Abstract
Ability to image cerebral metabolic rate of oxygen (CMRO2) is essential for studying the fundamental role of oxidative metabolism in brain function and disease. We have demonstrated recently that three-dimensional (3D) CMRO2 images can be obtained in the rat brain during a 2-min 17O2 inhalation using the 17O MR spectroscopic imaging (MRSI) approach at high field. The feasibility for establishing a completely noninvasive approach for imaging CMRO2 has also been demonstrated. In this study, we further explored the feasibility of 17O MRSI approach for performing repeated CMRO2 measurements within a short period of time and evaluated the reproducibility of the repeated measurements. Subsequently, we applied the 17O MRSI approach to measure CMRO2 and cerebral blood flow (CBF) values at two brain temperatures in the α-chloralose anesthetized rat brain at 9.4 T. Finally, we tested the validity of simplified model for noninvasively determining CMRO2 in normothermic and hypothermic rat brain. The results show (i) an excellent reproducibility among repeated measurements of 3D CMRO2 images under the same physiologic condition; (ii) a 44% decrease of CMRO2 across the rat brain at mild hypothermic (32°C) condition as compared with normothermic (37°C) condition; and (iii) a close correlation between CMRO2 and CBF within a relatively wide physiologic range. This study demonstrates the capability of 17O MRSI approach for noninvasively imaging CMRO2 and its changes caused by physiologic perturbation. This approach, thus, should provide a promising neuroimaging modality for studying oxidative metabolism and bioenergetics associated with brain functions and diseases.
Introduction
The cerebral metabolic rate of oxygen utilization (CMRO2) is tightly associated with brain physiology and pathology (Siesjo, 1978). Determination of CMRO2 is important for understanding the essential role of oxidative metabolism in brain function and brain diseases. The most straightforward nuclear magnetic resonance (NMR) method for directly measuring CMRO2 is the use of 17O NMR technique to determine the dynamic change of metabolic H217O, which is formed in mitochondria through oxygen metabolism of inhaled 17O2 (Arai et al, 1990; Fiat and Kang, 1992; Mateescu and Fercu, 1994; Mateescu et al, 1991; Pekar et al, 1991; Zhu et al, 2002). However, the major challenges of 17O NMR technique are its low intrinsic NMR sensitivity because of the low gyromagnetic ratio of 17O spins and relatively small change in the metabolic H217O concentration generated during an 17O2 inhalation. Recently, we have demonstrated a substantial sensitivity gain for detecting 17O NMR signal at high/ultrahigh magnetic field (Zhu et al, 2001). This crucial sensitivity improvement at higher field can be attributed to the field-independent relaxivity of the 17O spins in H217O, which leads to a significant sensitivity gain according to a quadratic power relation with the magnetic field strength (Zhu et al, 2001, 2005). This gain makes it possible to use the
One potential and important merit of the 17O MRSI approach is its ability allowing repeated CMRO2 measurements during the same experimental session. Because in the brain tissue, the metabolic H217O generated during a brief 17O2 inhalation can be washed out quickly and reach a new steady-state concentration within approximately 10 mins after the ceasing of 17O2 inhalation (Zhu et al, 2002). This characteristic allows subsequent measurements of CMRO2, which are particularly valuable for studies aiming at CMRO2 changes induced by physiologic or pathologic perturbations. However, to fully realize this advantage, the reproducibility of the repeated CMRO2 measurements has to be assessed and validated. Therefore, the first part of this paper was to evaluate the reproducibility of
It is well documented that the basal CMRO2 is sensitive to the brain temperature (see (Erecinska et al, 2003; Siesjo, 1978) and the references cited therein). However, most studies reported in the literature were based on the global CMRO2 measurements of entire brain using the Kety—Schmidt method (Kety and Schmidt, 1948a,
b
), which was lack of spatial information regarding regional CMRO2 variation inside the brain. In the second part of this study, we investigated the feasibility of using 3D 17O MRSI at 9.4 T for imaging and quantifying absolute CMRO2 values in the rat brain at normal brain temperature (37°C) (i.e., normothermia) and mild hypothermia (32°C) conditions. We also compared CMRO2 calculation results based on two different models: a
Finally, the measured CMRO2 results in this study were correlated with the cerebral blood flow (CBF) results, which were experimentally determined via bolus injection of H217O tracer into the brain through the internal carotid artery (Zhu et al, 2001). These data were obtained from α-chloralose anesthetized rat brains under a wide range of physiologic conditions including both normothermia and hypothermia.
Materials and methods
All NMR experiments were conducted on a 9.4 T horizontal animal magnet (Magnex Scientific, Abingdon, UK) interfaced to a Varian INOVA console (Palo Alto, CA, USA). A multinuclear surface-coil probe consisting of a butterfly-shape 1H coil (400 MHz) and an oval-shape 17O coil (~1 cm × 2 cm, 54.25 MHz) was used for acquiring anatomic 1H images and
Reproducibility Study
Five male Sprague—Dawley rats (body weight: 234 ± 23 g) were used for studying the reproducibility of two repeated CMRO2 measurements in the same rat and experimental session. The rats were anesthetized with ~ 2% (v/v) isoflurane in a mixture of O2 and N2O gases (2:3) during surgery. After oral intubations, femoral artery and vein were catheterized for physiologic monitoring and/or blood sampling and chemical administration. Four of the five rats also underwent a surgical procedure for CBF measurements via a bolus injection of H217O directly into the brain through the internal carotid artery (see surgical details in Zhu et al, 2001). After surgery, anesthesia was switched to α-chloralose by a bolus injection (40 mg/kg) and following continuous infusion (~ 20 mg/kg/h) of α-chloralose solution. Blood gases were sampled for monitoring physiologic conditions (pH = 7.39 ± 0.05, pCO2 = 38 ± 6 mm Hg, pO2 = 148 ± 13 mm Hg,
For 17O2 inhalation experiments, the 17O-labeled O2 gas (68.3% enrichment, ISOTEC) was mixed with N2O gas (~ 2:3) and stored in a cylindrical gas reservoir. The 3D 17O MRSI acquisitions were performed with the normal gas mixture with nonlabeled O2 gas first; these natural abundance 3D 17O MRS images were used as references for quantifying metabolic H217O concentrations in the rat brain during and after 17O2 inhalation (Zhu et al, 2002). Then the respiration gas was quickly switched to the 17O2-labeled gas mixture while the continuous 17O MRSI acquisitions were not interfered. After ~ 2 min of 17O2 inhalation, the gas line was switched back to the normal gas mixture, and the 3D 17O MRSI acquisitions were continued for ~ 15 mins after the ceasing of 17O2 inhalation. The second 17O2 inhalation and CMRO2 measurement were performed in the same fashion within ~ 30 to 60 min after the completion of the first 17O2 inhalation. The CBF measurement was performed after the second 17O2 inhalation experiment via a bolus injection of H217O tracer (~0.05 to 0.1 mL, 50% enrichment, ISOTEC) into the brain via an internal carotid artery. The physiologic conditions of the animal during these measurements were kept as close as possible.
The acquisition time for each 3D 17O-MRSI data set was 11.5 secs with following parameters:
Normothermia and Hypothermia Comparison Study
Five male Sprague—Dawley rats (body weight: 283 ± 25 g) were used for this study. The general surgical procedures were the same as described in the previous section. In addition, three of the five rats underwent a surgical preparation for the CBF measurement; and two of these three rats were also implanted a vascular 17O RF coil, tuned at the 17O Larmar frequency and wrapped around the intact carotid artery, allowing simultaneous measurement of arterial input function of H217O (
Under stable normothermic condition (37°C), a short 17O2 gas (72.1% or 89.9% enrichment) inhalation (~ 2 min) was performed on each rat, followed by a CBF measurement via bolus injection of H217O tracer (~ 0.05 mL, 50% enrichment) if it was applicable. After these measurements, the water pump for the circulating water blanket were switched to refrigerant mode and allowing the animal temperature to gradually decrease. When the body temperature of the rat reached the desired mild hypothermic (32°C) condition, the water pump was switched back to the heating mode to maintain the temperature at 32°C. This cooling process took approximately one and half hours. When the animal reached a stable hypothermic condition, second 17O2 inhalation experiment was performed on each rat. Therefore, the averaged time interval between the two CMRO2 measurements for this study was longer than 2 h.
The parameters for acquiring 3D 17O MRSI data were varied slightly among the rats in this group but were kept same for each rat. The acquisition time for each image was 10 to 12 secs with
Comparison of Complete Model and Simplified Model for CMRO2 Calculation
The dynamic change in the cerebral H217O concentration during an 17O2 inhalation is determined by three parallel processes: (i) cerebral oxygen utilization for generating the metabolic H217O in the brain tissue, (ii) blood perfusion resulting in H217O washout from the brain, and (iii) blood flow recirculation bringing the metabolically generated H217O in the entire living body into the brain. The mass balance of the 17O-labeled H217O concentration in the brain tissue during the period of 17O2 gas inhalation can be derived as (Kety and Schmidt, 1948a; Zhang et al, 2004; Zhu et al, 2002)
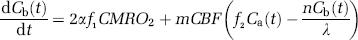
where
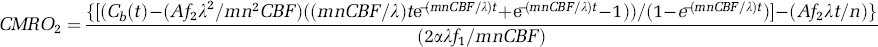
Therefore, the CMRO2 value at
In contrast, the invasive measurements could be eliminated by using the
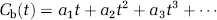
In this expansion, the first-order coefficient of
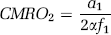
where α and
For the rats performed with multiple measurements of
Data and results are reported as means ± standard deviation (s.d.).
Results
In vivo 3D 17O MRSI of Rat Brain
Figure 1 demonstrates an example of 3D 17O MRS images (~ 11 secs data acquisition and 75 μL voxel size) of natural abundance H217O and corresponding 1H anatomic images from a representative rat brain acquired at 9.4 T. Five adjacent coronal slices covering the 17O RF coil-sensitive region of the rat brain are shown for both 17O and 1H images. The relative 17O RF coil size and position (cross-section) on each slice are depicted in the anatomic images. The nonuniform spatial distributions of 17O NMR signal in the 17O MRSI data are because of the inhomogeneous RF field (
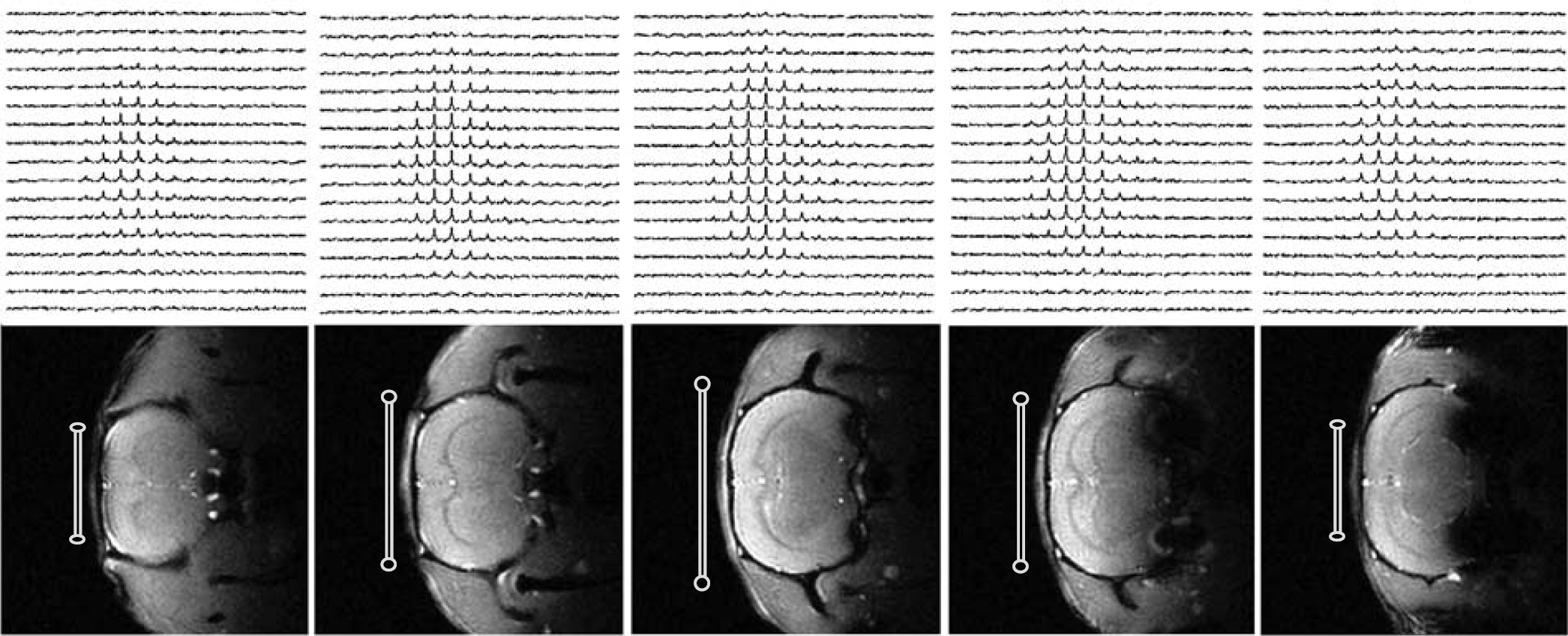
3D 17O MRSIs of natural abundance H217O (top row) and corresponding 1H anatomic images (bottom row) of rat brain at 9.4 T. The 17O RF surface coil positions and cross-sections are indicated in the images. The parameters for acquiring these 3D 17O MRSI data were:
Reproducibility Study of Repeated CMRO2 Measurements
Figure 2A shows the stacked plots of H217O spectra from a central voxel of 3D 17O MRS images acquired before, during, and after two consecutive 2-min 17O2 inhalations from a representative rat brain. The excellent sensitivity of 17O signal and the stability of magnetic resonance imaging scanner are clearly demonstrated in this figure, which are important for detecting the dynamic change of the metabolic H217O content in the rat brain during and after 17O2 inhalation. Figure 2B and Figure 2C are the linear fittings of the H217O concentration change in the same voxel during first and second 17O2 inhalations, which were used for calculating CMRO2 (Zhang et al, 2004; Zhu et al, 2002, 2005). The CMRO2 values in this particular voxel were almost identical between the first and second measurements (2.65 and 2.63 μmol/g/min) under the same physiologic condition.
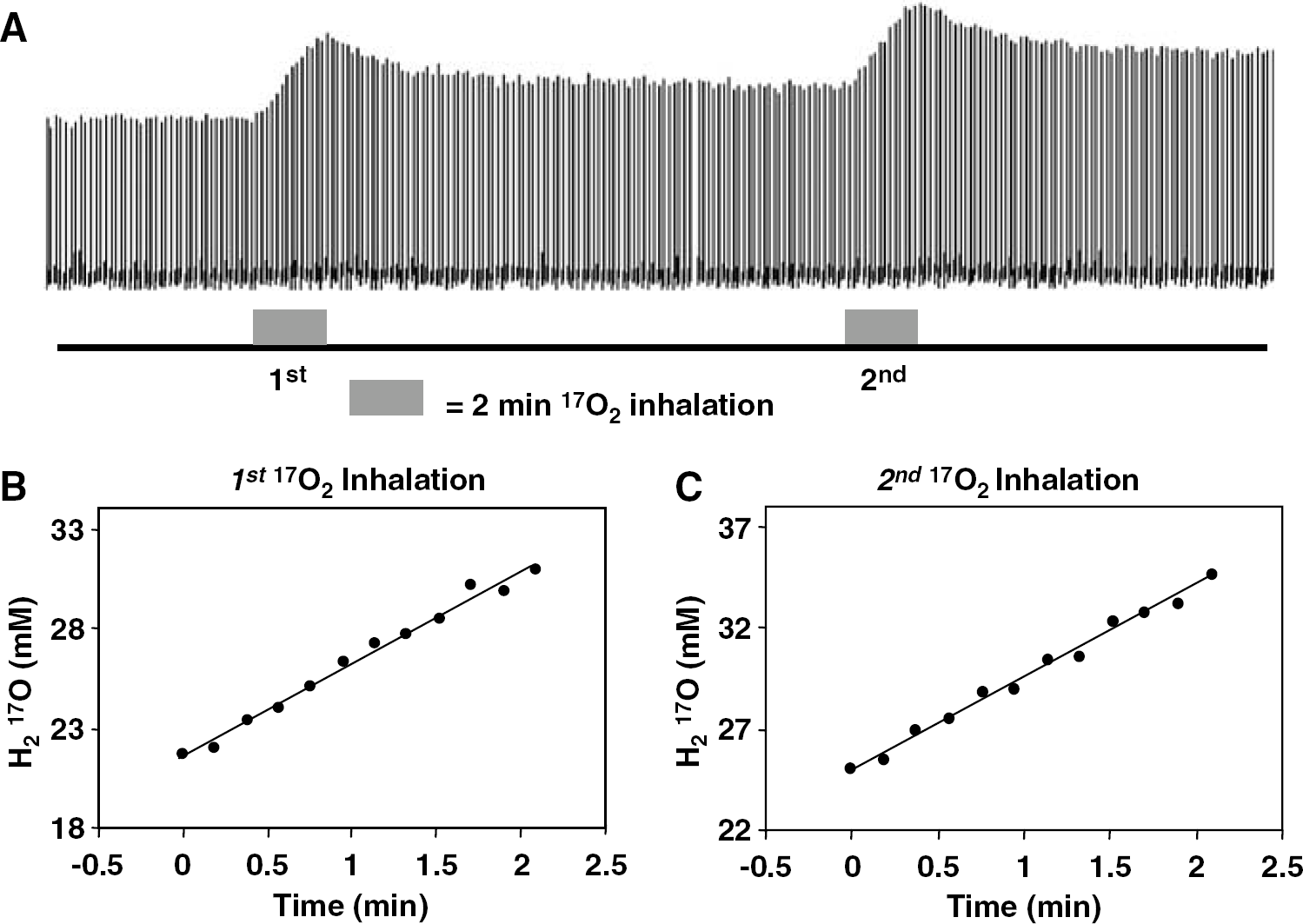
(
Figure 3 provides a voxel-by-voxel-based comparison of two CMRO2 measurements in a representative rat brain under the same physiologic condition. Total 305 voxels located in the 17O RF coil-sensitive regions (similar as shown in Figure 1) were used for the comparison. The ratios of the first measured CMRO2 to the second measured CMRO2 for each voxel were calculated. The linear correlation coefficient (
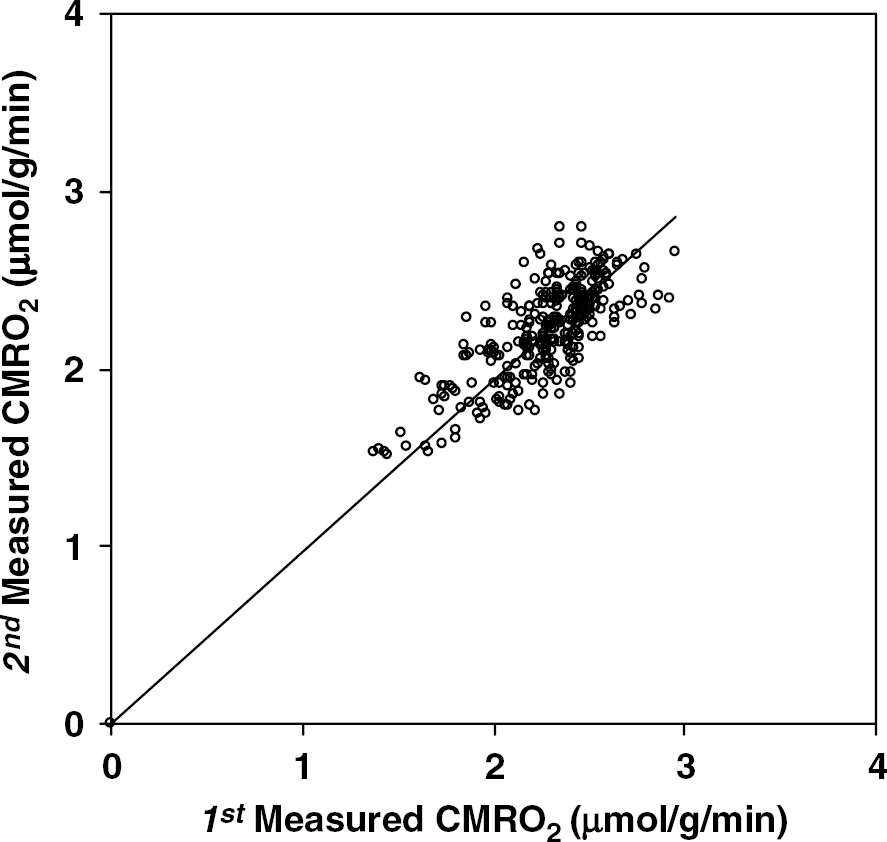
A voxel-by-voxel-based comparison of two consecutive 3D CMRO2 measurements in a representative rat brain. CMRO2 ratio (1st/2nd) = 1.02 ± 0.09 and correlation coefficient = 0.77 (total voxel number = 305).
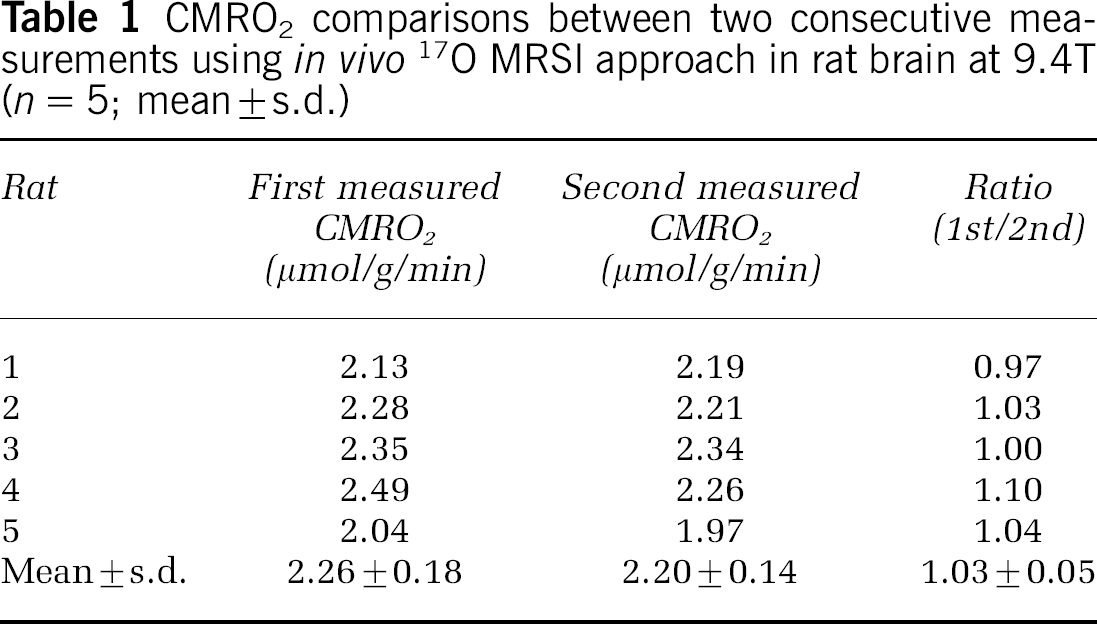
The same data analysis method was applied for each of the five rats that underwent reproducibility study. The results are summarized in Table 1. The mean ± s.d. values of the first and second measured CMRO2 values of these five rats were 2.26 ± 0.18 and 2.20 ± 0.14 μmol/gmin, respectively; and the CMRO2 ratio (1st/2nd) was 1.03 ± 0.05 (
CMRO2 comparisons between two consecutive measurements using
CMRO2 Measurements at Normothermia and Hypothermia Conditions
Physiological parameters of the rats under normothermia (37°C) and mild hypothermia (32°C) conditions are summarized in Table 2. While the blood gas data (pH, pCO2, and pO2) were similar for the rats at two brain temperatures, the mean arterial blood pressure and the heart rate of the animal decreased significantly under the hypothermia condition.
Physiological data of rats at normothermia and hypothermia condition (
Figure 4 illustrates the cerebral concentration changes of metabolic H217O in a single voxel taken from the 3D 17O MRS images as a function of inhalation time during the first 17O2 inhalation at 37°C and second 17O2 inhalation at 32°C, respectively. The accumulation rate of metabolic H217O during an 17O2 inhalation was significantly decreased under the hypothermia condition. The linear fittings to the two time courses of
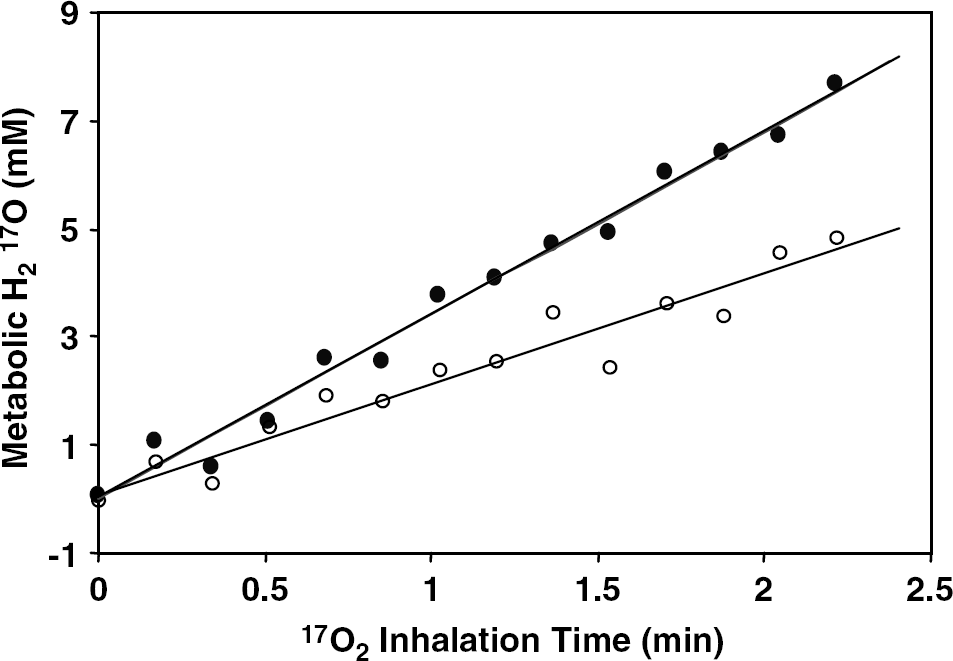
Dynamic changes of metabolic H217O concentration during 17O2 inhalation from a representative voxel (~ 105 μL, similar location as in Figure 2) in the rat brain at normal temperature (37°C, full circles) and during mild hypothermia (32°C, open circles). The linear fitting of the data was used for calculating CMRO2 values at two temperatures (= 1.9 μmol/g/min at 37°C and 1.1 μmol/g/min at 32°C).
We have demonstrated previously that under normal physiologic condition, CMRO2 calculations based on the
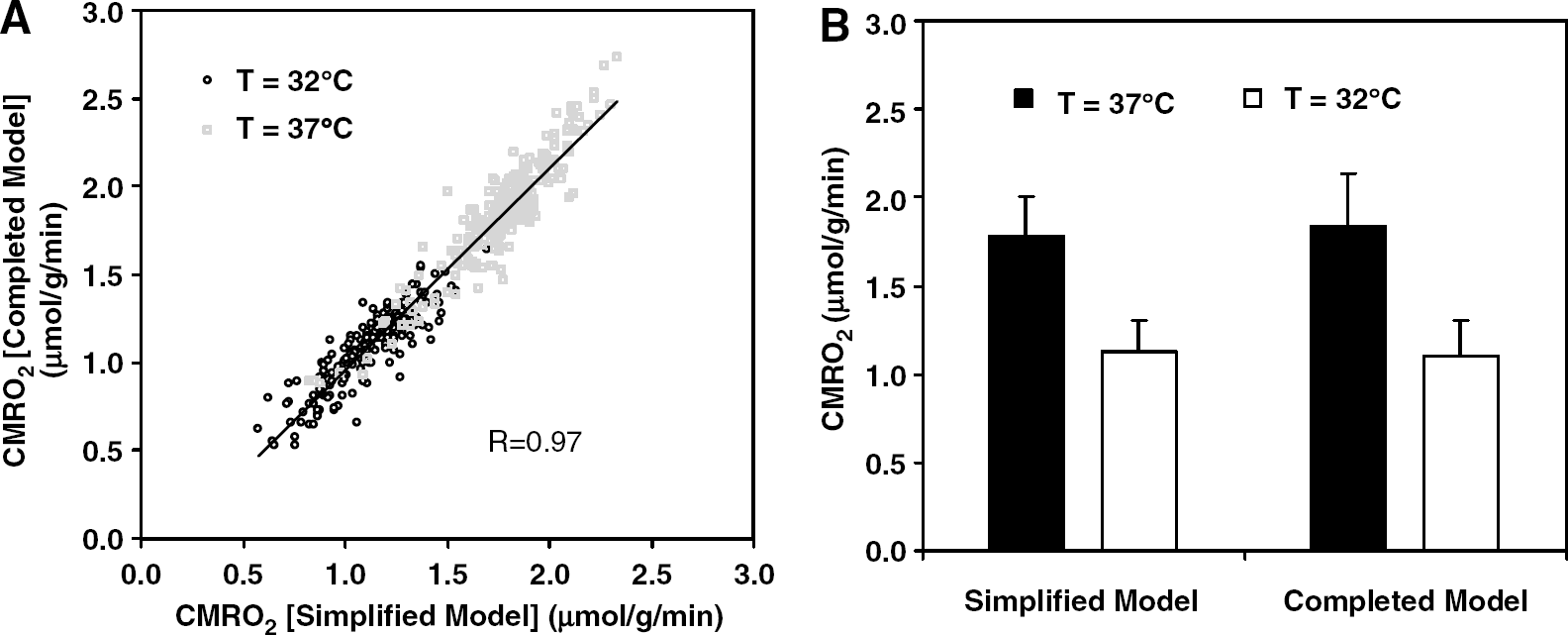
(
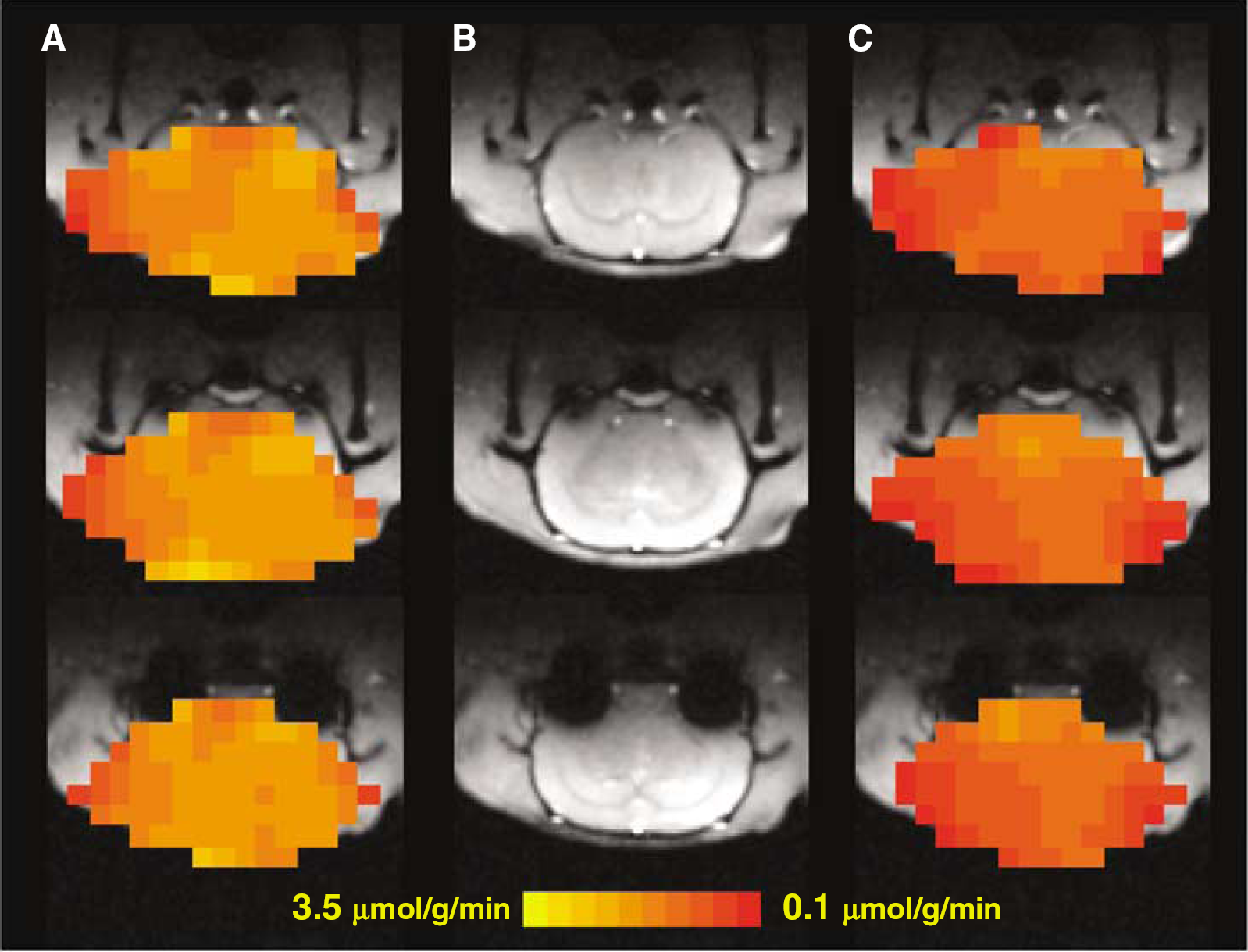
Three-dimensional CMRO2 maps of a representative rat brain at (
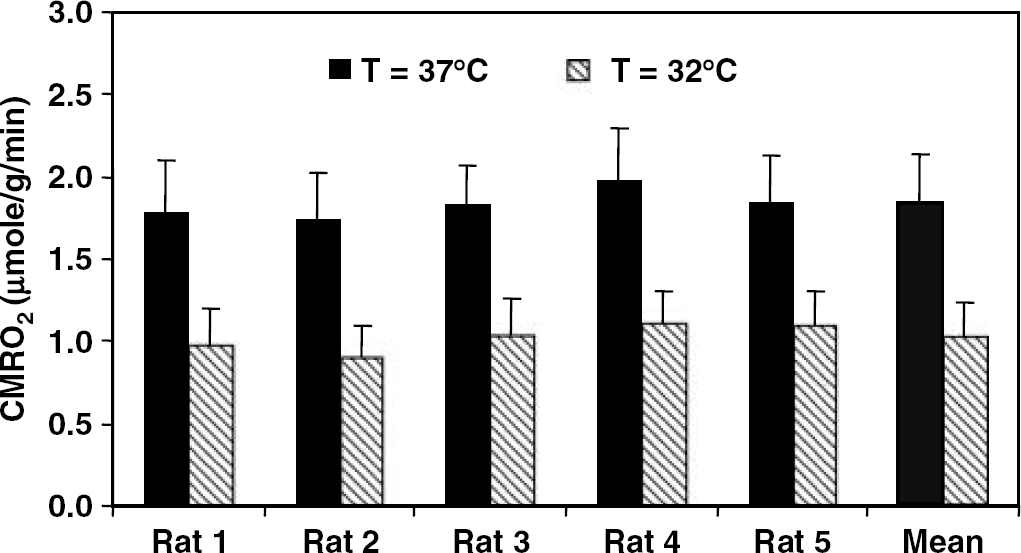
Measured CMRO2 values of individual rat calculated with the
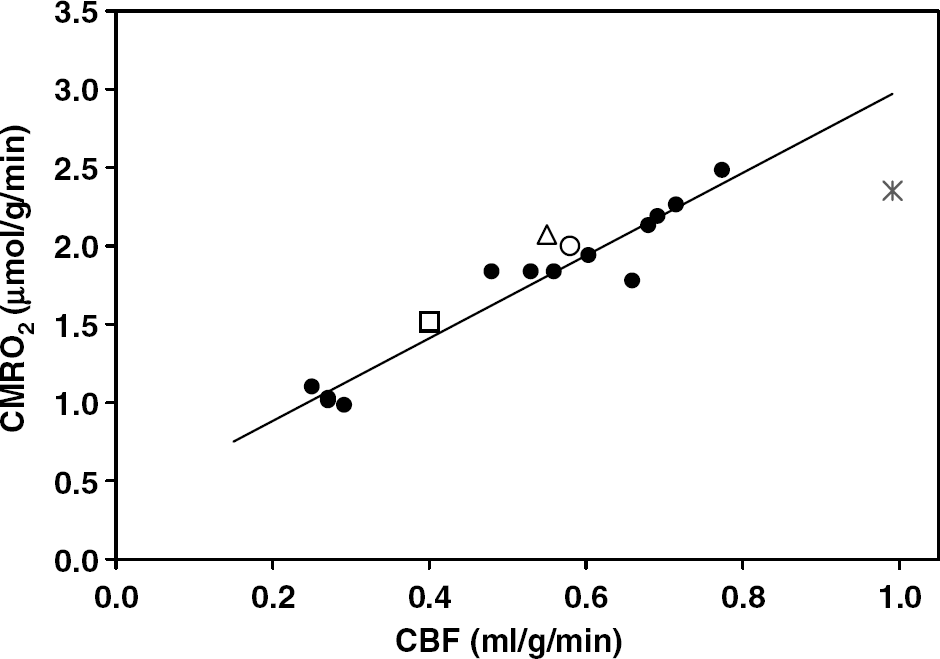
Correlation of CBF and CMRO2 values in the rat brains anesthetized with α-chloralose at brain temperature range of 32°C to 37°C (•). The linear correlation coefficient (
Correlation Between CMRO2 and CBF
In addition to mapping CMRO2 in the rat brain, the 17O MRSI approach was used to determine absolute CBF in a subgroup of rats in both reproducibility study (CBF = 0.72 ± 0.07 mL/g/min (
Discussion and conclusion
The superior 17O NMR sensitivity available at ultrahigh field makes it possible for detecting and imaging small dynamic changes of metabolic H217O during a brief inhalation of 17O2, and ultimately, for imaging CMRO2 in three dimensions in the rat brain (Zhu et al, 2001, 2002). In this study, we further extended our investigation to address one important aspect of 17O MRSI approach, that is, the reproducibility of CMRO2 measurements.
Our results show that in the rat brain, especially for those voxels with high SNR as illustrated in Figure 2, the reproducibility of two consecutive CMRO2 measurements is extreamly high. When considering all the voxels located in the 17O RF coil-sensitive region as shown in Figure 1, the majority of the voxels present highly reproducible CMRO2 values when voxel-by-voxel-based analysis was performed. This is evident from the ratio between two repeated CMRO2 measurements, which is closed to unity in most of the voxels inside the rat brain. Because these repeated measurements were performed in the animal under stable physiologic condition without any perturbation, therefore, we conclude that the
The feasibility of 3D 17O MRS imaging for determining CMRO2 changes because of physiologic perturbation was examined in the rat brain at normal brain temperature and under hypothermia condition. Our results show that in the same rat brain anesthetized with α-chloralose, a significant decrease of CMRO2 was observed across the entire brain when the brain temperature decreased from 37°C to 32°C. The measured CMRO2 ratio between normothermia and hypothermia conditions was 1.80 ± 0.10 (
In our previous work, we had demonstrated the feasibility to establish a completely noninvasive 17O NMR approach for imaging CMRO2 in the rat brain under normal physiology condition (Zhang et al, 2004). In an effort of extending the applicability of this noninvasive CMRO2 imaging approach to various physiologic conditions and/or in different species, we evaluated the validity of this method in the normothermic and hypothermic rat brains. Our results have clearly demonstrated that the CMRO2 calculation based on the
There are only few methods that are able to determine absolute CBF and CMRO2 values in a living brain without killing animal. Among them, the Kety—Schmidt method (Kety and Schmidt, 1948b) can provide global CMRO2 and CBF information and has led to much of our knowledge about the circulation and metabolism in animal and human brain under a variety of physiologic or pathologic conditions. This method is considered as the most robust and reliable way to determine CMRO2 and CBF values without information of their spatial distributions (see (Siesjo, 1978) and references therein). The positron emission tomography approach has been widely applied for imaging CMRO2 and CBF in humans (Mintun et al, 1984; Ter-Pogossian et al, 1970) by using radioactive 15O2 gas and H215O and C15O tracers. Positron emission tomography can provide information regarding the distribution of the CBF and CMRO2 values with limited spatial resolution and hence mainly applied for human brain studies. The NMR method capable of directly determining absolute CMRO2 and CBF values in the animal brain would be the 17O MRS approach alone (Pekar et al, 1991; Zhu et al, 2002, 2005) or in combination with other MR technique for CBF measurements (e.g. Arai et al, 1998; Pekar et al, 1995; Tailor et al, 2003). At ultrahigh fields such as 9.4 T, we were able to obtain 3D CMRO2 and CBF images of the rat brain with ~ 50 to 105 μL vexel size as demonstrated in this study. Furthermore, by comparing the CMRO2 and CBF values obtained from each rat brain at varied physiologic conditions, we were able to extrapolate the relation between CMRO2 and CBF in the α-chloralose anesthetized rat brain. Our data indicate that CMRO2 and CBF obey an approximate linear relationship (correlation coefficient:
In conclusion, the developed
Footnotes
Acknowledgements
We thank Run-Xia Tian for the assistance in animal preparation, John Strupp for the assistance in data analysis.
