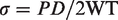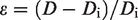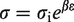Abstract
Age-related increases in large artery stiffness are associated with cerebrovascular dysfunction and cognitive impairment. Pyridoxamine treatment prevents large artery stiffening with advancing age, but the effects of pyridoxamine treatment on the cerebral vasculature or cognition is unknown. The purpose of this study was to investigate the effects of pyridoxamine on blood pressure, large artery stiffness, cerebral artery function, and cognitive function in old mice. Old male C57BL/6 mice consumed either pyridoxamine (2 g/L) or vehicle control in drinking water for ∼7.5 months and were compared with young male C57BL/6 mice. From pre- to post-treatment, systolic blood pressure increased in old control mice, but was maintained in pyridoxamine treated mice. Large artery stiffness decreased in pyridoxamine-treated mice but was unaffected in control mice. Pyridoxamine-treated mice had greater cerebral artery endothelium-dependent dilation compared with old control mice, and not different from young mice. Old control mice had impaired cognitive function; however, pyridoxamine only partially preserved cognitive function in old mice. In summary, pyridoxamine treatment in old mice prevented age-related increases in blood pressure, reduced large artery stiffness, preserved cerebral artery endothelial function, and partially preserved cognitive function. Taken together, these results suggest that pyridoxamine treatment may limit vascular aging.
Introduction
Increasing age is the leading risk factor for chronic conditions such as cerebrovascular and neurodegenerative diseases, but effective treatments for age-related morbidities are limited.1,2 One potential treatment target is age-related stiffening of the large arteries. Stiffer aorta and carotid arteries are strongly associated with cerebrovascular disease and cognitive impairment.3,4 However, it is unknown whether prevention of age-related increases in large artery stiffness leads to preserved cerebrovascular and cognitive function.
Pyridoxamine is a form of vitamin B6 that attenuates age-related large artery stiffening in rodents, 5 and thus is a potential therapeutic to prevent cerebrovascular and cognitive impairments that occur with advancing age. Traditionally, the benefits of pyridoxamine are attributed to its ability to stop the formation of advanced glycation end-products (AGEs).6,7 Pyridoxamine also has antioxidant properties that may also contribute to its beneficial effects on arterial stiffness and function.8,9 Other forms of vitamin B6, including the most common pyridoxine, do not have these same actions as pyridoxamine.7,10,11 While the evidence for cardiovascular benefits of vitamin B6 supplementation are inconsistent,12–14 these large clinical trials are all performed with pyridoxine and are missing the benefits specific to pyridoxamine. 8 Thus, by preventing increases in large artery stiffness, pyridoxamine is a potential treatment for chronic age-related diseases.
Large artery stiffness causes increased pulsatile pressure in smaller cerebral arteries, elevating the risk of endothelial cell damage and dysfunction. 3 Animal models of large artery stiffness indicate a causal relationship between arterial stiffness and cerebrovascular dysfunction. 15 Specifically, findings in transgenic and acute surgery mouse models demonstrate that greater large artery stiffness leads to cerebral artery endothelial dysfunction and reduced nitric oxide (NO) bioavailability.16,17 Additionally, mouse models with greater large artery stiffness have lower cerebral blood flow, higher blood-brain barrier permeability, higher oxidative stress, higher inflammatory markers, and greater cognitive deficits.18–21 However, the effects of pyridoxamine treatment on cerebral artery function are unknown.
The purpose of this study was to investigate the effects of pyridoxamine treatment on blood pressure, large artery stiffness, cerebrovascular function, and cognitive impairment in old mice. In this study, we hypothesized that pyridoxamine would attenuate the age-related increase in systolic blood pressure and large artery stiffness. Additionally, we hypothesized that pyridoxamine treated old mice would have better cerebral artery endothelial function characterized by greater NO bioavailability. Importantly, we hypothesized that pyridoxamine would preserve cognitive function compared with old control mice. We further hypothesized that the beneficial effects of pyridoxamine would also be found in another resistance artery, with mesenteric artery endothelial function being greater with pyridoxamine treatment in old mice. Finally, we hypothesized that pyridoxamine-treated old mice would have lower AGEs content in the aorta, lower gene expression of pro-oxidant/inflammatory factors, and higher gene expression of antioxidant enzymes in the cerebral arteries and hippocampus. To test these hypotheses, we treated old male C57BL/6 mice with pyridoxamine or vehicle control for ∼7.5 months, and measured blood pressure, large artery stiffness, middle cerebral artery (MCA), and mesenteric artery endothelium-dependent dilation (EDD), cognitive function, artery histology, gene expression, and protein expression.
Methods
A more detailed description of procedures can be found in the Supplemental Methods.
Mice and treatment
Male C57BL/6 mice were obtained from the National Institute on Aging colony at Charles River (old) or purchased from Charles River (young). All mice were fed a normal chow diet with ad libitum food and water and were housed in an animal care facility on a 12/12-hour light-dark cycle at 24°C. The experimental groups consisted of old mice treated with pyridoxamine orally via drinking water (2 g/L, 20–27.5 months), old control mice receiving vehicle control of normal drinking water (20–27.5 months), and young mice (4–9 months old).5,22,23 Outcomes were measured after a minimum of 6 months of treatment. Given the extensive number of outcomes, treatment length ranged from 6–8 months. All in vivo and ex vivo studies described below were conducted after treatment except for hemodynamic outcomes, which were measured on a subset of mice pre- and post-treatment. Investigators were blinded to treatment group when analyzing data. All animal procedures conform to the Guide to the Care and Use of Laboratory Animals (8th edition, revised 2011) and were approved by the Institutional Animal Care and Use Committees at the University of Oregon and University of Utah. This research complies with the Animal Research: Reporting in Vivo Experiments (ARRIVE) guidelines for reporting animal experiments.
Frailty
Frailty Index and Frailty Inferred Geriatric Health Timeline (FRIGHT) and Analysis of Frailty and Death (AFRAID) were assessed.24,25 Mouse frailty was assessed using a previously established 31-item Frailty Index based on established clinical signs of deterioration in C57BL/6J mice. 25 Frailty assessment included evaluation of the integumentary, musculoskeletal, vestibulocochlear/auditory, ocular, nasal, digestive, urogenital, and respiratory systems as well as signs of discomfort, body weight, and body surface temperature. The severity of each deficit was assessed and assigned either a 0, 0.5, or 1, with the higher score indicating more severe frailty. Each mouse was examined at approximately the same time of day, following the same order of assessments within 14 days of their planned vascular reactivity study. Vision loss was too difficult to assess and therefore not included in the index. FRIGHT and AFRAID scores were calculated using the individual components of the Frailty Index data and the webapp: http://frailtyclocks.sinclairlab.org. 24
Blood pressure
Blood pressure and heart rate were assessed in conscious mice by tail-cuff (CODA System, Kent Scientific, Torrington, CT) as previously described in detail. 26 Blood pressure and heart rate recordings were made in a quiet and warm (room temperature ∼24°C) environment. Mice were placed in restrainers on a heating unit and given 15–20 min to acclimate and reach a steady tail skin temperature of 30–35°C. Each session consisted of 5–10 acclimatization measures and documentation of stable values, followed by 20 experimental measures. Measures with aberrant movement/behavior or inadequate tail volume/flow values were excluded and the remaining values were used to calculate a mean value for each animal.
Pulse wave velocity (PWV)
Aortic stiffness was measured by in vivo pulse wave velocity (PWV) as previously described.27,28 Mice were anesthetized by 2% inhaled isoflurane in 100% oxygen and situated on a temperature-controlled heating pad (37°C) to maintain body temperature. Electrodes were placed on the distal parts of limbs to record ECG information, while 20 MHz Doppler transducers were placed on the aortic arch and abdominal aorta. Indus Doppler Flow Velocity System (Webster, Texas) was used to record and analyze data. The time for each beat to travel from aortic arch to abdominal aorta was measured and was divided by the distance between the two locations to determine the PWV value. Two researchers analyzed each Doppler recording and inter-observer confidence intervals were measured, and PWV measurements with confidence intervals over 15% were excluded. We measured PWV for all mice at the end of treatment, and for a subset of old control and old pyridoxamine mice pre-treatment.
Tissue collection
At the end of the treatment period, mice were anesthetized via isoflurane and euthanized via cardiac puncture in accordance with IACUC guidelines. After euthanasia, the heart, liver, spleen, soleus, gastrocnemius, and white adipose tissue (WAT) were collected, and their wet weights were recorded. In addition, MCA, carotid artery, aorta, cerebral cortex, and hippocampus were excised and frozen in −80°C for preservation.
Artery histology
Sections of thoracic aorta and carotid arteries were frozen in optimal cutting temperature (OCT) compound and then sliced into 8 µm sections using a cryostat (Nikon, Minato, Tokyo, Japan) and adhered to a charged slide. 16 A Verhoeff-van Gieson stain was applied to aorta sections by standard procedures for measurement of wall thickness. Carotid elastin images were captured by the autofluorescence in the FIT-C channel on slides mounted with Prolong Gold with DAPI (Life Technologies, Grand Island, NY, USA) and imaged with an Olympus Fluoview FV1000 (Tokyo, Japan) confocal microscope. Arterial wall thickness was analyzed using FIJI by ImageJ software.
Endothelial function and passive stiffness
EDD was assessed ex vivo in isolated, pressurized MCAs and mesenteric arteries as previously described in detail. 16 Briefly, MCAs and mesenteric arteries were excised and placed in myograph chambers (DMT Inc., Denmark) with physiological salt solution, 145 mM NaCl, 4.7 mM CaCl2, 1.17 mM MgSO4, 1.2 mM NaH2PO4, 5.0 mM glucose, 2.0 mM pyruvate, 0.02 mM EDTA, 3.0 mM MOPS buffer, and 1 g/100 mL BSA, pH 7.4 at 37°C, cannulated onto glass micropipettes, and secured with nylon (11-0) sutures. Once cannulated, arteries were warmed to 37°C, pressurized to 50 mmHg, and allowed to equilibrate for approximately 1 hour. All arteries were sub maximally pre-constricted with phenylephrine (1–6 μM to obtain ∼20–40% pre-constriction). Lumen diameter was measured in response to increasing concentrations of the endothelium-dependent dilator, acetylcholine (ACh: 1 × 10−9 to 1 × 10−4 M) or endothelium-independent dilator, sodium nitroprusside (SNP: 1 × 10−10 to 1 × 10−4 M).
For the MCA, ACh dose responses were repeated after incubation with N omega-Nitro-L-arginine methyl ester hydrochloride (L-NAME), 0.1 m
Passive stiffness
Following a 60 minute incubation in calcium free solution to eliminate effects of myogenic tone, passive arterial stiffness was measured ex vivo in carotid arteries and MCAs. Lumen diameter and medial wall thickness were measured following increasing intralumenal pressure.16,29 Arteries were isolated and cannulated between glass pipette tips on a myograph as previously described.
16
Measurements for each artery were recorded from 5 to 100 cmH2O (3.7–73.5 mmHg) in 5 cmH2O increments. Stress was calculated as:
Strain was calculated as:
Data for each artery were fit to the curve:
Cognitive testing
The Morris Water Maze cognitive test was used to assess learning and spatial memory. Mice underwent a 3-day training period, including 2 sessions per day, of 3 trials per session. On the fourth day, a probe trial was performed, followed by a beacon trial. All sessions were recorded and analyzed with EthoVision XT12 software (Noldus, Wageningen, the Netherlands). 33
A nest building test was used to assess instinctual cognitive behavior. 34 Mice were singly housed overnight in a cage with food, water, and a condensed cotton nestlet. In the morning, a researcher placed mice back in communal housing and scored each nest on a scale of 0–5 based on the quality of the nest.
An open field test was used to assess mouse anxiety levels. 33 Mice were placed in a 40 cm2 square arena with a white matte floor and walls. Mice were in the arena for 10 minutes and their exploration was recorded using EthoVision XT12 software. An 20 cm2 square was marked with Ethovision Software to denote the inner area of the arena. The exploration criterion was defined as the amount of time the center of the mouse’s body was inside the inner square area. Mice that spent more time exploring were considered to have lower levels of anxiety. 33
Gene expression
RNA from cerebral arteries and hippocampus was isolated by a standard protocol using Qiazol and RNAeasy Micro/Mini Kits (Qiagen, Hilden, Germany) and then quantified using a NanoDrop 2000 (Thermo Fisher). Reverse transcription was performed to produce cDNA with a Qiagen Quantitect Reverse Transcription Kit. The cDNA samples underwent real-time qPCR using ThermoFisher PowerUp Sybr Green. RNA expression was calculated using the 2−ΔΔCT method. 18S rRNA was used as a housekeeping gene transcript to control for tissue concentration, and values were normalized to the young group. Specific genes measured include: NADPH oxidase (Nox2), superoxide dismutase (Sod)1, Sod2, Sod3, AGE-specific receptor (Ager), and tumor necrosis factor (Tnf). Primers sequences used for mRNA expression are detailed in Supplemental Table 2.
AGEs abundance
AGEs abundance in the aorta was estimated via Western blot. Protein was extracted from aorta samples and concentrations of protein were determined using the Pierce BCA protein assay kit (Thermo Fisher). Proteins were separated on to a 4–20% Precast Criterion TGX gel using the Bio-Rad Criterion Cell system and transferred to a nitrocellulose membrane. Membranes were incubated with anti-AGEs antibody (1:500, ab23722, Abcam) at 4°C overnight. Membranes were then incubated in 1.5% milk with secondary antibody (1:10,000, ab6721, Abcam) for 45 min. Pierce ECL Plus Western Blotting Substrate (Thermo Fisher) was used to develop the membrane, and Bio-Rad Gel Doc system was used to image the membrane. Image Lab Software was used to determine band density for bands between 25–75 kDa for AGEs. Each sample was normalized to total protein via Ponceau intensity and expressed relative to the mean of the young group.
Statistical analysis
Statistical analyses were completed using SPSS 26.0 software, and GraphPad Prism 9.0. All data were examined for normal distribution according to Shapiro-Wilks tests. Gene expression data were log-transformed to achieve normality. For non-normally distributed data, a non-parametric Kruskal-Wallis test was performed for animal characteristics, tissue weights, nest building, and open field velocity data. A one-way ANOVA between groups was used for 6-month PWV, aortic wall thickness, EDD maximum dilation (mesenteric artery only), cognitive tests, gene expression, and supplemental data. A repeated measures ANOVA was used for within-group time comparisons for all pre-post analyses including hemodynamics and PWV. A 2-way ANOVA was used for Morris Water Maze statistical analysis. A student’s t-test was used for all comparisons between only old groups including hemodynamics and β-stiffness. To compare EDD maximum dilation in MCAs, a 1-tailed student’s t-test with a Bonferroni correction for 18 comparisons was used, making a significant p-value for this comparison p < 0.0028. Lastly, a nonlinear regression analysis normalized to Log was calculated to determine EC50 values and differences and using GraphPad Prism 9.0. All data are represented as the mean ± SD.
Results
Animal characteristics
Detailed animal characteristics can be found in Table 1. There were no differences between old control and old pyridoxamine treated mice for age (p = 0.680). Additionally, there was no difference between young, old control, or old pyridoxamine treated mice for body mass (p > 0.05 for all, Table 1). On average, old control mice drank more water per day compared with old pyridoxamine treated mice (p < 0.001), indicating that drinking preferences may have been altered due to pyridoxamine supplementation. Mass was greater for several tissues in old mice compared with young mice (p < 0.05), but all tissue masses were similar between the old groups (p > 0.05, Table 1). No differences in frailty index, FRIGHT, or AFRAID scores were found between old control and old pyridoxamine treated mice (p > 0.05 for all), indicating that qualitative outward expressions of health remained similar between treatment groups.
Animal characteristics for young, old control, and old pyridoxamine treated mice.
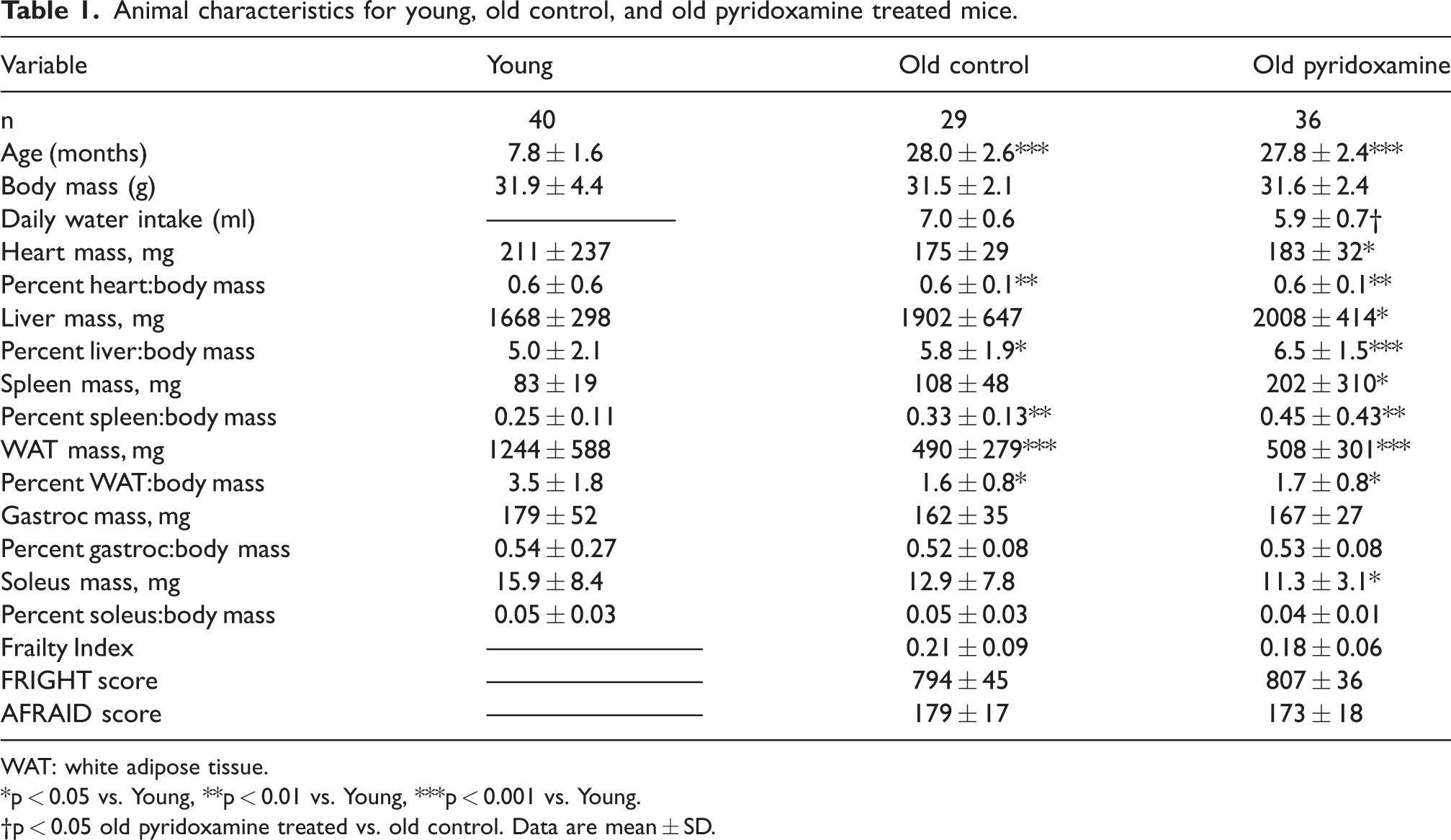
WAT: white adipose tissue.
*p < 0.05 vs. Young, **p < 0.01 vs. Young, ***p < 0.001 vs. Young.
†p < 0.05 old pyridoxamine treated vs. old control. Data are mean ± SD.
Blood pressure
In a subset of mice, hemodynamics were measured pre- and post-treatment. From pre- to post-treatment, old control mice had increased systolic blood pressure (SBP, p = 0.013), diastolic blood pressure (p = 0.043), and mean arterial pressure (p = 0.029, Table 2, Figure 1(a)). In contrast, blood pressures did not change in old pyridoxamine treated mice pre- to post-treatment (p > 0.05 for all, Table 2, Figure 1(a)). Old pyridoxamine treated mice had lower diastolic blood pressure compared to old control mice post-treatment (p = 0.048, Table 2). There were no other differences in blood pressure post-treatment between old groups, or between young and old mice (p > 0.05 for all, Table 2).
Hemodynamic measures for old control and old pyridoxamine treated mice pre- and post-intervention.
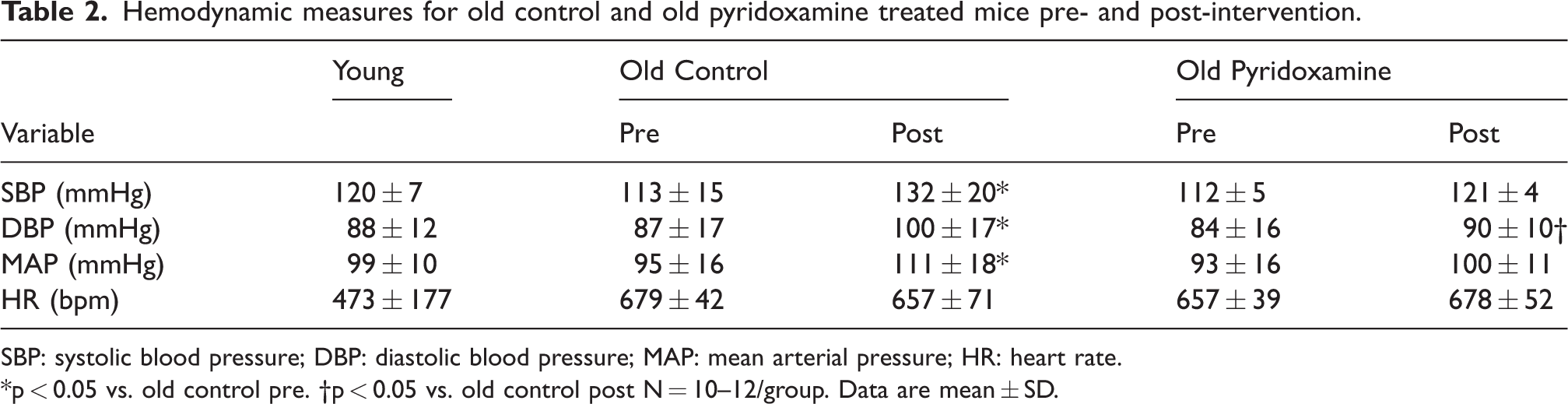
SBP: systolic blood pressure; DBP: diastolic blood pressure; MAP: mean arterial pressure; HR: heart rate.
*p < 0.05 vs. old control pre. †p < 0.05 vs. old control post N = 10–12/group. Data are mean ± SD.
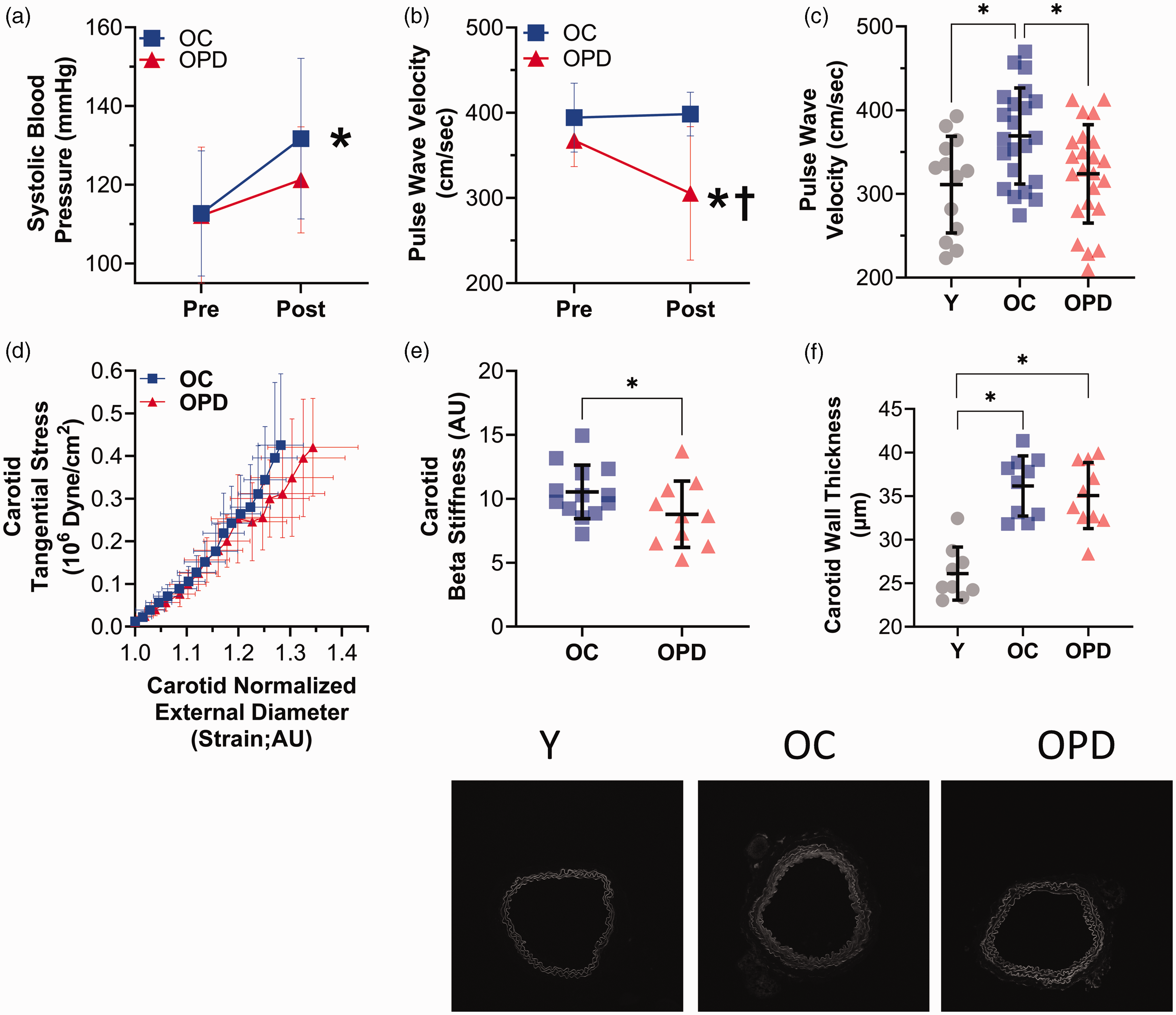
Pyridoxamine treatment attenuates increases in systolic blood pressure and large artery stiffness with age. (a) Systolic blood pressure measurements in old control (OC) and old pyridoxamine treated (OPD) mice from pre- to post-intervention, *p < 0.05 OC-pre vs. OC-post, N = 12–13/group. (b) Aortic pulse wave velocity in OC and OPD mice from pre- to post-intervention, *p < 0.05 OPD-pre vs. OPD-post, †p < 0.05 OC-post vs. OPD-post, N = 5–9/group. (c) Comparison of pulse wave velocity measurements in young (Y), OC, and OPD mice post-intervention, *p < 0.05 (d) Comparison of ex vivo stress-strain curves in carotid arteries of OC and OPD mice, N = 9–13/group. (e) Ex vivo β-stiffness measurements in carotid arteries of OC and OPD mice, *p < 0.05 and (f) Carotid wall thickness in Y, OC, and OPD mice with representative Elastin fluorescent images below, *p < 0.05. Data are mean ± SD. Statistics include a one-way ANOVA, RM-ANOVA, and student’s t-test using GraphPad Prism 9.0.
Large artery characteristics
Baseline PWV was measured pre-treatment in a subset of mice and did not differ between old control and old pyridoxamine treated mice (p = 0.092). With pyridoxamine treatment, PWV decreased from pre- to post-treatment (p = 0.028) but remained constant in with control treatment (p = 0.436, Figure 1(b)). When comparing the larger cohorts post-treatment, old control mice had higher PWV compared with both young mice (p = 0.016) and old pyridoxamine treated mice (p = 0.030, Figure 1(c)). Similarly, in common carotid arteries studied ex vivo post-treatment, old control mice had greater β-stiffness compared with old pyridoxamine treated mice (p = 0.044, Figure 1(d) and (e)). The carotid and aorta walls were less thick in young mice compared with old control (p < 0.05) and old pyridoxamine treated (p < 0.05) mice, but thickness was not different between the old groups, indicating that large elastic artery wall thickness was not congruent with stiffness (p > 0.05, Figure 1(f), Supplemental Figure 1A). Overall, these results indicate that pyridoxamine decreases large artery stiffness in old mice.
Small artery structural characteristics
There were no differences between young, old control, and old pyridoxamine treated mice in the MCA for maximal diameter, wall thickness, and β-stiffness index (p > 0.05 for all, Supplemental Table 1, Supplemental Figure 1B, C). These results suggest that pyridoxamine does not modulate small artery stiffness or structure.
Cerebral artery endothelial function
MCAs from old control mice had impaired dose response and maximum dilation to ACh compared with young and old pyridoxamine treated mice (p < 0.001 young vs. old control, p < 0.001 old control vs. old pyridoxamine), but there was no difference in ACh response between young and old pyridoxamine treated mice (p = 0.417, Figure 2(a) and (b)). L-NAME incubation reduced MCA dilation to ACh in all groups (p < 0.001 for young, p = 0.001 for old control, p < 0.001 for old pyridoxamine treated). Maximum dilation to ACh in the presence of L-NAME was greater in old control compared with old pyridoxamine treated mice (p = 0.005, Figure 2(b)). These results suggest that reduced NO bioavailability was the cause of the lower EDD in old control mice. SNP responses were similar among all three groups (p > 0.05 for all, Figure 2(c)), confirming that impaired ACh response in the old control group is caused by impaired endothelial function. Percent pre-constriction and EC50 values for all MCA responses were not different between groups (p > 0.05 for all, Supplemental Table 1). These results indicate that pyridoxamine treatment preserves cerebral artery endothelial function in old mice by maintaining NO bioavailability.
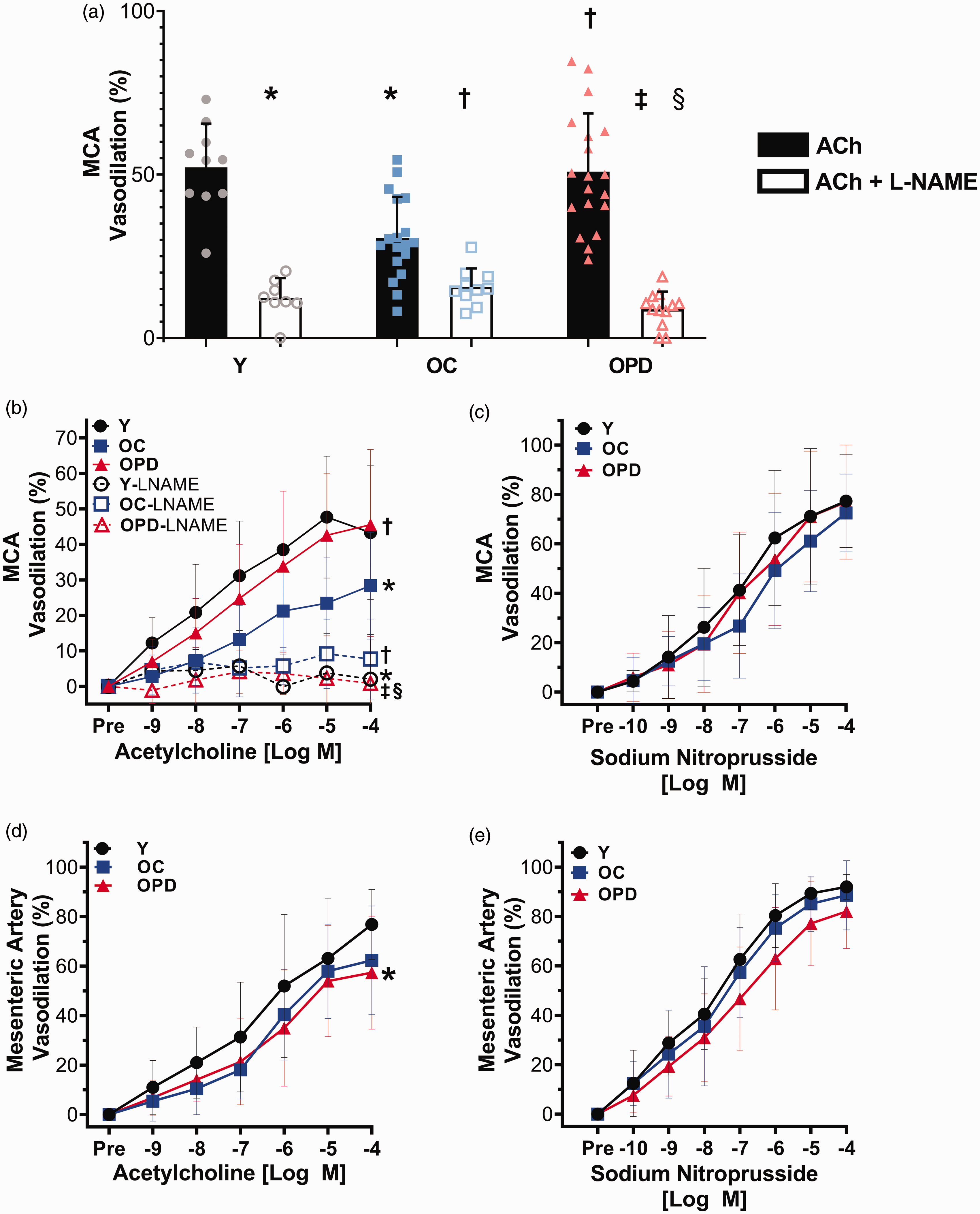
Pyridoxamine treatment prevents age-related endothelial dysfunction in cerebral arteries, but not mesenteric arteries. (a) Comparison of maximal vasodilation in the middle cerebral artery (MCA) between young (Y), old control (OC), and old pyridoxamine (OPD) treated mice in response to increasing concentrations of Acetylcholine (ACh) alone, and after incubation with L-NAME, *p < 0.05 vs. Y-ACh, †p < 0.05 vs. OC-ACh, ‡p < 0.05 vs. OPD-ACh, §p < 0.05 vs. OC-ACh + L-NAME. (b) MCA dose response curves for increasing concentrations of ACh before and after incubation with L-NAME, *p < 0.05 vs. Y-ACh, †p < 0.05 vs. OC-ACh, ‡p < 0.05 vs. OPD-ACh, §p < 0.05 vs. OC-ACh + L-NAME. N = 8–19 per group. (c) MCA dose response curves for increasing concentrations of sodium nitroprusside. N = 6–17 per group. (d) Mesenteric artery dose response curves for increasing doses of ACh. *p < 0.05 vs. Y-ACh. N = 14–18 per group and (e) Mesenteric artery dose response curves for increasing concentrations of sodium nitroprusside. N = 9–12 per group. Data are mean ± SD. Statistics include repeated measures ANOVA and student's t-test with Bonferroni correction using SPSS 26.
Mesenteric artery function
In mesenteric arteries, responses to ACh were similar between old control and old pyridoxamine treated mice (p = 0.349), and young mice had a greater ACh response compared with old pyridoxamine treated mice (p = 0.005, Figure 2(d)). No differences in response to SNP were observed between groups in mesenteric arteries (p > 0.05 for all, Figure 2(d)). Percentage pre-constriction and EC50 values for ACh and SNP were not different among groups for the mesenteric arteries (p > 0.05 for all, Supplemental Table 1). These results indicate that, unlike cerebral arteries, endothelial function in mesenteric arteries is not preserved with pyridoxamine treatment in old mice.
Cognitive function
During the final training trial of the Morris Water Maze, old control mice took longer to reach the escape platform compared with young mice, indicating an impaired learning ability in the old control mice (p = 0.030, Figure 3(a)). Old pyridoxamine treated mice did not differ from old control or young mice for time to reach the escape platform during the final training trial (p = 0.122 young vs. old pyridoxamine, p = 0.617 old control vs. old pyridoxamine, Figure 3(a)). In the 60 second probe trial, in contrast to young mice, neither group of old mice showed a clear bias towards the target zone; however, there were no differences within any group for time in each quadrant (p > 0.05 for all, Figure 3(b)). As the old mice appeared to have a more labile memory, we reanalyzed the probe trial for the period between 10 and 50 seconds. During the probe trial, from seconds 10–50, both young and old pyridoxamine treated mice spent more time in the target quadrant compared with the average of the other quadrants (young, p < 0.001, old pyridoxamine, p = 0.020, Figure 3(c) and (d)). There was no difference between target and other quadrants for old control mice (p = 0.684, Figure 3(c) and (d)). Old control and old pyridoxamine mice had slower swimming velocity compared with young mice (p = 0.001 young vs. old control, p = 0.005 young vs. old pyridoxamine, Supplemental Figure 2 A), but there were no group differences in the average time to the visible platform during a beacon trial (p > 0.05 for all, Supplemental Figure 2B), suggesting that swimming ability was not a confounding factor for the probe trial results.
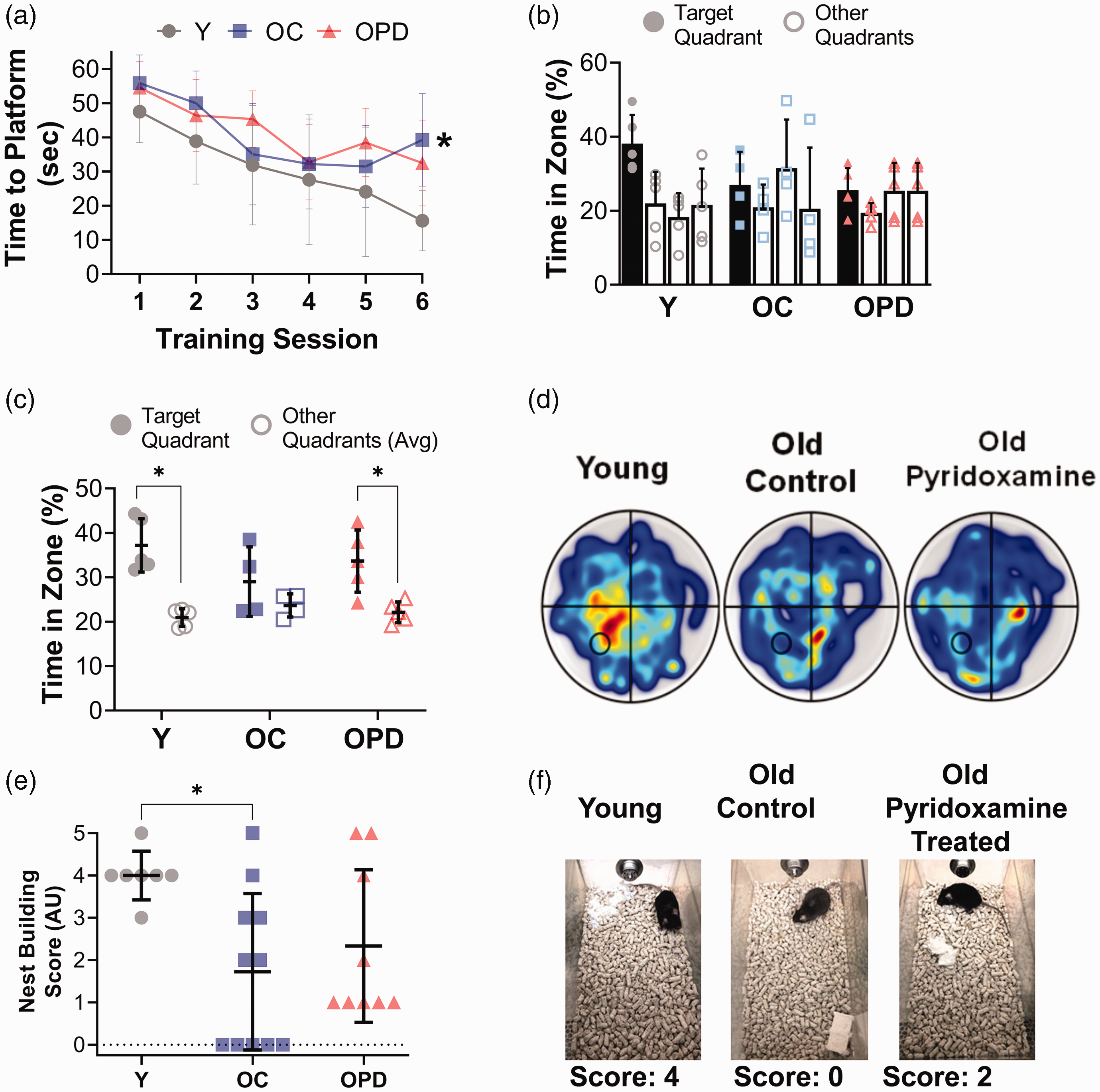
Pyridoxamine partially prevents age-related cognitive decline. (a) For the Morris Water Maze test, average time to platform during 6 training sessions for young (Y), old control (OC), and old pyridoxamine (OPD) mice. *p < 0.05 vs. Y, N = 4–5 per group. (b) Percentage of time spent in target and other quadrants during the 0–60 second interval of the Morris Water Maze probe trial. N = 4–5 per group. (c) Percentage of time spent in target quadrant and the average of other quadrants during the 10–50 second interval of a probe trial. *p < 0.05. (d) Representative heat maps of Y, OC, and OPD mice during the 10–50 second interval of the Morris Water Maze probe trial. (e) Comparison of nest building scores in Y, OC, and OPD mice, *p < 0.05 and (f) Representative nests and scores. Data are mean±SD. Statistics include a one-way ANOVA and a non-parametric Shapiro-Wilks (nest building) using GraphPad Prism 9.0.
For the nest building test, young mice had higher scores compared with old control mice (p = 0.0312, Figure 3(e) and (f)). However, old pyridoxamine treated mice had scores that were not different from either young or old control mice (p = 0.121 young vs. old pyridoxamine treated, p = 0.684 old control vs. old pyridoxamine treated, Figure 3(e) and (f)), suggesting that instinctual nest building behavior is impaired with age, but pyridoxamine may partially attenuate this age-related impairment.
The open field test of anxiety revealed that young mice spent more time exploring the center region of an open arena compared with old control and old pyridoxamine treated mice (p = 0.024 young vs. old control, p = 0.004 young vs. old pyridoxamine treated, Supplemental Figure 2D), indicating higher levels of anxiety in old mice that are not prevented with pyridoxamine treatment. There were no differences in velocity between groups during the open field test (p > 0.05 for all, Supplemental Figure 2C), indicating that there were greater effects on motor ability during swimming compared with walking with old age.
Hippocampus and cerebral artery gene expression
In the hippocampus, Nox2 gene expression was lower in young mice compared with old mice (p = 0.002 young vs. old control, p < 0.001 young vs. old pyridoxamine treated mice), but there were no differences between the old control and old pyridoxamine treated mice (p = 0.537, Figure 4(a)). Sod1 hippocampal gene expression was greater in old pyridoxamine treated mice compared to young mice (p = 0.015), but not different from old control mice (p = 0.207, Figure 4(a)). Additionally, old pyridoxamine treated mice had higher hippocampus antioxidant Sod3 expression compared with old control mice (p = 0.032) and not different from young mice (p = 0.555, Figure 4(a)). No differences in Ager and Sod2 gene expression were observed between groups in the hippocampus (p > 0.05 for all, Figure 4(a)). These results indicate that pyridoxamine treatment increases antioxidant Sod1 and Sod3 gene expression in the hippocampus of old mice but does not prevent the age-related increases in pro-oxidant Nox2 gene expression.
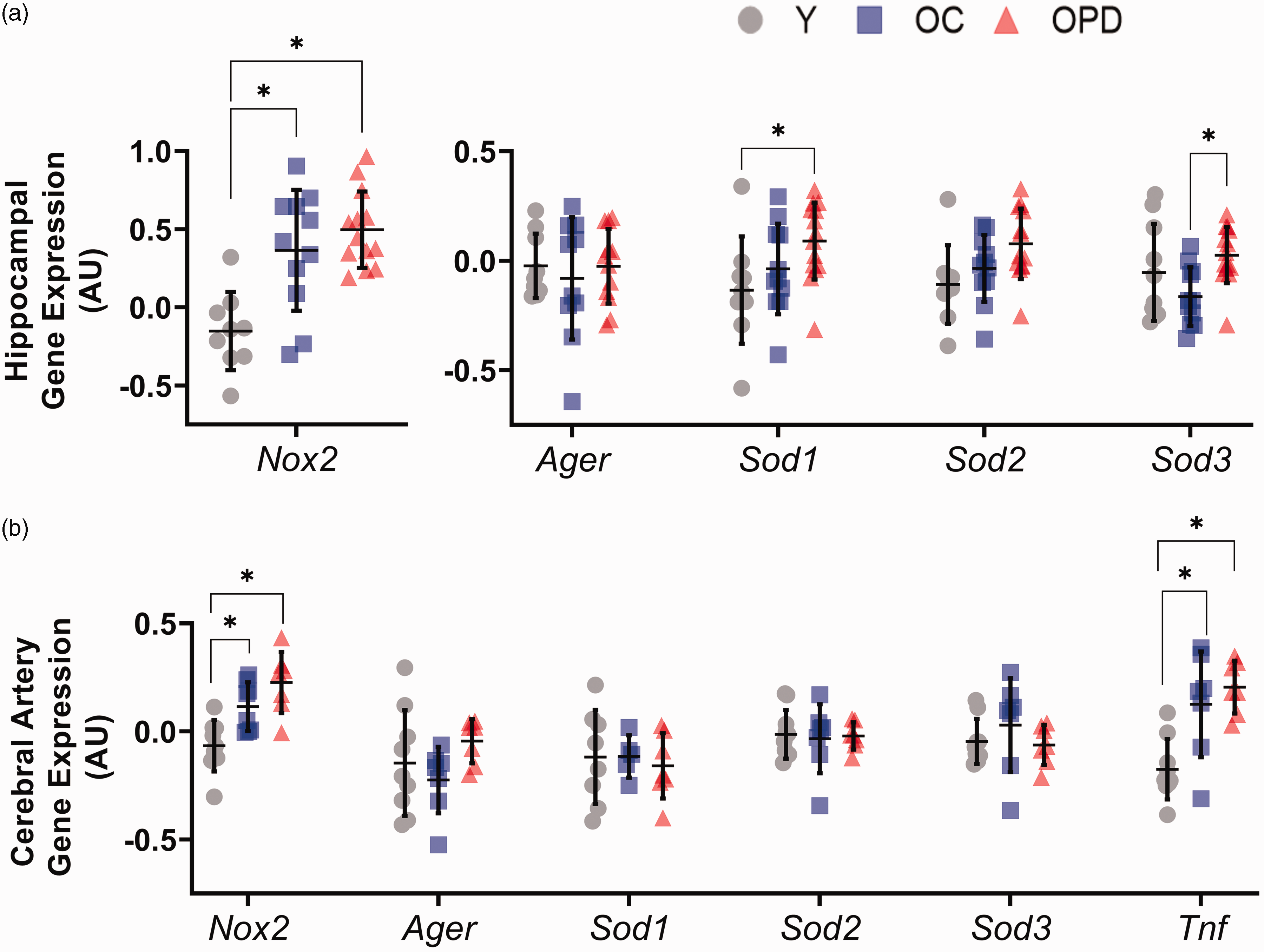
Pyridoxamine treatment increases Sod1 and Sod3 expression in the hippocampus but does not change cerebral artery gene expression. (a) Hippocampus and (b) cerebral artery gene expression of Nox2, Ager, Sod1, Sod2, and Sod3 in young (Y), old control (OC), and old pyridoxamine treated (OPD) mice. *p < 0.05. Data are mean ± SD. Statistics include one-way ANOVA using GraphPad Prism 9.0.
In cerebral arteries, both old control and old pyridoxamine treated mice had a higher gene expression of pro-oxidant Nox2 compared with young mice (p = 0.046 young vs. old control, p < 0.001 young vs. old pyridoxamine treated, Figure 4(b)). Expression of pro-inflammatory cytokine Tnf was greater in cerebral arteries from the old mice compared with young mice (p < 0.001 young vs. old control, p < 0.001 young vs. old pyridoxamine treated), and there was no difference between the old groups (p = 0.601, Figure 4(b)). No other differences in gene expression were found between groups for Ager, Sod1, Sod2, and Sod3 in the cerebral arteries (p > 0.05 for all). These results indicate that old mice have greater cerebral artery pro-inflammatory Tnf expression and greater pro-oxidant Nox2 expression that is not modulated by pyridoxamine treatment.
AGEs abundance
There were no differences in the AGEs abundance in the aorta between young, old control, or old pyridoxamine treated mice (p > 0.05 for all, Figure 5).
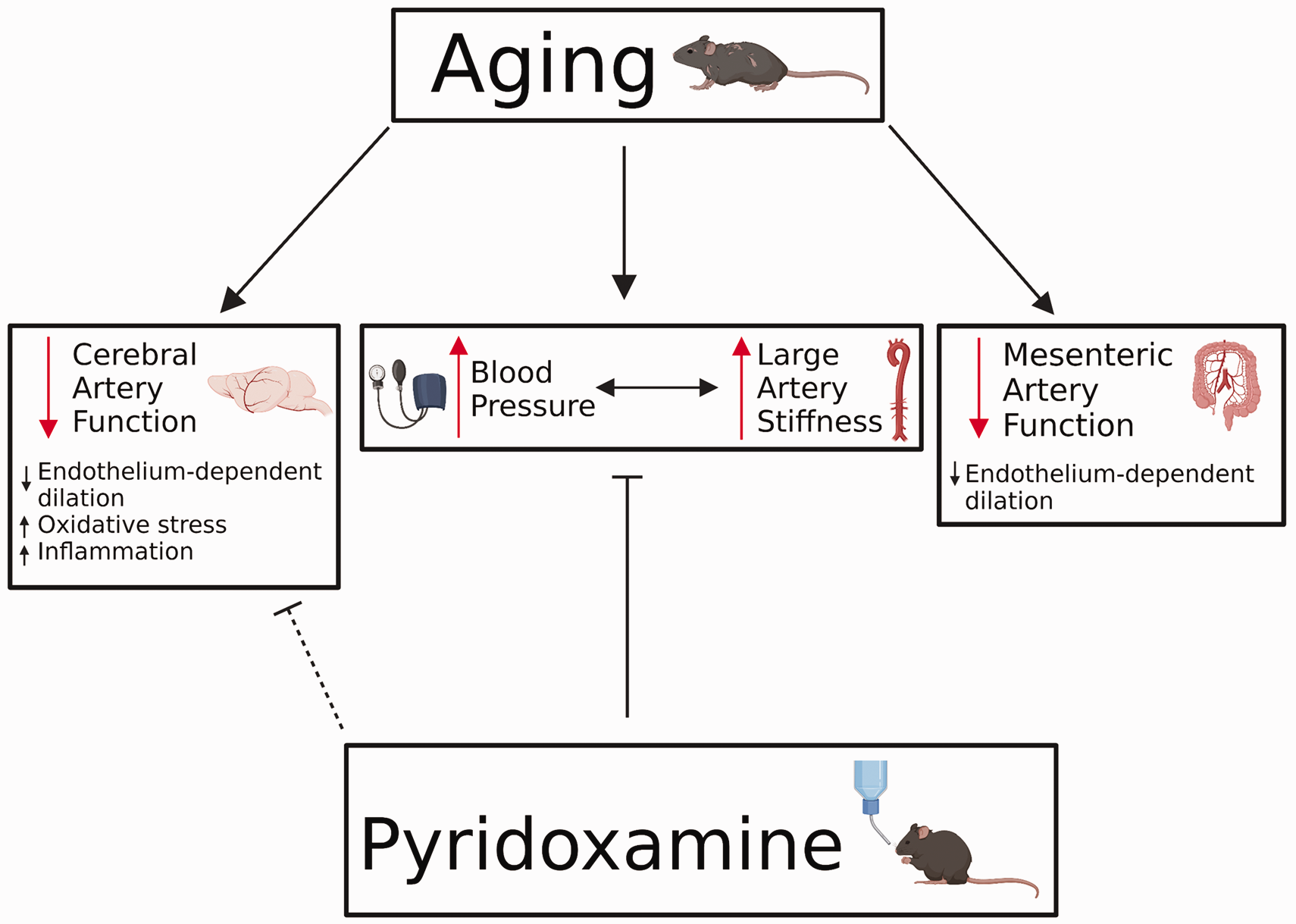
Schematic of the effects of age and pyridoxamine on the vasculature. Aging leads to adverse effects in both large elastic arteries and smaller, resistance arteries. In large arteries, aging contributes to increases in blood pressure and large artery stiffness. In mesenteric and cerebral arteries, aging leads to decreases in endothelium-dependent dilation. Additionally, aging leads to increased oxidative stress and inflammation in cerebral arteries. Pyridoxamine treatment attenuates the age-related increases in blood pressure and large artery stiffness. As a result of pyridoxamine treatment, endothelium-dependent dilation and oxidative stress are rescued in cerebral arteries. However, it is unknown if this effect in cerebral arteries is due to the changes in large arteries or via direct effects of pyridoxamine. Pyridoxamine does not affect mesenteric artery function.
Discussion
The results of this study indicate that pyridoxamine treatment in old mice reduces large artery stiffness and prevents age-related increases in systolic blood pressure.5,35 Pyridoxamine treatment also attenuates age-related declines in cerebral artery endothelial function by preserving NO-mediated vasodilation. This effect of pyridoxamine is specific to the cerebral circulation as there are no improvements in a different resistance artery: the mesenteric arteries. Pyridoxamine treatment also appears to partially attenuate age-related cognitive decline; however, these effects are modest. While pyridoxamine treatment leads to greater gene expression of antioxidant enzyme Sod3 in the hippocampus, there are no effects on expression of other genes or a measurable effect on AGEs abundance in the aorta. In sum, these data suggest that pyridoxamine is a potential treatment to prevent the deleterious effects of aging on the vasculature, but these vascular improvements may not sufficiently mitigate age-related cognitive decline (Figure 5).
Large artery stiffness and blood pressure
Our finding of lower blood pressure after pyridoxamine treatment align with previous findings in spontaneously hypertensive rats where pyridoxamine treatment resulted in lower SBP and attenuated aortic remodeling.5,36 In the present study, we are unable to conclude if the effects of pyridoxamine on large artery stiffness are causing the changes to blood pressure or vice versa. Pyridoxamine reduced aortic stiffness as indicated by lower PWV post- compared with pre-treatment, and this decrease in aortic stiffness may have directly prevented the age-related increase in SBP. Greater aortic stiffness is known to increase SBP and pulse pressure through changes to wave reflection and impaired pulse dampening. 37 Alternatively, the lessening of the age-related increase in SBP with pyridoxamine treatment may have influenced arterial stiffness. For example, mouse models of induced hypertension demonstrate that increased SBP contributes to large artery stiffening. 38 Therefore, with the concomitant changes in SBP and PWV with pyridoxamine treatment and aging, we cannot conclude which of these had a greater influence. Of note, there were no differences in cerebral artery stiffness between groups, suggesting that the effects of pyridoxamine on stiffness were isolated to the large arteries. We also found that pulse pressure was not different with aging or pyridoxamine treatment in this study, and that this is counter to the proposed effects of large artery stiffness. However, tail-cuff measurements tend to be less accurate for diastolic blood pressure compared with SBP and may explain the lack of differences in pulse pressure. In addition, the blood pressure of the young mice was not different from either old group in this study. This lack of age-related differences in blood pressure could be due to the variability of tail-cuff measurements and a reduced power to detect difference due to multiple comparisons. There also appears to be a cohort effect on blood pressure as the young mice trend towards higher blood pressure compared with the middle-aged (pre-treatment) mice. Anecdotally, the young mice appear more “stressed” during the measurement of blood pressure compared with the old mice and this likely contributes to their higher blood pressure. Importantly, our primary outcome is the paired pre- to post-treatment measurement of blood pressure and aortic stiffness, and this longitudinal design eliminates the cohort effect. Therefore, we find that pyridoxamine has beneficial effects on the age-related changes to both arterial stiffness and blood pressure.
Unlike previous studies, we did not detect a reduction of AGEs with pyridoxamine treatment.39,40 There are a few potential explanations for this discrepancy. First, there are many forms of AGEs, such as N(6)-Carboxymethyllysine (CML), glucosepane, and pentosidine. We used a Western blot to measure total AGEs, but the use of high performance liquid chromatography or immunohistochemical staining may have produced more accurate results. 41 Standard techniques for measuring AGEs content are not yet fully established. Second, there was high variability in our measurement of AGEs that may have impacted our ability to detect group differences. Lastly, we did not detect higher AGEs content in the aorta of old mice compared with young mice, unlike previous studies. 28 Thus, the potential anti-AGEs effect of pyridoxamine may have been limited by a lack of age-related elevation in AGEs.
An alternative mechanism for the beneficial effects of on large artery stiffness is by the antioxidant effects of pyridoxamine.8,9 Antioxidants are known to reduce large artery stiffness in older humans and animals.42,43 Due to the lack of differences in AGEs between treatment groups in this study, we surmise that the mechanism of pyridoxamine is mainly mediated via its antioxidant properties. Therefore, in this study pyridoxamine reduced large artery stiffness in old mice, but the mechanism for this effect remains unclear.
Effects of pyridoxamine on cerebral artery endothelial function
Previous studies established the link between large artery stiffness and cerebrovascular dysfunction. Greater large artery stiffness is associated with reduced cerebral blood flow in humans.3,44 In a transgenic mouse model, the Eln heterozygous knockout, greater large artery stiffness leads to impaired cerebral artery endothelial function associated with decreased NO bioavailability, increased oxidative stress and inflammatory signaling, and reduced cerebral perfusion.16,17,21 In a surgical model of carotid artery stiffness, increased carotid artery stiffness leads to reduced cerebral blood flow, increased blood-brain barrier permeability, and neuroinflammation.18–20 The present study is the first to examine the effect of reduced large artery stiffness on cerebrovascular function in old mice. Our results confirm our hypothesis, as pyridoxamine treatment did reduce large artery stiffness and preserve cerebral artery endothelial function by maintaining NO bioavailability in old mice.
It should be noted that we cannot definitively prove with the present study design that the effects of pyridoxamine on cerebral arteries were mediated by the modulation of large artery stiffness. First, the effects of blood pressure on cerebral artery endothelial function should not be ignored. Hypertension is linked to cerebral artery endothelial dysfunction and remodeling.45,46 Second, pyridoxamine may have acted directly on the cerebral arteries, such as by reducing local cerebral AGEs, but as of yet this effect has not been reported. In contrast, we find in the present study that pyridoxamine treatment had no effect on mesenteric artery endothelial function in old mice. Thus, our findings suggest that endothelial function improvements from pyridoxamine treatment may be specific to the cerebral vasculature with aging, but the study of additional tissue circulations is needed to support this finding.
Oxidative stress and inflammatory signaling
Oxidative stress is associated with aging and vascular impairment.47,48Our measurement of oxidative stress and inflammatory-related gene expression provides insights into the improvements mediated by pyridoxamine treatment. We found that Sod1 and Sod3 gene expression were elevated with pyridoxamine treatment in the hippocampus. This suggests that pyridoxamine has beneficial antioxidant effects in this region of the brain most important to memory. These increases in antioxidant gene expression were only in the hippocampus with pyridoxamine treatment and not in cerebral arteries. As the hippocampus sample contains a variety of cells (neurons, glia, vascular cells, etc.), we cannot identify which cell types are modulating Sod1 and Sod3 expression in response to pyridoxamine. Sod1 has protective effects against age-related tissue degradation and increases oxidative stress in muscle, and thus, it is possible that the upregulation of Sod1 in the hippocampus with pyridoxamine treatment aided in the mitigation of cognitive impairment. 49 Furthermore, as SOD3 is secreted from the cell to the extracellular space, the increase in its expression with pyridoxamine treatment could benefit multiple cell types. We also found that Nox2 and Tnf expression were greater with age, but pyridoxamine had no effect. Surprisingly, Pereira et al. (2020) found in a rat model of non-alcoholic fatty liver disease that Tnf expression was greater after pyridoxamine treatment in the liver. 50 This may indicate that pyridoxamine has deleterious effects on the liver but it does not appear to be more pro-inflammatory in vascular or brain tissues. In summary, pyridoxamine has beneficial effects on antioxidants with advancing age, but the effects on pro-oxidant and pro-inflammatory signaling appears limited.
Cognitive impairment
Greater large artery stiffness is associated with cognitive impairment, as supported by recent meta-analyses.4,51 Mouse studies demonstrate the causative nature of this relation, as increased carotid artery stiffness, induced by calcium chloride application, results in impaired memory. 18 At the same time, hypertension is associated with cognitive impairment, with hypertension occurring at mid-life typically having a stronger influence than late-life hypertension. 52 In our study, cognitive function in old mice appears to be modestly affected by pyridoxamine treatment. Instinctual nest building behavior and spatial memory were somewhat preserved. For the Morris Water Maze, old pyridoxamine mice spent similar amounts of time in all quadrants in 60 seconds. However, when observing just the 10–50 second time frame, old pyridoxamine mice showed a bias towards the target quadrant compared with the average of other quadrants. Furthermore, nest building scores for old pyridoxamine were not statistically different from young or old control mice, but trended toward higher scores compared with old control mice. Given sample size limitations with cognitive tests, we hypothesize that increased group size would demonstrate stronger effects for both age and pyridoxamine treatment. A factor limiting the cognitive effects of pyridoxamine in the present study is the old age of the mice. The treatment with pyridoxamine commenced at 20 months of age and spanned until 27.5 months of age. At these ages, irreversible cognitive decline may have already occurred. Thus, there is ample previous evidence that stiffening of the large arteries and hypertension are associated with cognitive decline and we find that pyridoxamine modestly improves memory when treatment begins in old age. Although we found only modest preservation of memory function, pyridoxamine treatment may be more effective if started at an earlier age.
Limitations
A major limitation of this study is that experiments were conducted in only male mice and need to be confirmed in female mice. There is evidence suggesting that sex differences exist in both humans and animals in relation to large artery stiffness.52–54 These sex differences could also contribute to increased risk of cerebrovascular dysfunction, and cognitive impairment seen in females with age. 55 Thus, it is important to note that results from this study may not be generalizable to the physiological changes that occur with age in females.
Additionally, we were unable to control for variation in individual consumption of pyridoxamine treatment as it was dissolved in communal drinking water. Thus, it is possible that some mice received a higher dose than others. The tail-cuff method used to measure blood pressure is variable and the accuracy may be limited, particularly for diastolic blood pressure. Finally, C57BL/6 mice are resistant to several age-related diseases, such as atherosclerosis and Alzheimer’s disease. Thus, to better understand the effects of pyridoxamine on aging, future studies should examine models that better recapitulate the complexity of human aging.
Clinical perspectives
Long-term supplementation with pyridoxamine is a tenable preventative intervention for humans. Pyridoxamine was previously used in human clinical trials without serious adverse events. 56 The chosen dosage, delivery, and duration of pyridoxamine treatment in this study is equivalent to oral supplement beginning in middle-age in humans as an 18-month-old C57BL6 mouse corresponds to a 55-year-old human. 57 Previous studies in humans treated with pyridoxamine lasted 24 or 52 weeks, indicating that longer term supplementation is possible.56,58,59 However, the outcomes of these studies were related to diabetes, and thus, the benefits of pyridoxamine on arterial stiffness and cerebrovascular function in humans remain to be investigated.
Conclusions
This study demonstrated that 7.5 months of pyridoxamine treatment in old mice prevents increases in blood pressure, reduces large artery stiffness and preserves cerebrovascular function, and modestly preserves cognitive function. Treatment, however, did not affect cerebral artery structure. Pyridoxamine treatment also resulted in greater Sod1 and Sod3 expression in the hippocampus. Overall, this study demonstrates pyridoxamine successfully attenuates the effects of aging on large arteries and the cerebral vasculature. Future studies are warranted to examine the effects of pyridoxamine treatment in models of age-related diseases.
Supplemental Material
sj-jpg-1-jcb-10.1177_0271678X221130124 - Supplemental material for Pyridoxamine treatment ameliorates large artery stiffening and cerebral artery endothelial dysfunction in old mice
Supplemental material, sj-jpg-1-jcb-10.1177_0271678X221130124 for Pyridoxamine treatment ameliorates large artery stiffening and cerebral artery endothelial dysfunction in old mice by Emily H Reeve, Elise K Kronquist, Julia R Wolf, Byron Lee, Aleena Khurana, Hanson Pham, Abigail E Cullen, Jessica A Peterson, Antonio Meza, R Colton Bramwell, Laura Villasana, Daniel R Machin, Grant D Henson and Ashley E Walker in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-jpg-2-jcb-10.1177_0271678X221130124 - Supplemental material for Pyridoxamine treatment ameliorates large artery stiffening and cerebral artery endothelial dysfunction in old mice
Supplemental material, sj-jpg-2-jcb-10.1177_0271678X221130124 for Pyridoxamine treatment ameliorates large artery stiffening and cerebral artery endothelial dysfunction in old mice by Emily H Reeve, Elise K Kronquist, Julia R Wolf, Byron Lee, Aleena Khurana, Hanson Pham, Abigail E Cullen, Jessica A Peterson, Antonio Meza, R Colton Bramwell, Laura Villasana, Daniel R Machin, Grant D Henson and Ashley E Walker in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-jpg-3-jcb-10.1177_0271678X221130124 - Supplemental material for Pyridoxamine treatment ameliorates large artery stiffening and cerebral artery endothelial dysfunction in old mice
Supplemental material, sj-jpg-3-jcb-10.1177_0271678X221130124 for Pyridoxamine treatment ameliorates large artery stiffening and cerebral artery endothelial dysfunction in old mice by Emily H Reeve, Elise K Kronquist, Julia R Wolf, Byron Lee, Aleena Khurana, Hanson Pham, Abigail E Cullen, Jessica A Peterson, Antonio Meza, R Colton Bramwell, Laura Villasana, Daniel R Machin, Grant D Henson and Ashley E Walker in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-jpg-4-jcb-10.1177_0271678X221130124 - Supplemental material for Pyridoxamine treatment ameliorates large artery stiffening and cerebral artery endothelial dysfunction in old mice
Supplemental material, sj-jpg-4-jcb-10.1177_0271678X221130124 for Pyridoxamine treatment ameliorates large artery stiffening and cerebral artery endothelial dysfunction in old mice by Emily H Reeve, Elise K Kronquist, Julia R Wolf, Byron Lee, Aleena Khurana, Hanson Pham, Abigail E Cullen, Jessica A Peterson, Antonio Meza, R Colton Bramwell, Laura Villasana, Daniel R Machin, Grant D Henson and Ashley E Walker in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-jpg-5-jcb-10.1177_0271678X221130124 - Supplemental material for Pyridoxamine treatment ameliorates large artery stiffening and cerebral artery endothelial dysfunction in old mice
Supplemental material, sj-jpg-5-jcb-10.1177_0271678X221130124 for Pyridoxamine treatment ameliorates large artery stiffening and cerebral artery endothelial dysfunction in old mice by Emily H Reeve, Elise K Kronquist, Julia R Wolf, Byron Lee, Aleena Khurana, Hanson Pham, Abigail E Cullen, Jessica A Peterson, Antonio Meza, R Colton Bramwell, Laura Villasana, Daniel R Machin, Grant D Henson and Ashley E Walker in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by NIH R01 AG064016, R00 AT010017, K01 AG046326, and a University of Utah Center on Aging pilot grant.
Authors’ contributions
EK, GH, and AW contributed to study concept and design. ER, EK, JW, BL, AK, HP, AC, JP, AM, CB, LV, DM, GH, and AW contributed to data collection and analysis. ER, GH, AC, and AW contributed to development of figures/tables. ER and AW drafted manuscript, and all other authors edited and approved final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.