Abstract
Pericytes, spatially isolated contractile cells on capillaries, have been reported to control cerebral blood flow physiologically, and to limit blood flow after ischaemia by constricting capillaries and then dying. Paradoxically, a recent paper dismisses the idea of pericytes controlling cerebral blood flow, despite confirming earlier data showing a role for pericytes. We show that these discrepancies are apparent rather than real, and depend on the new paper defining pericytes differently from previous reports. An objective definition of different sub-classes of pericyte along the capillary bed is needed to develop novel therapeutic approaches for stroke and disorders caused by pericyte malfunction.
Introduction
Pericytes are cells present at intervals along the walls of capillaries (and post-capillary venules). In the CNS, they are important for blood vessel formation, maintenance of the blood–brain barrier, regulation of immune cell entry to the central nervous system (CNS) and control of brain blood flow. 1 Classically, the blood flow increase evoked by neuronal activity, 2 which focuses the oxygen and glucose supply on active neurons and generates BOLD functional imaging signals, was thought to reflect relaxation of smooth muscle cells around arterioles. Recently, however, it has become clear that much of the flow increase is generated by dilation of capillaries, rather than of arterioles (which dilate more slowly), and that this is achieved by a relaxation of contractile pericytes.3,4 Pericytes also play a key role in pathology: in ischaemia pericytes constrict capillaries, trapping blood cells, which prevents microcirculatory reperfusion after clot removal in stroke.4,5 This has made pericytes an important therapeutic target.6,7
Paradoxically, a recent paper 8 dismissed the idea that pericytes are involved in the regulation of brain blood flow, despite confirming all the results of earlier work showing a role for pericytes. How can these conclusions be reconciled? We will show that the differences between these papers are apparent rather than real, and depend on how pericytes are defined. We conclude that a more objective definition of different sub-classes of pericyte along the capillary bed is needed in order to develop novel therapeutic approaches for stroke and other disorders with a vascular component, including Alzheimer’s disease.9,10
The definition and heterogeneity of pericytes
Pericytes were first characterised by Eberth
11
and Rouget
12
in the 1870s, as spatially isolated cells with a bump-on-a-log morphology present on the outside of capillaries, both on straight parts of the capillaries and at capillary branch points. They can be identified by their expression of the growth factor receptor PDGFRβ and of the proteoglycan NG2 which is a co-receptor for PDGF. They are surrounded by basement membrane, and extend processes both along and around capillaries, with more circumferential processes at the arteriole end of the capillary bed, more longitudinal processes in the middle of the capillary bed, and a stellate morphology at the venule end of the capillaries13–16 (see Figure 1, taken from Hartmann et al.
14
). Pericytes were first named by Zimmermann
13
in 1923 (translated into English by Krueger and Bechmann
17
). Zimmermann stated explicitly that he included all of these different morphology cells in the definition of pericytes, as follows13,17: ‘I will henceforth call these cells “pericytes” including their transitional forms to smooth muscle fibres’.
Schematic (reproduced from Hartmann et al.
14
with permission) showing the diversity of pericyte forms, based on imaging of the brain microvasculature in mice expressing fluorescent proteins under control of the NG2 and PDGFRβ promoters.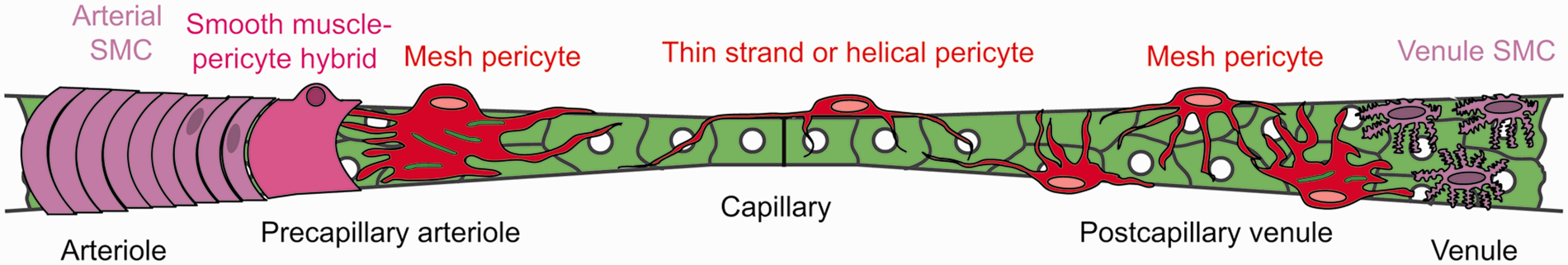
This definition of pericyte, embracing their different subtypes, has been used extensively in the subsequent literature examining the ultrastructure and function of pericytes. It has long been recognised that pericytes vary, not only in their morphology, but also in their protein expression, with the pericytes that give out more circumferential processes also expressing more smooth muscle α-actin (α-SMA), when assessed either with antibody labelling 18 or with expression of reporter dyes under control of the α-SMA promoter. 8 Nevertheless, a significant number of mid-capillary pericytes do also express α-SMA. 19
Control of blood flow by pericytes
In the retina,3,20 cerebellum 3 and cerebral cortex, 4 spatially isolated contractile pericytes were found to alter the capillary diameter in response to depolarization, neurotransmitter action or neuronal activity. Notably, in the cerebral cortex in vivo, almost as many capillaries dilated to neuronal activity as did penetrating arterioles, the capillary dilation occurred before the arteriole dilation and the occurrence of capillary dilation correlated with the presence of a pericyte on the vessel. 4 This implies that capillary dilation is caused by an active relaxation of pericytes, rather than being a passive response to an increase of local blood pressure produced by arteriole dilation.
Pericytes were also reported to contract during ischaemic conditions,3,4 constricting capillaries and trapping blood cells in their lumen, 5 and later dying.4,21 The resulting long-lasting capillary constriction 22 is expected (if it is not blocked pharmacologically5,7) to restrict microvascular reperfusion when a thrombus in a culprit upstream artery has been removed, which will contribute to the no-reflow phenomenon after stroke. In addition, death of pericytes is expected to result in the loss of their maintenance of the blood–brain barrier.23–25 Together, these pericyte malfunctions will promote neuronal death, which makes defining different subtypes of pericyte all the more important for the generation of therapies targeting these cells.
Definition drift
The definition of pericytes given above implies that the vascular bed within the brain parenchyma can be divided as follows:
arterioles, wrapped by a continuous layer of smooth muscle; capillaries, with pericytes of various morphologies on their surface; and venules, also with pericytes on them.
Normally, we define capillaries to be less than 10 µm in inner diameter, although the mean diameter in cortex in vivo is 4 <5 µm. While recognising that there are gradual transitions between these vessel types, we have omitted from this list the vague and confusing term pre-capillary arterioles. If a vessel is wrapped by a continuous layer of adjacent smooth muscle cells then we define it to be an arteriole, while if there is only an occasional spatially isolated contractile cell on a vessel then (by Zimmerman’s definition 13 ) that cell is a pericyte, and the lack of adjacent smooth muscle cells implies that the vessel cannot be an arteriole of any type.
In experiments using transgenic mice to label pericytes with fluorescent proteins driven by the promoters for NG2 or α-SMA, a radical redefinition has been proposed for some of the cells that are conventionally termed pericytes.
8
Beautiful imaging data in this paper
8
clearly show the morphology of the layer of adjacent smooth muscle cells around arterioles (Figure 2a) and of spatially isolated NG2-expressing pericytes on capillaries (Figure 2b). However, Hill et al.
8
used an unconventional nomenclature for the cells on the capillary walls. They asserted that the cells on capillaries near the arteriole end of the capillary bed (arrowheads in Figure 2b), which express more α-SMA and have more circumferential processes running around the capillaries, have a morphology more similar to smooth muscle cells (i.e. as in Figure 2a) than to the (visually almost identical) cells on capillaries further from the arteriole, which have fewer circumferential processes (arrows in Figure 2b). This is surprising because smooth muscle cells have an inconspicuous soma and extend broad processes (as much as 7 µm wide) that wrap around arterioles, whereas pericytes have isolated highly visible somata with a bump-on-a-log morphology and much thinner processes (2 µm or less in diameter
14
) extending around and along the capillaries. Because of this supposed similarity of morphology to smooth muscle cells, despite their obvious differences in appearance, Hill et al.
8
defined the NG2- and α-SMA-expressing (contractile) cells at the arteriole end of the capillary bed to be smooth muscle cells, rather than pericytes, as they would have been named under the original Zimmermann definition.13,17
(a) NG2-expressing cells on arteriole (smooth muscle cells) and venule from Hill et al.
8
(b) NG2-expressing cells on the microvasculature from Hill et al
8
: arteriole end of the capillary bed is at the left; venule end is at the right. Arrows denote pericytes. Arrow heads denote cells which Hill et al.
8
defines to be smooth muscle cells similar to those in panel a, but which we (and Zimmermann
13
) would define to be pericytes, like the cells labelled with arrows. (Both panels reproduced from Hill et al.
8
with permission; green channel enhanced for clarity).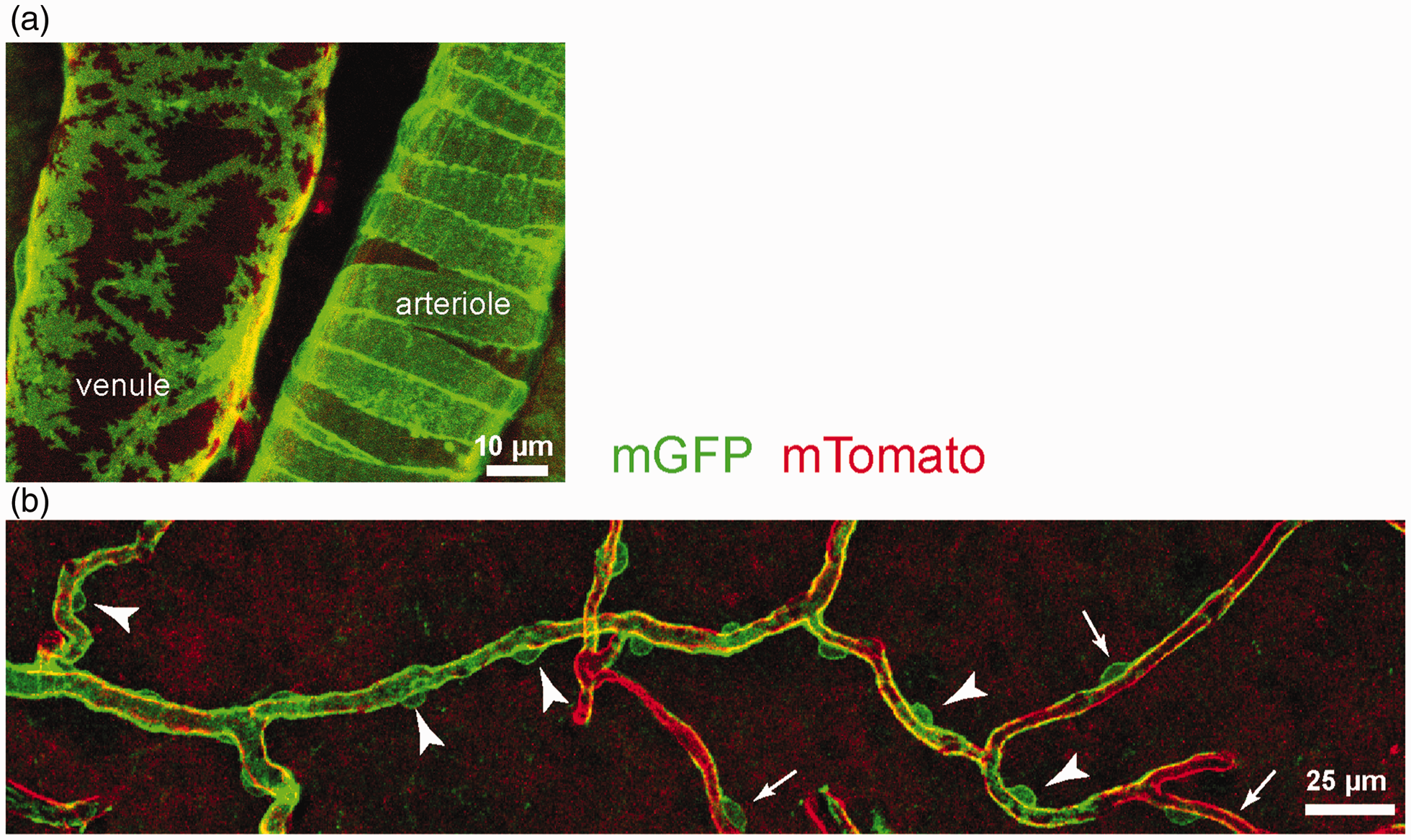
This means that when, in subsequent experiments, they examined capillary diameter changes occurring during spontaneous vasomotion, optogenetic activation, neuronal activity or spreading depression, they concluded that the dilations were produced by relaxations of smooth muscle cells rather than pericytes (see also ref. 26). 8 This was inevitable: if all contractile pericytes are re-defined to be smooth muscle cells, it has to be the case that the dilations are produced by “smooth muscle cells” and not pericytes. Nevertheless, the cells producing the dilations in the study of Hill et al. 8 and in earlier work3,4 are identical. Indeed, when examining the branching order of capillaries, with branch order 0 being the penetrating arteriole, 1 being the first capillary (sometimes termed a pre-capillary arteriole) coming off that, and so on, Hill et al. 8 found that their α-SMA reporter was expressed in pericytes down to at least the second order capillary in nearly 60% of cases, and in higher branch order capillaries in ∼30% of cases (see Figure 4K of their paper which shows α-SMA labelling on spatially separated pericytes along capillaries). This is consistent with pericyte-mediated capillary dilations occurring in response to neuronal activity in vivo out to at least the fourth branching order, as reported earlier. 4
Hill et al. 8 found that constriction of spatially isolated cells on capillaries blocked the capillaries during MCAO, and that this block was often prolonged. This is entirely consistent with previous work,4,5 with contractile pericytes on capillaries being responsible: the sole difference is that Hill et al. 8 define these cells, unconventionally, to be smooth muscle cells.
The need for a definition of different sub-classes of pericyte
Why does this difference of cell name matter? We believe it matters because targeting pericyte malfunction offers enormous therapeutic possibilities, but pericytes clearly exist in different subtypes, suggesting diversity of function.
Pericytes closer to the arteriole end of the capillary bed are likely to be preferentially involved in regulating cerebral blood flow because they express more α-SMA and have more circumferential processes. However, because red blood cells have to deform the walls of small capillaries to pass through the vessels, even contraction or relaxation of longitudinal processes along capillaries with no change of baseline diameter could affect blood flow, by altering the stiffness of the vessel wall. As well as altering total blood flow, contraction and relaxation of pericytes will alter capillary transit time heterogeneity, which affects nutrient delivery to the tissue even with constant total blood flow to a region. 27 It is the contractile pericytes that constrict capillaries during and after ischaemia,4,5 and thus these cells that need to be targeted therapeutically to reduce the no-reflow phenomenon after stroke.
Pericytes in the middle of the capillary bed that express less α-SMA may be more important for maintaining the blood–brain barrier, which is also damaged after stroke, and so these pericytes should be the subject of therapies aiming to maintain blood–brain barrier function. Finally, pericytes at the venule end of the capillary bed may regulate immune cell entry to the brain parenchyma (as in other tissues 28 ), which is also greatly increased after stroke. All of these pericyte sub-classes may also play varying pathophysiological roles in other CNS disorders such as Alzheimer’s disease 9 and CADASIL 29 (cerebral autosomal-dominant arteriopathy with subcortical infarcts and leukoencephalopathy).
The existence of these (at least) three sub-classes of pericyte, which were originally recognised by Zimmermann when he defined pericytes,13,17 and which make different contributions to cerebral malfunction in disease, implies that we need to be conceptually very clear about which cells we are referring to in publications. Lumping some pericytes into the same class as vascular smooth muscle cells, which they are very different from in morphology and function, will sow confusion in the field. The superficially different claims of the Hill et al. paper
8
and earlier papers3–5 highlight three points that are crucial for the field. We need to:
acknowledge that there is heterogeneity of pericytes in terms of morphology and contractile (and other) protein expression as a function of position along the capillary bed; quantify the differences in protein expression and function of different pericyte sub-types; invent suitable nomenclature for the different pericyte subtypes.
Only when all this is done will it be possible to target different pericyte subtypes (e.g. contractile pericytes30,31 versus those maintaining blood–brain barrier function) to generate novel therapeutic approaches for treating stroke, Alzheimer’s disease and other disorders in which pericyte malfunction is involved.
Footnotes
Funding
This study was supported by grants to DA from the Wellcome Trust, European Research Council and MRC and to TD from the Turkish Academy of Sciences and Hacettepe University Research Fund.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
