Abstract
Intracranial stenosis is a common vascular lesion observed in Asian and other non-Caucasian stroke populations. However, its role in cognitive impairment and dementia has been under-studied. We, therefore, examined the association of intracranial stenosis with cognitive impairment, dementia and their subtypes in a memory clinic case-control study, where all subjects underwent detailed neuropsychological assessment and 3 T neuroimaging including three-dimensional time-of-flight magnetic resonance angiography. Intracranial stenosis was defined as ≥50% narrowing in any of the intracranial arteries. A total of 424 subjects were recruited of whom 97 were classified as no cognitive impairment, 107 as cognitive impairment no dementia, 70 vascular cognitive impairment no dementia, 121 Alzheimer’s Disease, and 30 vascular dementia. Intracranial stenosis was associated with dementia (age/gender/education – adjusted odds ratios (OR): 4.73, 95% confidence interval (CI): 1.93–11.60) and vascular cognitive impairment no dementia (OR: 3.98, 95% CI: 1.59–9.93). These associations were independent of cardiovascular risk factors and MRI markers. However, the association with Alzheimer’s Disease and vascular dementia became attenuated in the presence of white matter hyperintensities. Intracranial stenosis is associated with vascular cognitive impairment no dementia independent of MRI markers. In Alzheimer’s Disease and vascular dementia, this association is mediated by cerebrovascular disease. Future studies focusing on perfusion and functional markers are needed to determine the pathophysiological mechanism(s) linking intracranial stenosis and cognition so as to identify treatment strategies.
Keywords
Introduction
Cerebrovascular diseases (CeVD) have been increasingly implicated as a cause and contributor to cognitive impairment and dementia with a strong association reported between infarcts and white matter hyperintensities (WMH) with cognitive decline. 1 In this context, intracranial stenosis (ICS) has gained increasing attention due to its role in causing ischemic damage and hence cognitive dysfunction. 2 The occurrence of ICS has been attributed to vascular risk factors such as hypertension and diabetes3,4 – the same risk factors which are also linked with Alzheimer’s disease (AD) and vascular dementia (VaD). Previous post-mortem studies have shown that severe arterial atherosclerosis of the Circle of Willis is common in dementia, being found in 53% of VaD and 34% of AD cases. 5 Furthermore, a high prevalence of ICS (>50% stenosis) on magnetic resonance angiography (MRA) is also observed in-vivo in VaD compared to AD patients (53% vs. 18%). 6 A recent study has reported an ICS prevalence of 70% (when defined as >20% stenosis on computed tomography angiography) in mild cognitive impairment which increased the risk of conversion into AD dementia. 7 However, these studies lacked a disease-free comparison group and have not taken into account other markers of CeVD such as infarcts, WMH, and cerebral microbleeds.
Thus, the mechanism(s) of how ICS leads to cognitive impairment and dementia remains under-explored. Specifically, the association of ICS with cognitive impairment and dementia in the presence of CeVD markers has not been determined previously. Hence, we examined the association of ICS in relation to cognitive impairment and dementia and their subtypes in patients recruited from a memory clinic in Singapore.
Materials and methods
For the present study, a case-control design was employed. Cases (cognitive impairment no dementia (CIND) and dementia) were recruited from two study sites in Singapore (memory clinics in the National University Hospital and Saint Luke’s Hospital). Controls, defined as those who were cognitively normal on objective neuropsychological assessment, were recruited from both memory clinics and the community (with a similar catchment area as cases). Details of this study have been described previously. 8 All subjects underwent physical, clinical, and neuropsychological assessments and neuroimaging at the National University of Singapore.
Ethics approval was obtained from the Singapore Eye Research Institute, and National-Healthcare Group Domain-Specific Review Board. The study was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained in the preferred language of the participants by bilingual study coordinators prior to their recruitment into the study.
Demographic and cardiovascular risk factor assessment
A detailed questionnaire was administered to all participants for information on age, gender, race, education, and smoking history. Previous medical history of hypertension, hyperlipidemia, and diabetes mellitus was also noted and subsequently verified by medical records. Physical examination included height, weight, and blood pressure. Systolic and diastolic blood pressures were measured using a digital automatic blood pressure monitor after the subject rested for five minutes. Blood pressure was measured twice, five minutes apart. The mean of two readings was considered as the relevant blood pressure. Hypertension was defined as systolic blood pressure ≥140 mmHg and/or diastolic blood pressure ≥90 mmHg, or use of anti-hypertensive medication. Mean arterial blood pressure was calculated as two-thirds of the diastolic blood pressure plus one-third of the systolic blood pressure. Diabetes mellitus was defined as glycated hemoglobin ≥6.5%, or use of anti-diabetic medication. Hyperlipidemia was defined as total cholesterol levels ≥4.14 mmol/l, or use of lipid lowering medication. Cardiovascular diseases included ischemic heart disease, atrial fibrillation, and congestive heart failure. Smoking was categorized into ever (past and current smokers) and never smokers.
Neuroimaging
MRI was performed on a 3 Tesla Siemens Magnetom Trio Tim scanner, using a 32-channel head coil, at the Clinical Imaging Research Centre of the National University of Singapore. The three-dimensional time-of-flight MRA images (3D TOF MRA) were acquired with a repetition time of 22 ms, time to echo of 3.40 ms, flip angle of 20°, 192 mm field of view, 218 × 256 acquisition matrix, slice thickness of 0.80 mm, distance factor −22.73%, and an acquisition time of 6 min and 28 s. Subjects with claustrophobia, contraindications for MRI, or those who were unable to tolerate the procedure were excluded.
Intracranial stenosis on MRA
ICS was defined based on the criteria published previously.
9
Briefly, arterial narrowing exceeding 50% of the luminal diameter in any of the intracranial vessels were assessed on 3D TOF MRA images. These images were first visually assessed on the reconstructed coronal sequences. The final decision on stenosis (>50%) was based on the source images (Figure 1). The arteries that were assessed were the vertebral, basilar, internal carotids, posterior cerebral, posterior communicating, middle cerebral, and anterior cerebral arteries. One grader (SH), who was blinded to clinical data, graded each participant’s MRA. The previously established intra-rater reliability was excellent (intraclass correlation coefficient, ICC = 0.88). As MRA was only able to assess intracranial vessels, the extracranial arteries were not graded for stenosis.
Intracranial stenosis (ICS) on 3D time-of-flight magnetic resonance angiography. (a) Shows occlusion of left-internal carotid (white-arrow) and middle cerebral arteries (red-arrow) in coronal view which is further confirmed on source images (b and c).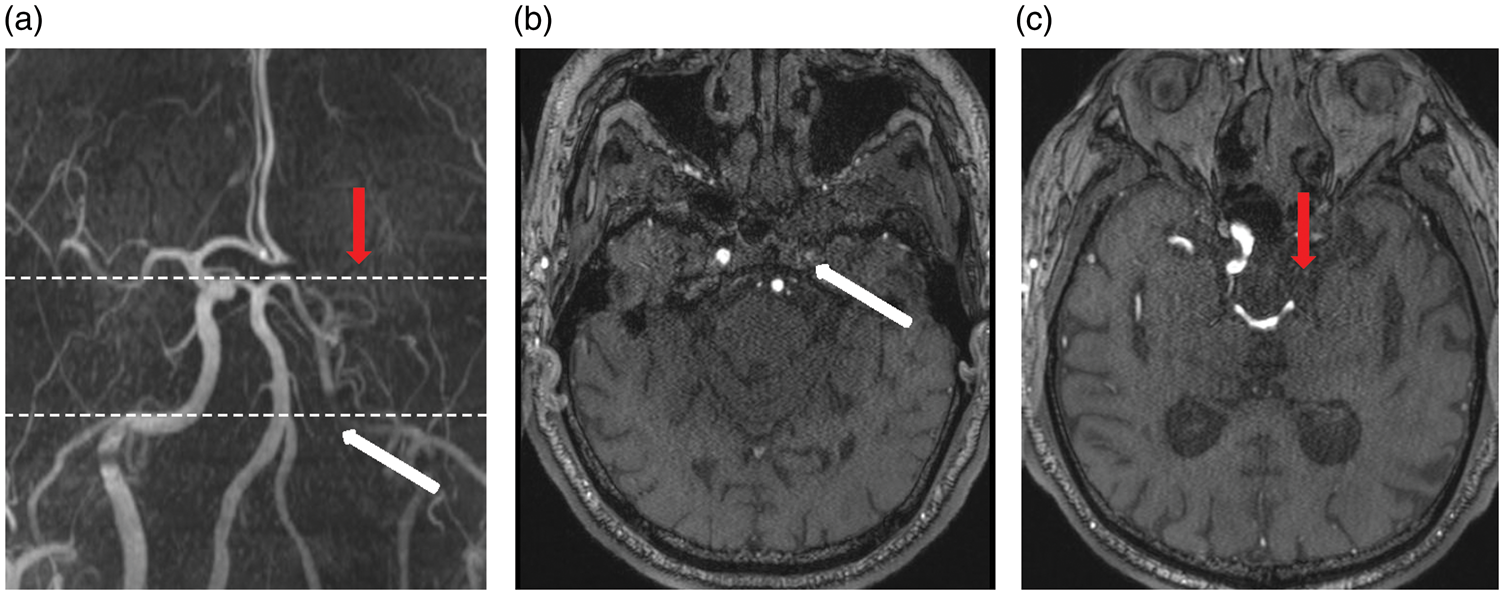
Other MRI markers
Other markers of CeVD and neurodegeneration were also graded on MRI using the following sequences and scales:
Infarcts (cortical and lacunar) were defined on fluid attenuated inversion recovery (FLAIR) and T2 sequences using STRIVE criteria.
10
Cerebral microbleeds were defined on susceptibility-weighted imaging (SWI) sequences using brain observer micro bleed scale (BOMBS).
11
Total intracranial volume and WMH volume were quantified by automatic segmentation at the Erasmus MC, University Medical Center Rotterdam, The Netherlands. Brain tissue segmentation was quantified using the Proton density-weighted T1 sequence and T2-weighted images, whereas the WMH volume was segmented using FLAIR, as described previously.
9
Briefly, as a marker of global atrophy, total brain volume was calculated as the sum of gray matter and white matter volumes of the five regions; frontal, parietal, occipital, temporal, and central regions. WMH volume was the summation of the WMH in the above-mentioned five regions.
Cognitive impairment and dementia assessment
Neuropsychological test battery
Trained research psychologists administered brief cognitive tests; the mini-mental state examination (MMSE) and the Montreal cognitive assessment (MoCA) and a formal detailed neuropsychological test battery that has been locally validated in Singapore. This battery assesses seven domains, five of which are non-memory domains.
The non-memory domains tested were:
Executive function: Frontal assessment battery and maze task, Attention: Digit span, visual memory span and auditory detection, Language: Boston naming test and verbal fluency, Visuomotor speed: Symbol digit modality test, digit cancellation, Visuoconstruction: Weschler Memory Scale – Revised (WMS-R) visual reproduction copy task, clock drawing and Weschler Adult Intelligence Scale – Revised (WAIS-R) subtest of block design.
The memory domains tested were:
Verbal memory: Word list recall and story recall, Visual memory: Picture recall and WMS-R visual reproduction.
The assessment was administered in the subject’s habitual language and was completed in approximately an hour.
Diagnosis of cognitive impairment and dementia
Research diagnoses of the participants were made at a weekly consensus meeting into the following groups:
Subjects with no objective evidence of neuropsychological deficits were classified as having no cognitive impairment (NCI). CIND was determined by clinical judgment and was defined as no significant loss of independence in daily activities and impairment in at least one domain of the neuropsychological test battery. Participants were considered to have failed a test if they scored 1.5 SD below education-adjusted cut-off values on each individual test. Failure in at least half of the tests in each domain was considered as impairment in that domain. Vascular cognitive impairment no dementia (VCIND) was defined as a history of ischemic stroke within the past 6–24 months and neuroimaging evidence of cerebral infarction with objective evidence of neuropsychological deficits. The diagnosis of dementia was made according to DSM-IV criteria. The etiological diagnoses of dementia were based on internationally accepted criteria; AD was diagnosed using the National Institute of Neurological and Communicative Disorders and Stroke and the AD and Related Disorders Association (NINCDS-ADRDA) criteria. According to this criteria, patients with slow and gradual onset of memory problems, impairment in objective neuropsychological assessment, and loss in activities of daily living were classified as AD.
12
VaD was defined using the National Institute of Neurological Disorders and Stroke and Association Internationale pour la Recherché et l' Enseignement en Neurosciences (NINDS-AIREN) criteria. Briefly, patients with history of stroke temporally related to onset or worsening of cognitive impairment, neuroimaging evidence of extensive or multiple infarcts, and with loss in activities of daily living were classified as VaD.
13
Statistical analysis
In order to compare the baseline characteristics between cases (CIND and dementia) and controls (NCI), analysis of covariance or chi square tests were used. In case of non-uniform data (WMH), differences between the groups were determined using the Kruskal–Wallis test. WMH volume was logarithmically transformed due to the skewed distribution for further analysis. The association between ICS and CIND/dementia was determined initially using logistic regression models with odds ratios (OR) and 95% confidence interval (CI). Further regression analyses were then constructed separately for subtypes of CIND (vascular vs. non-vascular) and dementia (AD and VaD). All models were initially adjusted for age, gender, education and additionally for hypertension, hyperlipidemia, diabetes, cardiovascular diseases, and smoking. Finally in order to examine whether the association between ICS and CIND/dementia remained independent of other CeVD, we adjusted logistic models additionally for each MRI marker. P-value <0.05 was considered statistically significant. Statistical analysis was performed using standard statistical software (Statistical Package for Social Science, SPSS V23, SPSS Inc., USA).
Results
Baseline demographic and clinical characteristics of the subjects (n = 424).
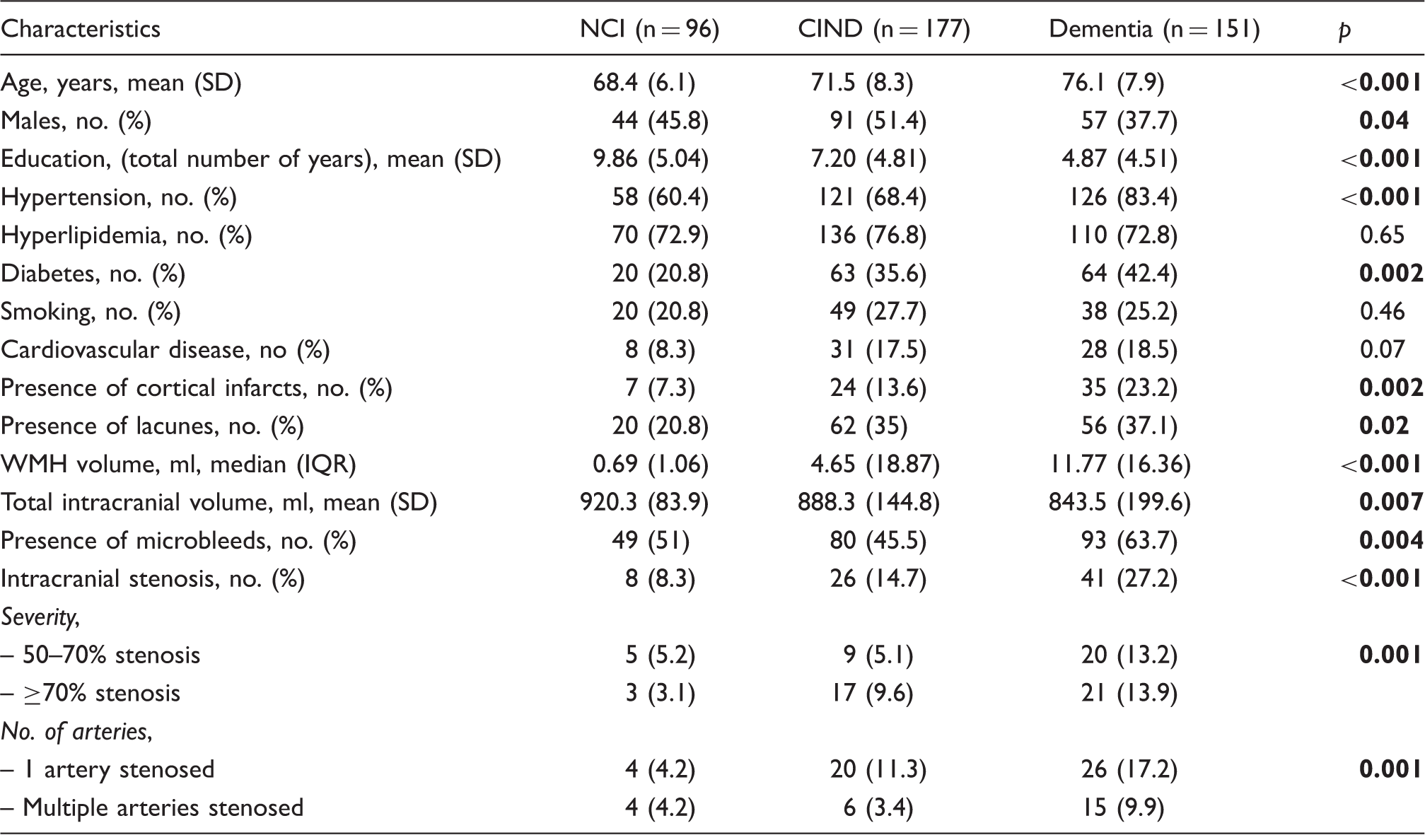
NCI: no cognitive impairment; CIND: cognitive impairment no dementia; SD: standard deviation; no: number; ml: milliliters; WMH: white matter hyperintensities; IQR: interquartile range.
Distribution of intracranial stenosis in different arteries based on diagnostic groups.
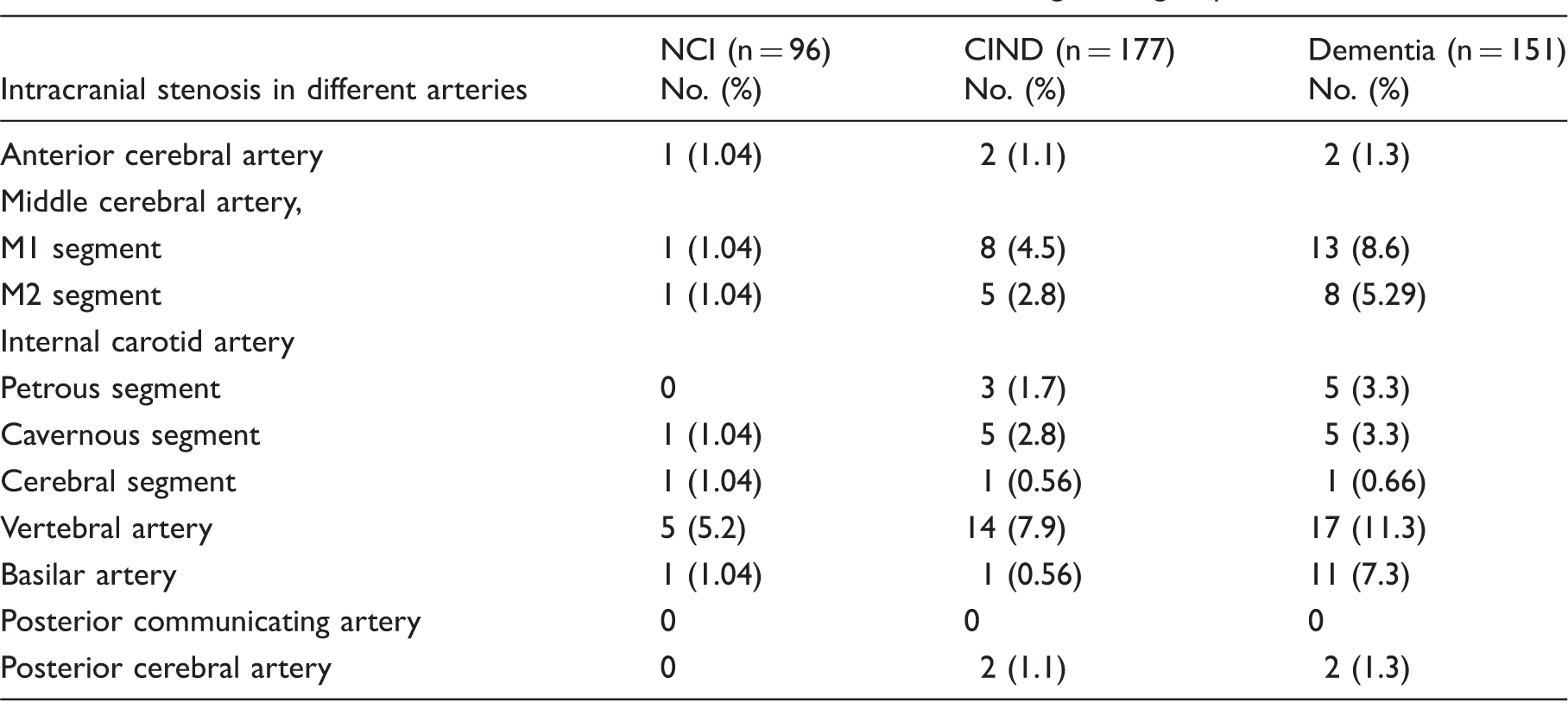
NCI: no cognitive impairment; CIND: cognitive impairment no dementia; no: number.
Association of intracranial stenosis with subtypes of cognitive impairment and dementia.
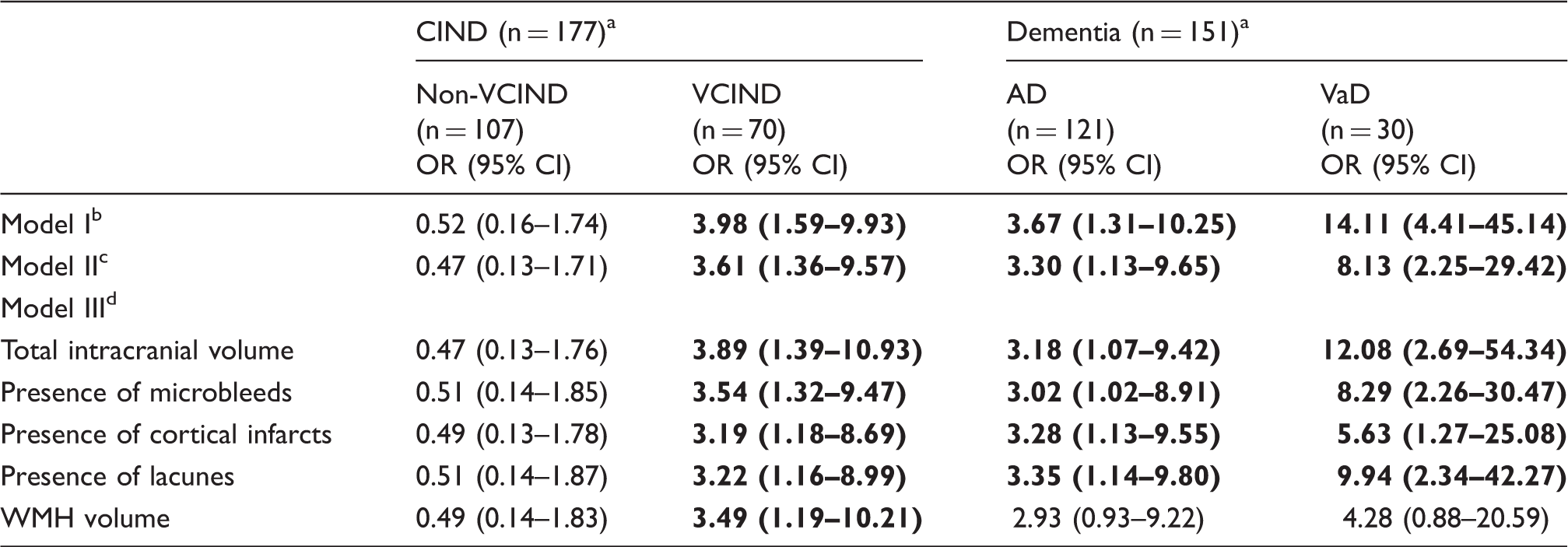
CIND: cognitive impairment no dementia; non-VCIND: non-vascular cognitive impairment no dementia; VCIND: vascular cognitive impairment no dementia; AD: Alzheimer’s disease; VaD: vascular dementia; OR: odds ratios; CI: confidence interval; WMH: white matter hyperintensities.
Compared against NCI.
Model I: Adjusted for age, sex and education.
Model II: Model I + hypertension, hyperlipidemia, diabetes, cardiovascular diseases, smoking.
Model III: Model II + individual MRI markers.
Discussion
This study showed that the persons with ICS are more likely to have VCIND and dementia independent of cardiovascular risk factors. Additional adjustments with MRI markers revealed that ICS possibly induces more severe cognitive dysfunction through CeVD.
Several studies have previously reported a higher prevalence of ICS in stroke subjects among Asians (11–85%)14–16 compared to Caucasians (8–10%).17,18 A higher prevalence of ICS was also observed among Asian dementia patients (18–53%). 6 This wide difference in prevalence may be due to diversities in, (1) imaging modalities, (2) measures of neuropsychological function, (3) limited information on degree of stenosis, and (4) different criteria to define ICS. The ICS prevalence of 19.8% in AD, 28.6% in VCIND, and 56.7% in VaD in the present study is in concordance with the previous findings using MRA.6,19
With respect to cognition, it has been reported that cognitive deficits are commonly present in symptomatic (history of stroke) subjects with extracranial and intracranial arterial stenosis possibly through cerebral hypoxia.20–23 Moreover, it has been shown that subjects with carotid stenosis improved significantly on neuropsychological performance after carotid surgery. However, the majority of these studies did not control for other relevant neurovascular lesions such as cerebral infarction and WMH. Furthermore, limited data exist on the association of ICS with cognitive impairment/dementia and its subtypes. The present study extends these previous findings by showing that persons with ICS are more likely to have a diagnosis of VCIND independent of other structural MRI markers. This could be linked to subtle cortical and subcortical ischemic damage (increased resistance and reduced vascular reactivity of the small vessels) or reduction in anatomic connectivity and perfusion deficits secondary to ICS. Moreover in VCIND, due to probable lack of underlying AD pathology, cognitive reserve is preserved and hence may ameliorate effects on cognitive function. By contrast, in dementia, vascular ischemic damage alone or in addition to neurodegeneration leads to decline in cognitive reserve and significant impairment of cognitive function. The similar mediating factors for both AD and VaD in this study may be due to the several overlapping neuropathological features between the two diseases. 24 Moreover, the pathogenesis of AD and VaD may also involve separate but additive processes. In the case of AD, hypoxic injury may not only accelerate amyloid beta deposition (especially in the hemisphere with the ICS) but also could trigger secondary degeneration induced by inflammatory processes, whereas in VaD, hypoperfusion may interact with existing CeVD and initiate secondary neurodegeneration. 25 Thus, the multifactorial mechanisms underlying cognitive impairment in patients with large-artery atheroma may explain the link between ICS and dementia (AD and VaD) observed in this study.
Our study has some limitations. First, as this case-control study was examined cross-sectionally, it was not possible to establish the temporal association between ICS and the development of cognitive impairment. Second, the control group was relatively younger and had less burden of vascular risk factors compared to cognitively impaired individuals which could have resulted in selection bias and residual confounding. Also, there was a high burden of vascular risk factors (hypertension, hyperlipidemia and diabetes) in our sample which limited generalizability of the results to the general population. Third, due to overlapping symptoms and similar MRI features between AD and VaD, there was a possibility of misclassification bias. Fourth, some subjects were excluded from the analysis as 3D TOF MRA sequence was affected by motion artifacts during MRA acquisition rendering their data uninterpretable. Fifth, MRA may overestimates stenosis due to blood flow turbulence which was mitigated by confirming stenosis on source images. Finally, angiograms with intra-arterial contrast are considered to be the gold standard to diagnose occlusive disease of the intracerebral arteries; however, it would not have been feasible or ethical to perform such invasive investigations with contrast safety issues in the memory clinic population. Strengths of the study include extensive neuropsychological assessment to diagnose cognitive impairment and dementia, and availability of 3T MRA neuroimaging to grade and classify individuals with ICS which was feasible to perform in a large population-based study.
Conclusion
In conclusion, ICS was shown to be independently associated with VCIND, whereas in dementia, this association was mediated by cerebral ischemic damage. This further suggests that ICS is a marker of cerebral or generalized atherosclerosis and may be a viable treatment target. Further studies focusing on cerebral perfusion and functional markers are required to determine the pathophysiological link between ICS and cognition.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by the Singapore National Medical Research Council, Center Grant, National University Health System (grants NMRC/CG/NUHS/2010 and NMRC/CG/013/2013). Chen had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
SH participated in data acquisition, performed statistical analysis, drafting, and revising the manuscript. XX participated in data acquisition, and revised the manuscript for intellectual content. MKI, HV, and NV participated in data analysis and interpretation, and revised the manuscript for intellectual content. CC was responsible for the study concept and design, obtaining funding, drafting and revising the manuscript.
