes to brain atrophy and cognitive decline, while simultaneously promoting cerebral amyloid-β (Aβ) and tau accumulation, adversely impacting cerebrovascular function.6–8
Under normal physiological conditions, cerebral blood flow (CBF) is continuously regulated to ensure sufficient nutritional flow necessary to sustain adequate brain function. 9 The cerebral vasculature plays a pivotal role in these regulatory responses, including the maintenance or recovery of CBF in response to changes in arterial blood pressure (BP) or intracranial pressure (ICP). This mechanism, known as cerebral autoregulation (CA), protects the brain against variations in perfusion pressure (BP – ICP) that risk pressure damage or hypoxic injury.9,10 It operates by adapting cerebrovascular resistance (CVR) through a balance of vasodilation and constriction to keep cerebral perfusion relatively constant during slow, gradual long-term changes in BP, (‘steady-state’ or ‘static’ CA [sCA]), and to recover CBF towards baseline during more rapid changes in BP, known as dynamic CA (dCA).9–11
Numerous studies have demonstrated pronounced alterations in cerebral haemodynamics, including reduced CBF or perfusion deficits and increased cerebrovascular resistance (CVR), in patients with dementia (VaD and AD) or mild cognitive impairment (MCI), that may even be present long before clinical symptoms manifest.9,12–15 These findings of cerebrovascular pathology and reduced CBF raise the question whether CA may be less effective in patients with AD, such that CBF becomes compromised as BP levels fluctuate. Indeed, some studies have suggested that in these patients as well as in the general older population, low BP combined with antihypertensive treatment (AHT) is associated with an increased risk of dementia and greater cognitive decline.16,17 On the other hand, a recent meta-analysis found no effect of AHT and BP lowering on CBF in older adults, however, only a small number of studies included patients with cognitive impairment, and the meta-analysis was limited to static (steady-state) CA, not dCA. 18
In animal models of AD, severe impairment of CA has been demonstrated as a result of amyloid angiopathy.7,19 In contrast, research on CA in humans is limited and contradictory, with preliminary results suggesting its preservation in AD and MCI. 7 However, these studies often have small sample sizes and differ in their method of CA assessment (static vs. dynamic). Whether CA is truly impaired in these patients remains uncertain. Yet, about 40% of dementia patients also have hypertension and are often receiving AHT. 20 Moreover, inadequate CBF can have deleterious effects on the functional and structural brain integrity of these already vulnerable patients. Therefore, the aim of this systematic review was to summarize and evaluate the available data on autoregulatory functioning in adults with dementia or MCI, specifically focussing on the dynamic relationship between beat-to-beat BP alterations and CBF fluctuations (i.e. dCA).
Methods
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials. The review protocol was pre-registered with the International Prospective Register of Systematic Reviews (PROSPERO 2021, CRD42021264236). The study reporting followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) updated reporting guideline. 21 All aspects of record screening, data extraction and quality assessment were performed by three independent reviewers (RH, AD, DG), who were not authors of any of the articles being assessed, with access to a second reviewer (RdH or LB) to make final decisions in case of disagreements. The full text was evaluated if eligibility was not clear from the title and abstract.
Eligibility criteria
Studies included were any design (case-control, cross-sectional, data from randomized controlled trials and quasi-experimental studies) investigating dCA in dementia and MCI. All settings were considered: community or primary care-based, specialist and secondary care settings. Populations of interest were: any person (≥18 years of age) diagnosed with dementia (all-cause and specified subtypes such as AD, vascular, frontotemporal, Lewy body) or MCI (as defined by authors in the primary papers). Patients both on and off anti-dementia medication were included. Healthy controls (≥18 years of age) were included as a comparator group. We included studies that measured dCA during rest (spontaneous oscillations) as well as during orthostatic challenges (induced oscillations), such as repeated squat-stand or sit-to-stand manoeuvres (SSMs), using transcranial Doppler (TCD) ultrasonography to conduct measurements. Methods of dCA quantification considered for inclusion were: transfer function analysis (TFA), autoregulatory index (ARI), Mx and PRx, and rate of regulation. Articles not in English or with no full text available were excluded.
Sources and search strategy
A comprehensive search of PubMed, Embase (OVID), PsycINFO (OVID) and Web of Science was performed using the strategy detailed in Supplementary Text A in consultation with a librarian. Articles were limited to humans and the English language. All databases were consulted from database inception until January 2022.
Study selection and data extraction
Study selection was performed using the Rayyan QCRI webtool. 22 Data were extracted to Microsoft Excel and studies were grouped based on the studied population and outcome measure. Data on dCA estimates (primary outcome), including the ARI, and (normalized) gain, phase and coherence were extracted and grouped based on the assessment method (spontaneous vs. induced) and oscillation frequency. When dCA estimates were only presented in a figure, the values were read from the figure by hand where possible. Additionally, data on sample size, participant characteristics, exclusion criteria, method of dCA assessment, and end-tidal carbon dioxide (EtCO2) were extracted. Where necessary, we contacted the authors of relevant articles to request additional data. All data are presented as mean ± standard deviation (SD) unless noted otherwise. From confidence intervals (CI) the SD was calculated, and standard errors of the means (SEM) were converted to SD. We included BP and CBFv measurements using any metric but anticipated that most data would be in the form of BP in mmHg and CBFv in cm/s. If the dCA estimate phase was reported in degrees, we converted it to radians.
Quality assessment
Study quality was assessed using a modified version of the Newcastle-Ottowa scale for case-control study designs by three independent reviewers (RH, AD, DG). 23 See Supplementary Text B for the list of items used for assessment. Uncertainties were resolved by an additional reviewer (RdH or LB).
Statistical analysis
Meta-analyses were performed when there were data from three or more studies with sufficient homogeneity using Revman Version 5.4 software for Windows. 24 The inverse variance method for continuous variables with a random-effects model was used for all analyses. We calculated the pooled estimate of the mean difference (phase and the ARI), or standardized mean difference (gain), between diagnostic groups. Analyses were split according to disease severity (healthy, MCI, moderate to severe dementia), dCA measurement method (spontaneous, induced), and frequency interval (very low frequency [VLF], low frequency [LF]). Data are presented in forest plots with 95%-CIs and corresponding p-values. Statistical significance is assumed if p < 0.05. The heterogeneity between studies was explored with the I2 statistic, where I2 = 0–30% was considered low, I2 = 31–60% moderate, and I2 = 61–100% high.
Results
Study selection
Figure 1 shows the PRISMA flow diagram. The initial search yielded 1529 references. Following duplicate removal, 953 references were screened (title/abstract) for eligibility leaving thirteen for full-text assessment. Via contact with relevant authors (LB, RBP), an extra article (with additional unpublished data provided) was added to the review after the final search. Ultimately, eight studies were included in the qualitative synthesis and seven in the meta-analysis.
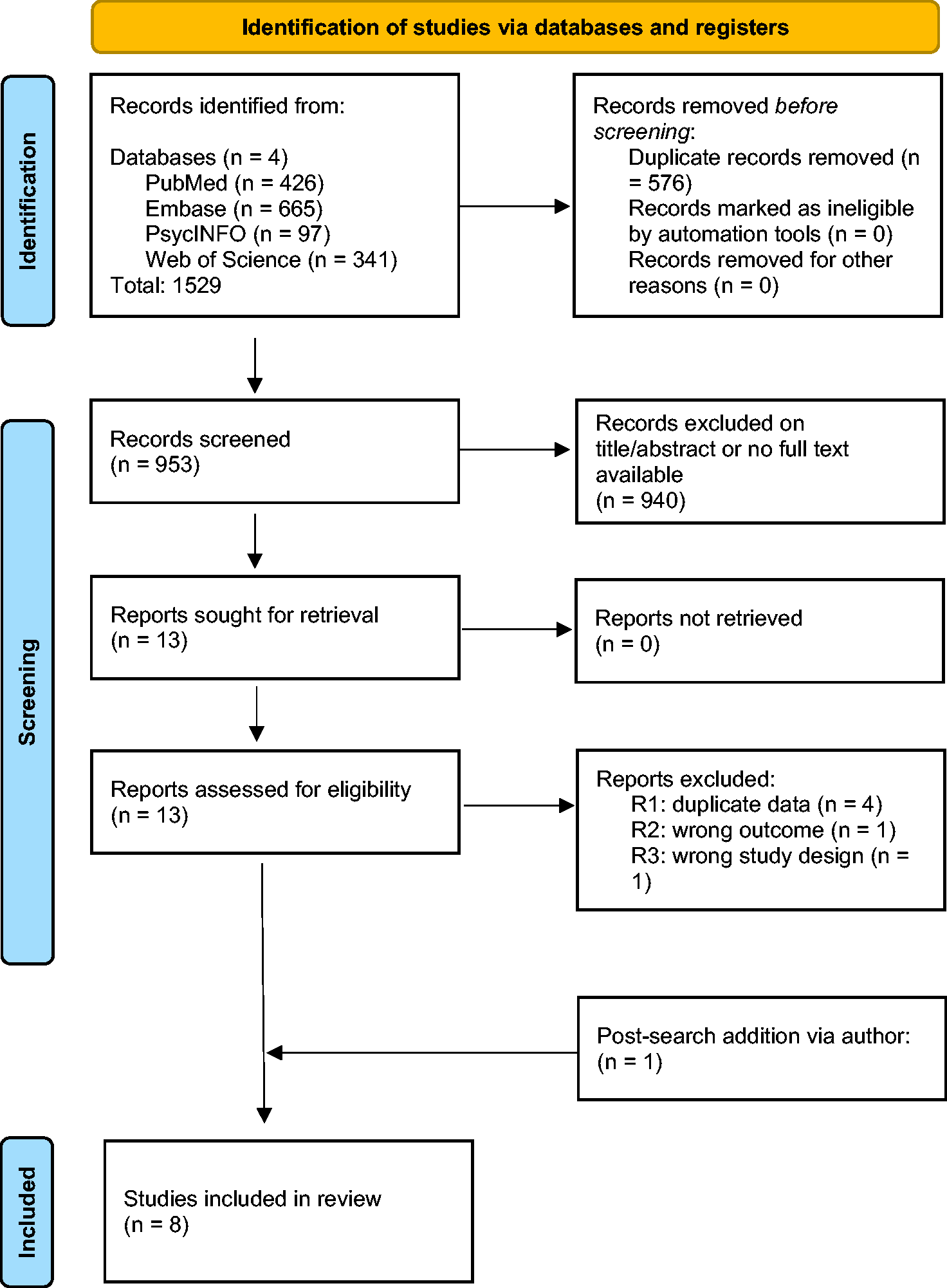
Flow diagram of included studies. N = number.
Study characteristics
An overview of the study characteristics of the eight included studies is presented in Table 1 and Supplementary Table S1. All studies were case control25–32 investigating dCA efficiency in patients with AD or MCI compared to healthy older adults. Despite the broad search, we found no studies that included other types of dementia. In total 279 patients (174 AD and 105 MCI) and 164 healthy controls participated in these studies. Few studies had an equal male to female ratio. The mean age varied from 65 to 76 years. Patients tended to be slightly older than controls, however, this difference was only significant in two studies.26,30
Characteristics of included studies.
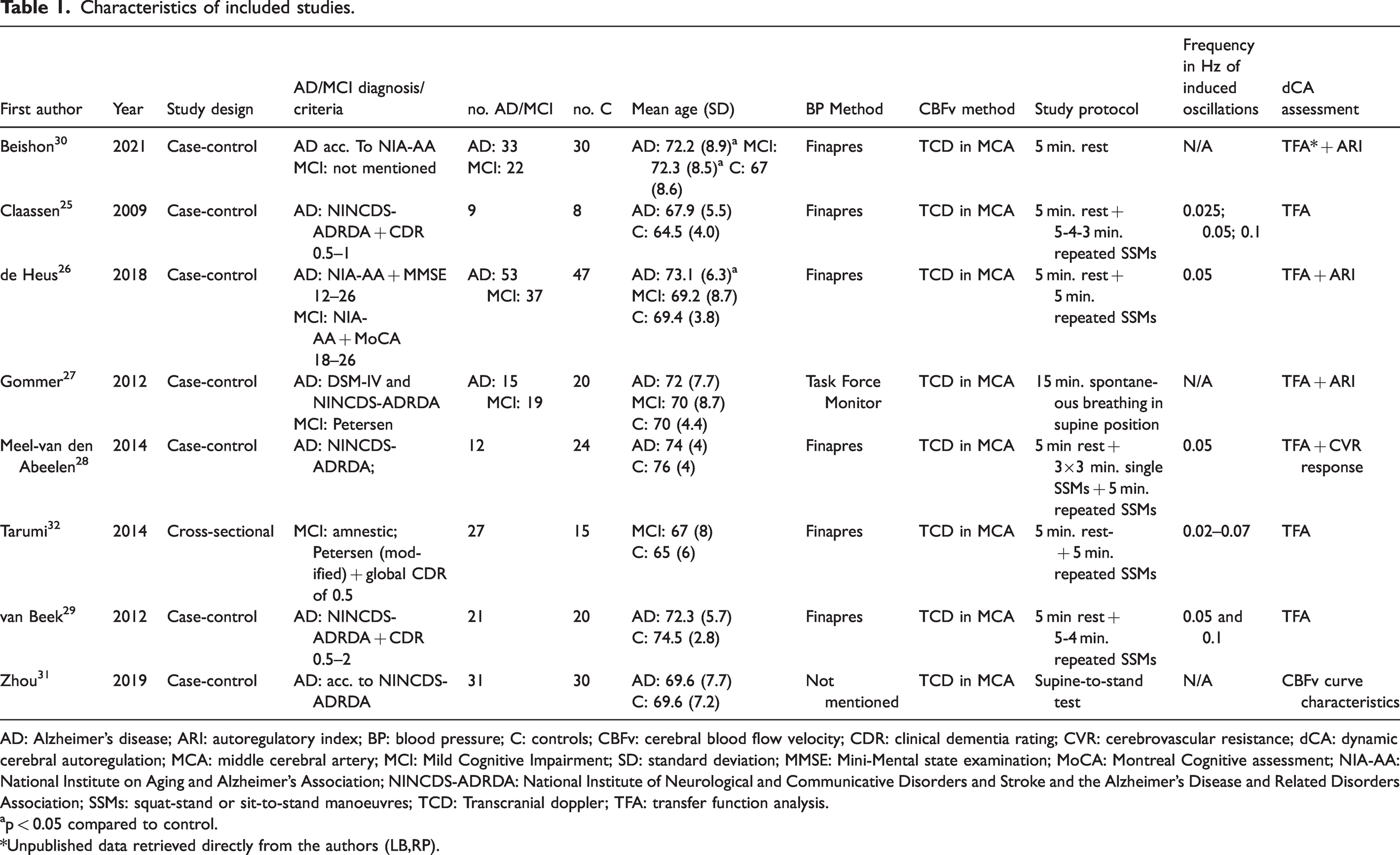
AD: Alzheimer’s disease; ARI: autoregulatory index; BP: blood pressure; C: controls; CBFv: cerebral blood flow velocity; CDR: clinical dementia rating; CVR: cerebrovascular resistance; dCA: dynamic cerebral autoregulation; MCA: middle cerebral artery; MCI: Mild Cognitive Impairment; SD: standard deviation; MMSE: Mini-Mental state examination; MoCA: Montreal Cognitive assessment; NIA-AA: National Institute on Aging and Alzheimer's Association; NINCDS-ADRDA: National Institute of Neurological and Communicative Disorders and Stroke and the Alzheimer’s Disease and Related Disorders Association; SSMs: squat-stand or sit-to-stand manoeuvres; TCD: Transcranial doppler; TFA: transfer function analysis.
ap < 0.05 compared to control.
*Unpublished data retrieved directly from the authors (LB,RP).
The diagnostic criteria used to classify AD and MCI patients varied between studies. The majority included patients with a diagnosis of probable AD according to the criteria of the National Institute of Neurological and Communicative Disorders and Stroke and the Alzheimer’s Disease and Related Disorders Association (NINCDS-ADRDA).25,27–29 Two studies used the National Institute on Aging and Alzheimer's Association (NIA-AA) criteria.26,30 These criteria were also used in one study to classify patients with MCI due to AD. 26 In two other studies, MCI was diagnosed according to the Petersen criteria,27,32 where one study restricted its population to solely amnestic MCI, 32 while one study did not specify their diagnostic method for MCI. 30
In all studies, participants underwent comprehensive clinical-neuropsychological evaluation. All studies used a validated cognitive assessment reporting scores for AD below validated thresholds, (e.g. MMSE score ≤25). In addition to age, sex, and education or cognition, data on BP, medication use, BMI, and vascular risk factors or comorbidities were collected. Most studies reported the use of antihypertensive drugs (AHDs) in patients and controls;25,26,28,29,32 some also reported the use of anti-dementia medication.25–27
Recently, preferred nomenclature was proposed for TCD assessments of CBF, as cerebral blood velocity (CBv) rather than flow-velocity (CBFv). 33 Here however we have followed the nomenclature used in the original publications, which was CBFv. In seven studies CBFv was measured in the MCA using TCD together with continuous BP measurement using arterial volume clamping of the finger artery; Additionally, EtCO2 and heart rate (ECG) were monitored. In these studies, estimates of (normalized) gain, phase and coherence were determined using TFA between mean BP, and mean CBFv. One study combined CBFv with brachial BP measurement in supine and standing positions without monitoring other vital signs. 31
In five studies, beat-to-beat BP and CBFv were measured both during rest (spontaneous breathing) and during orthostatic challenges.25,26,28,29,32 These orthostatic challenges consisted of repeated SSMs at a specific interval to induce oscillations in BP and CBFv at around 0.05 Hz (LF to VLF range), where dCA is most efficient. In addition to the TFA parameters, three studies determined the ARI,26,27,30 one study looked at the CBFv curve characteristics, and one study examined the response in CVR during induced changes in BP as a measure of dCA effectivity. 28
Quality assessment
The quality assessment is presented in Table 2. Overall quality was deemed good in four studies, fair in one study and poor in three studies. In all studies, cases or samples were clearly defined, however in two studies they were not clearly representative.27,29 Moreover, in three studies controls were not selected from the same community, or the definition was unclear or absent.25,26,28 Only one study reported sample size or power calculations, 26 although this is not a criterion in the Newcastle-Ottawa Scale. Three studies did not control for confounding variables by matching or covariate analysis.30–32 Generally, BP and CBFv were measured with reliable methods.
Quality assessment of the case-control studies.
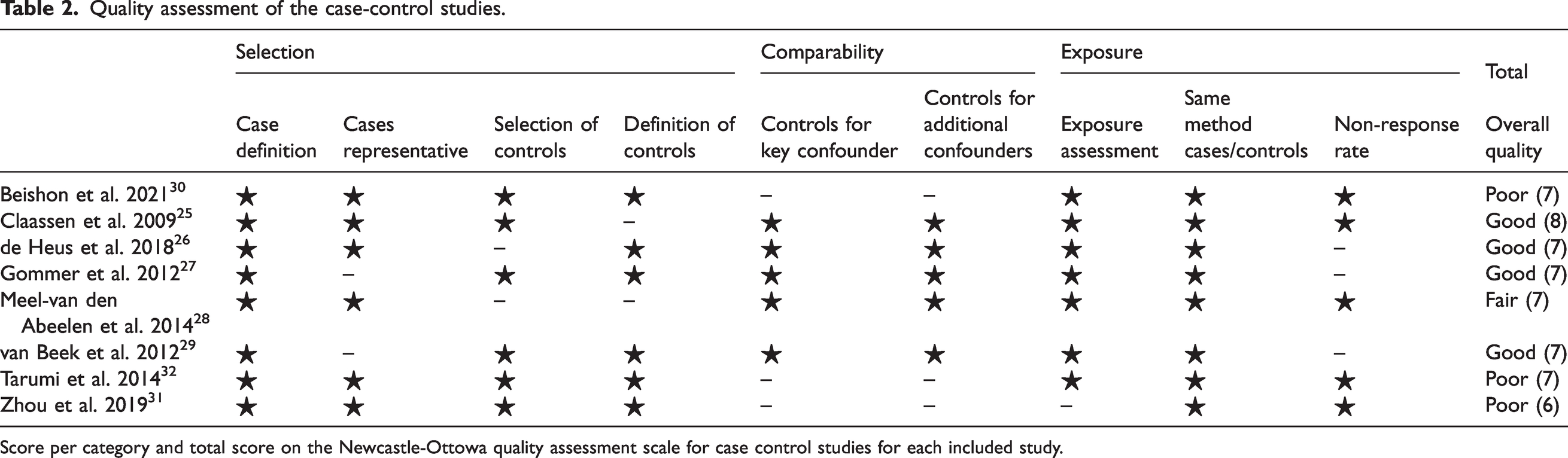
Score per category and total score on the Newcastle-Ottowa quality assessment scale for case control studies for each included study.
Qualitative synthesis
TFA quantifies how fluctuations in BP (input) are transmitted to CBFv (output) in three different parameters: gain, phase and coherence. Reduction in transfer function gain and increase in phase between BP and CBFv oscillations are interpreted as effective autoregulation. Coherence quantifies to what extent the changes in CBFv are associated with those in BP and is often used as a quality criterium. The ARI ranges between theoretical values of zero (no CA) and 9 (perfect CA), where a value ≥4 can be considered as functional CA. Hence, impaired dCA is indicated by higher gain, lower phase, and lower ARI.
In summary, two of the eight included studies reported impaired CA in AD patients compared to controls, each using non-TFA methods of dCA assessment.28,31 However, the TFA parameters phase and gain, and the ARI generally did not significantly differ between patients (AD and MCI) and healthy controls, nor between patients with AD and MCI. Four studies additionally demonstrated that orthostatic challenges resulted in greater TFA coherence between BP or MAP and CBFv compared to rest.25,26,29,32 None of the studies reported significant differences in coherence between groups. A more in-depth overview of the results is given below.
Dementia versus control
Four of the six studies investigating dCA in AD patients reported no alteration in autoregulatory function based on TFA outcomes or the ARI.25–27,29 Only one of these studies estimated the ARI during an orthostatic challenge, demonstrating a lower ARI in the control group compared to AD patients (p = 0.011), but no significant difference in (normalized) gain or phase. 26 Alternatively, Zhou et al. reported significant differences in the CBFv curve of AD patients compared to controls during the supine-to-stand test reflecting disease-related autoregulatory dysfunction. 31 Similarly, in the study by Meel-van den Abeelen et al. patients showed insufficient vasodilatory and constrictor responses (decrease/increase in CVR) to induced BP changes, resulting in larger fluctuations in CBFv compared to controls. 28 However, in this same study, there were no differences between patients and controls in the TFA parameters during spontaneous respiration.
MCI versus control
Three studies compared the dCA parameters phase, gain, and the ARI between patients with (amnestic) MCI and healthy controls during rest, showing no significant impairment in autoregulation in the patient group.26,27,32 In contrast, Beishon et al. reported a significantly lower ARI in patients with MCI compared to controls. 30 During orthostatic challenges, de Heus et al. found the ARI to be significantly lower (p = 0.015) and gain to be significantly higher (p = 0.017) in the control group compared to MCI patients, whereas phase was comparable across groups. 26 Tarumi et al. demonstrated no differences in gain or phase between patients with MCI and controls for induced BP fluctuations. 32
Dementia versus MCI
In the three studies assessing dCA in both AD and MCI patients, no significant difference in dCA efficiency during spontaneous BP fluctuations was found between these patient groups.26,27,30 Only in the study by de Heus et al. dCA parameters were estimated during orthostatic challenges, showing higher normalized gain in dementia patients, but no differences in phase or the ARI between both groups. They also performed stratification of AD and MCI patients by AHD use (yes or no), and high and low cerebrovascular or cardiovascular burden (based on cerebral vasomotor reactivity and orthostatic systolic BP recovery), but again found no differences in dCA parameters between these subgroups.
Meta-analysis
Dementia versus control
Spontaneous fluctuations
Neither the ARI (MD: 0.10 [95% CI, −0.78, 0.98], p = 0.82; Figure S3), nor TFA in the VLF range (phase MD: 0.02 [95% CI, −0.07, 0.11], p = 0.71, gain SMD: 0.10 [95% CI, 10.18, 0.38], p = 0.48) or LF range (phase MD: 0.04 [95% CI, −0.08, 0.16], p = 0.51, gain SMD: 0.18 [95% CI, −0.28, 0.64], p = 0.45; Figure 2 and S1) showed a significant difference between AD patients and controls. EtCO2 was comparable across groups (MD: −0.13 [95% CI, −2.16, 1.90], p = 0.90; Figure 3). Heterogeneity was low and non-significant in the VLF range (phase I2 = 3%, gain I2 = 0%), moderate and non-significant in the LF range for gain (I2 = 57%), but high and significant in the LF range for phase (I2 = 80%, p = 0.0005) and the ARI (I2 = 81%, p = 0.005). For EtCO2, heterogeneity was high and significant (I2 = 72%, p = 0.006).
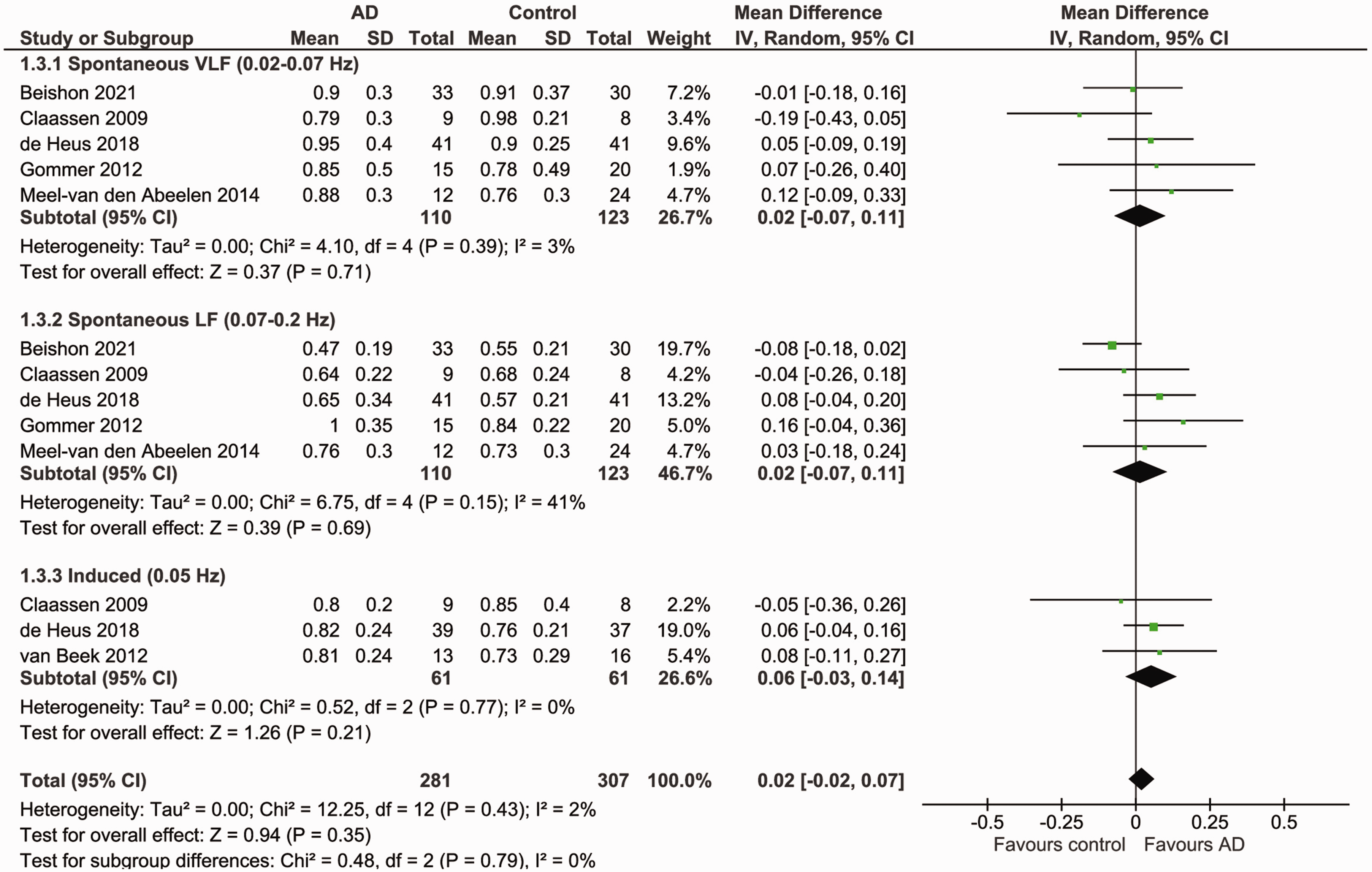
Forest plot for a meta-analysis of phase (radians) during spontaneous and induced BP fluctuations in AD patients versus controls. Data is pooled from a total of six studies. Results are reported as mean differences (95% CI). Statistical significance is set at p < 0.05. Higher phase indicates better dCA functioning. Results show no significant difference in phase between AD patients and controls during both spontaneous and induced BP fluctuations. Heterogeneity was low and non-significant in the spontaneous VLF range (I2 = 3%, p = 0.39) and for induced fluctuations (I2 = 0%, p = 0.77), and moderate and non-significant in the spontaneous LF range (I2 = 41%, p = 0.15). AD: Alzheimer’s disease; BP: blood pressure; dCA: dynamic cerebral autoregulation; LF: low frequency; VLF: very low frequency.
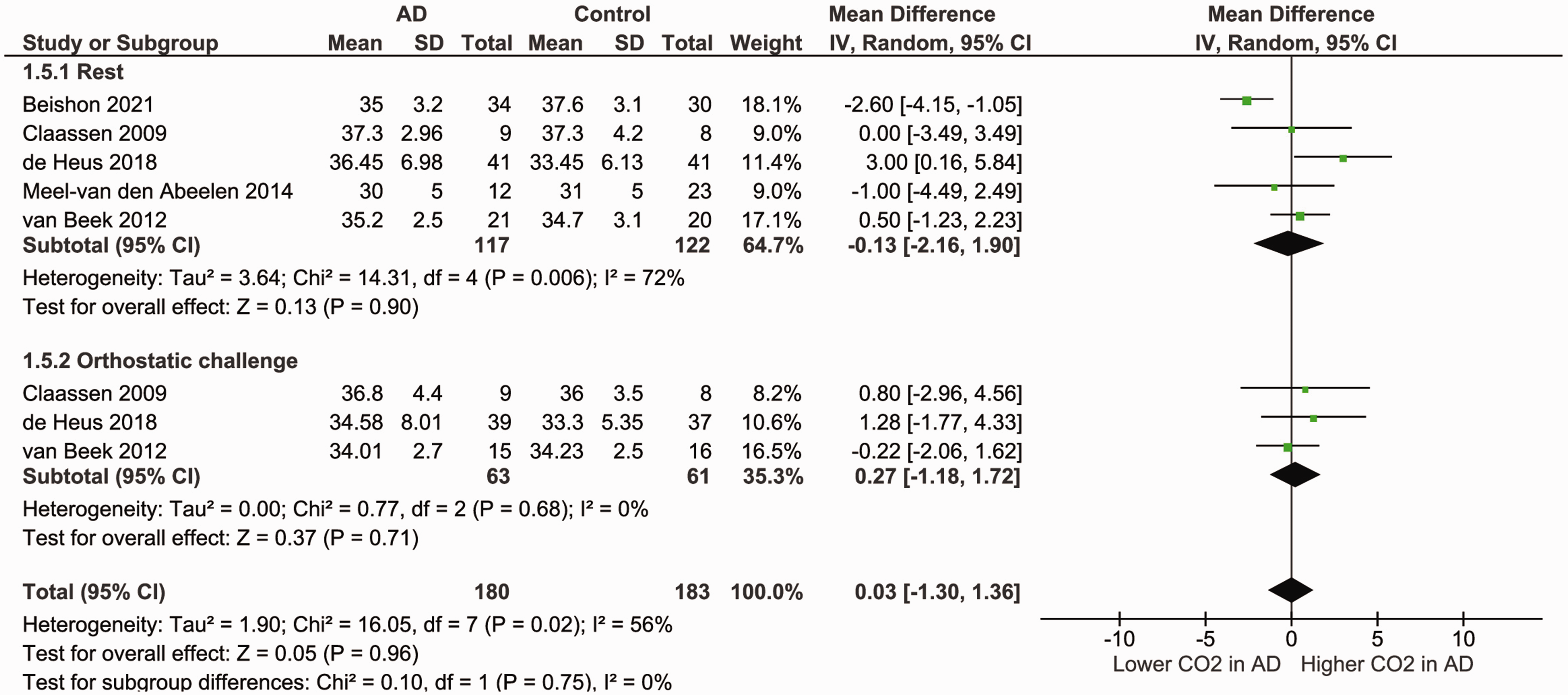
Forest plot for a meta-analysis of EtCO2 (mmHg) during rest and orthostatic challenge in AD patients versus controls. Data is pooled from a total of five studies. Results are reported as mean differences (95% CI). Statistical significance is set at p < 0.05. Results show no significant difference in EtCO2 between AD patients and controls during either rest or orthostatic challenge. Heterogeneity was high and significant during rest (I2 = 72%, p = 0.006), but low and non-significant during orthostatic challenge (I2 = 0%, p = 0.68). AD: Alzheimer’s disease; EtCO2: end-tidal carbon dioxide.
Induced fluctuations
Both phase and gain were comparable between patients and controls (phase MD: 0.06 [95% CI, −0.03, 0.14], p = 0.21, gain MD: −0.07 [95% CI, −0.29, 0.16], p = 0.56; Figure 2 and S2). There were also no differences in EtCO2 (MD: 0.27 [95% CI, −1.18, 1.72], p = 0.71). Heterogeneity was low and non-significant for phase (I2 = 0%, p = 0.77) and EtCO2 (I2 = 0%, p = 0.68), but high and almost significant for gain (I2 = 66%, p = 0.05).
MCI versus control
Spontaneous fluctuations
In both the VLF and the LF range, neither phase (MD: −0.12 [95% CI, −0.27, 0.03], p = 0.11, MD: 0.02 [95% CI, −0.10, 0.14], p = 0.74 respectively; Figure 4), nor gain (SMD: −0,02 [95% CI, −0.38, 0.34], p = 0.91, SMD: −0.04 [95% CI, −0.53, 0.45], p = 0.88 respectively; Figure S4) differed in patients with MCI compared to controls. Similarly, there was no significant difference in the ARI between both groups (MD: −0.07 [95% CI, −1.14, 0.99], p = 0.89; Figure S5) or in EtCO2 (MD:1.00 [95% CI, −2.39, 4.40], p = 0.56; Figure 5). Heterogeneity was moderate and non-significant in the VLF range for phase (I2 = 55%) and gain (I2 = 39%), but high and significant in the LF range (phase I2 = 89%, p < 0.00001, gain I2 = 66%, p = 0.03), for the ARI (I2 = 88%, p = 0.0003), and for EtCO2 (I2 = 87%, p = 0.0004). There were too few studies (< 3) for meta-analyses of TFA parameters phase and gain, and for EtCO2 for induced fluctuations.
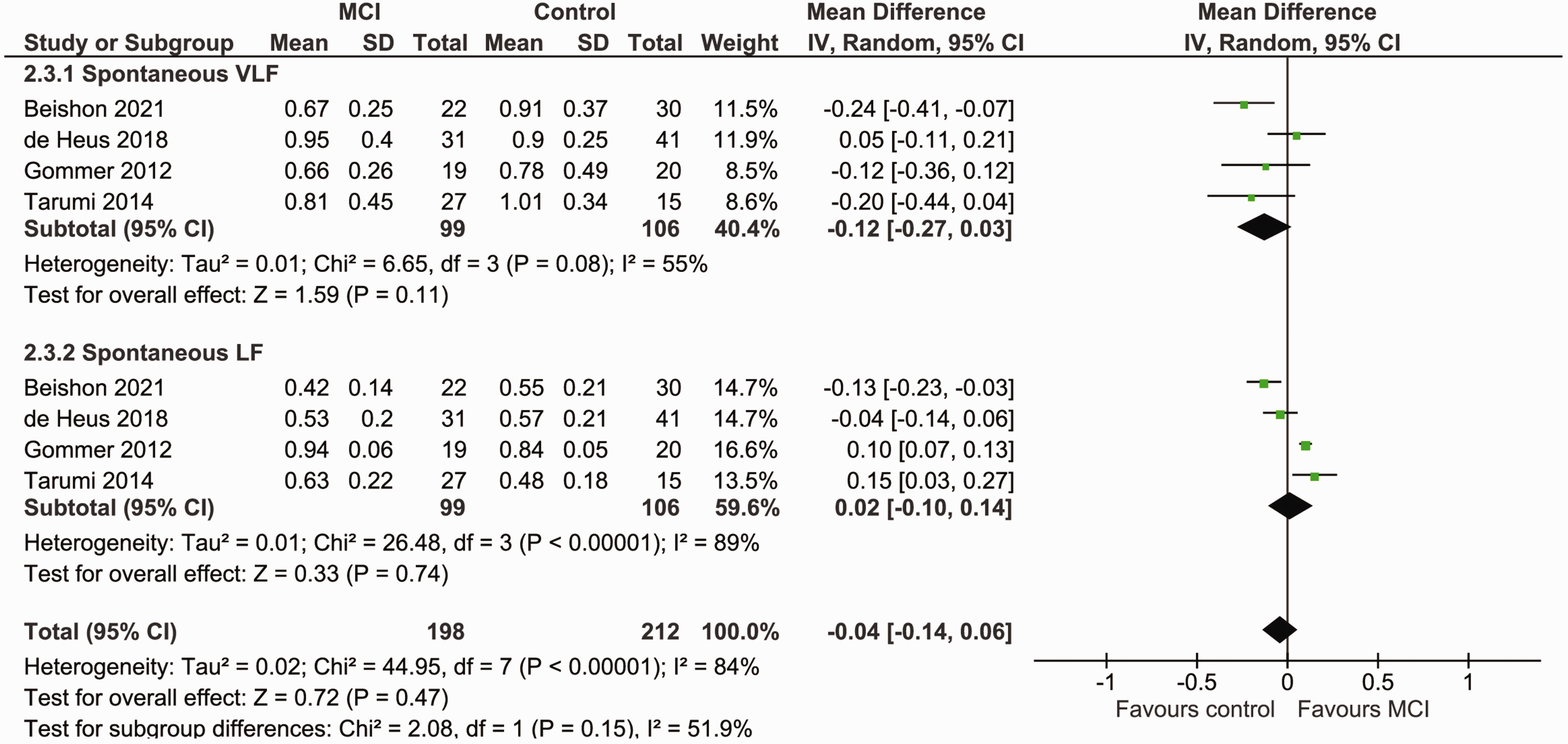
Forest plot for a meta-analysis of phase (radians) during spontaneous BP fluctuations in MCI patients versus controls. Data is pooled from a total of four studies. Results are reported as mean differences (95% CI). Statistical significance is set at p < 0.05. Higher phase indicates better dCA functioning. Results show no significant difference in phase between MCI patients and controls during spontaneous BP fluctuations. Heterogeneity was moderate and non-significant in the VLF range (I2 = 55%, p = 0.08), but high and significant in the LF range (I2 = 89%, p < 0.00001). BP: blood pressure; dCA: dynamic cerebral autoregulation; LF: low frequency; MCI: mild cognitive impairment; VLF: very low frequency.
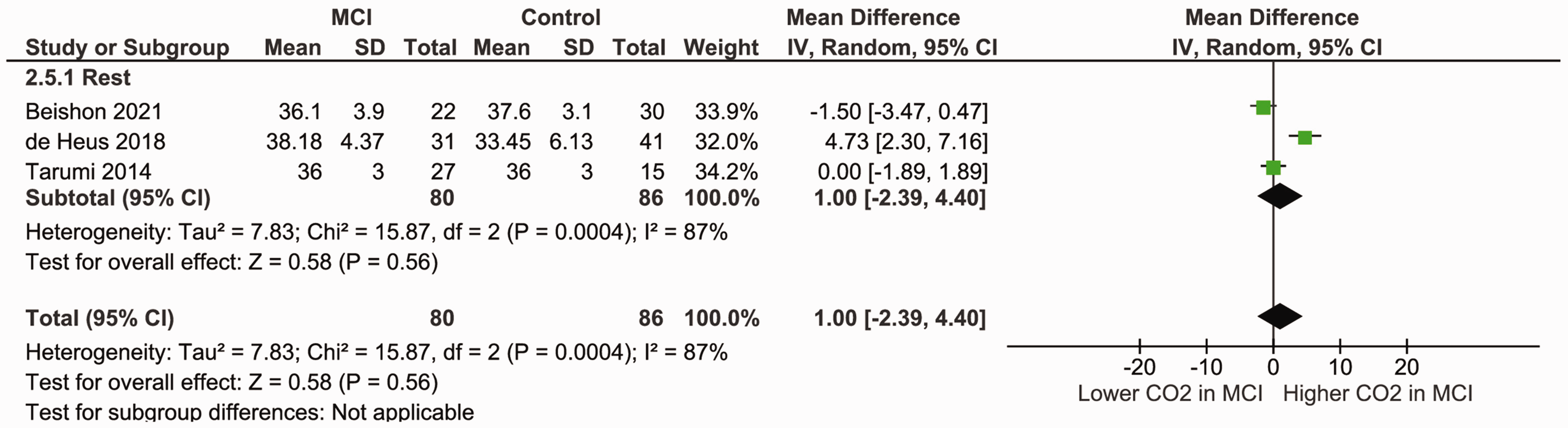
Forest plot for a meta-analysis of EtCO2 (mmHg) during rest in MCI patients versus controls. Data is pooled from a total of three studies. Results are reported as mean differences (95% CI). Statistical significance is set at p < 0.05. Results show no significant difference in EtCO2 between MCI patients and controls during rest. Heterogeneity was high and significant (I2 = 87%, p = 0.0004). EtCO2 = end-tidal carbon dioxide, MCI = mild cognitive impairment.
Dementia versus MCI
Spontaneous fluctuations
Both phase and gain showed no significant difference between patient groups in the VLF range (phase MD: 0.14 [95% CI, −0.01, 0.29], p = 0.07, gain SMD: 0.03 [95% CI, −0.50, 0.55], p = 0.92; Figure 6 and S6). In the LF range, no significant difference in gain could be demonstrated (SMD: 0.01 [95% CI, −0.46, 0.49], p = 0.95), but phase was significantly higher in AD patients (MD: 0.06 [95% CI, 0.02, 011], p = 0.003). The ARI was comparable between groups (MD: 0.21 [95% CI, −0.17, 0.60], p = 0.28; Figure S7). Heterogeneity was low to moderate and non-significant (I2 ≤ 62%) for all analyses. There were too few studies (<3) for meta-analyses of TFA parameters phase and gain for induced fluctuations, and for comparison of EtCO2.
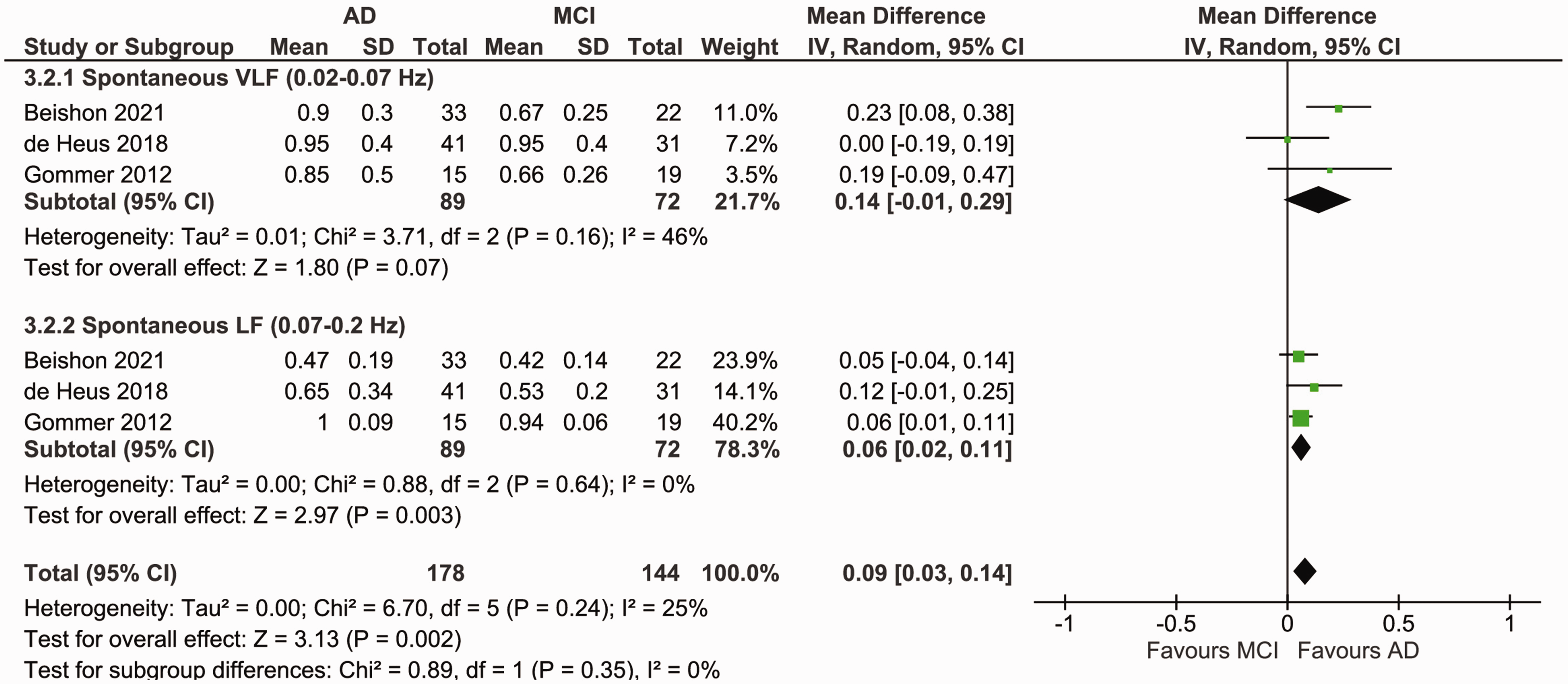
Forest plot for a meta-analysis of phase (radians) during spontaneous BP fluctuations in AD versus MCI patients. Data is pooled from a total of three studies. Results are reported as mean differences (95% CI). Statistical significance is set at p < 0.05. Higher phase indicates better dCA functioning. Results show no significant difference in phase between both patient groups in the VLF range, but significantly higher phase in AD patients in the LF range (p = 0.003). Heterogeneity was moderate and non-significant in the VLF range (I2 = 46%, p = 0.16), and low and insignificant in the LF range (I2 = 0%, p = 0.64). AD: Alzheimer’s disease; BP: blood pressure; dCA: dynamic cerebral autoregulation; LF: low frequency; MCI: mild cognitive impairment; VLF: very low frequency.
Discussion
Summary of results
This systematic review evaluated dynamic autoregulatory function in patients with dementia and MCI. Overall, we identified no difference in dCA efficiency during rest, estimated by the ARI and the TFA parameters gain and phase, between any of the participant groups. Data on autoregulatory function during orthostatic challenge were scarce but showed a trend towards better autoregulation in patients (AD and MCI) compared to controls. In contrast, two studies demonstrated that dCA was not intact during orthostatic challenge using alternative assessment methods, of which the reliability is unknown. These studies were therefore not eligible for the meta-analysis. None of the studies reported to have looked at EtCO2 as a co-factor to explain differences (or lack of) between patients and controls, however a meta-analysis of the available data on EtCO2 showed no differences between the groups during rest or postural changes. While these studies confirm the presence of reduced (global) CBF and increased CVR in (preclinical) AD, they give no reason to assume that dCA is affected by AD and MCI pathology, particularly in light of the meta-analysis findings.
Implications of findings
Evidence pointing to the involvement of cerebrovascular dysfunction in AD pathology has led to the hypothesis that CA may be impaired in this disease and other dementias. Multiple studies have shown that patients with AD and VaD present with reduced CBF, increased CVR, and impaired vascular CO2 reactivity and neurovascular coupling (NVC).5,9,11,34 These findings are already present in the preclinical stage and contribute to disease progression and accelerated cognitive decline.5,9 While the included studies confirm some of these vascular alterations (lower CBF, elevated CVR), this review shows that dynamic autoregulation is preserved in AD and MCI. Importantly, it should be noted that the quality of these results was graded as relatively low due to the observational nature of the included studies. However, for the purpose of establishing whether cerebral autoregulation is impaired or not in Alzheimer patients, observational case-control studies can still provide valid evidence.
MCI is often considered a prodromal phase for the development of different forms of dementia, depending on the pattern of cognitive deficit at presentation. For example, purely amnestic MCI has a greater risk of progressing to AD dementia, where-as multi-domain MCI is more likely to progress to vascular dementia. In this review, the majority of participants in the primary studies had amnestic sub-type of MCI, meaning they are more representative of AD prodrome. However, we identified very few differences between MCI and AD in dCA parameters, except phase at the LF range, which was higher in AD. This may represent a compensatory improvement in autoregulatory efficiency in people with dementia relative to MCI, but due to a lack of functional or cognitive data this is speculatory, and not a consistent pattern seen across parameters. Importantly, the pattern of dCA preservation appears to be consistent across both MCI and AD, suggesting it is maintained throughout a spectrum of cognitive dysfunction.
These findings suggest that patients with MCI or dementia will not experience perfusion deficits due to relatively fast changes in BP, for example during the BP fluctuations caused by postural changes. This dynamic autoregulation differs from static autoregulation, which describes the effect of much slower changes in BP on CBF, e.g. the gradual reduction in BP from hypertension to normotension over weeks. While it is conceivable that dynamic and static autoregulation are connected, the results of this review cannot directly be generalized to indicate the preservation of sCA in AD. This is supported by a study in healthy older adults that found no correlation between dCA and sCA parameters measured in the same subject. 35 However, the only study that investigated static autoregulation in AD, using BP lowering treatment, found no impairment in sCA. 36 Therefore, we postulate that sCA remains intact in AD and MCI based on earlier findings,9,36 and supported by the fact that many participants included in this review were using AHDs. Additionally, a comparison between patients using AHDs and patients not using these drugs did not change the results. 26
Contrary to earlier findings indicating possible harmful effects of BP-lowering treatment in patients with dementia and MCI,16,17 a recent clinical trial and meta-analysis reported a lower risk of developing MCI or dementia in those treated with AHDs.37,38 Notably, the beneficial effect of AHT in that study was dependent on baseline cognitive functioning; poorer functioning resulted in no or limited treatment effects indicating the importance of early AHT initiation. Thus, fear of causing perfusion deficits should not preclude treatment of hypertension, although further work on sCA in dementia and MCI is still required.
Different interconnected mechanisms, including CA, NVC, and vascular CO2 reactivity, together regulate CBF by altering CVR through vasoconstriction or vasodilation in response to different stimuli. It may therefore be unexpected to find that CA is preserved in AD, while CVR, NVC and CO2 reactivity are affected by this disease. Research indicates that autoregulation is profoundly affected by vascular tone, which in turn is largely dependent on partial pressure of arterial O2 and CO2. As a result, the presence of hypocapnia, inducing cerebral vasoconstriction, can augment the dCA response, potentially masking autoregulatory deficiency.39,40 However, the meta-analysis of EtCO2 data gave no evidence to support the presence of hypocapnia in dementia and MCI patients. Nevertheless, more research into the potential influence of hypocapnia on autoregulatory function in dementia patients is necessary, since none of the studies included in this review reported to have controlled for the influence of CO2 in their analyses.
Not all segments of the vascular tree contribute equally to overall vascular resistance, placing greater importance on the microvasculature than on larger vessels.9,26 A possible explanation could therefore be that AD pathology predominantly affects the microvasculature and not the proximal vessels, such that total CVR is increased while CA remains functional. Alternatively, some studies in this review reported better CA in patients compared to controls, particularly for induced (larger) BP fluctuations, without reporting differences in EtCO2 between these groups. Possibly, the autoregulatory mechanism may have improved efficiency in the early stages of the disease to compensate for the progressive loss of function, as seen with other parameters such as compensatory hyperperfusion and NVC in people with MCI.13,14,41 As the participants included in this study were mostly diagnosed with mild-to-moderate AD with mean age <80 years, it is possible that in later stages of the disease, CA will also be notably affected.
Moreover, evidence points to endothelial involvement in NVC and CO2 reactivity, while CA is mostly dependent on the myogenic response of vascular smooth muscle.9,42,43 It should be noted, though, that the exact biological mechanisms underlying each of these mechanisms are still to be elucidated. Interestingly, in cerebral amyloid angiopathy (CAA), Aβ accumulation causes toxicity to vascular cells, predominantly affecting smooth muscle cells instead of endothelial cells. 44 Since at least half of AD patients are expected to have concomitant moderate-to-severe CAA, 45 it would be interesting to further investigate whether CA remains intact when CAA is present.
Our observations are in contrast with findings from animal models,7,19 but important differences exist that may explain the discrepancy between these results. Most importantly, all included participants had sporadic late-onset AD, while animal models of AD are rather models for the rare early-onset genetic form of the disease. 46 Moreover, autoregulation in rodents is commonly studied using drug-induced stepwise elevation or reduction in MAP, which more resembles sCA instead of dCA. Lastly, animal models often do not fully reproduce the complete pathology of the disease, which has hampered successful translation of the results to the clinic in the past more often than not. 47
Strengths and limitations
Study quality was not always good, and the observational nature of the studies, small sample sizes and the methods used inherently have their own biases. All but one study combined continuous BP measurements and TCD with TFA as the method of dCA assessment, reducing methodological heterogeneity. TCD measurement can be seriously influenced by exogeneous noise signals, therefore some studies included repeated SSMs to increase the reliability of the CA parameters. Changes in velocity measured using TCD are assumed to reflect changes in CBF provided that the vessel diameter remains relatively constant, despite fluctuations in BP or CO2. Moreover, these measurements do not take into account brain tissue weight and only reflect changes in flow of the perfused area of the insonated vessel (the MCA), which both may be affected by AD and MCI pathology. 10 Additionally, in all studies, cerebral perfusion was solely measured in the MCA, which may not be reflective of the whole cerebrovascular bed. However, a study investigating changes in CBF using PET after BP lowering in AD found no regional differences. 36 Most studies did use similar protocols, including a resting period before starting TCD measurements during similar periods of spontaneous breathing or repeated SSMs. All but one study additionally recorded EtCO2 and did not report significant changes in CO2 or significant differences between groups that may have been present in rest or occurred as a result of postural changes.
The diagnostic criteria for AD and MCI and inclusion and exclusion criteria varied between studies, although MMSE scores were comparable. Moreover, in the majority of the studies, correct selection or definition of controls was absent. Together, this may have introduced heterogeneity in the studied populations, hampering reliable comparison of study results. Individual study sample sizes were small and power calculations were generally missing. By performing meta-analyses, however, sample sizes were increased to assume we were able to reliably detect (mild) impairment in CA in the absence of differences in EtCO2. 48 Lastly, with our search we found no studies looking at CA in other forms of dementia besides AD, limiting the scope of our review, albeit revealing a significant gap in the literature.
Further work
There is no clear evidence to assume that dynamic autoregulation is impaired during spontaneous BP fluctuations in patients with AD and MCI. Data on dCA efficiency during induced BP fluctuations are more limited but indicate at least preserved and possibly better autoregulatory function in people with AD or MCI. Moreover, there seems to be no significant difference in the presence of hypocapnia between patients and controls. More studies are needed to confirm this, preferably expanding the studied population to other forms of dementia and more advanced stages of AD. Ideally, to avoid methodological differences future studies should perform repeated within-subject comparisons while controlling for EtCO2 changes in the analyses to investigate autoregulatory functioning. Moreover, study protocols should adopt orthostatic challenges such as repeated SSMs to increase the reliability of dCA assessment and determine whether CA may function differently in rest compared to postural changes.
In summary, studies of dCA in dementia and MCI are limited and predominantly observational in nature with varying study quality and small sample sizes. Based on the available evidence however, this review indicates intact dCA in patients with (mild-to-moderate) AD or MCI.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X231173449 - Supplemental material for Dynamic cerebral autoregulation in Alzheimer’s disease and mild cognitive impairment: A systematic review
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X231173449 for Dynamic cerebral autoregulation in Alzheimer’s disease and mild cognitive impairment: A systematic review by Rachel Heutz, Jurgen Claassen, Sanne Feiner, Aaron Davies, Dewakar Gurung, Ronney B Panerai, Rianne de Heus and Lucy C Beishon in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
