Abstract
Glycosylation of lipids and proteins significantly increases the molecular diversity in the brain. Membrane-localized glycoconjugates facilitate critical neuro-immune interactions. Therefore, glycodysregulation is increasingly recognized as a novel hallmark of various acute and chronic neurological diseases. Although RNAs are heavily modified, they are never thought to be substrates for glycosylation due to their inaccessibility to the glycosylation machinery in the Golgi apparatus. The astonishing discovery of cell surface glycoRNAs opened new avenues for glycomedicine. This review highlighted the key features of GlycoRNAs and further discussed their potential immunomodulatory role in the brain, particularly focusing on post-stroke neuroinflammation.
Introduction
All the fundamental building blocks of the cell, including nucleic acids, proteins and lipids, can undergo modifications to produce functionally diverse macromolecules. 1 Particularly, glycoconjugates formed by the covalent linkage of carbohydrate chains to lipids and proteins are abundant in the brain. 2 Glycosylation occurs in a highly regulated and non-templated fashion in the endoplasmic reticulum (ER) and Golgi apparatus, and the glycoconjugates are either trafficked to the cell membrane forming glycocalyx or secreted to mediate cell-cell interactions. 3 Glycosylation is a reversible process where the glycan chains are synthesized by one or more unique glycosyltransferases depending on the linkage monosaccharide unit and removed by glucosidases. 2 Cellular glycome diversity is primarily attributed to the type of glycosidic linkage (α- or β-forms), degree of branching, length and monosaccharide composition. 4 Importantly, glycosylation patterns differ significantly across brain regions, age and sex.5–7 Glycosylation regulates developmental (neural cell adhesion and axon targeting), physiological (synaptic transmission) and pathological (neuroinflammation) processes in the brain.8–10
Although proteins and lipids are extensively glycosylated, RNAs are never thought to be pervasively glycosylated as they are predominantly localized in the nucleus or cytoplasm and do not have access to glycosylation machinery. However, certain tRNAs from rabbit liver were observed to be modified with galactose or mannose at the wobble base position. 11 A recent study showed a high prevalence of stable glycosylated RNAs (glycoRNAs) in cultured cells and tissues. 12 A staggering finding of this study is the cell surface display of glycoRNAs, resulting in their direct interaction with immune cell receptors. 12 We presently discuss the immunomodulatory potential of glycoRNAs in the post-stroke brain.
RNA glycosylation is a novel epitranscriptomic modification
Of a plethora of chemical modifications RNAs can undergo, methylation is the most abundant and widely studied epitranscriptomic modification.
13
Notably,
In mammals, most glycans are composed of 10 kinds of monomeric carbohydrate units, including glucose, galactose, mannose, sialic acid, fucose,
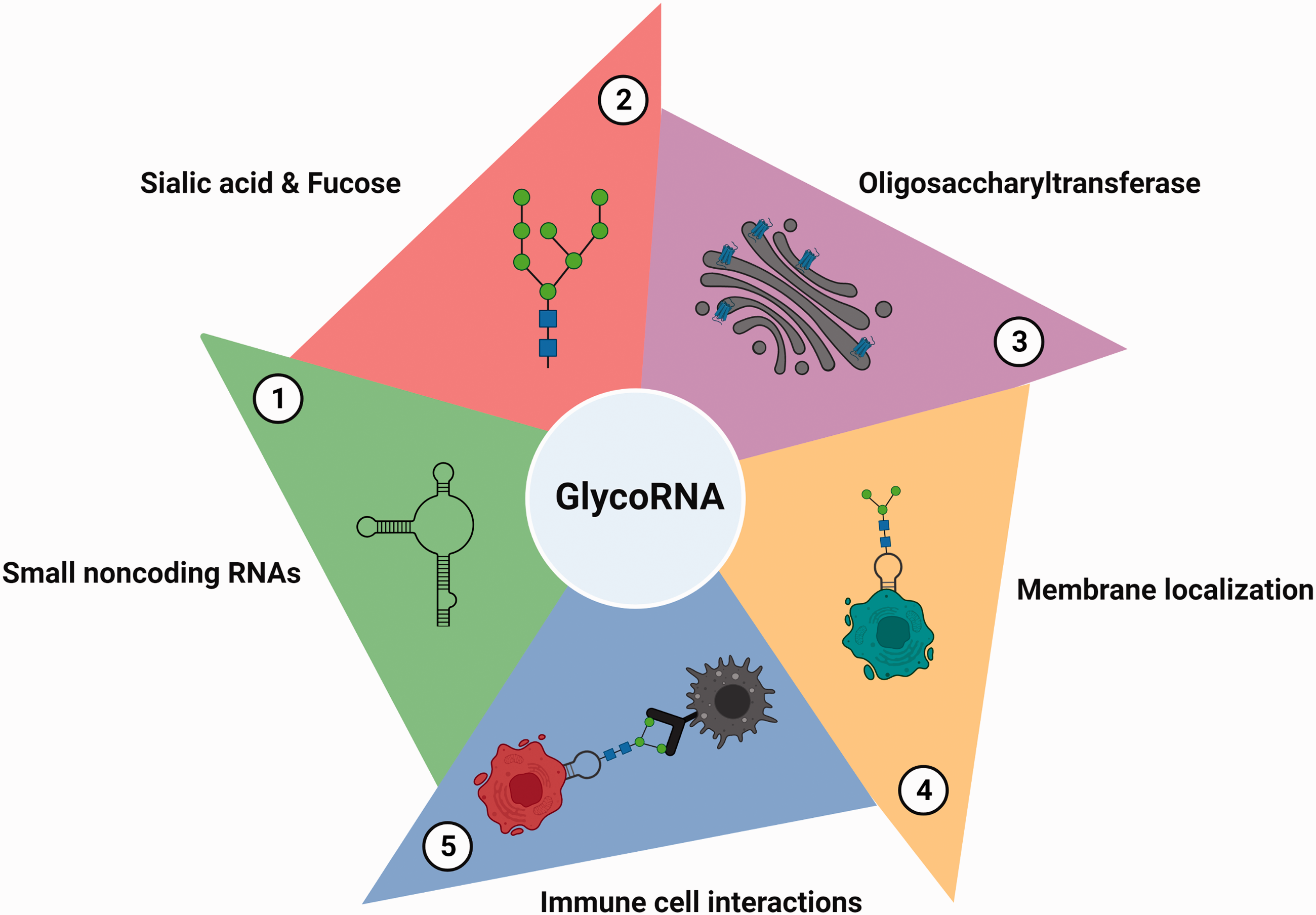
Y-RNAs are major targets for glycosylation as deletion of Y-RNA gene Y5 decreased Ac4ManNAz incorporation by ∼30% in RNA extracts from HEK 293 T cells. 12 Y-RNAs form a class of highly conserved small noncoding RNAs (∼110 bps) with a precedential role in immunoregulation. 24 Interestingly, Y-RNAs represent ∼63% of the circulating RNAs in humans and their levels correlate positively with the progression of atherosclerosis, which is a major risk factor for ischemic stroke. 24 Specifically, Y-RNA fragments activate macrophages and promote inflammation during atherosclerosis. 25 The human genome encodes 4 Y-RNA transcripts (Y1, Y3, Y4 and Y5), which are highly expressed in the brain and heart. 26 Although relatively short in length, Y-RNAs fold into characteristic stem-loop secondary structures and bind to several proteins like Ro60, La, IGF2BP1 and PUF60 to control DNA replication, stress response, ribonucleoprotein surveillance and mRNA stability. 26 Precise role of Y-RNAs in the brain is not understood, but their levels were shown to be altered in many neurological diseases. For instance, Y-RNA fragments are enriched in the exosomal RNA from the patient-derived glioma cells. 27 Whereas Alzheimer’s disease patients showed a decreased abundance of Y-RNA fragments in the frontal cortex compared to healthy subjects. 28 Additionally, plasma levels of Y4 and Y5 correlated with platelet function in patients with myocardial infarction. 29 Considering these reports, it will be interesting to analyze the Y-RNA glycosylation and its function after stroke and other CNS injuries.
Functional significance of glycoRNAs in the brain
Aberrant protein glycosylation is linked to various neuropathological conditions, including amyotrophic lateral sclerosis, Alzheimer’s disease, Huntington’s disease, gliomas and stroke.10,30–32 For example, stroke induces global protein glycosylation, specifically the linkage with β-
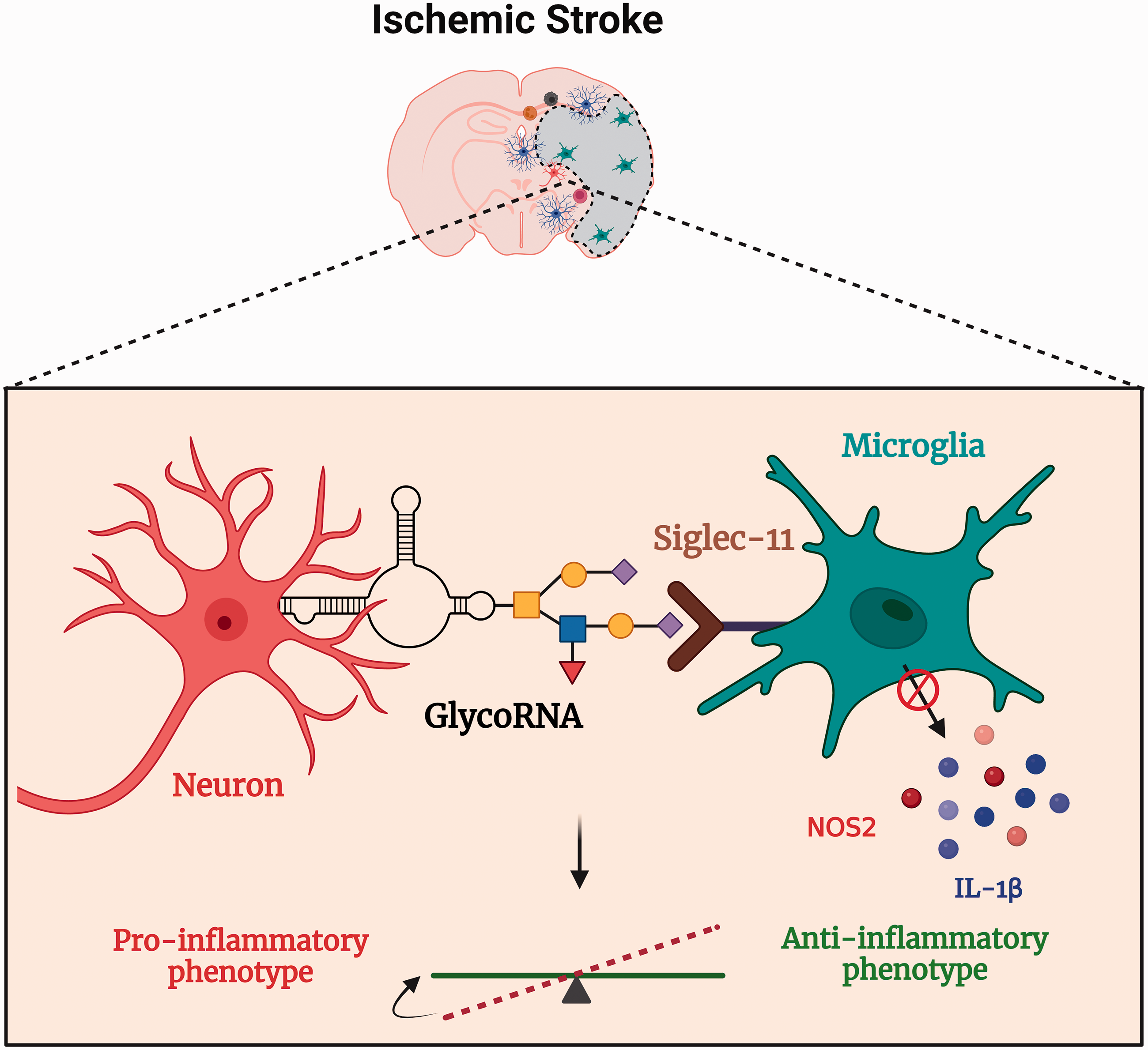
Mechanistically, glycan alterations are thought to fine-tune neuroinflammatory responses in conjunction with GBPs expressed on microglia. 10 Microglial GBPs recognize glycopatterns and initiate either pro- or anti-inflammatory cascades. Neuroinflammation after stroke is a double-edged sword triggering detrimental effects in the acute phase and beneficial outcomes in the chronic phase. 38 A plethora of immune cells and their mediators are shown to orchestrate this response. However, none of the treatments targeting acute immune response after stroke have proven beneficial in clinical settings. 39 Therefore, a better understanding of the post-stroke immunomodulatory mechanisms is needed to find efficacious therapeutic targets. Epitranscriptomic modifications are undoubtedly emerging as novel regulators of the immune response, with a myriad of downstream targets. 40 For example, we recently showed that the FTO-dependant m6A methylation regulates inflammatory transcripts like Tnf-α and shapes the post-stroke functional outcome. 41
GlycoRNAs emerge as vital epitranscriptomic regulators with tremendous potential in modulating the immune response in the brain due to high affinity for Siglec-11 and Siglec-14. 12 Siglecs are a class of GBPs with 14 members that recognize the sialic acids on the cell membrane. 42 Notably, Siglec-11 is highly expressed in the microglia and preferentially binds to neuronal glycoproteins such as polysialylated neuronal cell adhesion molecule (PSA-NCAM).43–45 More importantly, overexpression of Siglec-11 in the microglia diminished LPS-induced phagocytosis of apoptotic neurons, attenuating neurotoxicity. 43 Additionally, microglial Siglec-11 and neuronal PSA-NCAM interaction significantly decreased the expression of pro-inflammatory cytokine IL-1β and the nitric oxide synthase-2 isoform. 43 Given these observations, neuronal glycoRNAs might act parallel to glycoproteins and serve as ligands for microglial Siglec-11 at the immune synapses to initiate the anti-inflammatory cascades after stroke (Figure 2). Protective remodeling of microglia by glycoRNA/Siglec-11 axis could also be explored in neurodegenerative diseases like Alzheimer’s disease, Parkinson’s disease and multiple sclerosis. 46
Concluding remarks and future outlook
Future studies need to validate the role of glycoRNAs in post-stroke neuroinflammation by profiling glycoRNAome, capturing the glycoRNA/Siglec-11 interactions and assaying neuroinflammatory response after glycoRNA disruption with OST inhibitor NGI-1 in the ischemic brain. It is plausible that small glycoRNAs require less energy for biosynthesis than glycoproteins and hence can efficiently shift inflammatory disequilibrium in the brain after stroke. Glycosylation generates far more biomolecular diversity than any other chemical modification due to the polymeric glycans formed by the linkage and branching of distinct monosaccharide units in multiple permutations and combinations. More than 70% of the proteins and a significant fraction of lipids in the brain are glycosylated. 4 Being an immune-privileged organ, the brain utilizes glycoconjugates to potentiate intercellular communication during pathological conditions. 10 The newly discovered glycoRNAs add the exciting possibility of a higher order epitranscrptomic control of pathological mechanisms like inflammation after a CNS injury that helps to design novel therapies. Further research is necessary to define the nature of RNA-glycan linkage, their trafficking mechanisms, reversibility and extracellular interactome. Current glycoRNA profiling relies on specific reporter sugar probes, and hence untargeted strategies are necessary to identify the novel conjugates. Additionally, the bioactivity of the glycoconjugates can be modulated by the attachment of labile functional groups like sulfate or phosphoryl groups and this aspect remains to be tested for glycoRNAs. 47 As microbes forage for host glycans, it will be worth exploring the relationship between glycoRNAs and the gut microbiome. Several studies assessed the efficacy of glycans as tumor biomarkers due to their high abundance in eukaryotic body fluids.31,47 For instance, human plasma contains ∼2 mM protein-bound sialic acids and their levels correlate positively with brain tumor malignancy.31,47 Therefore, mapping the sialylated glycoRNAome of patients with neurological diseases will provide more insights regarding their clinical utility.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies were supported in part by the Department of Neurological Surgery, University of Wisconsin-Madison and the National Institute of Health (RO1 NS109459). Dr. Vemuganti is the recipient of a Research Career Scientist Award (IK6BX005690) from the US Department of Veterans Affairs.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
