Abstract
To expand previous understanding of age-related vascular changes, we examined the association between aging and characteristics of cerebral arteries among 1133 participants aged 35 to 75 years recruited from Shanghai, China. Characteristics of the cerebral vessels including arterial branch density, mean radius, and mean tortuosity were quantified using MR angiography. The radius, tortuosity, and length of the basilar artery (BA) and the M1 segment of middle cerebral artery (MCA) were also accessed. Linear regression model was used to examine the association between age and vasculature features. The sample was divided into four subgroups by age and the association was analyzed in each subgroup. Age was found to be a significant predictor for cerebrovascular modifications after adjusting for vascular risk factors. Further analysis in subgroup revealed that the associations were due to the predominate effect of the vascular modifications happened during the younger years (35–54 years). The radius of either BA or MCA was associated with aging only in subjects aged 45–54 years. In conclusion, rapid alterations in all three morphological features assessed have been noticed to be associated with aging in the 45–54 subgroup, suggesting the potential importance of the 5th decade for early preservation method for vascular aging.
Introduction
The power-hungry nature of the human brain requires a consistent supply of oxygen and nutrients by the cerebral vascular system. 1 Indeed, a synergistic relationship between cerebrovascular dysfunctions and certain neurological symptoms has been suggested.2,3 For instance, reduced cerebral blood flow (CBF) has been implicated in dementing diseases such as vascular dementia and Alzheimer’s disease.4,5
During one’s lifetime, it is known the maturation of the brain protracts into the third decades with continuous structural and functional modifications.6,7 In such processes, cerebral vessels undergo dynamic modifications, normative changes and exposure to conventional cerebrovascular risks (CVRs), such as aging and hypertension, drastically reshaping the vessels, which lead to significant impairments associated with various diseases.8,9 Among them, it is widely agreed that aging, dramatically increasing the incidence of cardiovascular or cerebrovascular diseases, has the strongest impact on the vascular system, leading to deteriorations of the vascular services, including generalized endothelial dysfunction and increase in central arterial stiffness.10–12 To further elucidate the influence of aging on the cerebral vascular system, a previous study assessed several vessel features including tortuosity, radius, and vessel number with time-of-flight magnetic resonance angiography (TOF MRA). 13 Their results suggested that age was associated with decreased vessel number and a general trend of increasing vessel radius in a group of healthy subjects. Similarly, Chen, et al., reported age related decline in the number of branches and increase in vessel tortuosity. 14
Nevertheless, previous studies investigating the effects of aging or general CVRs on the morphometric features of cerebral vessels usually suffered from a relatively smaller sample size with a restricted age range, minimizing their power to detect such changes.13,14 To date, only one study has reported, with a large dataset, in healthy subjects the associations of CVRs and cerebrovascular morphometric features, but careful investigations of such associations with different age specifications is lacking. 15 Such investigations are necessary for a better and more comprehensive understanding of the structural modifications of the cerebral vasculature during one’s lifespan and could provide preliminary data for age-specific management strategies for the preservation of cerebrovascular health.
In this study, we set out to test whether 1) as reported previously, 13 similar patterns of age-related modifications of cerebrovascular structures, including increased tortuosity and mean radius exist in our population, and 2) whether such changes differ for different age specifications. A large natural population in China aged from 35 to 75 was recruited, and participants were further divided in different age subgroups to recapitulate different stages of life. Morphometric features of the cerebral vessels, including the basilar and middle cerebral arteries (BA and MCA respectively) were quantified from 3D-TOF scans. The impact of aging as well as the conventional CVRs on vascular morphologies in different age groups was also assessed to explore their relationships.
Material and methods
Participants
This cross-sectional study initially enrolled 4813 participants (aged 35 to 75 years) who completed the survey of unruptured cerebral aneurysms in Chinese adults between June 2007 and June 2011 from two different Shanghai communities in China. 16 Ten years later (2017 to 2021), participants received call-backs, checking on their health status. As shown in Figure 1, 3198 participants at the first survey who were free of cardiac disease, stroke, arteriostenosis or a history of cancer were opted for the call back. 1751 participants lost at the callback, and 207 participants who reported severe cardiovascular disease (CVD) events, including myocardial infarction, unstable angina, heart failure, coronary heart disease or stroke during the callback were excluded. 1240 participants were included in final the analysis. Chronic conditions, including hypertension and diabetes, were well managed for the participants. The study was approved by the institutional review board of the Sixth People's Hospital, Shanghai, China. All participants provided written informed consent before data collection. All procedures were conducted according to the Declaration of Helsinki.
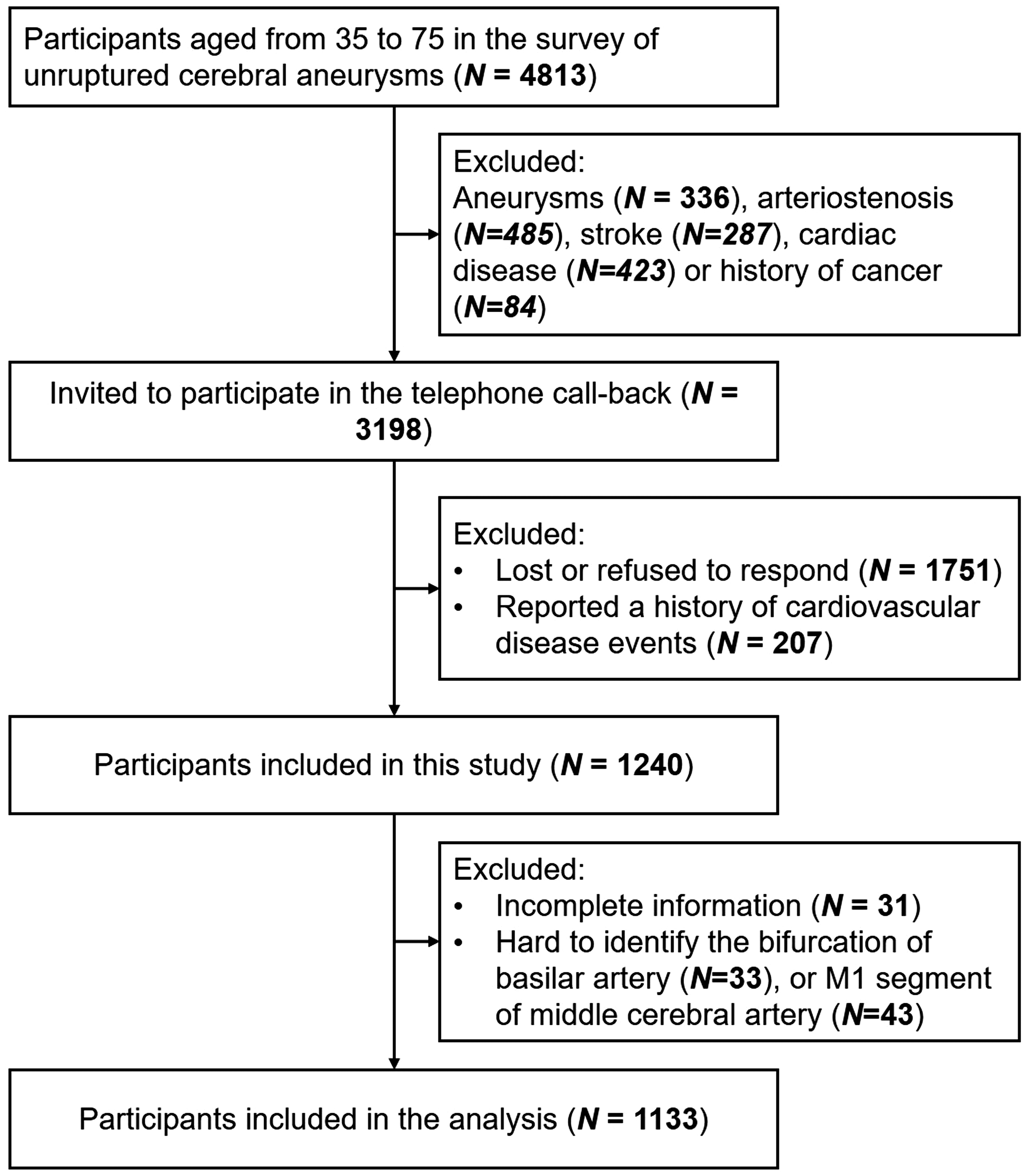
Flow chart of recruitment in the current study.
Assessment of risk factors
Initially, demographic characteristics, personal and family medical history, and lifestyle risk factors were collected in the clinic. Information about cigarette use, alcohol consumption, hypertension, diabetes, hyperlipidemia, stroke, coronary heart disease, myocardial infarction, and arrhythmia was obtained. The height, weight, and blood pressure were also recorded. Venous blood samples were taken by certified nurses between 7 AM and 8 AM after overnight fasting. The presence of hypertension was defined as systolic blood pressure ≥140 mm Hg or diastolic blood pressure ≥90 mm Hg, a previous diagnosis of hypertension, or use of anti-hypertensive medications. Smokers were defined as smoked >100 cigarettes in the past, and drinkers were defined as consuming >30 g of alcohol per week for 1 or more years. Diabetes was defined by a fasting level of plasma glucose ≥7.0 mmol/L, a previous diagnosis of diabetes, or treatment with antidiabetic drugs. Hyperlipidemia was defined by total plasma cholesterol (TC) ≥5.2 mmol/L, triglycerides (TG) ≥1.7 mmol/L, a previous diagnosis of hyperlipidemia, or use of lipid-lowering drugs. During the call-back, the medical history within the past 10 years was also obtained to further confirm the baseline characteristics.
MRI scanning and processing
MRI scanning was performed on one 3.0 T MRI system (Philips Healthcare, Amsterdam, The Netherlands). 3D-TOF MRA was completed using 3-dimensional T1-weighted fast-field sequences with the following settings: repetition time/echo time, 35/7; flip angle, 20 degrees; field of view, 250 × 190 ×108 mm3; 4 slabs (180 slices); slice thickness, 0.8 mm; matrix, 732 × 1024; acquisition time, 8 minutes and 56 seconds. Any adverse events during the MRI examination, including panic attack, claustrophobia, dizziness, or falling, were monitored and evaluated by the investigators.
Cerebral vessel segmentation and feature extraction were completed on TOF MRA scans using the existing method with following steps: 17 1) vascular structures were enhanced using the Jerman Enhancement Filter; 18 2) the level-set based method was used to obtain vessel segmentation; 3) segmentation results were firstly checked by B.Y.Z and B.W. Segmentations with too few vascular branches, nonvascular tissue remnants, or incorrect vascular branch adhesions were re-segmented by adjusting the segmentation parameters. Then all segmentation results were visually checked twice by two radiologist J.L and Y.H.L with over 3 years’ experience in neuroimaging interpretation. The re-segmented vessels that were still showing too few branches, remnants of nonvascular tissue, or severe vascular branch adhesions were defined as poor vascular segmentation and excluded from the analysis; 4) vessel centerlines were extracted using the Skeleton 3D toolbox; 19 5) cerebrovascular morphologic measurements, including vessel density, radius and tortuosity were calculated. The vessel density was defined as the ratio of intracranial artery branch numbers normalized to the brain volume. The vessel tortuosity represents the averaged tortuosity for each segment and was calculated as the ratio of Euclidean distance and the actual length of a given vessel segment. The vessel radius was determined at each centerline points and then averaged over each individual participant. The smaller vessels were defined as vessels with a mean radius less than a certain threshold (global thresholding based on the radius distribution of the whole intracranial arteries). 17 All data processing processes were accomplished on the Matlab2019a (MathWorks, Natick, Massachusetts, USA).
Local metrics of vascular modifications were also accessed, including morphological features of BA and M1 segment of MCA. The internal carotid artery was not included in the analysis as its origin cannot be clearly defined in the reconstructed images. The segmented vessels were reconstructed into 3D space to allow for interactive manual selection of start and end points of the BA and the first segment (M1) of the MCA. 20 B.Y.Z and J.L performed the processes independently to ensure the accuracy of selections. For the inconsistent results, re-selection was performed by Y.H.L and consensus of the three was made. The mean radius, length and tortuosity of the BA and MCA were then calculated based on the segmentation and the centerline information between each pair of start and end points. (Detailed workflow can be seen in Supplemental Method and Supplementary Figure 1) The mean morphological features of the left and right M1 segment were used (Supplementary Table 1). Supplementary Figure 2 shows the representative manually drawn points.
Statistical analysis
Bivariable and multivariable analyses were completed to investigate association between cerebrovascular morphologies (3 whole-brain measurements: cerebral vessel density, radius and tortuosity; 3 MCA measurements: radius, length and tortuosity; 3 BA measurements: radius, length and tortuosity) and CVRs (age, body mass index, smoking, alcohol consumption, diabetes, hypertension and hyperlipidemia). Each bivariable model consisted of one of the three whole-brain cerebrovascular morphologies as dependent variable and one of the CVRs as independent variable. Each multivariable model consisted of one of the nine cerebrovascular morphologies as dependent variable and all of the CVRs as independent variable. Bonferroni correction was used in the bivariable model for multiple comparison correction.
In addition, linear analysis was completed to investigate the correlations between aging and cerebrovascular morphologies in different age subgroups. In each subgroup, age was used as the independent variable and nine morphologies were separately used as the dependent variable. Participants were divided into different age subgroups according to their age: 35 to 44 years, 45 to 54 years, 55 to 64 years and 65 to 75 years. Since the number of individuals in the four groups was not balanced, with the 45–54 years group having the largest size, 50 simple random samples of 200 individuals were drawn from subgroup and regression analysis was performed to test whether the age effects in the 45-54 years subgroup were due to its larger sample size. Age distribution of all individuals are presented in the Supplementary Figure 3.
The individuals' combined CVR score representing the exposure to 6 risk factors was also analyzed, as existing evidence indicated combined risk factor score was correlated with the brain health in young adults. 21 The score was cumulative based on each of the following factors: body mass index ≤24; no smoking; not a drinker; no hyperlipidemia; no hypertension; no diabetes, with a score of six indicating no cerebrovascular risks. The relationships between the CVR score and MRA findings were studied using the linear regression model. In addition, linear analysis was completed to investigate the correlations between aging and cerebrovascular morphologies in different age subgroups.
All statistical analysis was completed using Matlab2019a. Normality of continuous variables was confirmed by Shapiro-Wilk test. Results of normally distributed continuous variables are presented as mean and standard deviation (SD) or as median and interquartile range (IQR). For categorical variables, frequencies, and proportions are presented. One-way analysis of variance and chi-square test were used to evaluate group differences according to requirement. All tests were 2-sided and a probability value <0.05 indicates significance.
Results
The participant characteristics are presented in Table 1. Figure 1 summarizes the recruitment process of the current study. A total of 1240 participants were included; of these, 76 participants failing to identify the bifurcation of BA or MCA and 31 participants with incomplete CVRs information were excluded. Therefore, 1133 participants were included in the subsequent analysis.
Characteristics of the study participants.
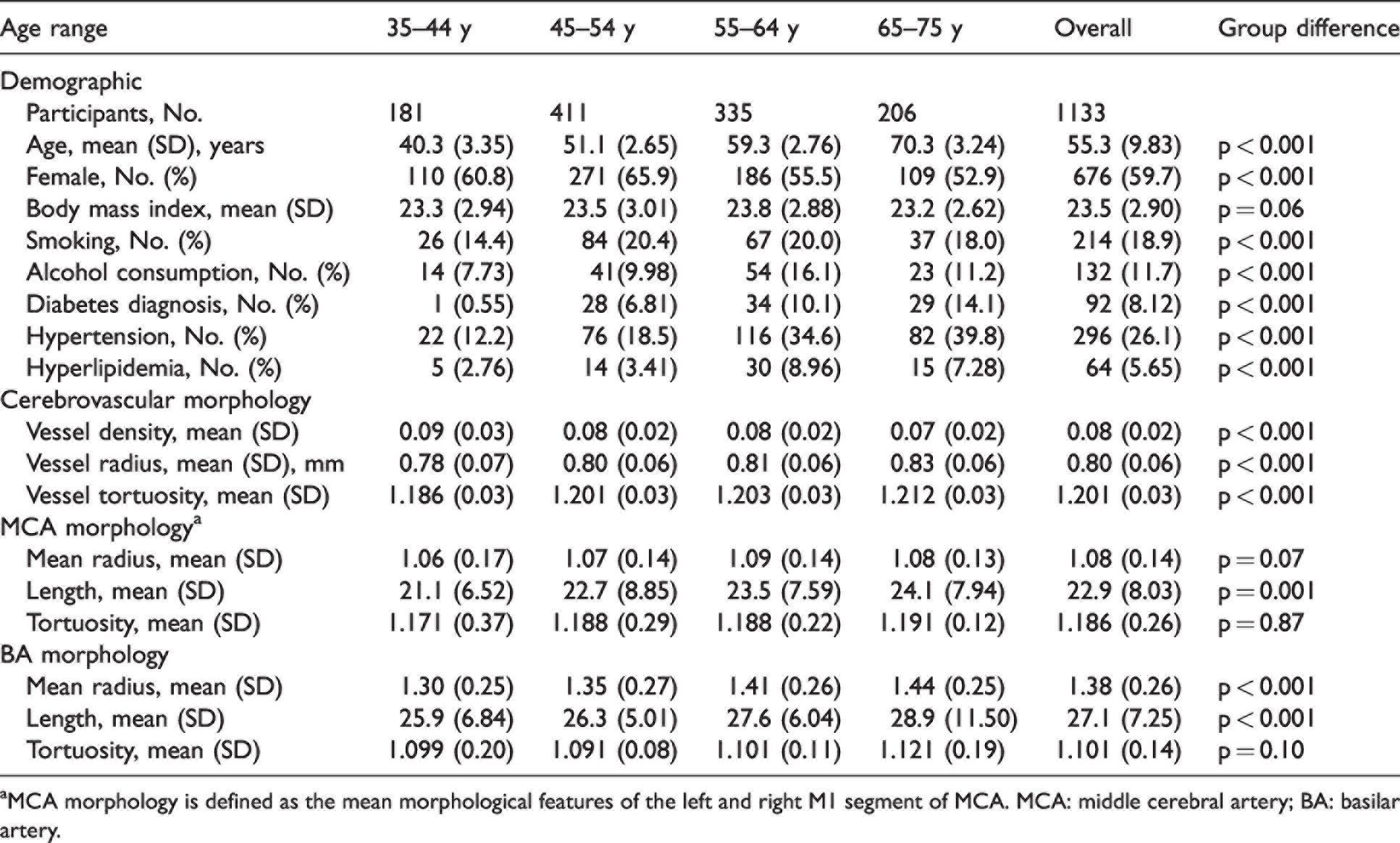
aMCA morphology is defined as the mean morphological features of the left and right M1 segment of MCA. MCA: middle cerebral artery; BA: basilar artery.
Association between cerebrovascular morphology and aging
Age was found to be significantly associated with decreased vessel density (β = −5.6 × 10−4, 95% CI −0.001 to −4.2 × 10−4, p < 0.001), increased vessel radius, (β = 0.002, 95% CI 0.002 to 0.002, p < 0.001), and increased vessel tortuosity, (β = 8.1 × 10−4, 95% CI 0.001 to 0.001, p < 0.001) after adjusting for CVRs, as shown in Figure 2. Vessel density negatively associated with aging in participants aged 45–54 after adjusting for CVRs (β = −0.001, 95% CI −0.002 to −0.001, p < 0.001). Similarly, increased vessel radius was only found to be associated with aging in participants aged 45–54 after adjusting for CVRs, (β = 0.005, 95% CI 0.003 to 0.007, p < 0.001). Increased vessel tortuosity was found to be significantly associated with age in subjects aged 35–44 (β = 0.002, 95% CI 3.6 × 10−4 to 0.003, p = 0.01) and 45–54 (β = 0.001, 95% CI 3.3 × 10−4 to 0.002, p = 0.01). adjusted for CVRs. No significant correlations of aging with vascular morphologies were observed before and after adjusting for CVRs in the 55–64 years and 65–75 yeares subgroups (Table 2 and Figure 2). Supplementary Figure 4 shows the scatter plot of association between age and cerebrovascular morphologies. Significant correlations remained in most of the 50 simple random samples, as presented in the Supplementary Table 2. Particularly, the correlation between aging and mean radius remained significant in more than 90% of the 50 samples.
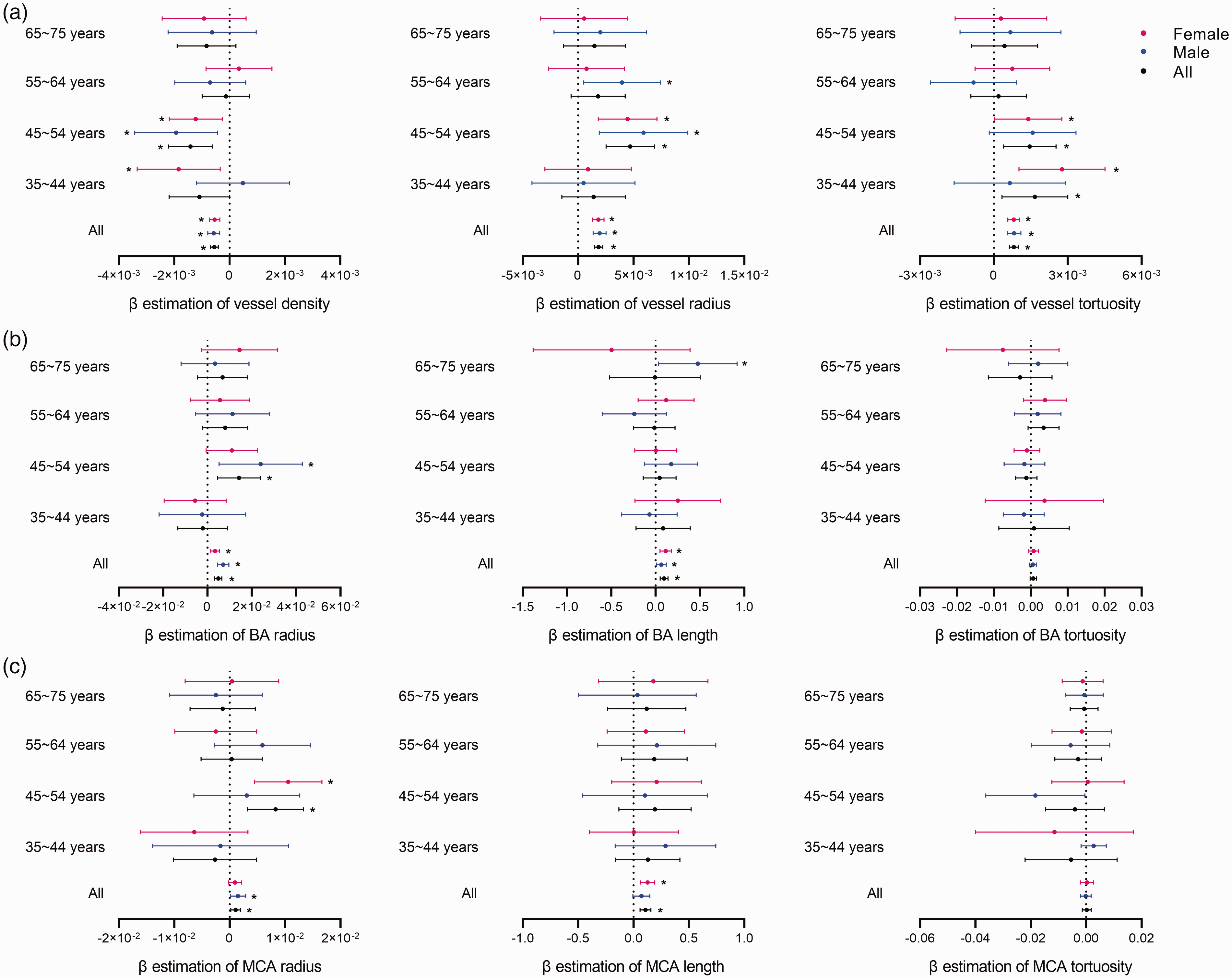
Association of cerebrovascular morphology with age in subgroups. (a) β estimation of association between age and vessel density (left), vessel radius (middle), and vessel tortuosity (right); (b) β estimation of association between age and BA radius (left), BA length (middle), and BA tortuosity (right) and (c) β estimation of association between age and MCA radius (left), MCA length (middle), and MCA tortuosity (right). BA: basilar artery; MCA: middle cerebral artery. β values of age in multivariable adjusted model were used which adjusted for cerebrovascular risk factors including body mass index, smoking status, alcohol consumption, diabetes status, hypertension status and hyperlipidemia status.
Association of cerebrovascular morphologies with sex and cerebrovascular risk factors.
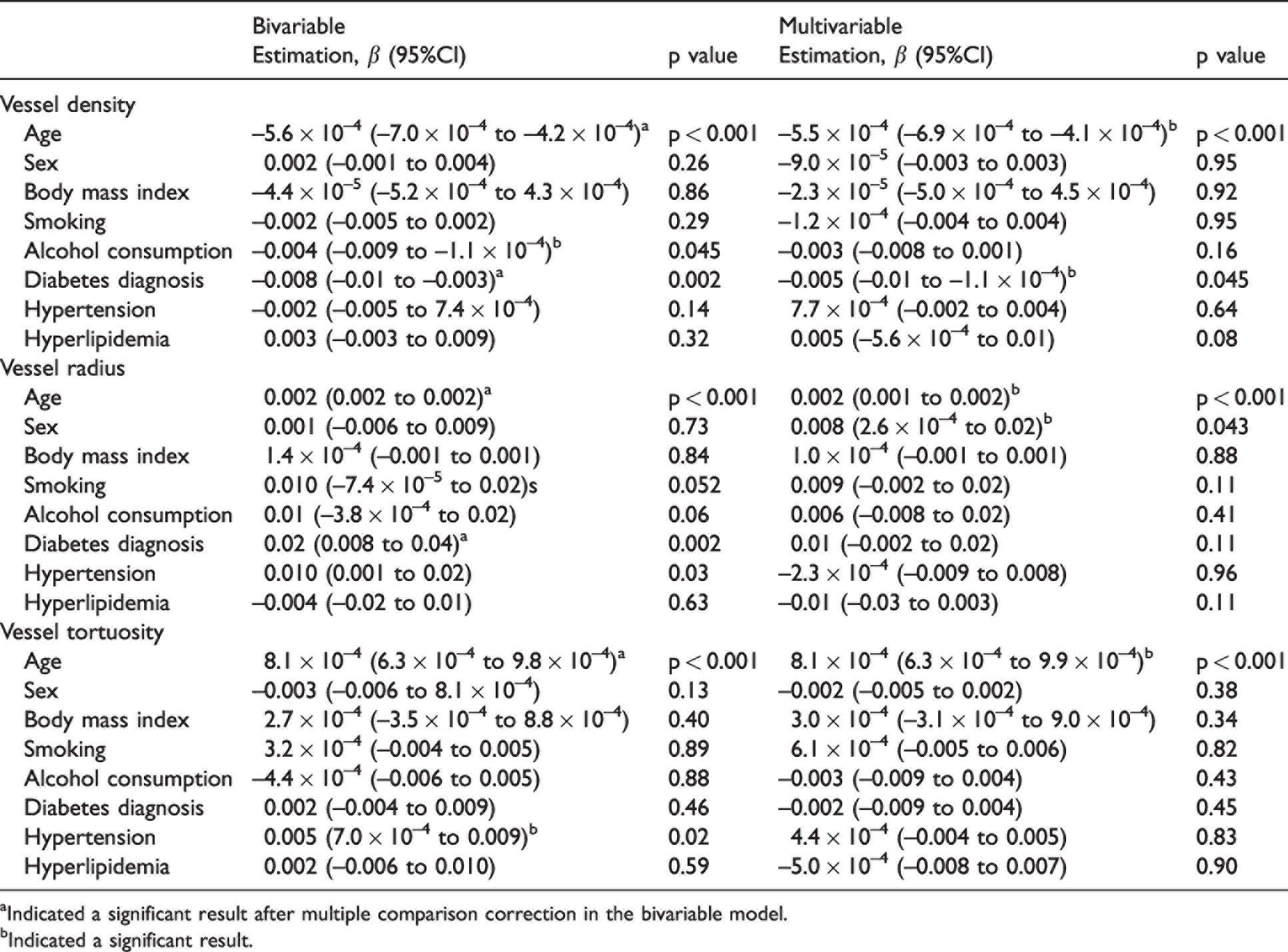
aIndicated a significant result after multiple comparison correction in the bivariable model.
bIndicated a significant result.
To better delineate the underlying mechanisms for dilated arteries, we further assessed the proportion of the smaller vessels visualized by MRA. As shown in Supplementary Table 3, a general decrease in the proportion of the smaller vessels with advancing age was identified, (β = −3.7 × 10−4, 95% CI: −6.1 × 10−4 to −1.3 ×10−4, p = 0.03). The decrease in mean vessel radius reported before, however, remained significant after adjusting for the proportion of the smaller vessels, (β = 2.7 × 10−4, 95% CI: 6.6 × 10−5, 4.8 × 10−4, p = 0.01).
Association of the characteristics of BA and MCA with aging
As shown in the Figure 2, aging was found significantly associated with increased mean radius (BA: β = 0.005, 95% CI: 0.004 to 0.007, p < 0.001; MCA: β = 0.001, 95% CI: 1.5 × 10−4 to 0.002, p = 0.02) and length (BA: β = 0.10, 95% CI: 0.06 to 0.14, p < 0.001; MCA: β = 0.10, 95% CI: 0.06 to 0.15, p < 0.001) of both BA and MCA among all individuals. However, in subgroups, aging only correlates with increased BA and MCA radius in participants aged 45–54 after adjusting for CVRs, (BA: β = 0.01, 95% CI: 0.005 to 0.02, p = 0.004; MCA: β = 0.008, 95% CI: 0.003 to 0.01, p = 0.001). Detailed data are shown in Supplementary Table 4 and 5.
Similar associations of aging with features of the left or right MCA were observed (Supplementary Table 6 and Table 7). Results from regression analysis for 50 simple random samples drawn from the 45–54 years group were presented in the Supplementary Table 8 and Table 9 and confirmed that the identified correlations were not due to the larger sample size.
Association between cerebrovascular morphologies and CVRs
The relationships between cerebrovascular morphologies and CVRs were further examined. Mean vessel tortuosity were found not to be associated with any of the individual CVRs listed in Table 2. Lower vessel density was found in participants with diabetes after adjusting for age and other CVRs (β = −0.005, 95% CI −0.01 to −0.001, p = 0.045). The interaction between age and smoking was found to significantly impact vessel density (β = −6.8 × 10−4, 95% CI: −0.001 to −2.6 × 10−4, p = 0.002) and vessel radius (β = 0.001, 95% CI: 2.8 × 10−4 to 0.003, p = 0.01). The interaction of age and alcohol consumption seems to result in a higher vascular density (β = 6.2 × 10−4, 95% CI: 8.7 × 10−4 to 0.001, p = 0.02) and decreased vessel radius (β = −0.002, 95% CI: −0.003, −4.5 × 10−4, p = 0.01) as shown in Figure 3. No significant interaction between age and other CVR factors was found (Table 3).
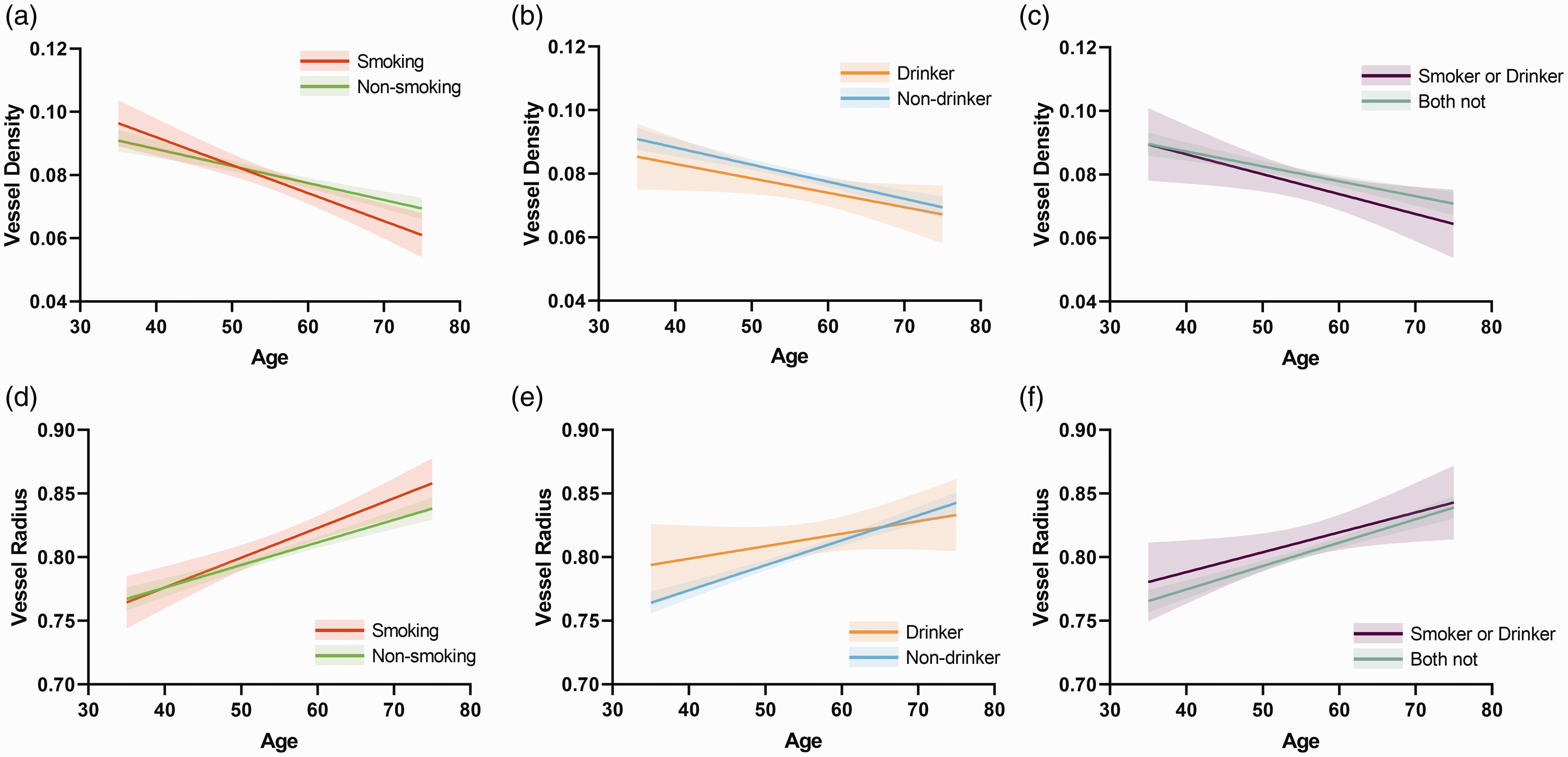
Interactions between age and vascular risk factors. (a)–(c) Association between vessel density and age in different groups (smoking and non-smoking, drinker and non-drinker, smoker or drinker and both not). Shaded area represents the 95% confidence interval and (d)–(f) Association between vessel radius and age in different groups.
Interaction between age and cerebrovascular risk factors.
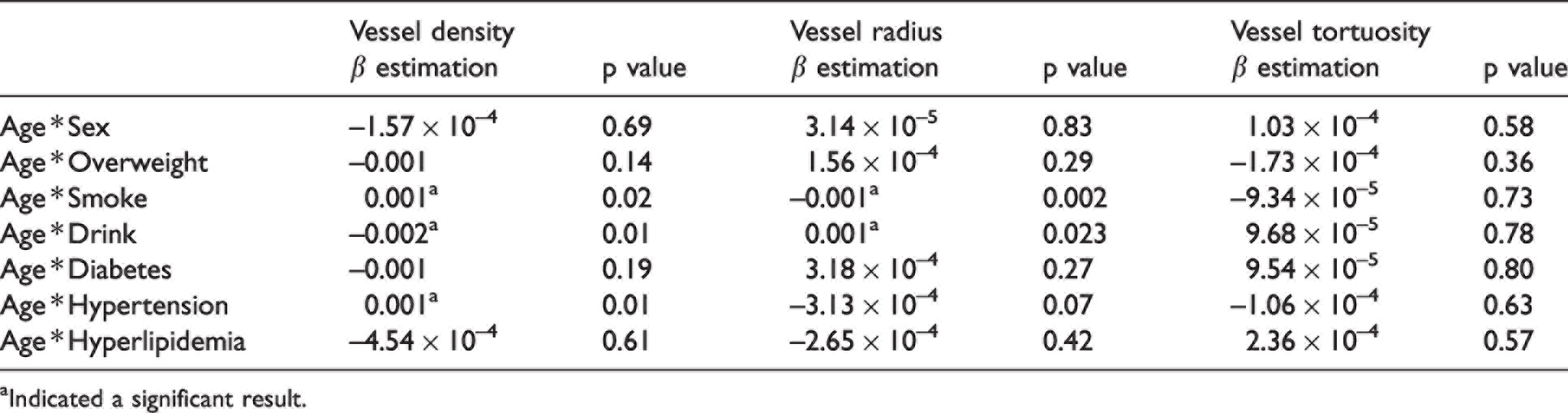
aIndicated a significant result.
A cumulative CVR score of 0 to 6 was assigned accordingly, with a higher score indicating less CVRs. The associations between CVR score and vascular morphologies have been reported in the Supplementary Table 10. Overall, a significant association was identified between CVR score and vessel density (β = 0.002 95% CI: 3.8 × 10−4 to 0.003, p = 0.01), and vessel radius (β = −0.006 95% CI: −0.009 to −0.002, p = 0.001). However, when participants were divided into subgroups, CVR score was only found to be correlated with an increase in vascular density adjusted for age and sex (β = 0.002, 95% CI: 2.4 × 10−4 to 0.004, p = 0.03) in the 55–64 group.
Discussion
In this cross-sectional study, the effects of aging and other conventionally defined CVRs on the cerebral arterial morphologies quantified from 3D TOF MRA scans were investigated. Per previous studies, advanced age was found in our research to be associated with increased vessel radius, increased vessel tortuosity and decreased branch density in general.13,14 Given the features are expressed as mean values which reflect more global modifications, local changes were also assessed. The characteristics of BA and MCAs were therefore analyzed, since that BA and MCAs could account for about 60% of the total blood supply to the brain. 22 Aging was again found associated with structural alterations including increased radius and length, except for tortuosity, of BA and MCAs, supporting that our results could reflect a general trend of age-related vascular modifications.
Upon testing, one very interesting group, consisting of subjects aged 45–54 years, only in which age remained significantly associated with all three vascular characteristics after adjusting for other CVRs was identified. More interestingly, although we failed to detect significant associations of aging with modifications of BA or MCA in most of the subgroups, possibly due to the relatively smaller sample sizes, age was significantly correlated with mean radius of both MCA and BA, again, only in the middle-aged subjects (45–54 years). It is worth noting that the large proportion of female subjects in this particular age subgroup could have potentially affected our results since a previous study suggested that females show significantly higher artery density and lower mean arterial radius, 15 and females in this age specification could be experiencing major changes in hormones due to menopause. Nonetheless, since the association between age and cerebrovascular morphological features remained statistically significant after adjustment for biological sex, it is unlikely our observations arose due to the uneven sex ratio in this particular group. However, such question remains to be an interesting research idea, and interactions between major hormonal changes and vascular features during menopause are worth further investigation, although a larger sample of ethnically diverse female participants who are experiencing menopause is required.
Unlike previously reported, 14 age ceased to be associated with previously investigated features in the older subgroups (55–64, and 65–75 years respectively) after adjusting for CVRs. The results suggested that the observed age-related changes in the morphometric features of the vessels were mainly driven by the substantial modifications happened during the 5th decade of life, and the effect of aging possibly has reached a plateau later on with CVRs having a larger influence. Nevertheless, since the size of our 45–54 subgroups is the largest (411 individuals), it is possible that our results simply were due to the larger sample size of this subgroup. 50 simple random samples of 200 people were therefore drawn from the 45–54 subgroup to test whether the significant associations remained. In half of 50 random samples, similar results were obtained, suggesting that the observed changes not likely to be solely due to the larger size of this particular subgroup.
Previous studies have reported age related reduction in cerebral perfusion may have already happened in people aged 31–50.21,23 Our study also identified several age-related vascular modifications happened rapidly at a very young age, with tortuosity markedly increased with aging in subjects aged 35–44. Increased arterial tortuosity was reported to have a presumed origin in connective tissue degradation or/and elongation of arteries,24,25 and more tortuous vessels are inevitably faced with higher flow resistance, contributing to decreased perfusion. Nonetheless, a causal relationship between age related reduction in cerebral perfusion and vascular modifications cannot be teased out simply based on current results, and future studies investigating the causal relationships between these would provide beneficial information for treatment or prevention methods for the pathological aspects of aging.
In this study, decreased vessel density was also evident. Nonetheless, due to the inability of the current MRA to detect under-perfused vessels, the age-related reduction in the branch density may also be the result of impaired cerebral perfusion. Nevertheless, as suggested previously, 13 such reduction in branch density with age may also result from a physical loss of smaller vessels. In view of the restrictions posed by the current imaging method, it is not plausible to attribute the observed reduction in branch density to either one of the causes. Nonetheless, the fact that lower branch density was identified in the older population (45–75) suggests a gradual decrement in vascular services in the aging population.
Our results also showed that advanced age was significantly associated with an increase in the mean vessel radius, in agreement with the results from previous MRA studies.13,14 Such results may arise from the rarefaction of smaller branches, resulting in a relative increase in the proportion of larger branches, or/and compensatory vasodilation, presumably with an ischemic origin. Since adjusting for the proportion of smaller vessels did not affect the significance of the results but only gave rise to a smaller beta value, it becomes more likely that although both may contribute to the age-related increase in mean vessel radius, the results may be dominated by the huge effect of compensatory vasodilation. This view was further supported by our results that BA and MCA radius increased with aging. Coincidentally, dolichoectasia, a dilative arteriopathy, has also found to be only correlated with advanced age. 26
Our results might, at the first glance, suggest that vascular risk was less related to cerebrovascular modifications after adjusting for age. However, consistent with previous reports that reducing vascular risk factors and sustained lifestyle intervention can reduce cardiovascular burden,27,28 and improve cerebral blood flow, 29 maintaining a healthy lifestyle is definitely helpful in preserving the soundness of the vascular system since our results showed a higher vascular score is tied to higher vessel density.
Taken together, our results implied age-related disturbances of the cerebral hemostasis, and such disturbances might arise as early as midlife (around the 4th decade). Further investigation of the relationships between vascular features and neurological diseases would provide helpful information as to whether prevention methods are necessary during midlife to combat the pathological aspect of aging.
This study suffered from several limitations: 1) only one MRI modality, TOF MRA, was available in the study, which makes further investigation of the potential causes of the discussed vascular alterations implausible. For example, to extract the general trend of the age-related cerebral perfusion changes in different age subgroups, arterial spin-labeling would be necessary. 2) Risk factors were coded dichotomously indicating whether the subject had the chronic condition at the time when their data was collected, and their medical history was not available. Caution is warranted, since the time of exposure to CVRs might just be longer in the older subjects, which means that CVRs inherently have bigger impact on vascular features. With current dataset, the effect of the duration of the chronic conditions cannot be examined. Future studies would benefit from having access to the medical history and more detailed information of the subjects’ chronic conditions such as the duration. 3) The configuration of the circle of Willis was not included in the analysis. The effect of the circle of Willis on the cerebrovascular morphology still needs further studies.
In this study, differences in the cerebrovascular morphologies were identified between older subjects and younger subjects after adjusting for vascular risk factors. In age subgroups, age was associated with the alterations of all three vessel morphologies extracted by MRA only in the 45-54 subgroup. Aging was again found associated with structural alterations including increased radius and length, except for tortuosity, of BA and MCAs. These results suggested the potential importance of the 5th decade for early preservation method of vascular aging.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221111609 - Supplemental material for Modification of cerebrovascular morphologies during different stages of life
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221111609 for Modification of cerebrovascular morphologies during different stages of life by Boyu Zhang, Zidong Yang, Jing Li, Bei Wang, Huazheng Shi, He Wang and Yuehua Li in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 81971583, No. 81871329), National Key R&D Program of China (No. 2018YFC1312900), New developing and Frontier Technologies of Shanghai Shen Kang Hospital Development Center (No. SHDC12018117), Shanghai Municipal Education Commission-Gaofeng Clinical Medicine Grant (No. 2016427), Shanghai Municipal Science and Technology Major Project (No. 2018SHZDZX01, No. 2017SHZDZX01) and ZJLab.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
H.W and Y.H.L conceived the idea and designed the study; J.L and Y.H.L contributed to the data collection; B.Y.Z and B.W performed the data analysis; H.W, Y.H.L, B.Y.Z, Z.D.Y and H.Z.S contributed to the data interpretation; B.Y.Z and Z.D.Y prepared the figures; H.W and Y.H.L contributed to the data verification and supervised the study. All the authors participated in the preparation of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
