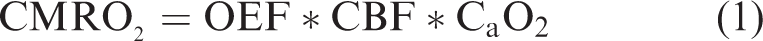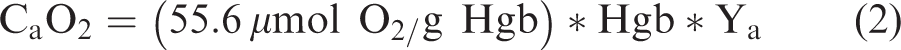Abstract
Oxygen extraction fraction (OEF) and cerebral metabolic rate of oxygen (CMRO2) are markers of cerebral oxygen homeostasis and metabolism that may offer insights into abnormal changes in brain aging. The present study cross-sectionally related OEF and CMRO2 to cognitive performance and structural neuroimaging variables among older adults (n = 246, 74 ± 7 years, 37% female) and tested whether apolipoprotein E (APOE)-ε4 status modified these associations. Main effects of OEF and CMRO2 were null (p-values >0.06), and OEF interactions with APOE-ε4 status on cognitive and structural imaging outcomes were null (p-values >0.06). However, CMRO2 interacted with APOE-ε4 status on language (p = 0.002), executive function (p = 0.03), visuospatial (p = 0.005), and episodic memory performances (p = 0.03), and on hippocampal (p = 0.006) and inferior lateral ventricle volumes (p = 0.02). In stratified analyses, lower oxygen metabolism related to worse language (p = 0.02) and episodic memory performance (p = 0.03) among APOE-ε4 carriers only. Associations between CMRO2 and cognitive performance were primarily driven by APOE-ε4 carriers with existing cognitive impairment. Congruence across language and episodic memory results as well as hippocampal and inferior lateral ventricle volume findings suggest that APOE-ε4 may interact with cerebral oxygen metabolism in the pathogenesis of Alzheimer’s disease and related neurodegeneration.
Introduction
Cerebral oxygen extraction fraction (OEF) reflects the percentage of oxygen extracted from the cerebral capillaries, and cerebral metabolic rate of oxygen (CMRO2) reflects the rate of oxygen consumption in brain tissue. These indices of cerebral oxygen homeostasis may offer insights into normal and abnormal changes in brain aging.1–3 OEF has been shown to increase over the lifespan to compensate for age-related decreases in cerebral blood flow (CBF)1,4 and meet cerebral oxygen demands, thereby maintaining 5 CMRO2. Compared to cognitively unimpaired peers, older adults with mild cognitive impairment (MCI) have similar global CBF but lower global OEF and CMRO2. 2 As low OEF has been linked to increased amyloid burden, 6 and hypometabolism precedes atrophy in MCI, 7 low OEF and CMRO2 may be important early correlates of an underlying Alzheimer’s disease (AD) neurodegenerative process.
The gold standard technique for measuring OEF and CMRO2 is 15O-based positron emission tomography (PET), which requires use of three radiotracers and sampling of arterial blood, limiting use of OEF and CMRO2 as AD biomarkers. 8 Early studies using 15O-based PET to quantify regional OEF, CBF, and CMRO2 have shown conflicting results as to whether changes in OEF or CBF primarily mediate declines in CMRO2 in AD, which may owe to differences in AD disease stage of the cohorts examined and region of interest (ROI) selection. Tohgi et al. 9 identified high OEF in the parietal lobe and low CBF and CMRO2 in parietal, temporal, and frontal lobes of those with AD. Frackowiak et al. 10 found that individuals with degenerative type dementia had no changes in regional OEF and that low CBF in parietal, temporal, and frontal lobes was linked to decreases in CMRO2 in those regions. Ishii et al. 11 found that among those with mild-to-moderate AD, medial temporal OEF was low and low medial temporal lobe CMRO2 was the most distinctive feature of AD. As stated above, discrepancies might owe to differences in ROI selection or between-study cohort heterogeneity, which is a particularly important consideration due to the compensatory interplay between OEF and CBF across the cognitive aging spectrum. 12
Among cognitively unimpaired older adults global OEF and CMRO2 have been inconsistently associated with cognition. Catchlove et al. 5 used noninvasive measurement of venous oxygenation (i.e., T2-Relaxation-Under-Spin-Tagging (TRUST) magnetic resonance imaging (MRI)) to quantify global OEF and grey matter CMRO2 and found that among cognitively unimpaired older adults there was no association between OEF or CMRO2 and cognitive performance. Nonetheless, emerging evidence suggests apolipoprotein E ε4 (APOE-ε4), the largest genetic susceptibility risk factor for sporadic AD 13 and modifier of damage to capillary pericytes 14 responsible for oxygen extraction efficacy, 15 moderates associations between OEF and cognition. Specifically, among cognitively unimpaired older adults, lower OEF related to worse executive function among APOE-ε4 allele carriers only. 3 Among adults with similar dementia severity, APOE-ε4 gene dose is associated with a more global pattern of glucose hypometabolism, 16 and cognitively unimpaired APOE-ε4 carriers unimpaired have glucose hypometabolism in AD-affected regions, 17 which has been associated with worse performance on a memory task. 18 Thus, early detectable associations between CMRO2 and cognition may also exist in a APOE-ε4 dependent manner.
In the present study, we investigated how OEF and grey matter CMRO2 cross-sectionally relate to neuropsychological performances and grey matter structural imaging markers among older adults with and without cognitive impairment. We hypothesized that lower OEF and lower CMRO2 would relate to poorer neuropsychological performances, smaller grey matter volumes, and a smaller AD imaging signature (reflecting low cortical thickness in a composite of regions susceptible to AD-related neurodegeneration).19,20 We also tested OEF and CMRO2 interactions with APOE-ε4 carrier status (present versus absent) and cognitive status (cognitively unimpaired versus impaired) on neuropsychological performances, grey matter volumes, and the AD imaging signature. Given previous research suggesting that lower OEF is associated with worse cognition among older cognitively unimpaired APOE-ε4 carriers, 3 and other evidence showing that individuals with MCI have lower OEF and CMRO2 but not CBF, 2 we hypothesized that associations between lower OEF and lower CMRO2 and worse brain health would be stronger among individuals with existing cognitive impairment and with increased risk for AD (APOE-ε4 carriers).
Methods
Study cohort
The Vanderbilt Memory & Aging Project (VMAP) is a longitudinal cohort investigating the complex intersection of vascular and brain health among aging adults. 21 Inclusion required participants to be at least 60 years old, speak English, have adequate auditory and visual acuity, and have a reliable study partner. At study entry, participants were excluded for MRI contraindication, history of neurological disease (e.g., stroke, dementia), clinical heart failure, major psychiatric illness, head injury with loss of consciousness >5 minutes, and a systemic or terminal illness that might affect follow-up participation. At study entry and each follow-up visit, participants underwent a comprehensive medical evaluation that included a fasting venous blood draw, physical examination, echocardiogram, cardiac magnetic resonance (CMR), neuropsychological assessment, brain MRI, and clinical interview. For the current cross-sectional study, the first available participant data from the 18-month (2014–2016) or 3-year follow-up visit (2015–2018) was used, as MRI variables relevant to this study were added to the protocol during the 18-month follow-up period. Participants were excluded for missing or unusable brain MRI, covariate, or neuropsychological data (Figure 1).
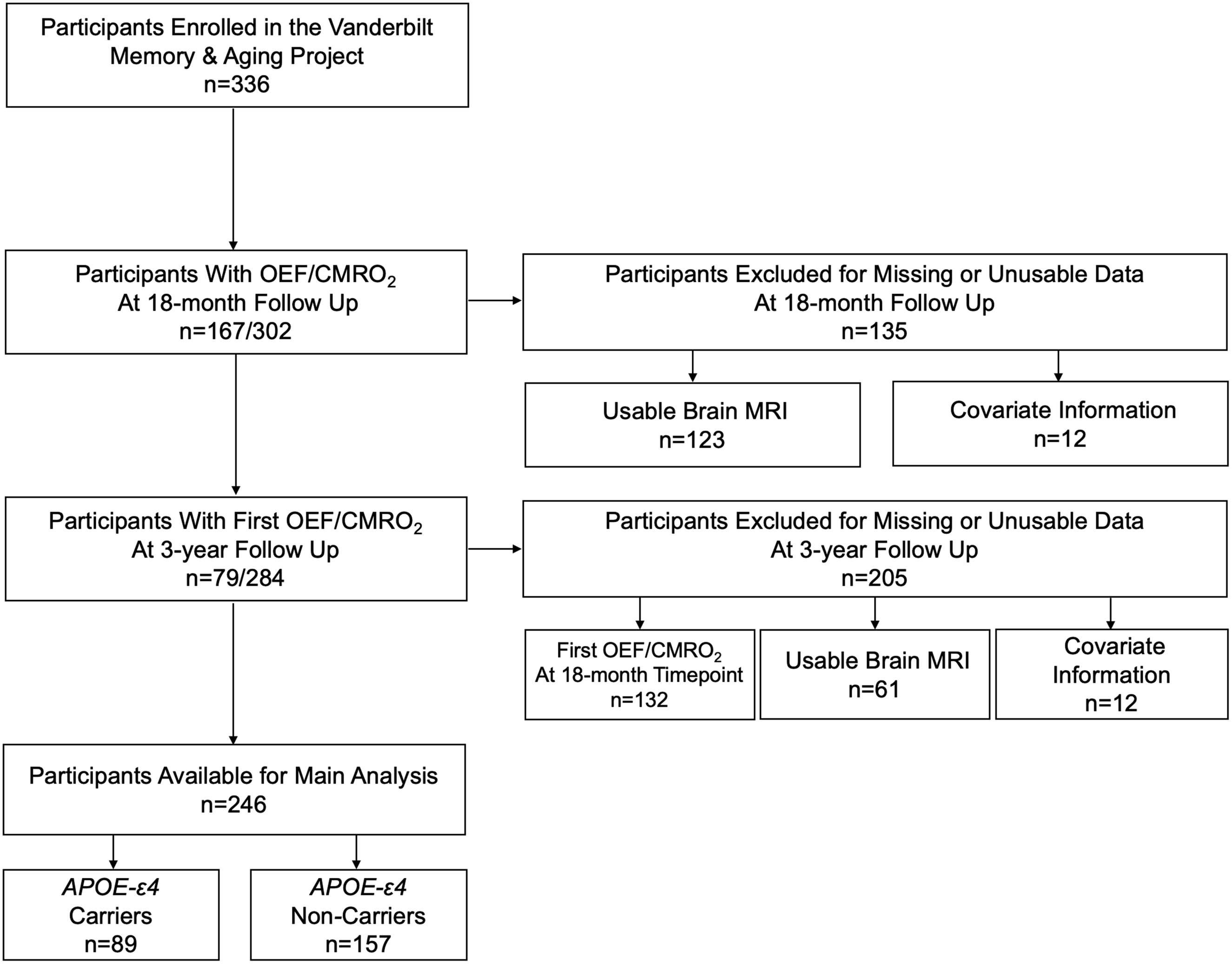
Participant inclusion and exclusion details. The first available participant data from the 18-month or 3-year follow-up visit was used, as MRI variables relevant to this study were added to the protocol during the 18-month follow-up period. Missing data categories were mutually exclusive. APOE-ε4, apolipoprotein E ε4; MRI, Magnetic Resonance Imaging.
The Vanderbilt University Medical Center Institutional Review Board approved the protocol (#120158). Procedures were followed in accordance with the ethical standards of the Vanderbilt University Human Research Protection Program and the Helsinki Declaration of 1975 (and as revised in 1983). Written informed consent was obtained from participants prior to commencement of data collection. Due to participant consent limitations in data sharing, only a subset of data is available to others for the purpose of replicating results or procedures. These data, analytic methods, and study materials can be obtained by contacting the corresponding author.
Neuropsychological assessment
At each time point, including the time points included in this study (18-month or 3-year follow up), participants underwent a detailed neuropsychological protocol assessing language, visuospatial skills, information processing speed, executive function, and episodic memory. As described in detail elsewhere, 22 to minimize multiple comparisons, two composites were calculated (executive function, episodic memory).
Brain MRI acquisition and post processing
Between 2014 and 2017, participants were scanned at the Vanderbilt University Institute of Imaging Science on a 3T Philips Achieva system (Best, the Netherlands) with an 8-channel phased-array SENSE receiver head coil. In 2017, the system was upgraded to a 32-channel dStream head coil. T1-weighted images (TR = 8.9 ms, TE = 4.6 ms, spatial resolution = 1 × 1 × 1 mm3) were acquired as part of a larger multimodal protocol and post-processed with an established Multi-Atlas Segmentation pipeline,23,24 as described previously. 25 After parcellation, 7 regions of interest were used in the present analyses, including whole brain, frontal, temporal, parietal, and occipital lobes, hippocampus, and inferior lateral ventricle. T1-weighted images were also separately postprocessed using FreeSurfer version 5.1.0 (http://surfer.nmr.mgh.harvard.edu/) to obtain cortical thickness measurements. 26 An AD imaging signature 19 was calculated by summing bilateral cortical thickness measurements from regions susceptible to AD-specific neurodegeneration. 20 Lower AD imaging signature values reflect greater cortical thinning in regions susceptible to AD-related neurodegeneration.
As previously described, 25 CBF (milliliters of blood per 100 g tissue per minute) was assessed using pseudo-continuous arterial spin labeling (label duration, 1.65 seconds; post-labeling delay, 1.525 seconds; spatial resolution, 3 × 3 × 7 mm3; TR/TE, 3900/13 milliseconds). Data were corrected for motion and baseline drift using the Functional MRI of the Brain Linear Image Registration Tool 27 and post-processed in MATLAB (MathWorks, Inc, Natick, MA). Images were corrected for slice timing, normalized to an equilibrium magnetization (M0) image (separately acquired using a 20 s repetition time with identical geometry), and converted to absolute CBF following recommended guidelines. 28 Transformation matrices were applied to the global CBF maps following co-registration to a T1-weighted anatomical scan and Montreal Neurological Institute standard template. 29 Mean resting CBF was then calculated in total grey matter.
Venous oxygen (Yv) was noninvasively measured from the superior sagittal sinus using TRUST MRI 30 with a spatial resolution of 3.44 × 3.44 × 5.00 mm3, TR = 1978 ms, flip angle = 90 degrees, and four T2-weightings with effective echo times (0 ms, 40 ms, 80 ms, 160 ms) and Carr-Purcell-Meiboom-Gill (CPMG) τ (i.e., T2-preparation interpulse spacing) = 10 ms. Two images were acquired per echo time, including a label image with signal from magnetically labeled venous blood water and a control image without a magnetic label. The labeling slab was positioned to cover the entire cerebrum and venous structures 25 mm above the imaging slice and was followed by a 1022 ms period for venous blood to flow into the slice. The label image was subtracted from the control image to create a difference image, on which a region of interest was drawn around the four voxels of highest signal intensity. Signal intensities were then plotted against effective echo times to generate a decay curve, which was then used to calculate the blood T2. Blood T2 was converted to Yv using participant-specific hematocrit values and a calibration plot. 31
OEF & CMRO2 calculation
As previously described, 32 OEF was calculated as OEF = (Ya–Yv)/Ya where arterial oxygenation (Ya) was the average of three pulse oximetry readings acquired during the scan. The venous oxygenation, Yv, was calculated from the TRUST data as described above.
Grey matter CMRO2 (µmol/100g/min) was derived using the Fick Principle
33
and the arterial oxygen content (CaO2),
34
where,
and, hemoglobin level (Hgb, g/dL) was captured as part of the fasting blood draw.
APOE genotyping
As previously described, 21 a TaqMan single‐nucleotide polymorphism genotyping assay from Applied Biosystems (Foster City, CA) was used to identify the 2 single‐nucleotide polymorphisms that define the APOE ε2, ε3, and ε4 alleles. Polymerase chain reaction in 5‐µL reactions was completed using a Life Technologies 7900HT real‐time polymerase chain reaction machine. Genotype information was analyzed using Life Technologies SDS 2.4.1 software.
Covariates
Covariates were selected a priori based on their potential to confound analytical models. Systolic blood pressure was the mean of 2 measurements. Diabetes mellitus was defined as fasting blood glucose ≥126 mg/dL, hemoglobin A1c ≥6.5%, or oral hypoglycemic or insulin medication usage. Medication review determined antihypertensive medication use. Left ventricular (LV) hypertrophy was defined on echocardiogram (LV mass index >115 g/m2 in men, >95 g/m2 in women). Self‐report atrial fibrillation was corroborated by any one of the following sources: echocardiogram, documented prior procedure or ablation for atrial fibrillation, CMR, or medication usage for atrial fibrillation. Current cigarette smoking (yes or no within the year before baseline) was ascertained by self‐report. Self‐reported prevalent cardiovascular disease (CVD) with supporting medical record evidence included coronary heart disease, angina, or myocardial infarction (note, heart failure was a parent‐study exclusion). Framingham Stroke Risk Profile (FSRP) score was calculated by applying points by sex for age, systolic blood pressure accounting for antihypertensive medication usage, diabetes mellitus, current cigarette smoking, LV hypertrophy, CVD, and atrial fibrillation. 35 Clinical Dementia Rating (CDR) 36 global score at the visit in which OEF and CMRO2 were acquired was used to define cognitive status as unimpaired (CDR = 0) or impaired (CDR ≥ 0.5). APOE-ε4 carrier status was defined using a dominant coding as positive (ε2/ε4, ε3/ε4, ε4/ε4) or negative (ε2/ε2, ε2/ε3, ε3/ε3) and additive coding with APOE-ε4 defined as 0, 1, or 2 ε4 alleles and modeled as a continuous variable.
Analytical plan
Linear regressions with ordinary least square estimates related OEF and CMRO2 individually to cross-sectional neuropsychological performances, grey matter volumes, and AD imaging signature (one test per model). Models were adjusted for age, sex, race/ethnicity, education, APOE-ε4 carrier status (positive, negative), FSRP (excluding points assigned for age), and CDR global score (0, ≥0.5) to account for cognitive status. For models with neuroimaging outcomes (except the AD imaging signature, which is a sum of cortical thickness measures) 19 intracranial volume was also included as a covariate.
To test the APOE-ε4 status hypothesis, models were repeated with an oxygen metabolism variable x APOE-ε4 status interaction term and then stratified by APOE-ε4 carrier status. Follow-up models assessed if significant oxygen metabolism variable x APOE-ε4 status interactions were similar among cognitively unimpaired participants (CDR = 0) and participants with mild cognitive impairment (CDR = 0.5) by repeating interaction analyses among cognitively unimpaired participants (CDR = 0) and participants with mild cognitive impairment (CDR = 0.5). Interaction and stratification models were also repeated using APOE-ε4 allele count. To determine if results differed by global cognitive status, models tested interactions with CDR global score followed by stratification by CDR (0, ≥0.5). Follow-up models assessed if cognitive status moderated associations between OEF or CMRO2 and outcomes among APOE-ε4 carriers by testing interactions with CDR followed by stratification by CDR (0, ≥0.5). To assess if associations between OEF or CMRO2 and outcomes were driven by any particular component variable (see OEF & CMRO2 Calculation above), significant models were repeated substituting each component variable individually in place of OEF or CMRO2.
Sensitivity analyses were performed (a) excluding participants with atrial fibrillation or CVD to assess if associations were independent of cardiovascular comorbidities (b) excluding outliers (values >4 standard deviations from the mean) (c) excluding participants with more severe cognitive impairment (CDR ≥1) and (d) excluding participants imaged using the 32-channel dStream head coil. Significance was set a priori at p < 0.05. To account for multiple comparisons, the Benjamini-Hochberg procedure 37 was used to control the false discovery rate (FDR), where thresholds were set at Q = 0.05 for all models. Statistical analyses were conducted using R 3.6.2 (https://www.r-project.org).
Results
Participant characteristics
Participants included 246 adults aged 62 to 94 years (74 ± 7 years); 37% were female; 85% self-identified as non-Hispanic white; 36% were APOE-ε4 carriers (29% ε4 heterozygous, 7% ε4 homozygous), and 58% were cognitively unimpaired based on CDR global score. As expected, OEF and CMRO2 were correlated (r = 0.53, p < 1 × 10−17) and OEF and CBF were negatively correlated (r = −0.35, p < 1 × 10−7) in the entire sample. Table 1 summarizes characteristics for the entire sample by APOE-ε4 carrier status.
Participant characteristics.
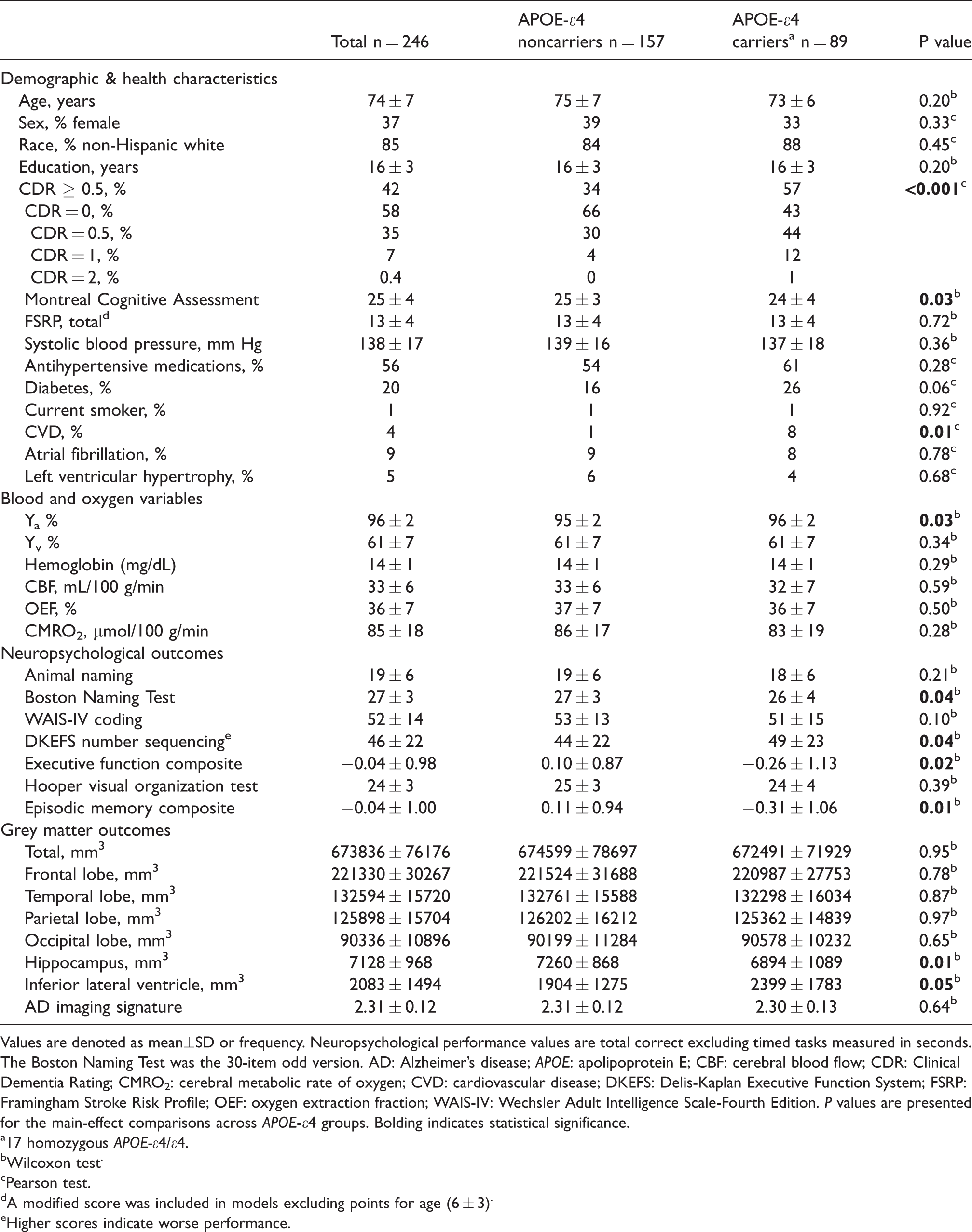
Values are denoted as mean±SD or frequency. Neuropsychological performance values are total correct excluding timed tasks measured in seconds. The Boston Naming Test was the 30-item odd version. AD: Alzheimer’s disease; APOE: apolipoprotein E; CBF: cerebral blood flow; CDR: Clinical Dementia Rating; CMRO2: cerebral metabolic rate of oxygen; CVD: cardiovascular disease; DKEFS: Delis-Kaplan Executive Function System; FSRP: Framingham Stroke Risk Profile; OEF: oxygen extraction fraction; WAIS-IV: Wechsler Adult Intelligence Scale-Fourth Edition. P values are presented for the main-effect comparisons across APOE
a17 homozygous APOE-ε4/ε4.
bWilcoxon test.
cPearson test.
dA modified score was included in models excluding points for age (6 ± 3).
eHigher scores indicate worse performance.
OEF and CMRO2 main effect models
Both OEF and CMRO2 were unrelated to all neuropsychological performances (p-values >0.08), grey matter volumes (p-values >0.06), or the AD imaging signature (p-values >0.08). See Supplementary Table 1 for details.
APOE-ε4 interaction and stratification models
OEF did not interact with APOE-ε4 carrier status on any neuropsychological or neuroimaging variable (p-values >0.06). See Figure 2 and Supplementary Table 2 for details. Results were similar in sensitivity analyses (data not shown). OEF modestly interacted with APOE-ε4 allele count on episodic memory composite performance (p = 0.005, FDR-adjusted-p = 0.04) and hippocampal volume (p = 0.01, FDR-adjusted-p = 0.08). While stratification models by APOE-ε4 allele count were null (p-values >0.07), the pattern of results suggest lower OEF was associated with worse memory function and lower hippocampal volume among participants with two APOE-ε4 alleles. See Supplementary Figure 1 for illustrations and Supplementary Table 3 for details. In sensitivity analyses excluding participants with CDR ≥ 1 (mild or moderate dementia), OEF interactions with APOE-ε4 allele count on episodic memory composite performance and hippocampal volume were attenuated but maintained significance (p-values <0.03). Results were similar to the primary analyses in all other sensitivity analyses (data not shown).
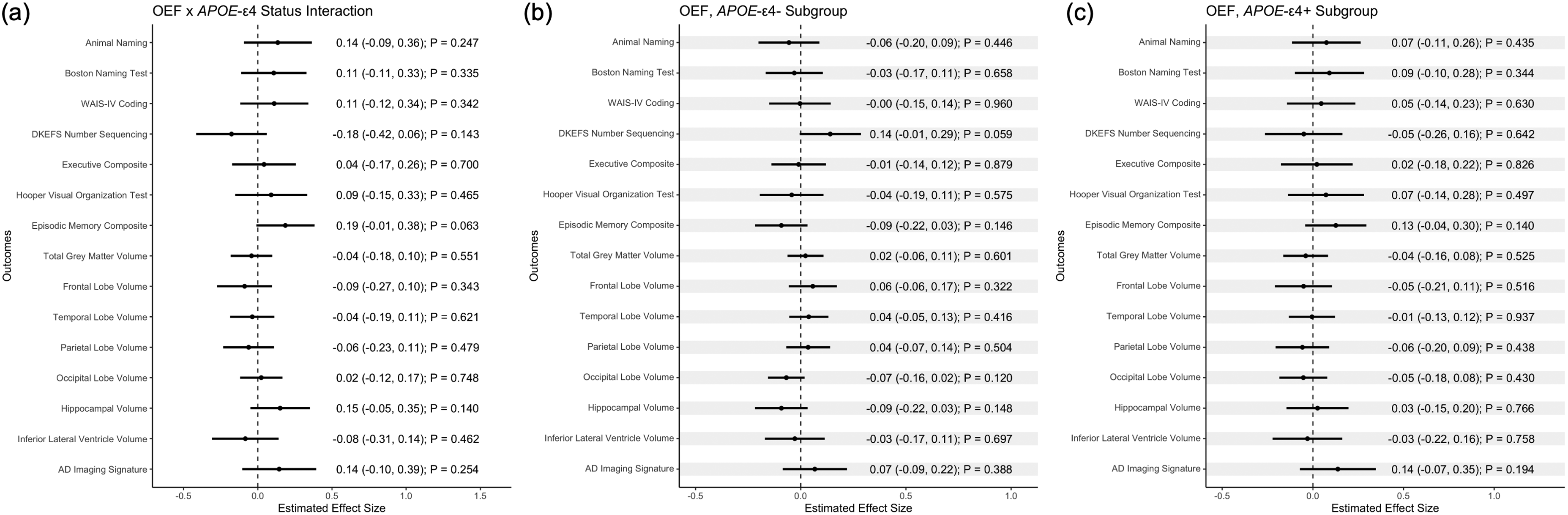
(a) OEF x APOE-ε4 allele status interaction and APOE-ε4 subgroup (b, c) forest plots. Models were adjusted for age, sex, race/ethnicity, education, FSRP minus age, and CDR global score (0, ≥0.5). Grey matter outcomes were additionally adjusted for ICV where appropriate. The Boston Naming Test was the 30‐item odd version. AD, Alzheimer’s disease; APOE, apolipoprotein E; DKEFS, Delis–Kaplan Executive Function System; OEF, oxygen extraction fraction; WAIS‐IV, Wechsler Adult Intelligence Scale, Fourth Edition. Higher scores indicate worse performance on DKEFS number sequencing.
CMRO2 interacted with APOE-ε4 carrier status on Boston Naming Test (p = 0.003, FDR-corrected-p = 0.02), executive function composite (p = 0.03, FDR-adjusted-p = 0.07), Hooper Visual Organization (p = 0.005, FDR-adjusted-p = 0.02), and episodic memory composite (p = 0.04, FDR-adjusted-p = 0.07), and hippocampal volume (p = 0.006, FDR-adjusted-p = 0.05) and inferior lateral ventricle volume (p = 0.02, FDR-adjusted-p = 0.07). Among APOE-ε4 carriers, lower CMRO2 related to worse performance on Boston Naming Test (p = 0.02, FDR-adjusted-p = 0.09) and episodic memory composite (p = 0.03, FDR-adjusted-p = 0.09). See Figures 3 and 4 for illustrations and Supplementary Table 4 for details. CMRO2 interactions with APOE-ε4 carrier status which were significant in the entire cohort were then examined in follow-up models to assess if CMRO2 x APOE-ε4 carrier status interactions on outcomes differed among participants with CDR = 0 compared to participants with CDR = 0.5. Interaction beta-values were relatively similar between the models, but greater in magnitude among those with CDR = 0.5, and models were generally not significant and limited by smaller sample sizes. See Supplementary Table 5 for details. In sensitivity analyses excluding participants with CDR ≥ 1, CMRO2 interactions with APOE-ε4 carrier status on outcomes were attenuated such that CMRO2 no longer significantly interacted with APOE-ε4 carrier status on executive function composite (p = 0.16) or episodic memory composite (p = 0.22). Further, in stratified models excluding participants with CDR ≥ 1, CMRO2 was no longer significantly related to Boston Naming Test (p = 0.07) or episodic memory composite (p = 0.18) among APOE-ε4 carriers. Results were similar to the primary analyses in all other sensitivity analyses (data not shown). Follow-up models investigating the association between lower CMRO2 and both worse Boston Naming Test and episodic memory composite performance among APOE-ε4 carriers revealed no significant association between any CMRO2 component variable and outcomes (p-values >0.07). See Supplementary Table 6 for details.
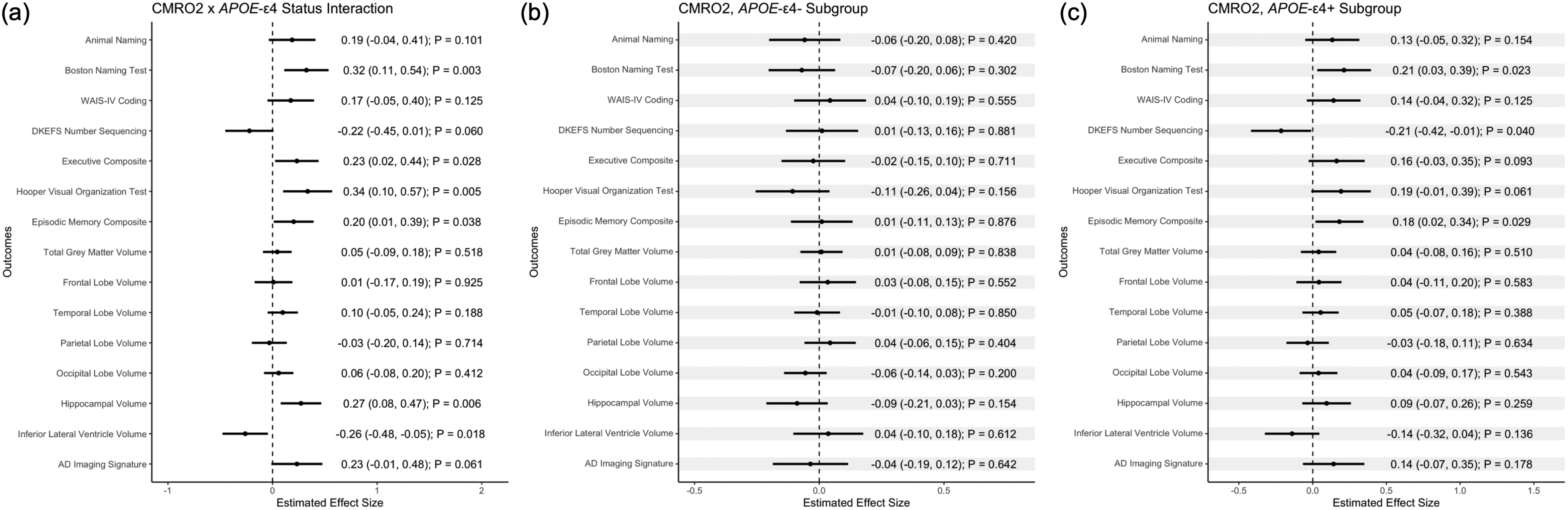
(a) CMRO2 x APOE-ε4 allele status interaction and APOE-ε4 subgroup (b, c) forest plots. Models were adjusted for age, sex, race/ethnicity, education, FSRP minus age, and CDR global score (0, ≥0.5). Grey matter outcomes were additionally adjusted for ICV where appropriate. The Boston Naming Test was the 30‐item odd version. AD, Alzheimer’s disease; APOE, apolipoprotein E; CMRO2, cerebral metabolic rate of oxygen; DKEFS, Delis–Kaplan Executive Function System; WAIS‐IV, Wechsler Adult Intelligence Scale, Fourth Edition. Higher scores indicate worse performance on DKEFS number sequencing.
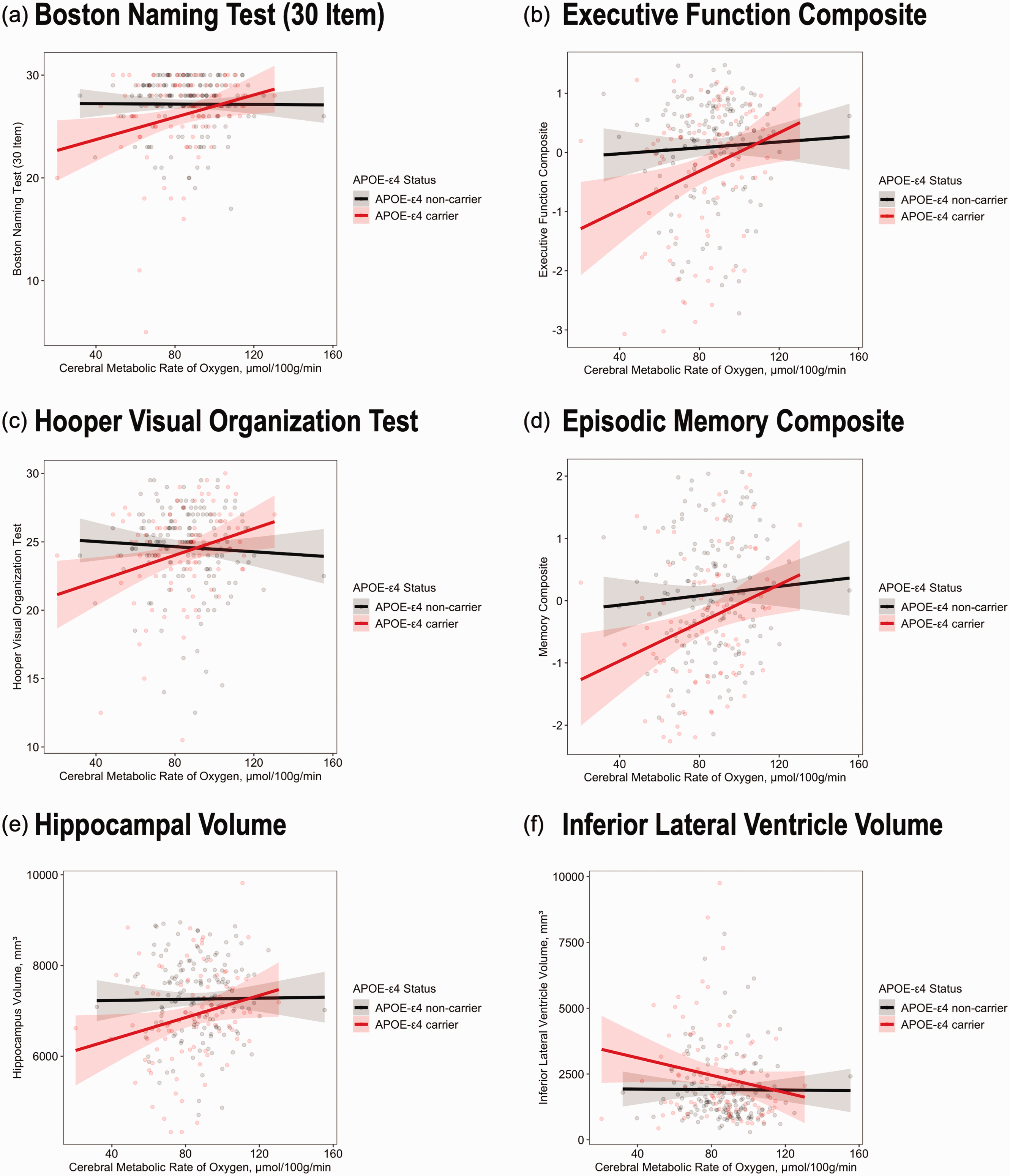
CMRO2 x APOE-ε4 allele status interactions. (a) Boston Naming Test (b) Executive Composite (c) Hooper Visual Organization Test (d) Episodic Memory Composite (e) Hippocampal Volume (f) Inferior Lateral Ventricle Volume. A: interaction p-value = 0.003, B: interaction p-value = 0.03, C: interaction p-value = 0.005, D: interaction p-value = 0.04, E: interaction p-value = 0.006, F: interaction p-value = 0.02. Results were similar in sensitivity analyses excluding outliers. APOE-ε4, apolipoprotein E ε4; CMRO2, cerebral metabolic rate of oxygen.
CMRO2 interacted with APOE-ε4 allele count on Animal Naming (p = 0.01, FDR-adjusted-p = 0.02), Boston Naming Test (p = 0.004, FDR-adjusted-p = 0.01), executive function composite (p = 0.01, FDR-adjusted-p = 0.01), Hooper Visual Organization (p = 0.004, FDR-adjusted-p = 0.01), and episodic memory composite (p = 0.003, FDR-adjusted-p = 0.01), and hippocampal volume (p = 0.007, FDR-adjusted-p = 0.05), inferior lateral ventricle volume (p = 0.01, FDR-adjusted-p = 0.05), and the AD imaging signature (p = 0.02, FDR-adjusted-p = 0.07). While stratified models were mostly null (p-values >0.04), lower CMRO2 tended to relate to worse cognitive performance or smaller grey matter volumes among APOE-ε4 carriers in a dose-dependent manner. See Supplementary Figure 2 for illustrations and Supplementary Table 7 for details. Results were attenuated in sensitivity analyses excluding participants with CDR ≥ 1 such that CMRO2 no longer significantly interacted with APOE-ε4 allele count on Animal Naming (p = 0.11), executive function composite (p = 0.16), episodic memory composite (p = 0.08), or the AD imaging signature (p = 0.17). Results were similar to the primary analyses in all other sensitivity analyses (data not shown).
CDR interaction and stratification models
CDR interacted with OEF on parietal lobe volume (p = 0.03, FDR-adjusted-p = 0.22), such that among cognitively impaired participants (CDR ≥ 0.5), lower OEF was associated with smaller parietal lobe volume (p = 0.02, FDR-adjusted-p = 0.17). The remaining interaction models were null (p-values >0.05). See Supplementary Figure 3 for illustrations and Supplementary Table 8 for details. Results were similar in sensitivity analyses (data not shown). In follow-up OEF component variable models, Ya was not significantly related to parietal lobe volume (p = 0.12) while higher Yv was associated with smaller parietal lobe volume (p = 0.05) among cognitively impaired participants (CDR ≥ 0.5). See Supplementary Table 6 for details.
CDR interacted with CMRO2 on total grey matter volume (p = 0.03, FDR-adjusted-p = 0.11), parietal lobe volume (p = 0.05, FDR-adjusted-p = 0.11), and inferior lateral ventricle volume (p = 0.04, FDR-adjusted-p = 0.11). The pattern of results suggests that among cognitively impaired participants (CDR ≥ 0.5), lower CMRO2 is associated with smaller total grey matter and parietal lobe volume and larger inferior lateral ventricle volume. See Supplementary Figure 3 for illustrations and Supplementary Table 9 for details. Results were similar in sensitivity analyses (data not shown).
CDR interaction and stratification models among APOE-ε4 allele carriers
Among APOE-ε4 allele carriers, OEF did not interact with CDR on any outcomes (p-values >0.15). See Supplementary Table 10 for details.
Among APOE-ε4 allele carriers, CDR interacted with CMRO2 only on Number Sequencing (p = 0.01, FDR-adjusted-p = 0.10). In stratified analyses, lower CMRO2 related to worse Number Sequencing (p = 0.01, FDR-adjusted-p = 0.05) among cognitively impaired (CDR ≥ 0.5) APOE-ε4 allele carriers, and the pattern of results was similar for other cognitive tests. See Supplementary Table 11 for details. Results were mildly attenuated in sensitivity analyses excluding participants with CDR ≥ 1 and similar to the primary analyses in all other sensitivity analyses (data not shown). Follow-up models investigating the association between CMRO2 component variables and Number Sequencing among cognitively impaired (CDR ≥ 0.5) APOE-ε4 carriers revealed no significant association between any CMRO2 component variable and Number Sequencing (p-values >0.16). See Supplementary Table 6 for details.
Discussion
We investigated the association between cerebral oxygen extraction and metabolic rate and neuropsychological and structural imaging markers of brain aging in a group of community-dwelling older adults. While main effect models were null, we observed significant connections linking lower CMRO2, and to a lesser extent lower OEF, with worse brain health among participants at increased risk for cognitive decline (APOE-ε4 carriers) and participants with existing cognitive impairment (CDR ≥ 0.5). Among APOE-ε4 carriers, the pattern of results suggested lower CMRO2 was associated with worse performance in several cognitive domains, including language, executive function, visuospatial, and episodic memory, with significant stratified effects in the domains of language and episodic memory. These cognitive associations are aligned with our neuroimaging findings where CMRO2 interacted with APOE-ε4 carrier status on indicators of hippocampal neurodegeneration (i.e., smaller hippocampal volume and larger inferior lateral ventricle volume). 38 Congruence between our cognitive and neuroimaging findings, and their localization to hippocampal structure and function, suggest that cerebral oxygen metabolism and APOE-ε4 may interact in the pathogenesis of a neurodegenerative disease, such as AD. Nonetheless, because our cohort contained a higher proportion of APOE-ε4 carriers with cognitive impairment than non-carriers with cognitive impairment, we are cautious about asserting that CMRO2 and APOE-ε4 pathogenically interact. Interactions between CMRO2 and APOE-ε4 carrier status on outcomes significant in our primary interaction analysis appeared to be greater in magnitude among those with mild cognitive impairment (CDR = 0.5) compared to cognitively unimpaired participants (CDR = 0), but associations were similar, and these models were generally not statistically significant and limited by smaller sample sizes. Importantly, when excluding participants with more severe cognitive impairment (CDR ≥ 1), associations between lower CMRO2 and worse language and episodic memory performance among APOE-ε4 carriers were no longer significant, but interactions between CMRO2 and APOE-ε4 carrier status on most outcomes significant in the primary analysis persisted.
To our knowledge, this study is among the first to investigate interactions between CMRO2 and APOE-ε4 carrier status on cognitive and structural neuroimaging outcomes. In addition, we investigated interactions between CMRO2 and cognitive status, which indicated potential associations between lower CMRO2 and smaller brain volumes (i.e., smaller total grey matter and parietal lobe volume and larger inferior lateral ventricles) but no associations between cognition and CMRO2 among participants with existing cognitive impairment (CDR ≥ 0.5). These results are not in agreement with one prior study which found a modest association between lower CMRO2 and worse cognitive performance among older adults with amnestic MCI. 2 Nonetheless, associations between lower CMRO2 and worse cognitive performance were present among individuals at increased risk of cognitive decline (APOE-ε4 carriers), and were primarily driven by APOE-ε4 carriers with existing cognitive impairment (CDR ≥ 0.5). Therefore, the apparent discrepancy between the aforementioned study and ours might be due to the fact that that we collected and utilized APOE genotyping information in our study, while the authors of the previous study did not. Our findings align with previous literature reporting that, among cognitively unimpaired individuals, CMRO2 is unrelated to cognitive performance. 5
In contrast to one prior study reporting that lower OEF relates to worse executive function among older cognitively unimpaired APOE-ε4 carriers, 3 we observed no interactions between OEF and APOE-ε4 carrier status on any cognitive outcome. Further, there were no associations between OEF and cognition among cognitively unimpaired or impaired APOE-ε4 carriers. We did observe modest interactions between OEF and APOE-ε4 allele count on episodic memory and hippocampal volume, which persisted when excluding participants with more severe cognitive impairment (CDR ≥ 1), but we are cautious when interpreting these results due to the small sample of APOE-ε4 homozygotes (n = 17) and are reluctant to infer that OEF and APOE-ε4 count pathogenically interact in a dose dependent manner.
Associations between lower CMRO2 and worse brain health may exist among APOE-ε4 carriers because possession of an APOE-ε4 allele is associated with an altered neurometabolic profile, even in young adulthood.39,40 Aged APOE-ε4 targeted replacement mice display a compensatory increase in entorhinal cortex fatty acid oxidation, 41 which is a metabolic pathway that requires more oxygen per ATP generated than glucose oxidation and produces more oxidative damage. 42 Reduction in glucose oxidation and compensatory oxidation of substrates other than glucose is known to occur in AD. 43 Whether driven by the influence of APOE-ε4 on fatty acid or glucose metabolism or another mechanism, our results suggest APOE-ε4 carriers might be more vulnerable to worse brain health with lower CMRO2. Previous findings suggest older APOE-ε4 carriers may have higher reliance on cortical energy metabolism to support cognition. Yi et al. 44 found that cognitively unimpaired APOE-ε4 carriers with compensatory frontotemporal glucose hypermetabolism performed better on an episodic memory task, and Arenaza-Urquijo et al. 45 similarly found associations between higher frontotemporal glucose metabolism and episodic memory among cognitively unimpaired APOE-ε4 carriers.
Both OEF and CMRO2 interacted with APOE-ε4 allele count on hippocampal volume and episodic memory performance. The congruent localization of these results to hippocampal structure and function may owe to the greater vulnerability of hippocampal ATP production to low oxygen conditions due to differences in hippocampal pericyte morphology. 46 APOE-ε4 is a molecular moderator of brain capillary pericyte degeneration.14,47 Capillary dysfunction reduces oxygen extraction efficacy 15 and loss of pericytes reduces tissue oxygenation, 48 which may lead to deleterious energy sparing adaptations 49 and AD-related reductions in brain health with lower OEF and CMRO2 in APOE-ε4 allele carriers.
Secondary analyses using an additive model of the APOE-ε4 allele (i.e., allele count) resulted in additional interactions with cerebral oxygen utilization variables on outcomes compared to the dominant genetic model (i.e., APOE-ε4 status). APOE-ε4 allele count interacted with OEF on episodic memory performance and hippocampal volume and additionally interacted with CMRO2 on the AD imaging signature. Several studies have shown that APOE-ε4 allele dose relates to regional cerebral glucose hypometabolism across the cognitive aging spectrum. 50 Among cognitively unimpaired older adults, APOE-ε4 allele dose relates to greater glucose hypometabolism in AD-related brain regions. 51 APOE-ε4 allele dose is associated with a more global pattern of glucose hypometabolism among individuals with AD, 16 and a lower ratio of peripheral APOE-ε3-to-APOE-ε4 among older cognitively unimpaired APOE-ε3/ε4 heterozygotes has been associated with associated with lower regional cerebral glucose metabolism 52 and higher plasma glucose levels, 53 suggesting dosage of peripheral APOE isoforms may also influence central and peripheral glucose metabolism. OEF has been observed to be slightly lower per APOE-ε4 allele in cognitively unimpaired older adults, 3 and while that finding is not observed in the present study (data not shown), we exclusively observed OEF interactions with APOE-ε4 in additive genetic models. Though limited by a small sample of APOE-ε4 homozygotes, our findings expand upon previous literature by indicating that oxygen metabolism variables may dose-dependently interact with APOE-ε4 allele count on AD-related cognitive performances and brain structures.
CMRO2 interacted with APOE-ε4 status and count on more cognitive domains (i.e., language, visuospatial, and executive function) and structural neuroimaging variables (i.e., inferior lateral ventricle volume, and AD imaging signature) than did OEF. These more wide-ranging cognitive and structural associations may be because CMRO2 takes into account not only oxygen extraction but also arterial oxygen content and perfusion (i.e., CBF). CMRO2 thus provides insight into the compensatory interplay between CBF and OEF that occurs with aging.1,5 Traditionally, lower OEF can reflect either hypometabolism, indicating less demand for oxygen extraction, or preserved blood flow, as OEF would be higher if there were lower blood flow but preserved oxygen demand. Thus, others have shown that interpretation of OEF may differ depending on the presence of vascular risk factors. 6 Interpretation of CMRO2, however, is less limited by such considerations due to its inclusion of both oxygen extraction and perfusion information.
Our study had several strengths, including robust neuroimaging methods for noninvasively measuring resting OEF and grey matter CMRO2, a comprehensive neuropsychological protocol, and inclusion of numerous covariates to adjust for potential confounding in our statistical models. Neuroimaging data and blood samples were processed by core laboratories with staff blinded to participant information. While our values for CMRO2 are somewhat lower than those observed in the literature, this discrepancy is likely primarily due to differences in CBF quantification, as other CMRO2 component variables are in similar ranges to values in previous studies. 2 One major strength of our study was our use of participant specific Ya, hematocrit, and hemoglobin values in the calculation OEF and CMRO2, which have often been assumed as reference values in previous studies associating brain health with TRUST MRI variables.2,3,6 In models where CMRO2 significantly related to an outcome, no CMRO2 component variable was significantly related to the outcome, indicating that associations between CMRO2 and outcomes were not driven by any single CMRO2 component variable, highlighting the potential importance of capturing all such variables. Nonetheless, it is possible that regional CBF may explain associations between CMRO2 and outcomes among APOE-ε4 carriers even though the component variable grey matter CBF did not.
Several limitations must be considered when interpreting our results. First, many analytical models were calculated, increasing the probability of a Type I error. While we applied an FDR correction to minimize false positive errors and many associations survived, it is possible that some significant results (though biologically plausible) are spurious. Replication is needed to confirm our results, particularly associations found in secondary APOE-ε4 count models with smaller sample sizes. Second, while robust, the TRUST technique used here captures venous oxygenation from the superior sagittal sinus, which is then used to calculate global OEF and grey matter CMRO2, so relevant regional differences in oxygen metabolism variables may exist that we were unable to capture. Future studies might employ emerging neuroimaging techniques54,55 to identify potential regional differences in these indices that may be tied to brain health. Third, we used grey matter CBF rather than whole brain CBF in our calculation of grey matter CMRO2, so there may be relevant differences in white matter CMRO2 that were not captured here. Finally, our cohort consisted of predominantly white, well-educated, and relatively healthy older adults, which limits generalizability of our findings. One recent study showed that associations between lower OEF and worse cognition are only present among older adults with fewer vascular risk factors. 6 Therefore, associations between lower OEF and cognition may differ in cohorts with a greater burden of vascular risk factors. Because our findings indicate associations are stronger in those adults at increased risk of cognitive decline and with existing cognitive impairment, associations might be more robust in other cohorts with greater AD risk or burden of comorbidities.
In conclusion, we found that non-invasively measured global OEF and grey matter CMRO2 were not associated with brain health among a cohort of older adults as a whole, but our interaction analyses indicated that global OEF and grey matter CMRO2 were associated with brain health in an APOE-ε4 and cognitive status dependent manner. These results provide evidence that cerebral oxygen utilization is a key marker of brain health, particularly among individuals at genetic risk for AD. Future studies should examine OEF and CMRO2 longitudinally to determine if compensatory increases, or a lack thereof, in either variable account for our cross-sectional findings.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X211056393 - Supplemental material for Lower cerebral oxygen utilization is associated with Alzheimer’s disease-related neurodegeneration and poorer cognitive performance among apolipoprotein E ε4 carriers
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X211056393 for Lower cerebral oxygen utilization is associated with Alzheimer’s disease-related neurodegeneration and poorer cognitive performance among apolipoprotein E ε4 carriers by W Hudson Robb, Omair A Khan, Humza A Ahmed, Judy Li, Elizabeth E Moore, Francis E Cambronero, Kimberly R Pechman, Dandan Liu, Katherine A Gifford, Bennett A Landman, Manus J Donahue, Timothy J Hohman and Angela L Jefferson in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Alzheimer’s Association IIRG-08-88733 (ALJ); R01-AG034962 (ALJ); R01-NS100980 (ALJ); K24-AG046373 (ALJ); R01-AG056534 (ALJ); Paul B. Beeson Career Development Award in Aging K23-AG045966 (KAG); T32-AG058524 (WHR); F30-AG064847 (EEM); T32-GM007447 (EEM); K01-AG049164 (TJH); UL1-TR000445 and UL1-TR002243 (Vanderbilt Clinical Translational Science Award); S10-OD023680 (Vanderbilt’s High-Performance Computer Cluster for Biomedical Research); P20-AG068082 (Vanderbilt Alzheimer’s Disease Research Center), and Vanderbilt Memory and Alzheimer’s Center.
Acknowledgements
The authors thank the dedicated Vanderbilt Memory & Aging Project participants and their loved ones.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TJH serves on the Scientific Advisory Board for Vivid Genomics. MJD is a paid consultant for Pfizer Inc, Global Blood Therapeutics, and LymphaTouch; is a paid advisory board member for bluebird bio; and receives research-related support from Philips Healthcare and research funding from Pfizer Inc.
Authors’ contributions
Author contributions included conception and study design (WHR, ALJ, KAG, MJD, TJH), data collection or acquisition (EEM, FEC, KRP), neuroimaging analysis (KRP, BAL, HAA), statistical analysis (OAK, DL), interpretation of results (WHR, JL, FEC, MJD, TJH, ALJ), drafting the manuscript work or revising it critically for important intellectual content (WHR, OAK, HAA, JL, EEM, FEC, KRP, DL, KAG, BAL, MJD, TJH, ALJ) and approval of final version to be published and agreement to be accountable for the integrity and accuracy of all aspects of the work (all authors).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.