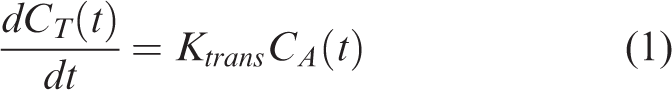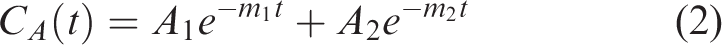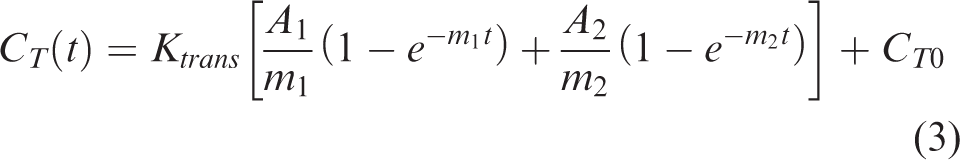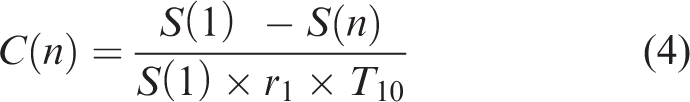Abstract
Cerebral edema and the subsequent increased intracranial pressure are associated with mortality and poor outcome following traumatic brain injury. Previous in vitro studies have shown that the Gibbs-Donnan effect, which describes the tendency of a porous, negatively charged matrix to attract positive ions and water, applies to brain tissue and that enzymatic reduction of the fixed charge density can prevent tissue swelling. We tested whether hyaluronidase, an enzyme that degrades the large, negatively charged glycosaminoglycan hyaluronan, could reduce brain edema after traumatic brain injury. In vivo, intracerebroventricular injection of hyaluronidase after controlled cortical impact in mice reduced edema in the ipsilateral hippocampus at 24 h by both the wet-weight/dry-weight method (78.15 ± 0.65% vs. 80.4 ± 0.46%; p < 0.01) and T2-weighted magnetic resonance imaging (13.88 ± 3.09% vs. 29.23 ± 6.14%; p < 0.01). Hyaluronidase did not adversely affect blood–brain-barrier-integrity measured by dynamic contrast-enhanced magnetic resonance imaging, nor did hyaluronidase negatively affect functional recovery after controlled cortical impact measured with the rotarod or Morris water maze tasks. Reduction of fixed charge density by hyaluronidase was confirmed in cortical explants in vitro (5.46 ± 1.15 µg/mg vs. 7.76 ± 1.87 µg/mg; p < 0.05). These data demonstrate that targeting the fixed charge density with hyaluronidase reduced edema in an in vivo mouse model of traumatic brain injury.
Introduction
Cerebral edema following traumatic brain injury (TBI) is associated with poor outcome and increased mortality, as swelling of the brain within the rigid skull can increase intracranial pressure (ICP) and result in coma, brain herniation, and death.1–6 Even a 1% increase in brain water content in humans can increase ICP above 20 mmHg. 7 While strategies to manage elevated ICP following severe TBI include administration of the hyperosmotic agents mannitol and hypertonic saline, hyperventilation, barbiturate coma, and decompressive craniectomy, each of these interventions is associated with adverse effects, and their efficacy is still a major topic of investigation and debate (for review, see Stocchetti and Maas 8 ).
Cerebral edema with a net increase in brain water content leads to an increase in brain volume. Marmarou et al. identified that edema drives brain swelling in human TBI, as opposed to vascular engorgement and an in increase in cerebral blood volume. 9 There are two major types of brain edema: vasogenic edema and cytotoxic edema. Vasogenic edema is an increase in water in the extracellular space due to an osmotic gradient hypothesized to be generated by extravasation of plasma-derived solutes from the vasculature as a result of blood–brain barrier (BBB) breakdown and increased permeability. Cytotoxic edema, or cell swelling, is defined as an increase in water in the intracellular compartment in response to accumulation of osmotically active solutes within the cell.
Regardless of the type of edema, the primary issue is a net accumulation of water in the tissue, increasing ICP, and reducing perfusion. Preclinical studies have identified a number of cellular and molecular mechanisms that contribute to the development of cytotoxic and vasogenic edema, including: excitotoxicity due to excessive glutamate release, mitochondrial dysfunction, ion pump failure, degradation of BBB components by matrix metalloproteinases, inflammation-induced release of vasoactive agents, insertion of aquaporin 4 water channels into the cell membrane increasing bi-directional flow of water driven by the net Starling force, and mechanical injury to the vasculature and tissue.10–12
We hypothesize that the fixed charge density (FCD) of brain tissue may also act as an osmotic driving force that contributes to the development of cerebral edema. Previous studies using triphasic mixture theory to model the swelling behavior of brain tissue support that dead brain tissue swells as described by the Gibbs-Donnan effect.13–16 The Gibbs-Donnan effect describes the tendency of a porous, negatively charged matrix to generate an osmotic gradient that attracts positive ions and water into the matrix causing the matrix to swell. In the brain, glycosaminoglycans (GAGs), such as chondroitin sulfate, are immobilized, negatively charged, matrix-molecules that contribute to the FCD of the tissue. Enzymatically degrading chondroitin sulfate both in vitro 14 and in vivo in a mouse model of TBI 17 reduced tissue swelling, identifying the FCD as a potential osmotic agent of edema.
Hyaluronan is a large, negatively charged matrix molecule that contributes to the FCD in the brain. Hyaluronan is composed of repeating disaccharides of glucoronic acid and N-acetylglucoasmine, can range in size from 2000 to 25000 disaccharides, and forms a backbone which binds other FCD constituents such as GAGs. 18 Additionally, each disaccharide unit can bind and retain 10–15 molecules of water, 19 making hyaluronan a key component for tissue hydration. Increased levels of hyaluronan have been implicated in the development of edema in several conditions, including experimental myocardial infarction 20 and experimental pulmonary edema. 21 Hyaluronan has also been shown to be directly related to extravascular water content in rabbit lungs with hyaluronidase infusion decreasing lung water content. 22
Although hyaluronan has been heavily investigated in other parts of the body and for its roles in other conditions, less is understood about hyaluronan in the brain. In the adult brain, hyaluronan is concentrated around mature neurons 23 and is a key component of perineuronal nets that regulate plasticity. Hyaluronan was originally regarded primarily as a structural molecule, but it is becoming increasingly clear that hyaluronan can mediate a diverse array of biological functions depending on its size and the type of cell and receptor. Hyaluronan may play a role in extracellular matrix remodeling, inflammation, the immune response, and in the proliferation, migration, and differentiation of cells (for review, see literature24–26).
As there are many FCD constituents in the brain, enzymatically targeting any may lead to an attenuation of edema after TBI. The purpose of this study was to investigate whether hyaluronidase, an enzyme that degrades hyaluronan, would reduce edema following controlled cortical impact (CCI) TBI in the mouse.
Methods
Animals
All experimental procedures were approved by the Institutional Animal Care and Use Committee at Columbia University (protocol AAAO6151) and carried out in accordance with the Guide for the Care and Use of Laboratory Animals (NIH). All mice were on a 12 h light/dark cycle, group housed before and after injury, and provided food and water ad libitum. Three month-old male C57BL/6J mice, 28–32 g (Jackson Laboratory, Bar Harbor, ME), were used for in vivo experiments, and brain tissue from 2–3-month-old male and female mice (28–32 g) were used for in vitro experiments. Experiments were conducted and reported per the ARRIVE guidelines.
CCI injury
Mice received 5 mg/kg carprofen intraperitoneally (i.p.) and were anesthetized using 4% isoflurane in oxygen. The scalp was shaved, cleaned, and 2 mg/kg bupivacaine administered subcutaneously. Using aseptic procedure, a midline incision was made, the soft tissues were reflected, and a 5 × 5 mm craniectomy was made between bregma and lambda and 1 mm lateral (left) to the midline. A moderate contusion injury was imparted to the left parietal cortex using a Leica One Stereotaxic Impact device (Leica, Houston, TX) with a 3 mm hemispherical tip with the following injury parameters: impact depth = 1.0 mm, velocity = 4.5 m/s, and dwell time = 0.3 s, as described previously. 27 Sham injury consisted of exposure to the same procedures without cortical impact.
Hyaluronidase administration
Sterile-filtered hyaluronidase (Type VI-S from bovine testes, Sigma Aldrich, St. Louis, MO) or vehicle (sterile PBS) was delivered 4 min after CCI or sham injury via stereotaxic intracerebroventricular (ICV) injection by an investigator blinded to the identity of the solution. For the injection, a second hole was drilled through the skull on the contralateral side, and a pulled glass pipette was inserted at –0.22 mm, 1.0 mm, 2.0 mm relative to bregma. A total of 1 U of hyaluronidase in 10 µl sterile PBS or vehicle was injected into the lateral ventricle at a rate of 1 µl/min using a 10 µl Hamilton syringe (Hamilton Company, Reno, NV) and microinfusion pump (KD Scientific, Holliston, MA). The pipette tip remained in place for 5 min before being withdrawn to minimize backflow. Following removal, the incision site was sutured, topical antibiotic was applied to the incision site, and the animal was placed on a heating pad to recover.
Brain water content
Animals (n = 4–5 per group) were euthanized 24 h after CCI injury via cervical dislocation following deep anesthesia. The brain was extracted and the hippocampi were collected. The ipsilateral and contralateral hippocampi were immediately weighed to obtain wet weight. Samples were then dehydrated for 24 h at 100°C and weighed again to obtain dry weight. Percent water content was calculated as (wet weight–dry weight)/wet weight × 100. Experiments and analysis were conducted by investigators blinded to group and treatment.
T2-weighted MRI for edema
Animals (n = 4 per group) were imaged 24 h following CCI injury using a Bruker Biospec 94/20 9.4 Tesla magnetic resonance imager (Bruker, Billerica, MA) at the Cancer Center Small Animal Imaging Shared Resource at the Columbia University Medical Center’s Herbert Irving Comprehensive Cancer Center. Animals were anesthetized using isoflurane, i.p. catheterized for injection, and positioned in the magnet where anesthesia was maintained and heart rate was monitored throughout scanning. T2-weighted images were obtained using 2D rapid acquisition with refocused echoes (RARE) with the following imaging parameters: pulse repetition time/echo time (TR/TE): 3300/44 ms; echo train: 8; number of excitations: 10; scan time: 13 m 12 s; matrix size: 256 × 196 pixels; spatial resolution: 86 × 86 µm voxels; slice thickness: 500 µm, no interslice gap. Scans were conducted by investigators blinded to group and treatment.
Dynamic contrast-enhanced T1-weighted MRI for BBB permeability
Following the T2-weighted magnetic resonance imaging (MRI) imaging sequence, dynamic contrast-enhanced (DCE) T1-weighted MRI was performed on the same animals without removal from the scanner or changing of alignment. Detection of a gadolinium-based contrast agent Gd-DTPA (Omniscan, GE Healthcare, Chicago, IL) over time was achieved using a 2D FLASH T1-weighted image sequence with the following parameters: TR/TE: 132.4/2.3 ms; number of excitations: 4; scan time: 76 s; matrix size: 256 × 196 pixels; spatial resolution: 86 × 86 µm voxels; slice thickness: 50 µm, no interslice gap; flip angle: 70°. The number of axial slices imaged of the brain ranged from 14 to 15 slices per animal, covering the top of the brain to the base of the neck to capture the carotid artery for determination of the arterial input function (AIF). Thirty-two dynamic acquisitions were taken over a total period of 40 min. Following collection of pre-contrast images, a 300 µl bolus injection of Gd-DTPA was administered through the i.p. catheter for detection of BBB permeability by calculation of Ktrans, the volume transfer coefficient of the Gd-DTPA from the vasculature to the extravascular extracellular space within the brain. Scans were conducted by investigators blinded to group and treatment.
T2-weighted image analysis for edema quantification
Edematous tissue appears hyperintense in T2-weighted MRI. To quantify the volume of edematous tissue, the ipsilateral and contralateral hippocampi were first traced in serial images (6–8 images per animal) using ImageJ (National Institutes of Health, Bethesda, MD). In each image, a circular region of interest (ROI) was placed in uninjured gray matter on the contralateral side to serve as a baseline tissue ROI. A custom program MATLAB r2017a (MathWorks, Inc., Natick, MA) was used to quantify hyperintense pixels in the ipsilateral and contralateral hippocampi indicative of edema. Pixels were considered hyperintense if their intensity value was greater than three standard deviations from the mean intensity of the baseline tissue ROI. The percent edema in a hippocampal ROI was calculated as the sum of the bright pixels in the hippocampal ROI divided by the total number of pixels in the hippocampal ROI. Analyses were conducted by investigators blinded to group and treatment.
T1-weighted image analysis for quantification of Ktrans
Image analysis to calculate Ktrans, indicative of BBB permeability, was performed using a custom MATLAB program and the hippocampal ROIs traced in the corresponding T2 images. For kinetic modeling, first-order unidirectional transport of Gd-DTPA across a semi-permeable BBB separating two compartments was assumed, described by the following differential equation
Following Vlachos et al., the time-course of the arterial tracer concentration (AIF) was modeled by a biexponential function
29
Substituting the expression in equation (2) for CA(t) in equation (1) and solving the resulting first-order linear differential equation for CT(t) yields the following analytical solution
Gd-DTPA concentration was determined from temporal changes in T1-weighted DCE-MRI signal intensities using the following expression
29
AIF parameters (A1, A2, m1, and m2) were obtained by fitting the expression in equation (2) to tracer concentration in the carotid artery for each animal. Ktrans was determined by fitting equation (3) to the tracer concentration over time in a tissue region. Analyses were conducted by investigators blinded to group and treatment.
Behavioral testing
Functional outcome was evaluated in a cohort of mice exposed to sham or CCI injury and treated with ICV injection of vehicle or hyaluronidase using the rotarod task for motor deficits and the Morris water maze task for hippocampal-dependent spatial learning and memory. Naïve mice were included as a control group to detect any changes in behavior due to ICV injection. Prior to injury, mice were acclimated to the rotarod (Rotarod/RS; Panlab/Harvard Apparatus; Barcelona, Spain), and baseline testing was performed to place mice into one of five experimental groups (naïve, sham vehicle, sham hyaluronidase, CCI vehicle, CCI hyaluronidase; n = 8–12 mice/group) so that the pre-injury baseline between groups was not significantly different. Mice that failed to grip the wheel, walked backwards on the wheel, or did not meet the minimum training criteria (an average latency to fall of at least 20 s on baseline testing) were excluded from further rotarod testing but were still used for the Morris water maze. For rotarod testing, four sequential trials were performed for each mouse, and average latency to fall was calculated from trials 2 to 4. The speed of the rotarod increased linearly from 4 to 40 r/min over the course of 60 s, the length of the trial. Animals were tested on the rotarod on days 3, 7, and 14 after injury.
Animals were evaluated in the Morris water maze task at days 16–20 after injury (n = 11–17/group). Spatial learning was evaluated on days 16–19 after injury by measuring the latency to find a 10 cm diameter platform submerged 0.5 cm below the surface in a 120 cm diameter circular pool of water using extra-maze visual cues. Learning trials consisted of four trials per day for four days. Mice were placed into the pool at an entry point to one of four quadrants. If a mouse did not find the platform in 60s, it was guided to and placed on the platform for 15s. The pool water was maintained at 22–25°C and made opaque using white paint. A video camera (Computar; Cary, NC) and ANY-maze behavioral tracking software (Stoelting Co., Wood Dale, IL) were used to detect the mouse’s body and record swim path, distance travelled, mean speed, and latency to the platform. The average latency to find the platform was calculated for each day from the four trials. A single probe trial for memory was conducted on day 5 of testing (day 20 after injury). The platform was removed, and the time spent in the target quadrant (the quadrant previously containing the platform) was recorded as well as the mean speed and distance travelled to assess swimming performance. Following the memory probe trial, a visible platform trial was conducted to rule out visual deficits. The platform was raised above the surface and flagged, and the latency to find the platform was recorded. All behavioral testing and data analyses were performed by investigators blinded to group and treatment.
1,9 Dimethylmethylene blue colorimetric assay for GAG content
Following deep anesthetization with isoflurane and cervical dislocation, the ipsilateral hippocampus was isolated from groups of sham and CCI mice, treated with hyaluronidase or vehicle (n = 4–5), and weighed (wet-weight). Additionally, cortical brain tissue was dissected from a separate group of naïve mice (n = 5), weighed (wet-weight), and then placed in 1 ml of Gey’s solution (control) or 1 ml of Gey’s solution containing 1 U/ml hyaluronidase to test its effects in explants. At 24 h, all samples were dehydrated at 100°C for 24 h to obtain their dry weight. All samples were digested in 500 µl papain digestion solution (100 mM sodium phosphate, 10 mM EDTA, 10 mM cysteine, 125 µg/ml papain, pH 6.3) overnight at 60°C. To quantify GAG content, 40 µl of samples and standards (chondroitin-6-sulfate from shark cartilage, Sigma Aldrich) were mixed with 250 µl of 1,9 dimethylmethylene blue (DMMB) dye (0.2% sodium formate, 0.2% formic acid, 0.001% DMMB, and 0.5% ethanol in distilled water, pH 3.5). Absorbance was measured at 595 nm with a reference wavelength of 540 nm and converted to µg GAG/mg dry weight. Experiments and data analysis were performed by investigators blinded to treatment.
Statistical analyses
Statistical analyses were performed using GraphPad Prism 7 (GraphPad Software, Inc., La Jolla, CA). The following statistical tests were used: one-way ANOVA with Tukey’s multiple comparisons post hoc test for in vivo water content, in vivo MRI edema, and Morris water maze percent time in target quadrant, mean speed and distance travelled; two-way repeated measures ANOVA with Tukey’s multiple comparisons post hoc test for latency to fall off the rotarod and latency to find the platform in Morris water maze learning trials; Student’s unpaired t-test for in vitro DMMB data and in vivo MRI Ktrans. Differences with p < 0.05 were considered statistically significant. Values are presented as mean ± standard deviation.
Results
Hyaluronidase reduced CCI-induced hippocampal edema
CCI increased the percent water content of the ipsilateral hippocampus in vehicle-treated mice (80.4 ± 0.46%) compared to sham mice treated with vehicle (77.37 ± 0.48%; p < 0.001) and hyaluronidase (78.66 ± 1.14%; p < 0.05). Treatment with hyaluronidase after CCI reduced edema compared to vehicle-treated CCI mice (78.15 ± 0.65% vs. 80.4 ± 0.46%; p < 0.01), so that the percent water content in hyaluronidase CCI mice was similar to sham mice (Figure 1).
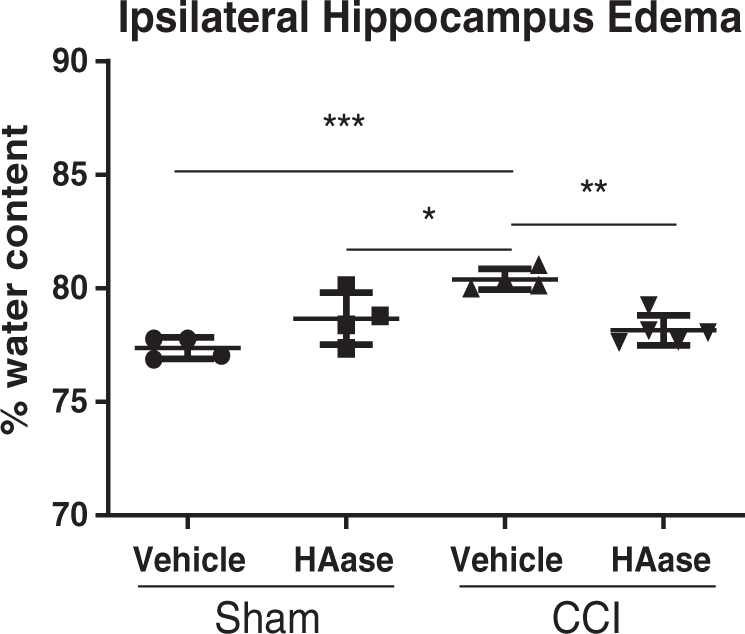
Hyaluronidase reduced hippocampal water content following CCI injury in mice. Percent water content in the ipsilateral hippocampus of vehicle-treated mice was increased at 24 h after CCI compared to sham-injured mice. Treatment with hyaluronidase (HAase) significantly reduced this increase in edema after CCI. *p < 0.05, **p < 0.01, ***p < 0.001; one-way ANOVA with Tukey’s post hoc multiple comparisons; n = 4–5 mice/group.
Hyaluronidase reduced post-traumatic brain edema in living mice without increasing BBB permeability
Hyaluronidase reduced the percentage of hyperintense, edematous voxels in the ipsilateral hippocampus 24 h after CCI so that the percent volume of edematous tissue in hyaluronidase-treated mice was less than half of that in vehicle-treated mice (13.88 ± 3.09% vs. 29.23 ± 6.14%; p < 0.01; Figure 2). Edema in the contralateral hippocampus was also reduced in hyaluronidase-treated CCI mice compared to vehicle-treated CCI mice (p < 0.01; Figure 2).
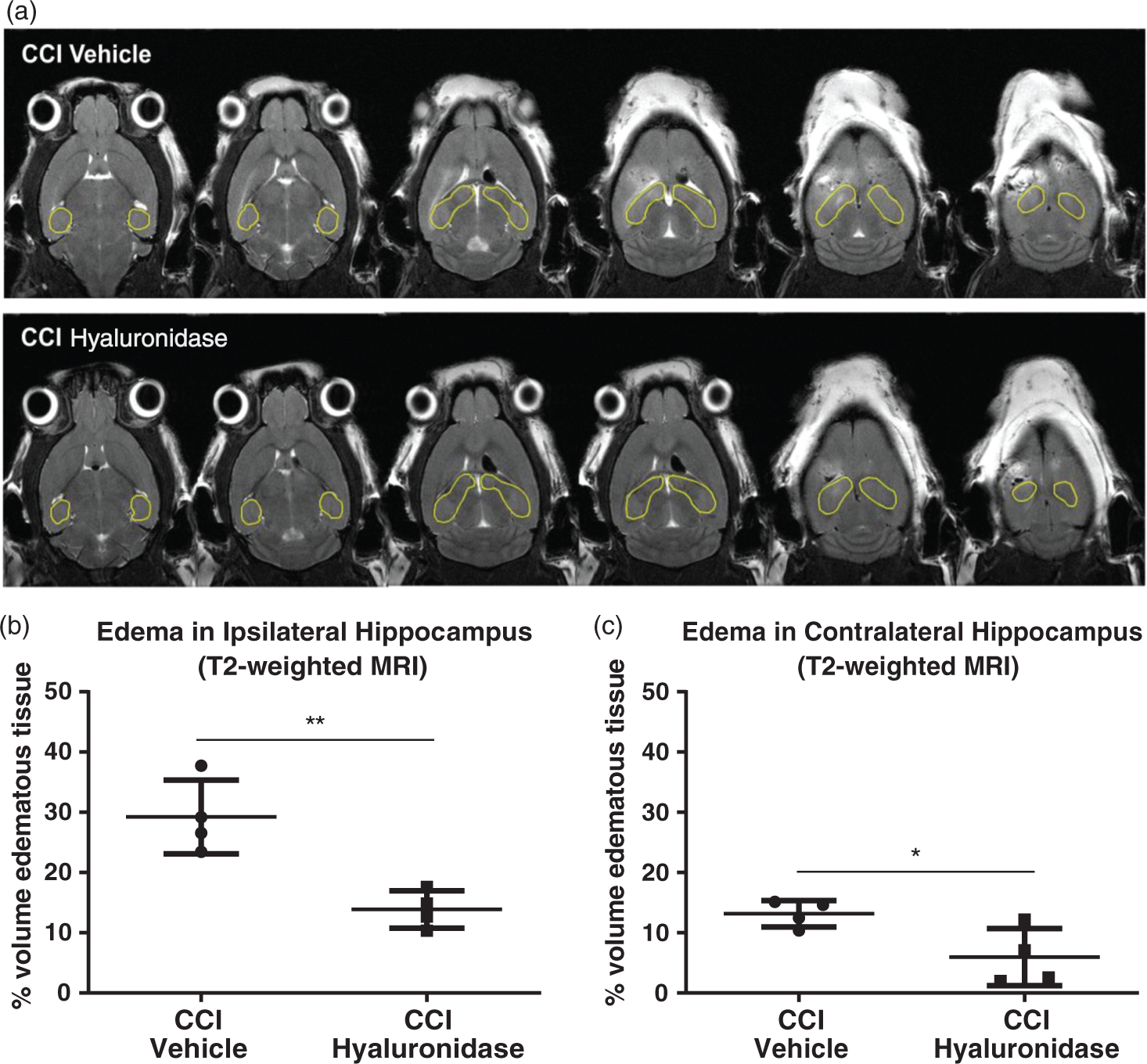
Hyaluronidase treatment following CCI injury in mice reduced edema by T2-weighted MRI. (a) Representative serial, axial T2-weighted magnetic resonance images from CCI vehicle and CCI hyaluronidase-treated mice showing hyperintense regions indicative of edema at different levels of the hippocampus in the same animal. (b) Hyperintense pixels (edema volume) in the ipsilateral hippocampus were significantly reduced in hyaluronidase-treated CCI mice compared to CCI mice treated with vehicle. (c) Hyaluronidase treatment significantly reduced edema volume after CCI in the contralateral side as well. *p < 0.05, **p < 0.01 unpaired t-test; n = 4 mice/group.
Ktrans did not differ between the ipsilateral hippocampus in vehicle- and hyaluronidase-treated CCI mice (63 ± 41 × 10−5 min−1 vs. 77 ± 78 × 10−5 min−1; n.s.; Figure 3). Ktrans values for the contralateral hippocampus were lower than that for the ipsilateral hippocampus in both groups, but were not significantly different from each other (7.5 ± 15 × 10−5 min−1 vs. 6.8 ± 12 × 10−5 min−1; n.s; Figure 3).
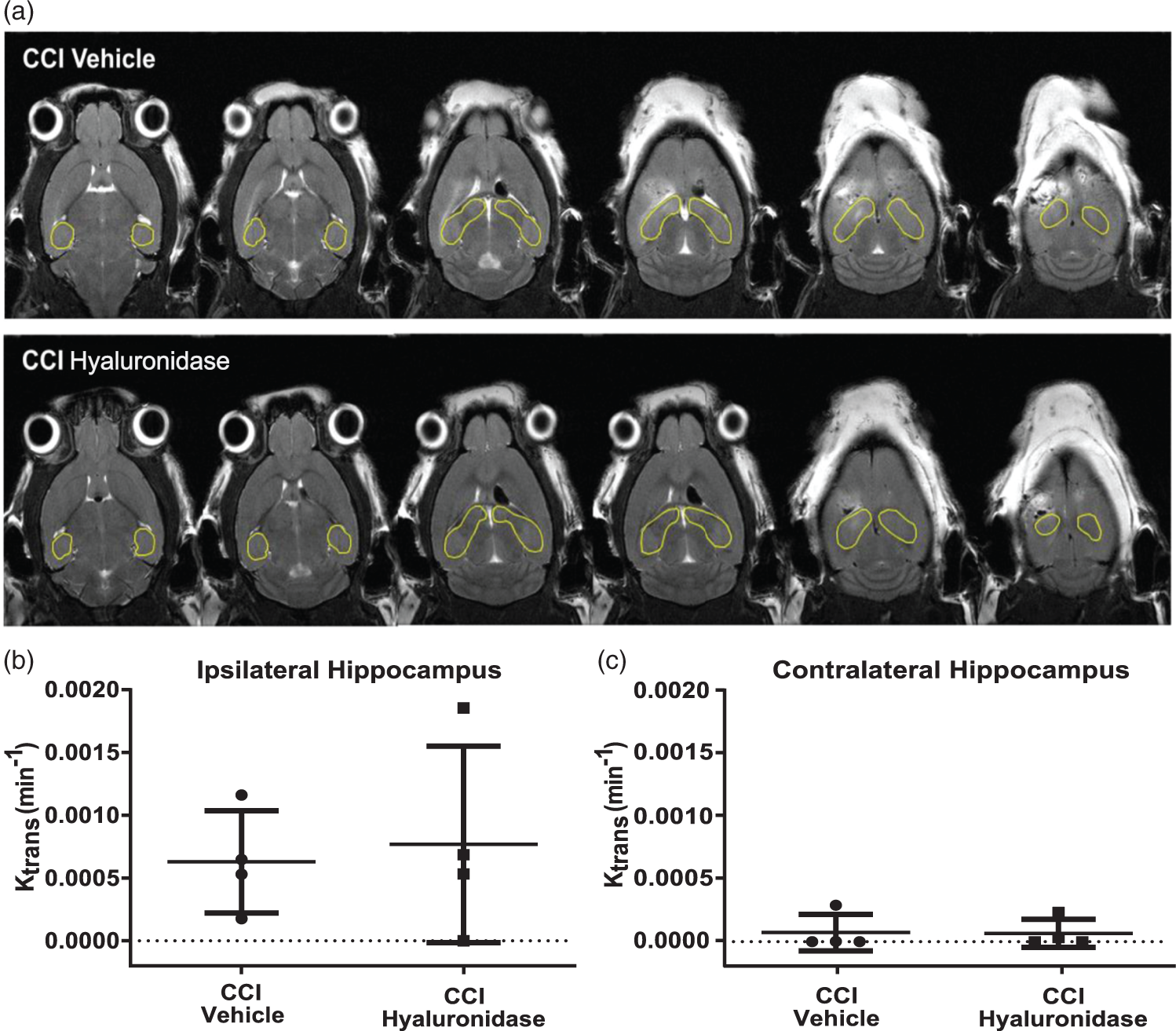
Hyaluronidase did not increase BBB permeability after CCI. (a) Representative axial T1-weighted magnetic resonance images from CCI vehicle and CCI hyaluronidase mice showing extravasation from the systemic vasculature of the contrast agent Gd-DTPA, which appears hyperintense. Ktrans, a measure of blood–brain barrier permeability, was not different between vehicle- and hyaluronidase-treated CCI mice (n.s.) in the ipsilateral hippocampus, (b) or contralateral hippocampus (c). Unpaired t-test; n = 4 mice/group.
Hyaluronidase treatment did not negatively impact recovery in the rotarod or Morris water maze tasks after CCI
Motor function was evaluated using the rotarod at 3 days, 7 days, and 14 days post-CCI. Two-way repeated-measures ANOVA with test day and group as the dependent variables found a significant main effect of test day (F3,126 = 51.44, p < 0.001) and a significant group × test day interaction (F12,126 = 2.95, p < 0.01), but the main effect of group was not significant (F4,42 = 1.4, n.s.; Figure 4(a)). Tukey’s post hoc testing for simple effects found a reduction in latency to fall at three days post-injury for sham vehicle (14.0 s ± 6.7) and CCI hyaluronidase-treated mice (12.7 s ± 8.6) compared to naïve mice (26.3 s ± 8.1) (p < 0.05). By day 14 after injury, the performance of all groups returned to or surpassed baseline with sham hyaluronidase-treated mice performing significantly better than at baseline (43.7 s ± 11.0 vs. 28.4 s ± 5.9; p < 0.001).
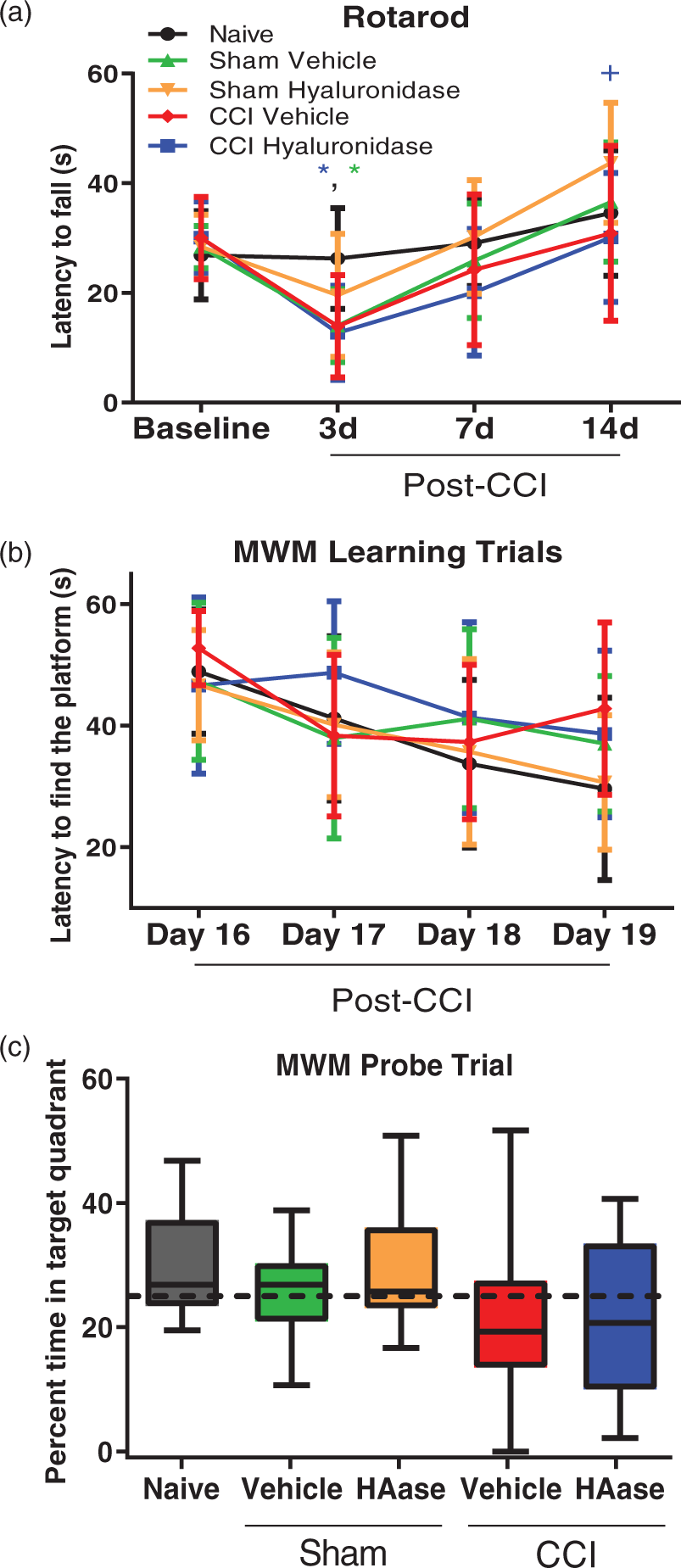
Hyaluronidase treatment did not negatively impact recovery of function after CCI injury. (a) Although performance in the rotarod task was significantly decreased at 3 days post-injury for sham vehicle and CCI hyaluronidase mice, all groups recovered to baseline performance by 14 days post-injury. *p < 0.05 for sham vehicle and CCI hyaluronidase mice vs. naïve mice; +p < 0.05 for sham hyaluronidase mice vs. CCI hyaluronidase mice; two-way repeated measures ANOVA with Tukey’s post hoc multiple comparisons; n = 8–12 mice/group. (b) The latency to find the platform in the Morris water maze decreased over training days in all groups, indicative of learning, and there was no difference between groups (two-way repeated measures ANOVA with Tukey’s post hoc multiple comparisons; n.s.; n = 11–17 mice/group). (c) The percent time spent in the target quadrant during the memory probe trial on day 20 after injury was not different between groups (one-way ANOVA with Tukey’s post hoc multiple comparisons, n.s.; n = 11–17 mice/group). Hyaluronidase is represented as HAase.
Hippocampal-dependent spatial learning was evaluated with the Morris water maze on days 16–19 after injury. Two-way repeated-measures ANOVA with test day and group as the dependent variables found a significant main effect of test day (F3,180 = 15.5, p < 0.001), while the main effect of group was not significant (F4,60 = 1.1, n.s.) nor was the group × test day interaction (F12,180 = 1.41, n.s.; Figure 4(b)). Tukey’s post hoc analysis comparing groups at different time points found no significant differences between any groups at any time point.
Memory was assessed in the Morris water maze task on day 20 after injury following conclusion of the learning trials. There was no difference in time spent in the target quadrant between groups (naïve: 30.6 ± 8.47%; sham vehicle: 25.8 ± 7.67%; sham hyaluronidase: 30.3 ± 11.38%; CCI vehicle: 21.7 ± 13.48%; CCI hyaluronidase: 22.2 ± 13.31%; n.s.; Figure 4(c)). Deficits in motor and visual function were not observed, as all groups were comparable in mean speed (naïve: 0.18 ± 0.04 m/s; sham vehicle: 0.17 ± 0.03 m/s; sham hyaluronidase: 0.17 ± 0.03 m/s; CCI vehicle: 0.15 ± 0.06 m/s; CCI hyaluronidase: 0.15 ± 0.05 m/s; n.s.) and distance travelled during the memory probe trial (naïve: 11.0 ± 2.17 m; sham vehicle: 10.3 ± 1.57 m; sham hyaluronidase: 10.2 ± 1.55 m; CCI vehicle: 9.2 ± 3.65 m; CCI hyaluronidase: 9.0 ± 2.76 m; n.s.), and all mice found the raised platform during the visible probe trial on the last day of testing.
Hyaluronidase reduced FCD in cortical explants but not in vivo
In cortical explants, hyaluronidase significantly reduced GAG content (5.46 ± 1.15 µg/mg vs. 7.76 ± 1.87 µg/mg; p < 0.05; Figure 5(a)) compared to control solution as measured with the DMMB assay. In vivo, GAG content in the hippocampus was not changed following injection of hyaluronidase into the ventricle (Figure 5(b)). These results suggest that hyaluronidase did degrade hyaluronan both in vivo and in vitro making it free to diffuse, thereby reducing its osmotic potential. However, only in the cortical explants was hyaluronan, and/or other negative molecules bound to its backbone, able to diffuse out of the tissue into the physiological salt solution thereby reducing total FCD.
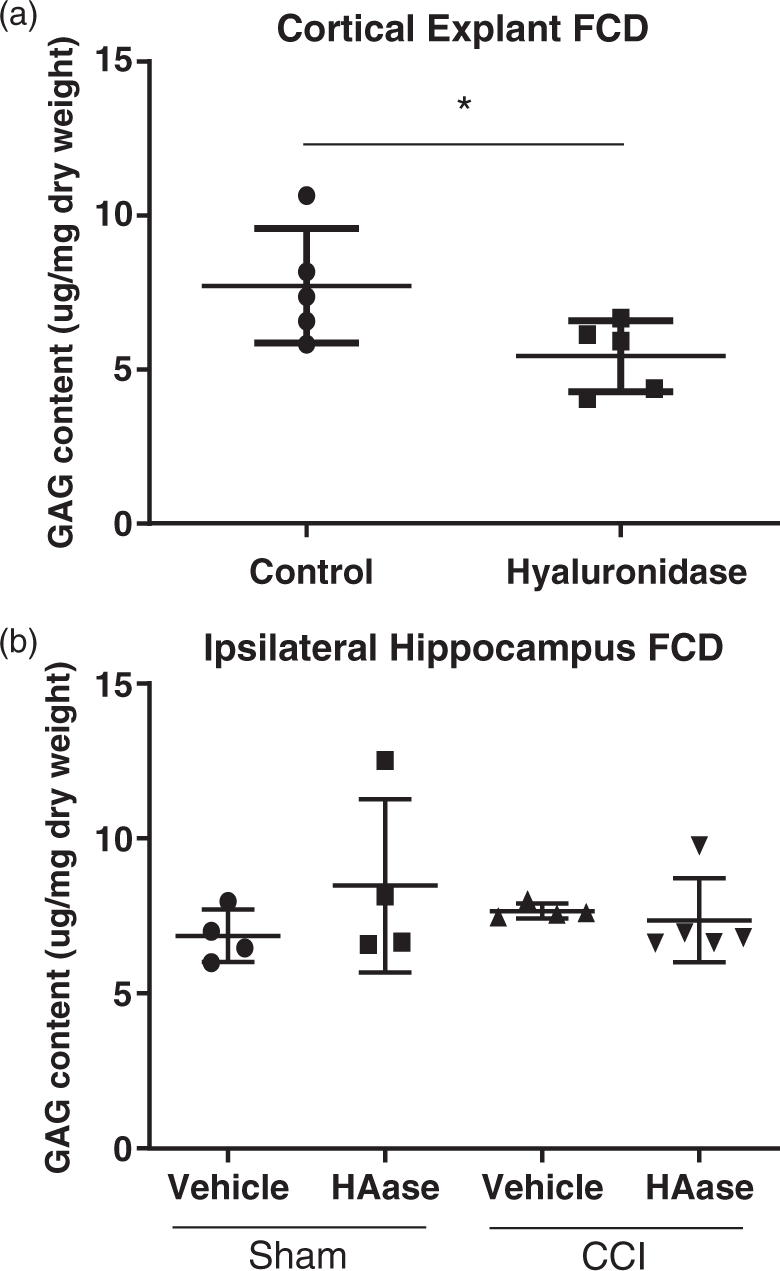
Hyaluronidase reduced FCD content in cortical explants. (a) Incubation in 1 U/ml hyaluronidase significantly reduced GAG content compared to control solution as measured by the DMMB assay. (b) Hyaluronidase treatment did not alter GAG content in the ipsilateral hippocampus of sham or CCI mice. Hyaluronidase is represented as HAase. *p < 0.05, unpaired t-test; n = 5/group.
Discussion
The FCD hypothesis of edema suggests that brain tissue swells according to the Gibbs-Donnan effect and that one of the osmotic forces driving fluid accumulation is the negative charge of the tissue.13,14,17 Therefore, we suggest that any negatively charged molecule that contributes to the FCD, such as hyaluronan, could serve as a target for reducing edema in addition to other contemporary targets. In the present study after CCI in the mouse, hyaluronidase reduced the water content of brain tissue and reduced the fraction of edematous volume in the hippocampus. These findings are consistent with the hypothesis that the FCD contributes to the development of cerebral edema after experimental TBI and that enzymatically targeting the FCD can reduce edema.
In humans, ICP increases exponentially in response to small increases in edema after TBI. A 1% increase in brain water content in TBI patients is associated with ICP levels of 20 mmHg or higher 7 compared to normal ICP values of 10–15 mmHg. 8 As increased ICP is associated with coma, brain herniation, and death,1–6 mitigating edema and reducing ICP may be advantageous for patient survival. Unfortunately, therapeutic strategies that have shown promise in preclinical studies, such as steroids, have failed to demonstrate efficacy or improve outcome in clinical trials, continuing the search for effective edema therapies.31,32
Previous studies of edema using the CCI model in rodents have shown that edema peaks 24 h after injury with an increase in water content of ∼1–3%.33–36 In contrast to edema, the time course of BBB breakdown after CCI is more variable, with studies reporting various results including BBB-opening to be biphasic with a relative dip in permeability at 24 h compared to earlier and later time points in both the ipsilateral cortex and hippocampus, 33 as well as BBB permeability increased at 4, 8, and 24 h after injury with the maximum at 24 h. 36 As edema was our primary interest, we evaluated edema and BBB at 24 h after injury, during the expected peak of edema.
Similar to previous studies, CCI increased water content in the ipsilateral hippocampus by 3%, which was reduced to a 1% increase by treatment with hyaluronidase. Sequential T2-weighted MRI to quantify edema and T1-weighted MRI with contrast agent to quantify BBB permeability in the same animal indicated both brain edema and BBB opening at 24 h after CCI. Our method did not directly measure water flux through the BBB, only the flux of the contrast agent. While it is not possible to determine whether this was the peak of BBB opening as we only assessed one time point, it is clear that there was BBB breakdown that could contribute to edema formation as accumulation of the MRI contrast agent was visible in the ipsilateral cortex around the injury site and the contralateral cortex at the site of ICV injection in both groups. However, Ktrans in the ipsilateral hippocampus was not different between vehicle- and hyaluronidase-treated CCI mice, and the low values support that hyaluronidase treatment did not further compromise BBB function nor exacerbate BBB permeability, at least for the gadolinium tracer.
In the in vitro cortical explant assay, the FCD content of hyaluronidase-treated explants was reduced compared to that of controls, indicating enzyme activity and reduced FCD. These findings are in agreement with previous in vitro studies, showing that incubation of brain explants in enzymes that reduced GAG content, including chondroitinase, heparinase and DNAse, reduced uptake of water and tissue swelling.13,14 Together, these studies suggest a relationship between the FCD of tissue and water content and that reducing the FCD of the tissue is a viable approach for reducing water content. Chondroitinase was further shown to reduce brain edema following CCI in mice. 17
Degradation of hyaluronan by hyaluronidase may reduce edema through a number of potential mechanisms including complete enzymatic degradation of hyaluronan, improving clearance of hyaluronan degradation products via the liver or the lymph nodes, 37 or mobilization and diffusion of previously fixed hyaluronan and hyaluronan-bound negative charges leading to restoration of ionic and osmotic equilibrium. The observed reduction in total tissue FCD in the in vitro assay suggests that hyaluronidase reduced tissue FCD through mobilization and diffusion of negative charges. Additionally, there was no change in the total tissue FCD in the ipsilateral hippocampus following the injection of hyaluronidase into the ventricles. Importantly, this result is consistent with our FCD hypothesis because hyaluronidase needs only to cleave hyaluronan so that its charge can diffuse freely within the tissue. It is not necessary for the hyaluronan or its charge to be cleared or eliminated from the tissue to reduce its osmotic potential and hence reduce swelling. Additionally, only the hyaluronan in damaged/dead cells (that draws in water) needs to be liberated, further reducing the requirements for limiting edema; therefore, it was not surprising that there was no discernible effect on FCD in vivo at this level of specificity.
BBB permeability was evaluated because hyaluronan is a component of brain endothelial surface glycocalyx, and a potential concern was that degrading a BBB component could increase BBB permeability. However, ICV administration of hyaluronidase did not exacerbate CCI-induced BBB permeability as shown with DCE MRI. The injection route may play a role, as intravenous injection of hyaluronidase has been shown to degrade the glycocalyx and has been used to increase capillary diameter and rescue blood flow after experimental subarachnoid hemorrhage. 38 Further, of the behavioral outcomes that we assessed, the performance of hyaluronidase- and vehicle-treated sham and CCI mice did not differ from that of each other or naïve mice, suggesting that any major off-target effects of hyaluronidase did not manifest as changes in motor function or hippocampal-dependent spatial learning and memory over the course of the testing period. Previous studies have shown that injection of hyaluronidase (different from the one used in this study) into the hippocampus 39 or infusion of hyaluronidase plus chondroitinase into the hippocampus 40 impaired fear conditioning, a hippocampal-dependent task. In the current study, ICV injection of hyaluronidase did not have deleterious effects on hippocampal function as evaluated by the Morris water maze.
Although hyaluronidase reduced edema after CCI, it was not possible to conclude from this study whether reduction of edema improved functional outcome after CCI. A mild-to-moderate severity CCI injury was used to keep the hippocampus mostly intact for edema measurements and therefore did not result in CCI-induced hippocampal deficits in the Morris water maze. This level of severity was chosen specifically to detect any potential deleterious effects of hyaluronidase treatment on hippocampal function, as opposed to detecting therapeutic efficacy in preserving hippocampal function. In general, the full benefit of reducing edema and ICP (and therefore maintaining blood flow) after injury may not be realizable in the mouse CCI model because the thin skull and compliant sutures of the mouse in conjunction with the craniectomy preclude the development of increased ICP that is typically observed in human severe TBI.
There are several limitations of the current study to be considered. First, edema and BBB permeability were evaluated at one time point after injury in response to one administration of a single dose of hyaluronidase. A more robust evaluation of potential dosing paradigms and off-target effects will be the focus of future experiments to determine the translational potential of hyaluronidase for edema. ICV injection as the administration route is relevant to the clinical severe-TBI population as external ventricular drains are often inserted to divert fluid and/or monitor ICP. Hyaluronidase is currently FDA approved and used clinically as an adjuvant to increase dispersion of injected drugs; however, preclinical and clinical research on the functions of hyaluronan in the brain and potential side-effects of hyaluronidase are limited.
Second, although the CCI model is well established and has been shown to recapitulate many features of TBI, it may not be the optimal model for evaluating edema and increased ICP because of the craniectomy in the model. Craniectomized mice have been shown to have less edema at 24 h following CCI compared to craniotomized mice (where the bone is replaced and the skull sealed). 36 In terms of clinical relevance, whether decompressive craniectomy improves outcome after TBI is still a major topic of debate with clinical trials demonstrating better outcomes 41 and similar or worse outcomes 42 compared to non-craniectomy patients. Although we did not monitor ICP in this study, measurements of ICP would also likely be influenced by the craniectomy. Although an increase in water content occurs after CCI in mice, experimental closed-head TBI in larger mammals with gyrencephalic brains, such as pigs, would allow for more representative modeling of post-traumatic edema, brain swelling, and increased ICP with its subsequent complications.43,44 Third, the level of brain edema could be affected by a change in metabolism as an unintended side-effect of hyaluronidase treatment, as no experiments were performed to evaluate brain metabolism. Last, the DMMB assay quantifies negative charges but does not specifically quantify hyaluronan directly nor does it differentiate between free (diffusible) and bound hyaluronan or other charges, of which only the bound contributes to the osmotic potential necessary for edema formation.
Increased ICP is a common and serious complication of severe TBI and is associated with poor outcome and increased mortality. There is an urgent need for therapeutic strategies that reduce ICP by directly targeting post-traumatic cerebral edema. Here, we demonstrate that ICV injection of hyaluronidase reduced edema in a mouse model of TBI and provide additional evidence to support the hypothesis that the FCD contributes to edema formation in the brain after injury.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Columbia University’s Research Initiatives for Science and Engineering (RISE) program sponsored by Columbia University’s Office of the Executive Vice President for Research (S.G. Kernie and B. Morrison III), the Taub Institute MRI Platform Pilot Scan/Analysis Award (B. Morrison III), and NIH 1R56NS089523 (S.G. Kernie).
Acknowledgements
The authors would like to thank Yangping Sun, Ph.D., MRI Physicist, and Christopher Damoci, Senior Staff Officer/Imaging Scientist, for their technical assistance in planning and executing the MRI studies, and Marilena Karakatsani, M.S. for her technical assistance with magnetic resonance image analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
PMW performed the experiments, analyzed data, and helped write the article. CL analyzed the imaging data and executed the compartmental modeling. MKRD analyzed data and helped write the article. EEK helped design the imaging experiments and assisted with data analysis. SGK helped design experiments and reviewed the article. BM designed experiments, analyzed data, and helped write the article.