Abstract
This study was aiming at investigating the extent of neuronal damage in cases of traumatic brain injury (TBI) with diffuse axonal injury (DAI) using 123I-iomazenil(IMZ) SPECT and MRI. We compared the findings in 31 patients with TBI without any major focal brain lesions and 25 age-matched normal controls. Subjects underwent 123I-IMZ SPECT and MRI, and also assessment by cognitive function tests. The partial volume effect of 123I-IMZ SPECT was corrected using MRI. In the patients with TBI, decreased spatial concentration of 123I-IMZ binding was detected in the medial frontal/orbitofrontal cortex, posterior cingulate gyrus, cuneus, precuneus, and superior region of the cerebellum. ROC analysis of 123I-IMZ SPECT for the detection of neuronal injury showed a high diagnostic ability of 123I-IMZ binding density for TBI in these areas. The decreased 123I-IMZ uptake density in the cuneus and precuneus was associated with cognitive decline after the injury. In the patients with TBI, brain atrophy was detected in the frontal lobe, anterior temporal and parietal cortex, corpus callosum, and posterior part of the cerebellum. Evaluation of the neuronal integrity by 123I-IMZ SPECT and MRI provides important information for the diagnosis and pathological interpretation in cases of TBI with DAI.
Introduction
Patients with traumatic brain injury (TBI) suffer from various kinds of neurological disorders, including neurobehavioral disabilities, such as impaired attention, memory disorders, executive function disorders, motivational deficits, and personality changes. Even patients with a history of mild or blunt head injury with no or subtle abnormalities detected on magnetic resonance imaging (MRI) may present with annoying complaints such as fatigue, poor concentration ability, and memory disorders. These symptoms/complaints often have unfavorable effects on the education and employment worthiness of the patients, followed by serious economic problems and unfortunate consequences in terms of the quality of life of the patients. A positive history of TBI is thought to be a risk factor for neurodegenerative diseases and psychiatric comorbidities, including Alzheimer’s disease. 1 Accurate diagnosis is needed to provide appropriate treatment or rehabilitation for the patients. In addition to clinical examinations and neuropsychological testing, image-based objective examination modalities are often required in patients with TBI to enable them to obtain financial support from insurance companies or public funds.
Although MRI is the diagnostic imaging modality of first choice for patients with TBI, diagnosis by MRI is difficult in some cases with diffuse axonal injury (DAI), where the apparent damage to the brain is small or a long time has passed since the injury. 123I-iomazenil (IMZ) single photon emission computed tomography (SPECT) can be used to visualize the distribution of benzodiazepine receptor (BZR) binding, which is thought to reflect neuronal integrity, in the brain. Because BZRs are expressed in the thin cortical grey matter, SPECT or positron emission tomography (PET) images of this tracer are subject to the partial volume effect, particularly in cases with brain atrophy, including in patients with TBI. In the present multicenter study, we attempted to clarify the extent of brain atrophy and loss of neuronal integrity commonly found in patients with TBI in whom the primary pathology is DAI by MRI and 123I-IMZ SPECT with partial volume effect correction (PVC).
Materials and methods
The present study was a multicenter research carried out at the following 8 facilities: Hokkaido University Hospital, Sapporo Medical University Hospital, Iwate Medical University Hospital, Tohoku University Hospital, Saitama Prefectural Rehabilitation Center, Tokyo Medical and Dental University Medical Hospital, Yamaguchi University Hospital, University of Occupational and Environmental Health, Japan, University Hospital.
Patients
Patients from any the above-mentioned facilities aged 20–60 years old with neurobehavioral deficits after traumatic brain injury, in whom DAI was considered as the central feature according to the accident history, symptoms, and absence of severe contusions, hematoma formation, or other pathologies on CT/MRI at the time of the hospital visit were recruited for the study. In this study, DAI was defined as TBI with evidence of cerebral microbleeds in the corpus callosum, brainstem, thalamus, or grey-white matter junction of the cerebrum on T2*-weighted or susceptibility-weighted MRI (SWI).
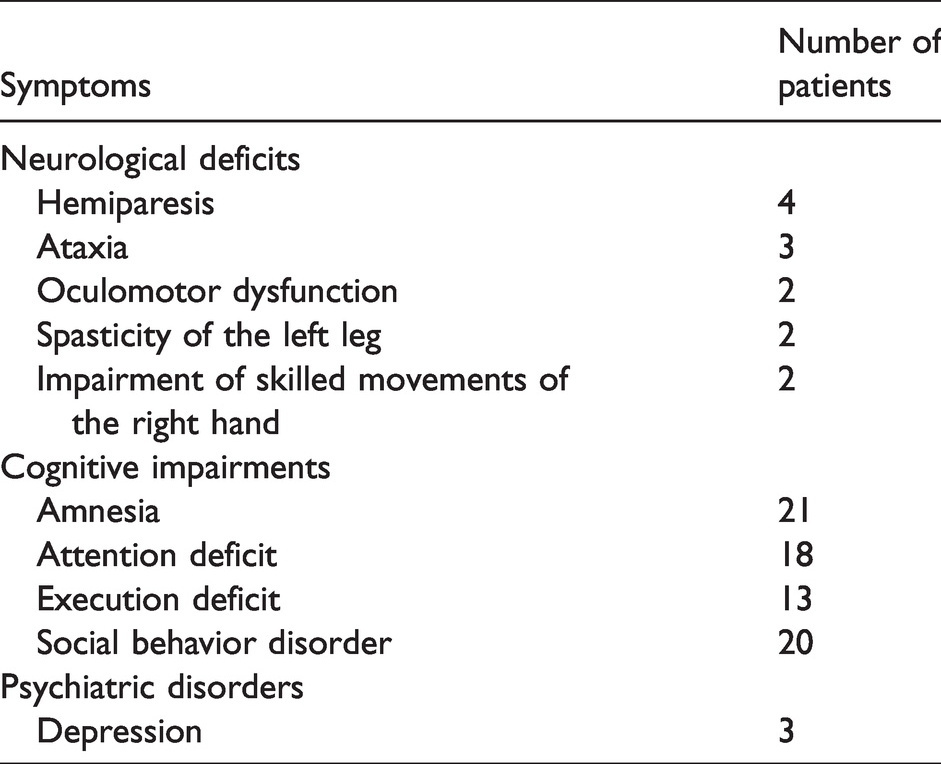
A total of 31 patients with TBI (age = 39 ± 12 years old; Male/Female = 25/6; disease duration = 21 ± 14 months (range, 4 to 59 months)) were enrolled in the study. Although no microbleeds were found on T2* or SWI in one patient, T2-weighted images after recruitment showed a small cystic lesion suspected as being post-thalamic hemorrhage. Their neurological manifestations at the time of recruitment into the study are shown in Table 1. At the time of the imagings for this research, none of the patients had any severe brain damage, which could potentially affect the image analysis, including segmentations or coregistration.
Neurological manifestations in the patients with TBI.
As the control group, 25 healthy subjects (healthy control (HC) group; age = 44 ± 13 years; M/F = 12/13) were also recruited at the Osaka University Hospital. None of these subjects had any history of neurologic or psychiatric symptoms; furthermore, none of them exhibited any abnormal findings on T1-weighted or T2-weighted MR imaging. Each subject was asked to undergo a cognitive function screening test conducted using the Japanese version of the Neurobehavioral Cognitive Status Examination Battery, Cognistat, 2 on the day of the SPECT examination.
All the subjects, except for one patient, were right-handed. As for the patients, the interval at which the imaging examinations for this study were conducted was 13 ± 15 days, and the maximum interval until all the examinations, including the cognitive test, were completed was 38 ± 61 days. The time interval from the onset of traumatic brain injury to the MRI and SPECT examinations was 21 ± 14 and 21 ± 13 months, respectively.
Written informed consent was obtained from the participants, in accordance with the principles laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Approval for the entire study was first obtained from the ethics committee of Tohoku University Hospital (2015-2-185), and then the study was performed with the approval of the institutional ethics committee of each of the aforementioned 8 facilities. The study in the healthy control group was conducted with the approval of the ethics committee of Osaka University Hospital. This study was registered to the University Hospital Medical Information Network (UMIN) Clinical Trials Registry, trial no. UMIN000008095 (https://upload.umin.ac.jp/cgi-open-bin/ctr/ctr_view.cgi?recptno=R000009528).
SPECT
All the subjects enrolled in the study, including the healthy subjects and patients with TBI underwent 123I-IMZ SPECT. 123I-IMZ (167 MBq) was first injected into the antecubital vein, and the brain SPECT scanning was started 3.5 hours after administration of the tracer. Images were acquired using 4 different scanners at the 9 facilities, using acquisition protocols specific to the scanners (Table S1).
Using the projection data acquired at each facility, images were reconstructed using the QSPECT image reconstruction package developed by Iida et al. 3 The objectives of QSPECT are to exclude variable factors that occur during SPECT imaging and image reconstruction, to minimize inter-facility differences which are larger than those in MRI, and to harmonize the images. This software, in brief, extracts head outlines from the projection data, makes µ maps, and performs scatter correction using transmission-dependent convolution subtraction. Attenuation correction with ordered subset expectation maximization is performed using the µ maps to reconstruct the images (voxel size = 1.6 mm, 1.6 mm, 2.0 mm). Gaussian point spreading function with the scanner-specific FWHM was applied to each image in order to unify the spatial resolution. The reconstructed SPECT images were corrected for PVE according to the method described below.
MRI
Three-dimensional (3D) MRI was performed using the T1-weighted gradient echo sequence (image matrix: 256 × 256 or larger, slice thickness: less than 1.4 mm) (Table S2). In the patients with TBI, T2-weighted images, T2* weighted images and fluid-attenuated inversion recovery (FLAIR) images (slice thickness: less than 5 mm) were also acquired for clinical evaluations.
Partial volume effect correction (PVC) of SPECT
PVC of SPECT images was performed using a method similar to that described for our previous study.4–6 3D T1-weighted MR images were first resliced into transaxial sections, segmented into grey matter and other compartments, and then spatially normalized using the “new segment” function of Statistical Parametric Mapping (SPM) 8 (Wellcome Department of Imaging Neuroscience: http://www.fil.ion.ucl.ac.uk/spm/). By this process, segmented tissue maps, including grey and white matter maps in original coordinates, normalized segmented tissue maps, and spatial normalization matrices were generated. The grey matter maps in original coordinates were convoluted with a 3D Gaussian function with a full-width at half maximum (FWHM) of 20 × 20 × 20 mm, which was assumed to be the same as the point spread function (PSF) of the reconstructed SPECT images, as described in previous studies.7,8 The resulting images were subsequently referred to as the “smoothed grey matter map.” The SPECT images were coregistered to the smoothed grey matter maps using the linear coregistration function in SPM8. The coregistered SPECT images were then divided using the smoothed grey matter map on a voxel-by-voxel basis to obtain PVE-corrected SPECT images.
SPM analysis
Spatially normalized SPECT images after PVC obtained from the TBI and HC groups were statistically compared. The “two-sample T-test” was performed using SPM8 with proportional scaling of the voxel values or explicit masking based on the corresponding segmented tissue map, to limit the areas for comparison. The statistical data were corrected for multiple comparisons using cluster-level inference. Correlation analyses between the SPECT images and the cognitive test scores were also performed using SPM8. The resultant T-score maps obtained from the SPM analysis were analyzed based on the Automated Anatomical Labeling (AAL) 9 using an in-house tool,5,10 in order to evaluate the distribution and extension of regions with significant changes in 123I-IMZ uptake.
Voxel-based morphometry (VBM)
Using the “new segment” function of SPM8, the following two types of normalized segmented tissue maps were generated: tissue maps without modulation, and modulated tissue maps obtained by multiplying the Jacobian determinants of the corresponding spatial normalization matrices, which corrects the local volumetric contraction or dilatation that the images undergo during warping. For the purpose of VBM, the modulated grey and white matter maps obtained in the TBI and HC groups were smoothed by a 3D Gaussian function with a FWHM of 20 × 20 × 20 mm (tissue-concentration-map), and statistically compared. Correlation analyses between the tissue-concentration-maps and cognitive test scores using SPM8 were also performed with the addition of age as a nuisance covariate. The resultant T-score maps obtained from the VBM analysis were analyzed by the same method as the SPM analysis.
ROC analysis
Spatially normalized SPECT images after PVC were assessed based on the AAL, to calculate the regional 123I-IMZ uptake ratios to the mean cerebral uptake, using an in-house tool.5,10 The mean cerebral uptake was measured using the SPM apriori/grey.nii mask thresholded at p > 0.5. ROC analysis of the regional 123I-IMZ uptake ratios was performed to clarify the diagnostic ability of 123I-IMZ SPECT for TBI using the software IBM SPSS Statistics, ver. 20.0. The optimal cutoff values were determined based on the Youden Index. 11
Cognitive tests
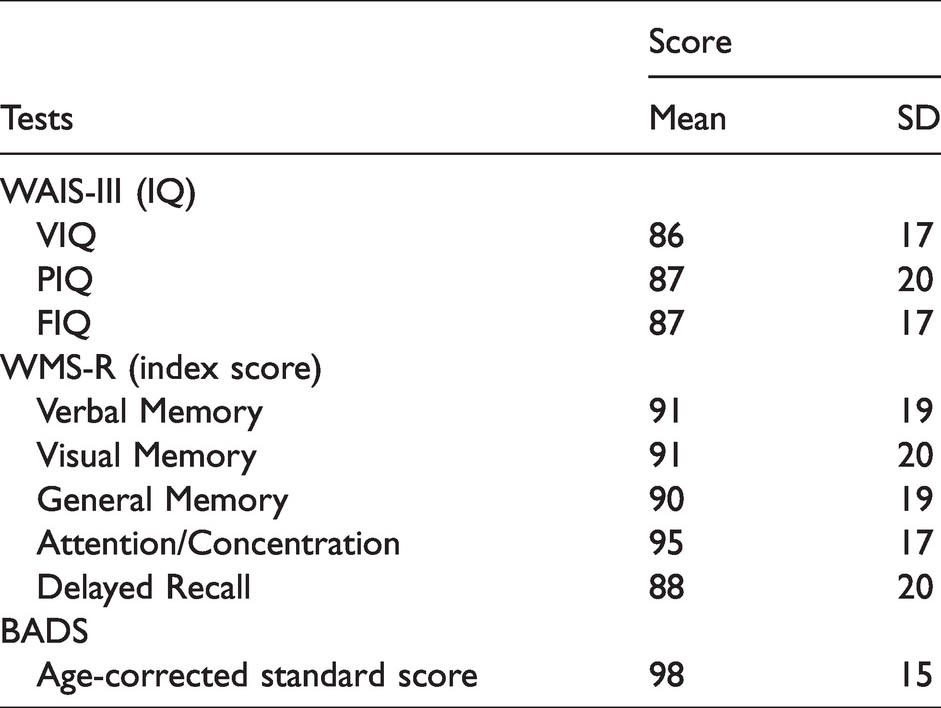
A total of 33 patients underwent evaluation by the Wechsler Adult Intelligence Scale—Third Edition (WAIS-III) and Wecheler Memory Scale-Revised (WMA-R), and 32 patients were examined by the Behavioral Assessment of the Dysexecutive Syndrome (BADS). These cognitive tests were performed within 2 months after recruitment into the study.
Image interpretations
Interpretation of the MR images was performed in a collaborative manner by an experienced psychiatrist and a well-trained neuroradiologist, for the purpose of assessing the brain lesions. Cerebral hemorrhage was evaluated in T2* images and brain contusions were diagnosed based on the findings in T1-weighted images, T2-weighted images, or FLAIR images. The psychiatrist and neuroradiologist performing the interpretation were blinded to clinical information about the patient.
Results
The results of the cognitive testing are shown in Table 2. Interpretation of the findings of MRI revealed that all the areas of cerebral hemorrhage and contusions were small lesions measuring less than 1 cm in diameter, and were predominantly located in corpus callosum, the lateral part of the frontal, parietal, and temporal lobes (Figure 1). In the SPECT, significant reduction in 123I-IMZ binding density in the bilateral cingulate gyrus, precuneus, cuneus, orbitofrontal cortex, medial frontal cortex, medial occipital cortex, and superior cerebellar region, and right mesial temporal cortex was demonstrated by SPM analysis in the TBI patients (Figure 2(a)) (Table S3). VBM analysis revealed significant volume reduction of the grey matter in the frontal regions bilaterally, anterior temporal cortex, angular cortex, superior parietal lobule, posterior cerebellar region, and right supramarginal gyrus in the TBI patients (Figure 2(b)) (Table S4-1). Atrophic changes of the white matter were significant in the frontal subcortical areas and posterior part of the corpus callosum (Figure 2(c)) (Table S4-2). Correlation analysis using SPM8 revealed a significant positive correlation between the 123I-IMZ binding density in the precuneus and cuneus and the score of BADS in the TBI patients (Figure 3). According to the ROC analysis based on the AAL regions, the area under the curve (AUC) was the highest (>0.9) in the middle cingulate gyrus, and moderately high (from 0.9 to 0.8) in the posterior or anterior cingulate gyrus, rectus, cuneus or precuneus, calcarine, and superior region of the cerebellum (Figure 4).
Cognitive test scores.
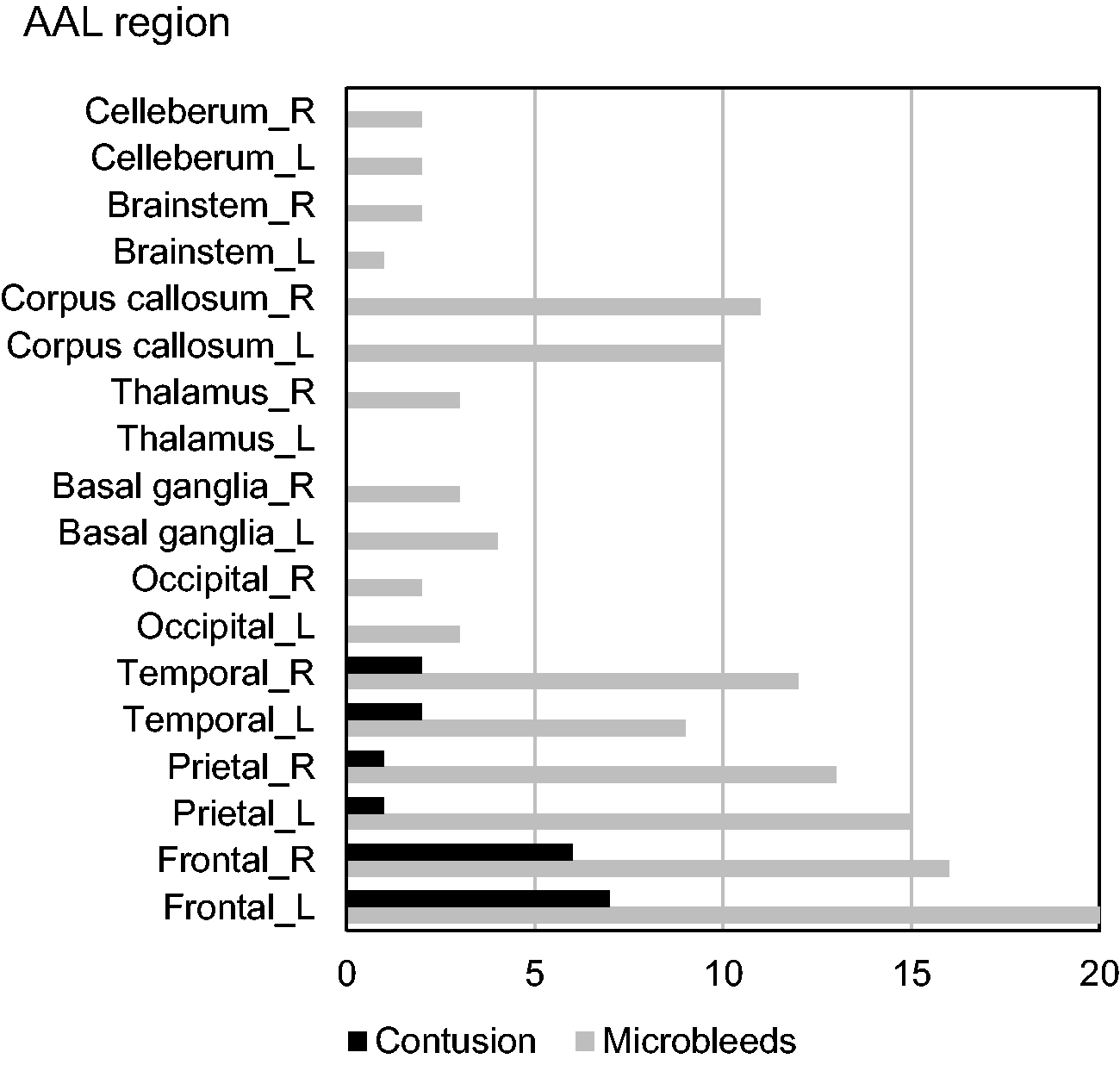
The bar graph shows the number of microbleeds and cerebral contusion in each area. Areas without lesions are omitted; no lesions were found in the medial frontal lobe, medial temporal lobe, or medial parietal lobe. All the lesions were less than 1 cm in diameter.
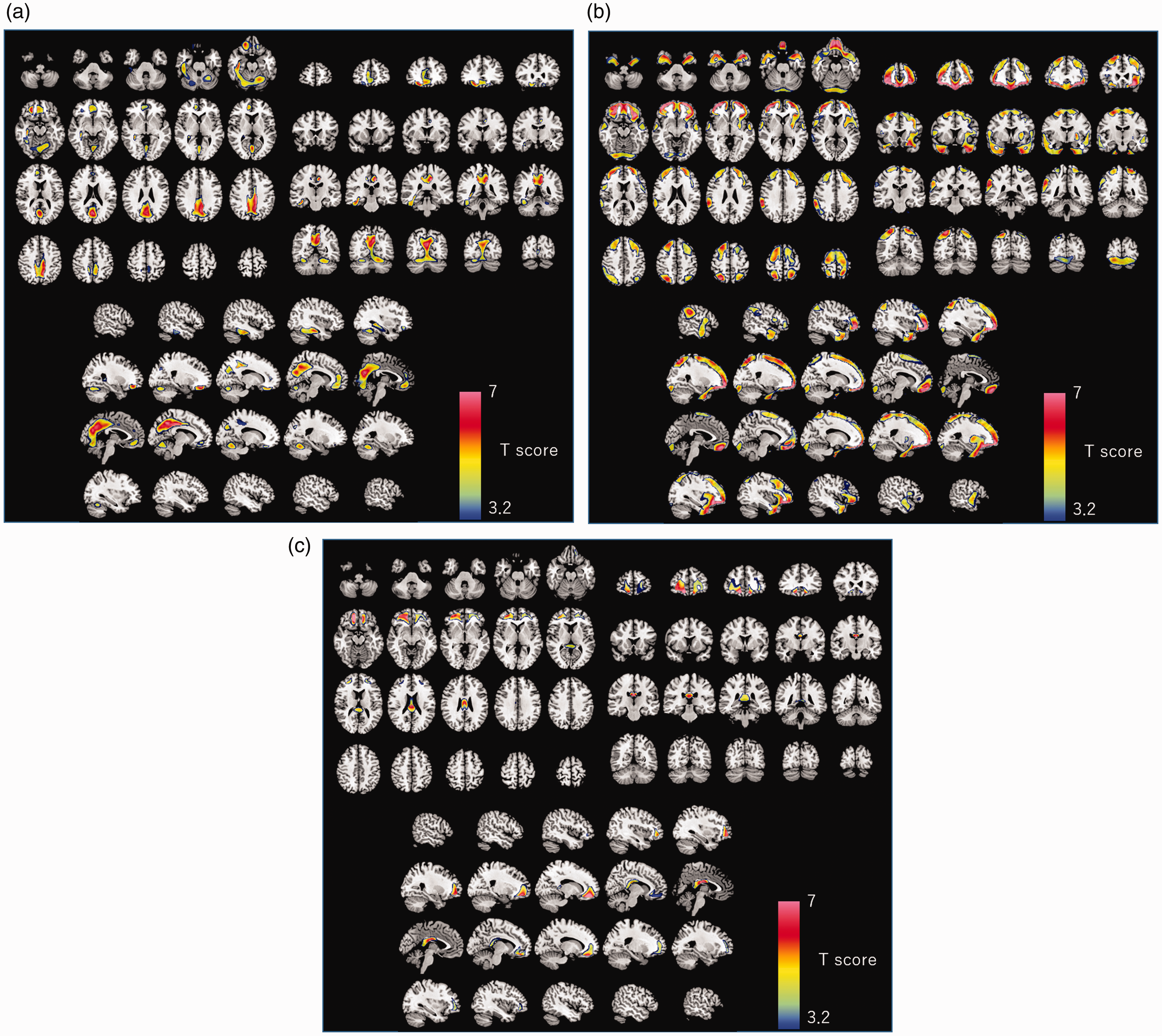
The results of the SPM analysis are overlaid on to the transaxial, coronal, and sagittal sections of the T1-weighted MRI template. Colored voxels indicate T scores showing significantly decreased 123I-IMZ binding (a), significantly decreased grey matter (b), and significantly decreased white matter volume (c) in the cases of TBI as compared to the normal controls. The significant threshold (T = 3.2) is corrected for multiple comparisons at the cluster level, with the p value set at 0.05 (family-wise error correction).
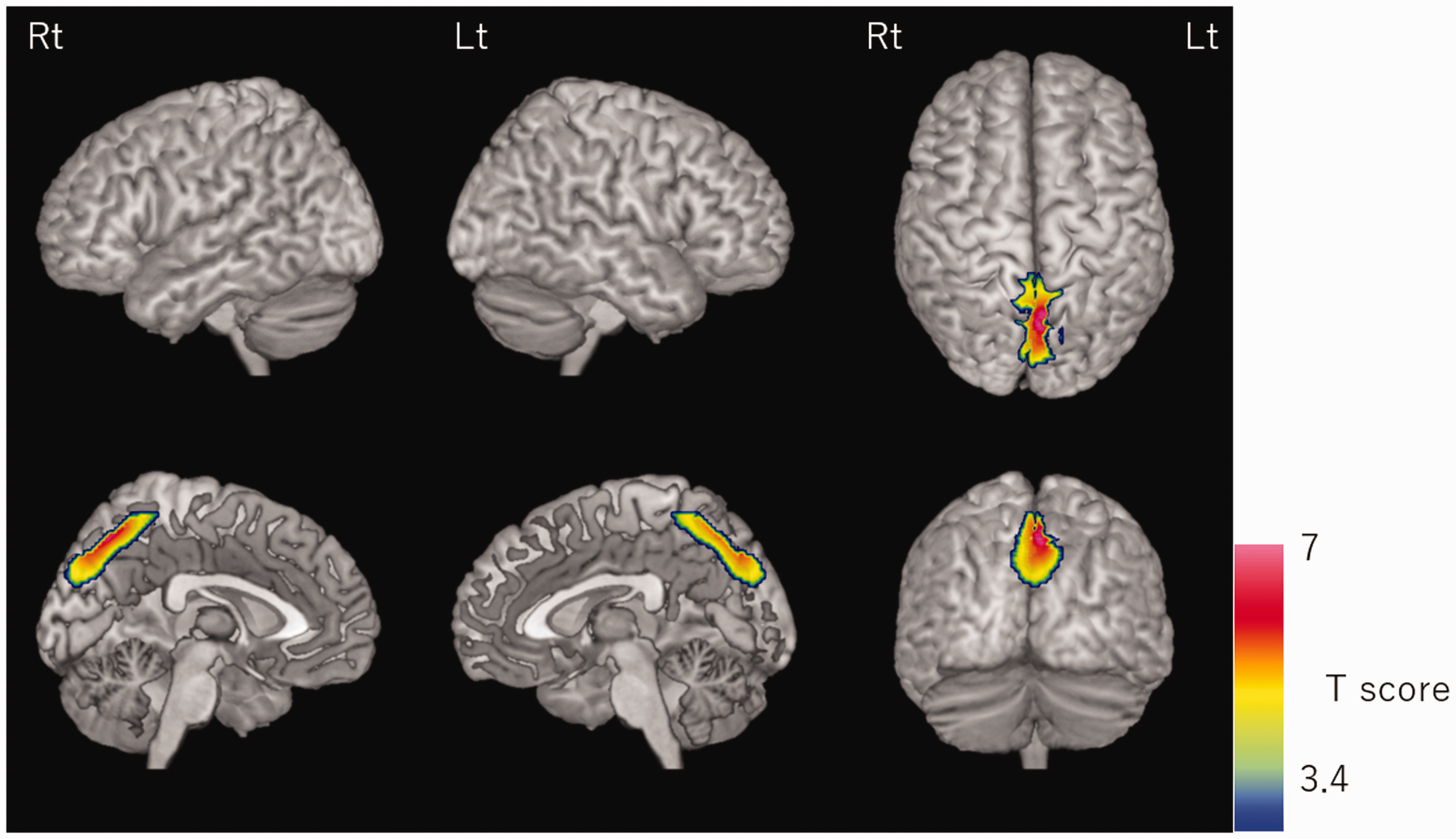
The results of the voxel-based correlation analysis between SPECT and the cognitive test scores was rendered on the 3D surface image of the template MRI. Colored voxels indicate T scores showing significant positive correlation between the 123I-IMZ binding and scores on the BADS test. In order to limit the analysis target to the region where the 123I-IMZ binding was significantly decreased in the cases of TBI, the significant clusters (height threshold was p = 0.05, uncorrected) obtained by SPM comparison of 123I-IMZ SPECT between the normal controls and the TBI group was used as the “explicit mask.” The significant threshold (T = 3.4) is corrected for multiple comparisons at the cluster level, with the p value set at 0.05 (family-wise error correction).
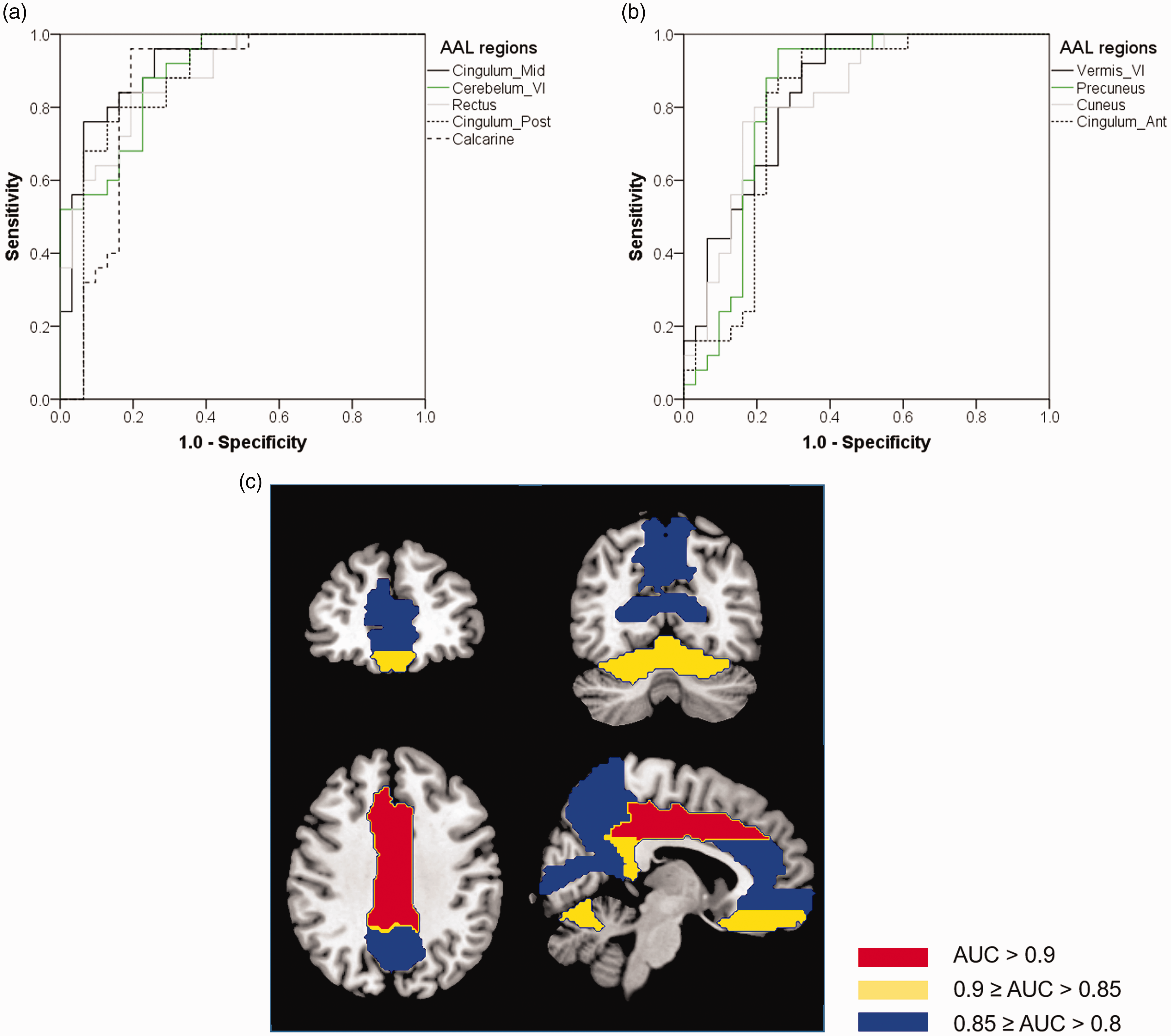
ROC analysis for regional 123I-IMZ uptake ratio based on the AAL was performed to clarify the key brain regions to diagnose TBI by 123I-IMZ SPECT. Each pair of left and right regions of AAL was combined into one region. The AUC of the corresponding curves, and sensitivity and specificity at the optimal cutoff values of the 123I-IMZ uptake ratio in (a) and (b) were as follows: (a) Cingulum Mid (AUC = 0.916, sensitivity/specificity = 0.960/0.742, cutoff value = 1.12); Cerebellum VI (0.893, 0.880/0.774, 0.913); Rectus (0.885, 0.840/0.806, 1.05), Cingulum Post (0.872, 0.800/0.839, 1.09); Calcarine (0.855, 0.960/0.806, 1.32), (b) Vermis IV (0.841, 1.00/0.613, 0.914); Precuneus (0.828, 0.960/0.742, 1.02); Cuneus (0.828, 0.800/0.806, 1.15); Cingulum Ant (0.800, 0.960/0.677, 1.08). The above regions were classified by AUC value and mapped on to the template MRI (c).
Discussion
123I-IMZ is a tracer that binds to the benzodiazepine-binding site of the GABAA receptor, and its signal reflects the density of the BZR, the GABAA receptor. GABAA receptors are located in axo-somatic and axo-dendritic synapses in the pyramidal cells and granule cells in the brain12,13 and are considered as a cortical marker of neurons. 14 The distribution of GABAA neurons in the cerebral cortex is thought to be uniform throughout the cortical layers, with mild variations among brain regions. 14 Since the total number of normal neurons is reflected in the number of GABAA receptors, it is considered that the density of normal neurons in the cerebral cortex can be estimated by measuring the density of GABAA receptors. Therefore, the local distribution of BZR binding is used as a surrogate marker of normal neuron density i.e., of neuronal integrity, in the brain. 15
In the present study, statistical analysis of the findings of 123I-IMZ SPECT or MRI was performed in a relatively large number of patients with TBI and normal healthy control subjects. According to the findings on the MR images, DAI was thought to be the main pathology in the patients enrolled in this study, because the discrete lesions in the brain were very small to explain the severe manifestations. Although the responsible lesions differ from case to case, abnormalities common to TBI could be detected in a large number of subjects by voxel or VOI-based statistical analysis. The voxel-based analysis revealed reduced the spatial concentration of 123I-IMZ binding in the inferior part of the frontal cortex, cingulum, medial parts of the parietal/occipital cortices, and superior part of the cerebellum. Grey matter atrophy was found over a broad area, including the frontal, parietal, temporal cortices, and cerebellum. White matter atrophy was also seen in the frontal subcortical area and posterior corpus callosum. In the case of TBI, the impact on the skull is probably transmitted directly to damage the adjacent cortical grey matter, eventually resulting in atrophic changes over a broad area of the cortical grey matter. The most remarkable atrophic changes were observed in the frontal area in the TBI patients, in both the grey and white matter. Particularly in the orbitofrontal regions, both reduction in the spatial concentration of 123I-IMZ binding as well as brain atrophy was found. According to the research based on diffusion tensor imaging, significant reduction in connectivity was evident within the rostral middle frontal gyrus, pars orbitalis, lateral, and frontal orbitofrontal regions in the patients with TBI. 16
Significant atrophy of the posterior part of the corpus callosum containing commissural fibers connecting the parietal cortices of both sides was observed in the patients with TBI in this study.17,18 In a research based on MRI, reduced fractional anisotropy in corpus callosum was reported in patients with TBI. 19
123I-IMZ SPECT showed decreased uptake density in the frontal lobe, medial part of the parietal lobe, floor of the temporal lobe, and cerebellum. Since these regions are in contact with the falx cerebri or tentorium cerebelli, the decrease in 123I-IMZ uptake density was considered to be caused by the impact on these structures. Using tagged MRI, the deformation of the human brain with applied acceleration can be measured in time series. According to a report from such a study, central dividing features, like the falx cerebri and tentorium cerebelli, have a strong impact on the brain. 20
Because the spatial resolution of SPECT is low, without partial volume effect correction (PVC), IMZ SPECT images represent a mixture of both a decrease in BZR affinity as an indicator of reduction of the neuronal integrity and of cerebral atrophy associated with advanced neuronal loss. In this study, the partial volume effect correction of the 123I-IMZ SPECT images eliminated the decrease in uptake proportional to the extent of cerebral atrophy. Therefore, SPECT with PVC allows visualization of the spatial concentration of 123I-IMZ binding, that is, of the density of viable (normal) neurons as an indicator of neuronal integrity.
VBM, on the other hand, exclusively detects atrophy or neuronal loss. If the degree of brain damage is mild, it does not immediately lead to neuronal loss, and it is thought that mainly neuronal integrity would be reduced, 21 in which case long-term neuronal loss would be expected. 15 DAI is characterized by damage not only to the cerebral cortex, but also to central structures such as the corpus callosum. The central structure is particularly vulnerable to damage by central dividing features, such as the falx cerebri. 20
According to an 18F-FMZ PET study whose findings showed good correlation with those of 123I-IMZ SPECT, 22 significant decrease of the regional FMZ uptake was seen in the medial part of the cerebral hemispheres, including the bilateral medial frontal gyri and anterior cingulate gyri, in patients with TBI. 23 The impact on the falx cerebri and tentorium cerebelli is weaker than that on the skull, therefore, only the neuronal integrity was thought to decreased, in the absence of grey matter atrophy. In addition, it is presumed that axonal damage due to central dividing features causes anterograde and/or retrograde degeneration in the proximal and distal regions of the damaged axons, resulting in a decrease in the neuronal integrity.
18F-FDG PET studies have revealed regional glucose hypometabolism in the medial frontal lobes bilaterally and the anterior cingulate cortex in patients with cognitive impairment after diffuse TBI, as compared to the normal controls24,25 Nakashima et al. clarified that dysfunction of the cingulate gyrus, lingual gyrus, and cuneus may play a crucial role in the development of the neuropsychological deficits after TBI. 25 In the present study, a significant positive correlation between the BADS scores and 123I-IMZ uptakes was found in the cuneus and precuneus. These regions are adjacent, with some overlap, to the region in which disease-specific reduction in the uptake was found in patients with TBI, and, therefore, account for the TBI-associated cognitive decline. Although a VBM study 26 reported the existence of a correlation between the cortical grey matter volume and attention in TBI, no association was found between brain atrophy and the cognitive test scores in this study. It is difficult to compare the results of these studies because the number of samples and the methods of multiple comparison corrections were different.
According to a report on the longitudinal patterns of brain atrophy in TBI, 27 no atrophy of the medial parietal lobe, posterior cingulate gyrus, or precuneus was detected at the baseline, although brain atrophy of the lateral cortices, mainly in the frontal lobe and temporal lobe, was evident at the baseline. On the other hand, significant progression of grey matter atrophy over a wide area was clarified in cases of TBI, including the inferior frontal lobe, medial parietal lobe, posterior cingulate gyrus, occipital lobes, and cerebellum, where TBI-related reduction in 123I-IMZ uptake was found in this study. The decreased BZR affinity detected by 123I-iomazenil SPECT may represent a change in neuronal integrity that precedes future brain atrophy and neurodegeneration.
The posterior cingulate gyrus, precuneus, and medial prefrontal cortex in which markedly decreased 123I-IMZ binding density was found are also the key hubs of the default mode network (DMN), in addition to the parietal cortex, which also showed atrophy in this study. In a 18F-FDG PET study, hypometabolism in the posterior cingulate gyrus was reported in cases of TBI. 25 In addition to the mild mechanical damage caused by central dividing features, major damage to the medial prefrontal cortex or parietal cortex by strong impact from the skull may have caused functional damage to the posterior cingulate gyrus and precuneus via the DMN. The posterior cingulate gyrus and precuneus are the pathologic sites at which hypometabolism and low cerebral blood flow are detected in Alzheimer’s disease and dementia with Lewy bodies.
In addition to group comparisons, in regard to the diagnosis of individual patients, ROC analysis showed that TBI can be diagnosed accurately based on the 123I-IMZ binding density in the cingulate gyrus, medial regions of frontal, parietal, and occipital lobes, and superior part of the cerebellum. 123I-IMZ SPECT with partial volume effect correction was shown to be significantly reliable as a clinical diagnostic imaging tool for patients with TBI.
The limitation of the present study was weak detectability of 123I-IMZ SPECT for deep cerebral structure lesions. Some previous studies have pointed out that reduced fractional anisotropy, 28 reduced cerebral blood flow, 29 or reduced FMZ binding 23 in the thalamus is observed in patients with TBI, and that these changes were associated with cognitive decline. Since 123I-IMZ binding in the deep regions of the cerebrum, such as the thalamus, is weak, the evaluation ability of 123I-IMZ SPECT in that part of the brain is limited.
In conclusion, in patients with TBI in whom the major pathology was DAI, decreased spatial concentration of 123I-IMZ binding, that is, decreased neuronal integrity, was detected in the medial frontal or orbitofrontal cortex, posterior cingulate gyrus, cuneus, precuneus, and superior region of the cerebellum. Brain atrophy, namely neuron loss, in the patients with TBI was detected in the frontal lobe, anterior temporal and parietal cortex, corpus callosum, and posterior part of the cerebellum. 123I-IMZ SPECT with PVC provides reliable diagnostic information and relevant insights into the pathology and mechanism of injury in cases of TBI.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221113001 - Supplemental material for Impaired neuronal integrity in traumatic brain injury detected by 123I-iomazenil single photon emission computed tomography and MRI
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221113001 for Impaired neuronal integrity in traumatic brain injury detected by 123I-iomazenil single photon emission computed tomography and MRI by Hiroki Kato Jyoji Nakagawara, Kenji Hachisuka, Jun Hatazawa, Katsunori Ikoma, Eiichi Suehiro, Hidehiko Iida, Kuniaki Ogasawara, Osamu Iizuka, Sumio Ishiai, Tadashi Ichikawa, Tadashi Nariai, Tetsuya Okazaki, Tohru Shiga and Etsuro Mori in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Foundation for Advancement of International Science (FAIS).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
The specific contribution of each author is noted below: Hiroki Kato: study conception and design, acquisition of data, analysis and interpretation of data, and drafting of manuscript; Jyoji Nakagawara: interpretation of data; Kenji Hachisuka: acquisition and interpretation of data; Jun Hatazawa: interpretation of data; Katsunori Ikoma: acquisition and interpretation of data; Eiichi Suehiro: acquisition of data; Hidehiko Iida: analysis and interpretation of data, and development of software; Kuniaki Ogasawara: acquisition and interpretation of data; Osamu Iizuka: study conception and design, and research secretariat; Sumio Ishiai: acquisition of data; Tadashi Ichikawa: acquisition of data; Tadashi Nariai: acquisition of data; Tetsuya Okazaki: acquisition and interpretation of data; Tohru Shiga: acquisition and interpretation of data; Etsuro Mori: study conception and design, and critical revision.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
