Abstract
A recent study by Bisht, Okojie, and Sharma, et al. characterizes a population of capillary-associated microglia (CAM) whose cell bodies are positioned along small blood vessels in the healthy mouse brain. Through elegant, longitudinal intravital imaging of brain vasculature and CAMs, the authors have uncovered the significance of microglia in cerebral blood flow regulation. Further investigation into the functions of this CAM population and how they interact with surrounding cells within the neurovascular unit will improve our understanding of vascular regulation and cerebrovascular diseases.
Microglia have been implicated in brain vascular development and in preserving vascular integrity under various central nervous system (CNS) injuries and diseases.1,2 Previous studies in brain development revealed a population of ‘juxtavascular microglia’ that migrate along the vasculature before colonizing the brain parenchyma, but become stationary along vessels in the adult brain. 1 Bisht et al. further characterize these seemingly stationary adult juxtavascular microglia. 3 It is noted that approximately one-third of all microglia are capillary-associated microglia (CAM) in the adult mouse, even after PLX3997-mediated microglia depletion and repopulation, which suggests active recruitment of microglia by vessels. In support, intravital imaging revealed that parenchymal microglia can become CAMs (and vice versa), indicating that the vessel association of microglia is indeed a dynamic process and may be vital for maintaining vascular homeostasis. These CAMs are different from the better-characterized perivascular macrophages (PVM) in that they express the microglia-specific markers Sall1 and P2RY12, but not the PVM marker CD206. CAMs are primarily defined by their position and morphology: they are associated with smaller vessels than PVM and positioned outside of the basement membrane, in contrast to the location of PVM within the basement membrane. 3 However, recent research has shown direct purinergic contacts between parenchymal microglia processes and the endothelial cell surface in both large vessels and microvessels. 4
The study also reveals the roles of CAMs in vasodilation and cerebrovascular perfusion. When microglia are depleted, there is a ∼15% increase in capillary diameter with a corresponding 20% increase in basal cerebral blood flow (CBF), assessed in vivo with two-photon microscopy and laser speckle contrast imaging, respectively. When challenged with CO2, untreated mice experienced an increase in CBF, while the CBF of microglia-depleted mice remained unchanged from its already elevated basal level. The clever use of longitudinal imaging allows the authors to observe the 3D structure of capillaries before and after microglial depletion in the same subjects, producing convincing results.
The mechanism underlying microglial attraction to capillaries is further described (Figure 1). Microglia use purinergic receptors (P2RY12) to detect ATP that is released from pannexin 1 gap junction channels (PANX1) expressed by cells in the neurovascular unit (NVU). A smaller proportion of microglia are associated with capillaries when either P2RY12 or PANX1 is knocked out (despite an increase in total microglial number). P2RY12-dependent sensing of ATP is responsible for microglial process outgrowth in other contexts, but the present study describes the relocation of microglial somata toward (and away from) blood vessels. The authors further show increased basal CBF and impaired vasodilation to CO2 in P2RY12 or PANX1 knockout animals, though the capillaries of knockout animals do not appear larger at rest. A recent study also reports the importance of microglial P2RY12 in CBF regulation and demonstrates decreased CBF response to hypercapnia and whisker stimulation-evoked neuronal activity in P2RY12 knockout mice. 4 Longitudinal in vivo imaging experiment using an inducible P2RY12 knockout mouse would be helpful to confirm the function of P2RY12/PANX1 in vasodilation.
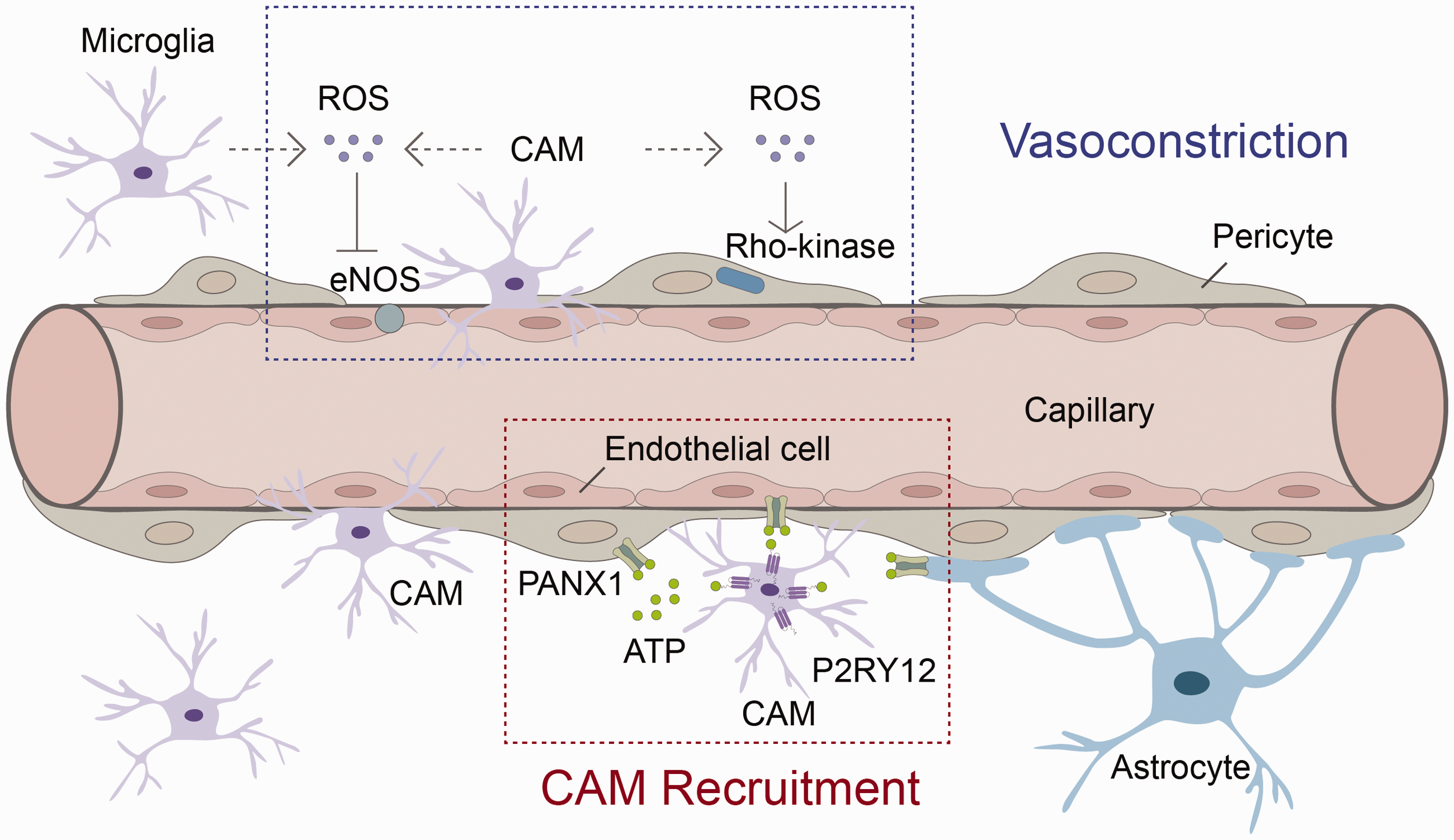
The interactions between capillary-associated microglia (CAM) and other cellular components within the neurovascular unit. Microglia use purinergic receptor P2RY12 to detect ATP that is released from pannexin 1 gap junction channels (PANX1) expressed by endothelial cells, astrocytes, and pericytes in the neurovascular unit (NVU). Such interactions maintain CAMs’ association with capillaries and their vascular regulatory functions. In addition, CAM-generated reactive oxygen species (ROS) may activate Rho-kinase in pericytes and inhibit endothelial nitric oxide synthase (eNOS) to promote capillary contractility.
These findings by Bisht et al. nicely characterize a novel CAM population and illustrate the importance of CAMs in regulating vascular tone in the healthy adult brain. The observations from this study will inspire future research to elucidate the behavior of lined, wrapped, and junctional CAMs and the mechanisms underlying their functions. For example, while P2RY12 is relatively specific to microglia, PANX1 is expressed by various cell types such as pericytes, endothelial cells, and astrocytes, that could be responsible for ATP release. In addition, microglial P2RY12 expression increases in the aged brain, 5 which may elicit age-dependent alterations in CAM behavior. Other recent work has shown that purinergic signaling from astrocytic connexin 43 (Cx43) hemichannels drives the microglial response to acute vascular injuries, 2 suggesting alternative mechanisms for sensing vascular injury.
Though the characteristics and attraction mechanism for CAMs are thoroughly described, the mechanism behind microglial control of capillary diameter has yet to be discovered. Although the Bisht study mainly focuses on CAM-endothelial cell direct interaction, it is highly plausible that other types of cells participate in CAM-mediated capillary control. Pericytes are well-known to control contractility in brain capillaries that lack smooth muscle cells.6,7 Optogenetic stimulation of capillary pericytes in vivo results in gradual but significant (∼20%) contraction of brain capillaries from their baseline diameter over the course of a minute. In contrast, ablation of individual pericytes induces a sustained increase in local capillary diameter with a corresponding increase in red blood cell flux, similar to what is observed with PLX3397 induced microglial depletion in the Bisht study. The Rho-kinase signaling pathway is necessary for pericyte-mediated contraction, as treatment with a Rho-kinase inhibitor abolishes optogenetically-induced capillary constriction. 6 It is known that Rho-kinase is strongly activated by reactive oxygen species (ROS), 8 while the activity of endothelial nitric oxide synthase (eNOS), a major vasodilator, can be inhibited by ROS. 9 Since microglia are a major source of ROS in the brain, 10 CAMs are thus perfectly positioned to activate Rho-kinase and inhibit eNOS while promoting capillary contractility (Figure 1). Further studies are warranted to explore whether CAM-mediated capillary dilation is due to direct CAM-endothelial cell crosstalk, indirect signaling through pericytes, or through complicated interactions among multiple cellular components within an integrated NVU.
These hypotheses are facilitated by the careful characterization of CAMs laid out in this paper. The study by Bisht et al. leads to an exciting discovery in understanding the importance of microglia in homeostatic brain capillary function. Elucidating the functional differences between PVM, which exist outside of the brain parenchyma, and CAM will serve as a foundation for the holistic understanding of vascular regulation and cerebrovascular diseases.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Xiaoming Hu is supported by a VA merit review grant (I01 BX003651).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
