Abstract
We aimed to demonstrate the feasibility of whole brain oxygen extraction fraction (OEF) mapping for measuring lesion specific and regional OEF abnormalities in multiple sclerosis (MS) patients. In 22 MS patients and 11 healthy controls (HC), OEF and neural tissue susceptibility (
Keywords
Introduction
Multiple sclerosis (MS) is an inflammatory demyelinating disease of the white matter (WM) with progressive neurodegeneration and is a leading cause of neurological disability in young adults. 1 MRI has been the standard in vivo imaging technique for diagnosing MS and monitoring disease progression.2,3 New acute MS lesions develop near the disrupted brain blood barrier (BBB) and appear hyperintense on Gadolinium-enhanced T1-weighted image (T1w+Gd)3,4 as a consequence of contrast agent leakage. Following BBB repair, lesions are characterized by a non-enhancing status on T1w+Gd; these lesions are typically regarded as chronic and appear hyperintense on T2-weighted fluid-attenuated inversion recovery (T2FLAIR) image. This is attributable to demyelination and axonal loss.2,3
Recently, an important subset of chronic MS lesions has been identified which exhibit low-grade sustained microglial activity and expanding demyelination at the lesion rim.5–7 These so-called chronic active lesions have been associated with increased tissue damage, neuroinflammation, and disability, 8 and may be a driving factor in progressive disease.9,10 Quantitative susceptibility mapping (QSM) 11 has been used to detect these lesions based on their characteristic paramagnetic rim appearance (QSM rim+) owing to its sensitivity to the iron-rich pro-inflammatory microglia and macrophages in the rim region.9,12–18 However, the metabolic activity of these immune cells has yet to be measured with current imaging techniques such as PET (due to limited spatial resolution) and conventional MRI (due to lack of sensitivity).
The primary objective of this study was to demonstrate the feasibility of whole brain mapping of oxygen extraction fraction (OEF) in MS patients using a widely available multi-echo gradient echo (mGRE) sequence and a novel integrated model of QSM phase signal and quantitative blood oxygenation level dependent magnitude signal (qBOLD) (QSM+qBOLD, or QQ).19,20 QQ separates the effect of deoxyheme iron in a cylindrical micron-scale venule (OEF effect) from that of neural tissue susceptibility (
In our study, we aim to demonstrate the potential of QQ to provide a quantitative marker of oxygen metabolism in the rim area of QSM rim+ lesions, which may be useful in the development of new therapeutic targets to reduce inflammation in chronic active lesions. As a secondary objective, QQ-based OEF mapping is used to investigate regional oxygen metabolism differences in cortical gray matter (CGM) and deep gray matter (DGM) between MS patients and healthy controls (HCs), which have been implicated in neurodegenerative processes and are regarded as major indicators of tissue injury and neuronal loss in MS.30–36
Materials and methods
Study cohort
This retrospective image analysis study was approved by the Weill Cornell Medicine local Institutional Review Board, and written informed consent was obtained from all individuals in accordance with the ethical standards of the Helsinki Declaration of 1975 and its later amendments. MS patients were selected from an ongoing imaging and clinical MS research database and were diagnosed according to the McDonald criteria. 3 A total of 22 relapsing-remitting MS patients with at least one chronic active QSM rim+ lesion was included (17 females, 5 males; mean age, 37 ± 6 years). In addition, 11 age-matched HCs (1 female, 10 males; mean age, 34 ± 12 years) were included for comparison. The demographic and clinical characteristics of the study participants, e.g. expanded disability status scale (EDSS) to evaluate the functional systems of the central nervous system (0 = normal neurologic status, 10 = death due to MS), 37 are summarized in Table 1.
Clinical and demographic characteristics of the study cohort. Age, disease duration, and expanded disability status scale (EDSS) score are shown as mean ± standard deviation (minimum-maximum).
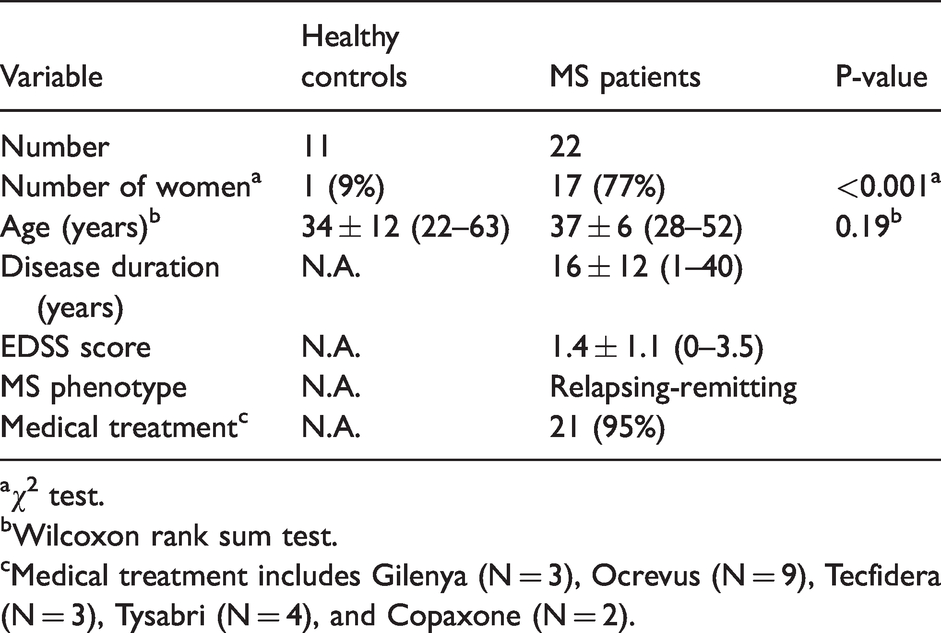
aχ2 test.
bWilcoxon rank sum test.
cMedical treatment includes Gilenya (N = 3), Ocrevus (N = 9), Tecfidera (N = 3), Tysabri (N = 4), and Copaxone (N = 2).
MRI protocol
All brain MRIs were performed on a 3 T GE scanner. The typical MS imaging protocol consisted of 3 D sagittal T1w and T1w+Gd (field of view (FOV) = 24 cm, TR = 8.8 ms, TE = 3.4 ms, TI = 450 ms, flip angle (FA) = 15°, voxel size = 1.2 × 1.2 × 1.2 mm3), 2 D axial T2w (FOV = 28 cm, TR = 5917 ms, TE = 88 ms, flip FA = 90°, voxel size = 0.7 × 1.2 × 3 mm3), and 3 D T2FLAIR (FOV = 24 cm, TR = 6050 ms, TE = 149.4 ms, TI = 1820 ms, FA = 90°, voxel size = 1.2 × 1.2 × 1.2 mm3) for anatomy and lesion detection, as well as 3 D mGRE for QSM and OEF mapping (FOV = 24 cm, TR = 57.5 ms, TE1/ΔTE = 4.5/4.8 ms, number of TEs = 11, FA = 20°, voxel size = 0.7 × 0.9 × 3 mm3, axial slice acquisition parallel to B0 direction).
The imaging protocol for HCs consisted of axial 3 D mGRE (FOV = 20 cm, TR = 30.5 ms, TE1/ΔTE = 2.3/3.9 ms, number of TEs = 7, FA = 15°, voxel size = 1.2 × 1.2 × 1.2 mm3, flow-compensated in all three directions, 38 axial slice acquisition parallel to B0 direction) and 3 D T1w (FOV = 20 cm, TR = 7.7 ms, TE = 2.9 ms, TI = 450 ms, FA = 15°, voxel size = 0.8 × 0.8 × 1.2 mm3).
Image processing and analysis
QSM reconstruction consisted of total field estimation from the mGRE phase data by linear fitting, 39 background field removal using Projection onto Dipole Fields (PDF) algorithm, 40 and inversion of the local field to obtain the susceptibility distribution using Morphology Enabled Dipole Inversion with automatic uniform cerebrospinal fluid zero reference (MEDI + 0) algorithm.11,41–43 In HCs, the total field was estimated using an adaptive quadratic-fit of the mGRE phase to benefit from 3 D flow-compensation. 38
OEF and neural tissue susceptibility (
To improve the robustness of QQ against noise in the data fitting, the original cluster analysis of time evolution (CAT) algorithm 20 was refined by incorporating a brain/CSF segmentation into clustering.48,49 This segmentation was obtained from the echo-combined magnitude mGRE image using the FSL FAST segmentation algorithm. 50 The CAT method groups voxels of the same tissue type with similar temporal mGRE magnitude evolutions into a cluster that are assumed to have similar model parameter values including OEF. Then, cluster-wise optimization is performed to solve for the model parameters. Total variation denoising 51 was applied to enhance the effective signal-to-noise ratio of OEF reconstruction.
For MS lesion-specific OEF analysis, two neuroradiologists (S.Z., 6 years of experience and W.H., 11 years of experience) identified lesions on T2FLAIR and manually traced QSM rim+ lesions on QSM based on their relative contrast with respect to the adjacent normal appearing white matter (NAWM).12,13 To exclude both regional variation within a subject and inter-subject variation, the average OEF within each lesion was referenced to the average of its adjacent NAWM, OEF contrast (OEFC) =
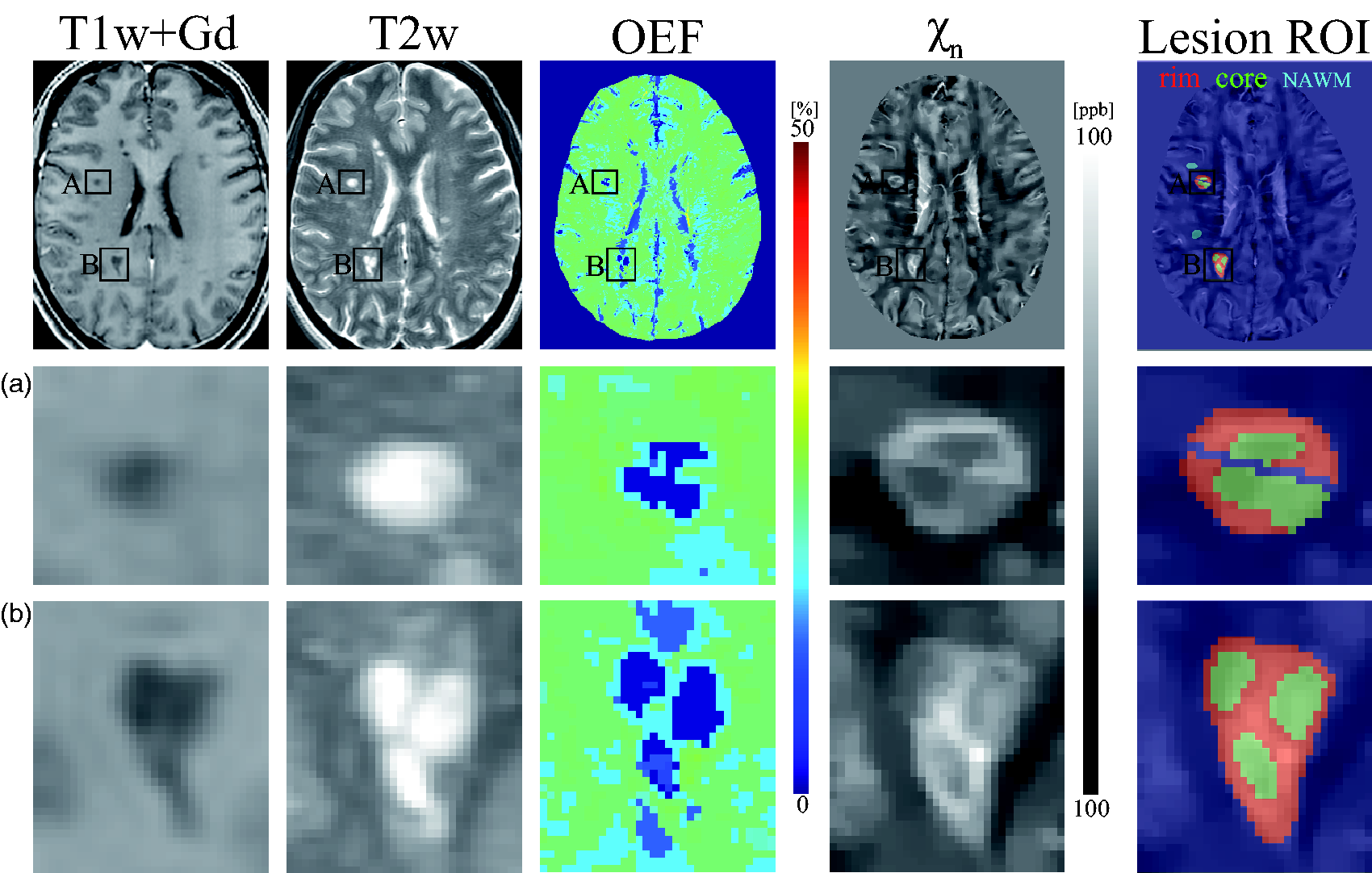
Example of T1w+Gd image, T2w image, OEF and neural tissue susceptibility (
Statistical analysis
For QSM rim+ lesions, a linear mixed-effect model with a random effect for patient was used to assess the OEFC between the rim and core regions (core = 0, rim =1), adjusting for lesion volume. The random effect for patient accounts for the correlation within a patient. For OEF comparison between HC and MS, linear regression models were fit for each ROI (whole brain, CGM, and DGM), with average OEF in the region as the dependent variable and an indicator of MS versus HC (0 = HC, 1 = MS), gender (female = 0, male = 1), and age as independent variables. Multiple comparisons correction was performed using the false discovery rate.
55
The Jarque-Bera test confirmed the normality of each ROI data distribution (all p-values > 0.09).
56
A p-value of less than 0.05 was considered statistically significant. The same analyses were performed for
Results
In MS patients, a total of 80 chronic QSM rim+ lesions were identified. Of these, the mean lesion volume was 440.0 ± 431.0 mm3, and the mean volume of rim and core regions was 267.0 ± 267.4 mm3 and 172.9 ± 185.4 mm3, respectively.
Figure 1 shows a representative case with two QSM rim+ lesions, showing higher OEF and
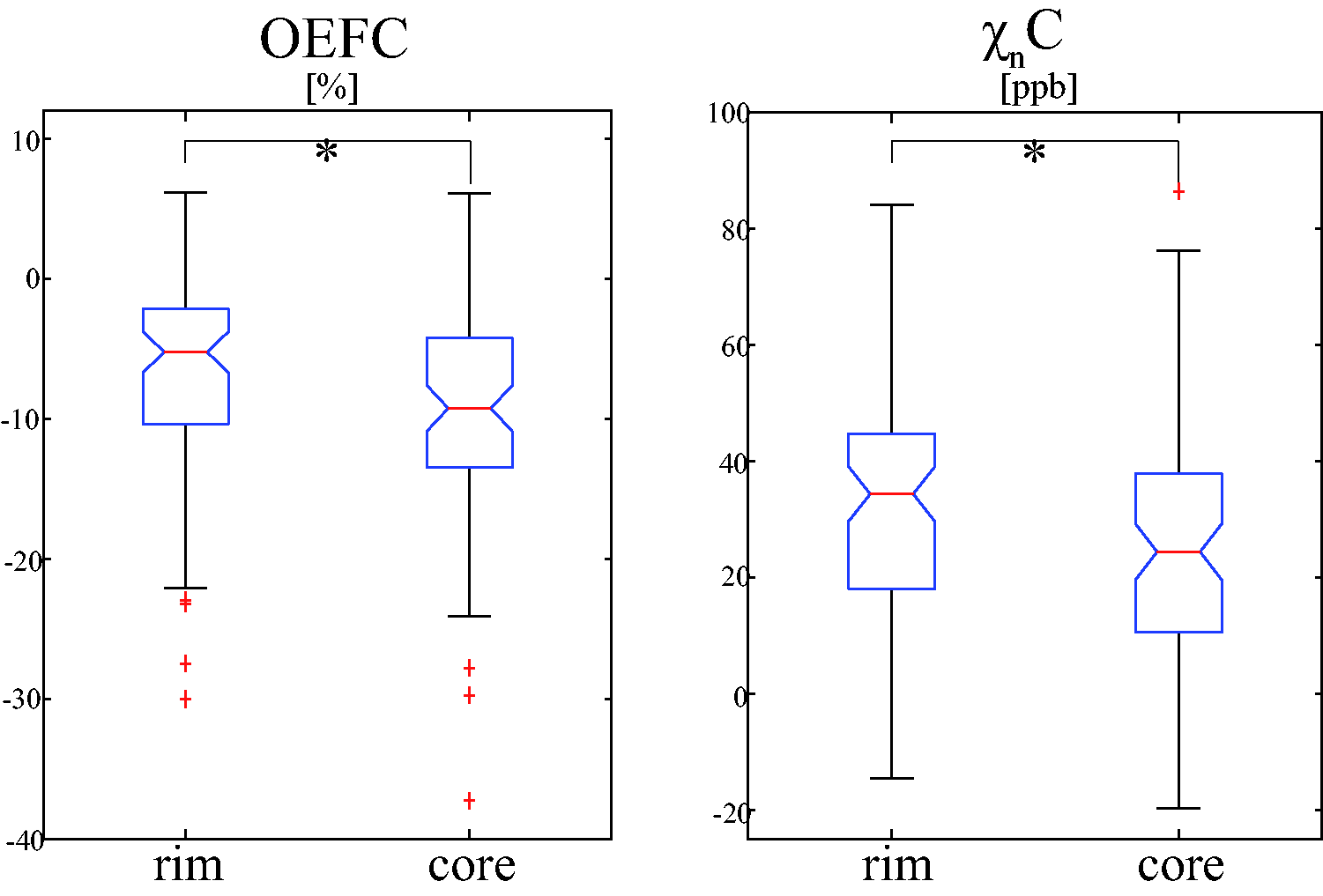
Comparison of mean OEF and neural tissue susceptibility (
The lesion volume remained a significant covariate for OEFC (
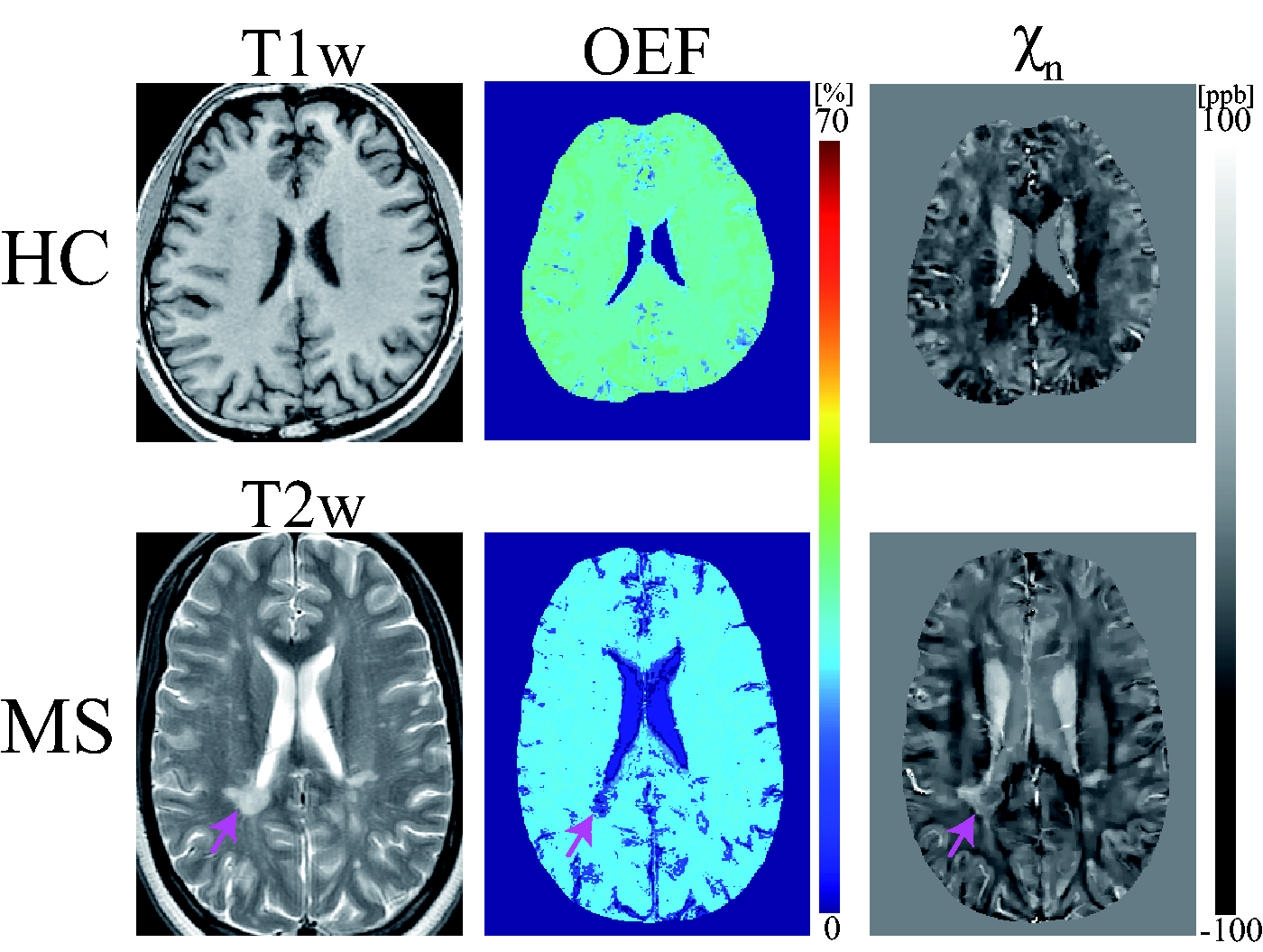
Exemplary OEF and neural tissue susceptibility (
Both HCs and MS patients generally show uniform OEF maps except for lesions and GM/WM contrast in
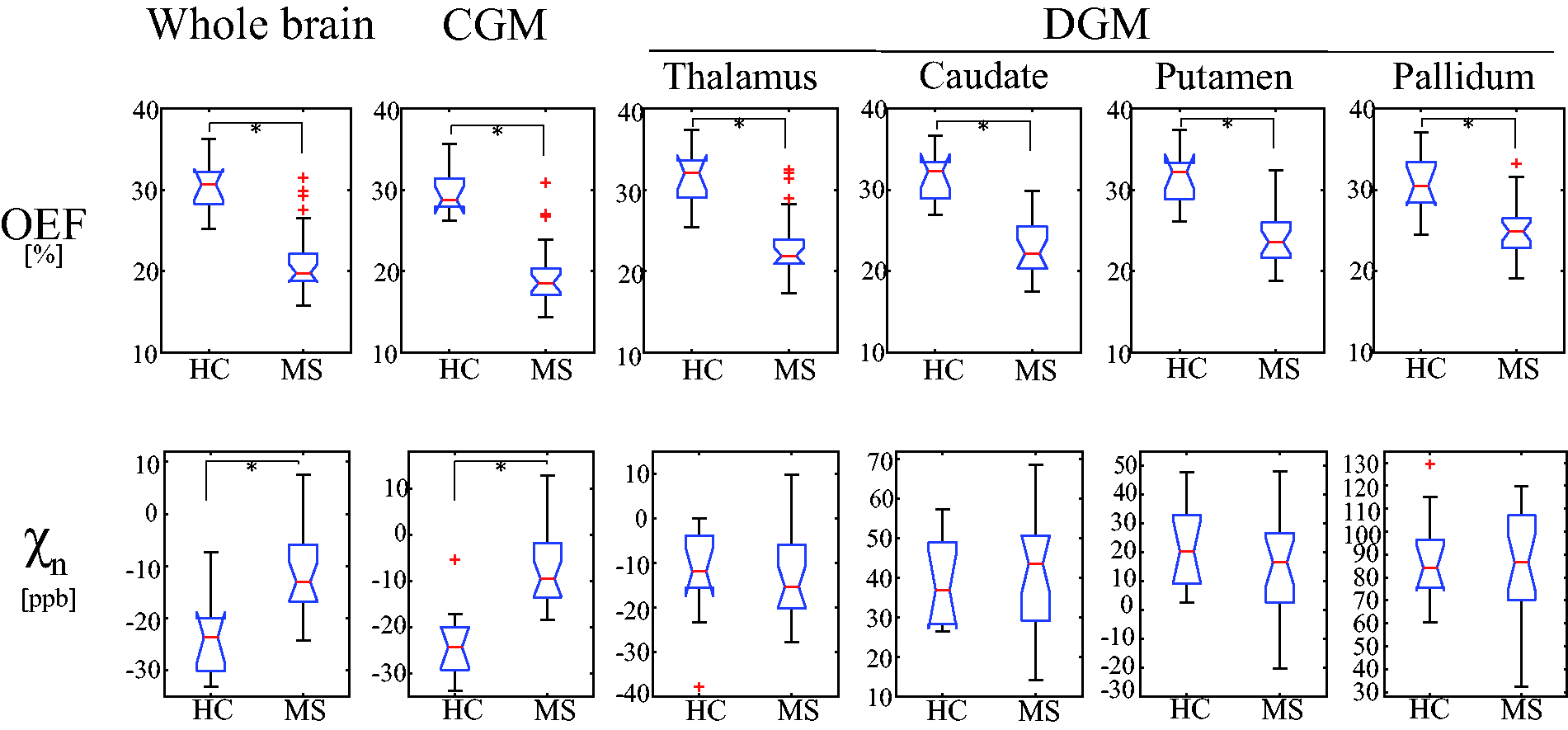
Comparison of average OEF and neural tissue susceptibility (
MS patients showed significantly higher
Discussion
Our feasibility study demonstrated that high-resolution OEF maps can be extracted from mGRE data using the QQ algorithm for the study of global and regional tissue oxygen utilization in MS brains. We showed for the first time an OEF increase in the rim region compared to the core region in chronic active MS lesions with paramagnetic rim appearance. As QSM has become a reliable imaging tool for measuring susceptibility changes in the brain, the ability to further separate the contributions of blood deoxyheme iron and neural tissue to tissue susceptibility by the QQ algorithm has the potential to provide a more comprehensive picture of both tissue oxygenation status and change in tissue iron and/or myelin content in chronic active MS lesion.9,12–17
In QSM rim+ lesions, the rim region was found to have significantly higher mean OEF and
In both HCs and MS patients, brain OEF maps generally appear uniform except in lesions (Figure 3). This observation is consistent with OEF maps that were obtained by the reference standard 15O PET,58,59 which supports the presence of an equilibrium between metabolic needs and blood flow in the resting state. 59 The observed lower global OEF obtained by the QQ algorithm in MS brains as compared to HCs (Figures 3 and 4) agrees well with the previously reported reduced OEF in MRI,34,35,60 near-infrared spectroscopy, 60 and PET studies. 61 Lower OEF may represent a unique tissue injury mechanism underlying progressive demyelination and neurodegeneration in the MS brain. Several studies have indicated that impaired mitochondrial function via nitric oxide overproduction likely induces chronic oxygen deprivation in neuronal cells, which may result in neuronal dysfunction.62–65
Compared to a previous MRI-based OEF study using vein susceptibility modeling, 35 cortical OEF values in our study were similar for HCs, 29.9 ± 2.9 vs. 31.5 ± 3.0%, but lower for MS patients, 19.9 ± 4.3 vs. 28.1 ± 3.0%, which may be explained by the longer disease duration of our cohort (16 vs. 8.5 years). 35 Significantly lower OEF in frontal, temporal, parietal, and occipital CGM in MS, compared to HC (Supporting Information Figure S2), agrees with lower OEF observed in regional cortices, e.g. sensorimotor, parietal, and prefrontal cortex, with using the vein susceptibility modeling. 35 Furthermore, our global OEF was lower for both HCs (30.4 ± 3.3 vs. 38.6 ± 4.1%) and patients (21.1 ± 4.4 vs. 32.8 ± 5.2%) compared to a study using venous T2 modeling. 34 This discrepancy may be explained by the complexity in estimating oxygenation from T2 modeling. For instance, performance of T2 oxygenation modeling is strongly dependent on accurate calibration of its model parameters. 66
In DGM, MS patients have lower OEF values than HCs (Figure 4), which may be associated with tissue loss in those regions in MS brains.30,32,67 For instance, the lower OEF in the thalamus, a central link for cortical-subcortical circuits, 68 may be related to thalamic neuronal loss 33 and atrophy, 69 which are predictors of long-term disability progression in MS. 70
QSM sensitivity to tissue iron71–73 has been applied to study MS brain tissue.57,74 The susceptibility values of several deep gray nuclei (e.g. caudate and globus pallidus) in MS brains have been reported to be higher compared to HC.67,74 This study furthers our understanding of QSM as it separates QSM into the contribution of oxygen metabolism (OEF effect) and neural tissue (
Both HC and MS showed generally higher
This study has several limitations. First, the patient cohort is small and limited to relapsing-remitting MS with low EDSS. For instance, OEF was not significantly correlated with disease duration in all the ROIs (uncorrected p-values > 0.538, Spearman correlation analysis) (Supporting Information Figure S3) unlike a previously reported negative correlation. 35 This may be caused by a small number of patient cohort. Also, no significant correlation was found between OEF and EDSS in this study (Supporting Information Figure S3), whereas cortical OEF negatively correlated with EDSS in the previous venous OEF study. 35 This may be caused by a limited and low range of EDSS in this study (0-4).
Second, HC subjects had a different gender ratio from MS subjects which may affect the comparison result between HC and MS. To address the lack of female subjects in the HC group, we performed additional analysis to compare OEF only in the male subjects between HC (n = 10) and MS (n = 5) groups adjusting for age. We found that the MS group still showed significantly lower whole brain OEF than the HC group: 30.9% ± 3.2% vs. 18.7 ± 1.3% (
Third, QQ has some modeling assumptions: the random cylinder orientation of the venous microvasculature in qBOLD of QQ may not be valid in MS lesions and therefore induce bias in OEF estimations. Although intracranial and extracranial vascular abnormalities have not been found in MS compared to HC,81–83 microvascular abnormalities should be further investigated and potentially incorporated into QQ. Also, QQ does not consider detailed microstructure of the brain tissue including myelin, which may lead to a bias in OEF estimation. This may be a confounding factor in the comparison between MS and HC due to the demyelination and axonal loss in MS. QQ assumes two source compartments in a voxel, deoxyhemoglobin in venous structure and medium, such as diffusively distributed non-blood tissue ferratin or protein. If other structured strong sources including myelin in WM exists, additional introvoxel field variation would be induced, which may lead to greater MRI signal decay than the sole deoxyhemoglobin effect of venous blood. Consequently, QQ-based OEF may be overestimated in WM. However, in a validation study of QQ against the reference standard 15O-PET, the OEF difference between QQ and PET was not significant in WM. 29 This may suggest that the myelin effect in QQ-based OEF estimation may not be significant.
Fourth, vein voxels (blood only) were treated the same way as brain voxels (mixture of neural tissue and blood), which could influence the accuracy of OEF and
Fifth, different scan protocols between HC and MS patients, e.g. longer TE range, no flow compensation, and lower slice resolution in MS, may affect the OEF comparison between HC and MS patients. Additional OEF processing was performed to investigate the effects of scan parameters on OEF comparison. When MS data were processed using a similar TE range (TE1/ΔTE/TE6 = 4.7/4.8/28.5 ms) compared to that of HC (TE1/ΔTE/TE7 = 2.3/3.9/25.7 ms), we found lower OEF values in MS than HC, e.g. 23.0 ± 3.2% vs. 30.4 ± 3.5% for whole brain average OEF, in good agreement with the original findings (Supporting Information Figure S4). Lack of flow compensation may cause large veins with high blood velocity to shift due to mis-encoding. 84 In this study, this problem was alleviated by manually excluding the central veins from the lesion ROI (identified and segmented by neuroradiologists experienced with brain QSM, S.Z., W.H, and I.K) from the MS lesion ROI. For capillary level vessels, flow-related issue is not expected as they exhibit very slow blood flow of less than 1 mm/sec. Spatial resolution, which scales linearly with SNR, could have an impact on OEF quantification. To investigate this issue, a numerical simulation was performed to compare OEF obtained at SNR =1000, 100, and 50 using CAT algorithm used in this study, and negligible difference in mean OEF value was found. 20 The CAT may alleviate the OEF dependency on SNR by substantially improving the effective SNR through signal averaging in clusters. 20
Sixth, the medication effect has not been considered in the statistical analysis due to the limited sample size and variation of treatment drugs.
Seventh, though the comparison of venous oxygenation in lesion draining veins and its corresponding normal veins would be valuable to study the source of decreased OEF in the lesion core, it is difficult to perform because it is hard to specify the exact draining veins and venous oxygenation estimation may be biased due to partial volume effect. For instance, in an additional comparison, a neuroradiologist (I.K., 23 years of experience) identified central veins in 17 out of 80 lesions with thickness of 1∼2 voxels and its corresponding normal veins on contralateral side. Then, venous oxygenation in the central veins was calculated using mean QSM values
85
and compared with that in the normal veins. The venous oxygenation was similar and not significantly different between the lesion central veins and its corresponding normal veins: 88.9 ± 1.5% vs. 89.1 ± 1.1% (
Finally, this study used a cross-sectional design to investigate chronic active rim lesions. We plan to perform longitudinal studies in our future work.
In conclusion, brain OEF mapping is feasible in MS brains and can detect within-lesion difference between the rim and core regions of MS lesions with QSM hyperintense rim as well as global and regional gray matter differences between MS and HC. OEF maps may provide useful information on tissue oxygenation and viability for MS monitoring and treatment.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X211048031 - Supplemental material for Brain oxygen extraction fraction mapping in patients with multiple sclerosis
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X211048031 for Brain oxygen extraction fraction mapping in patients with multiple sclerosis by Junghun Cho, Thanh D Nguyen, Weiyuan Huang, Elizabeth M Sweeney, Xianfu Luo, Ilhami Kovanlikaya, Shun Zhang, Kelly M Gillen, Pascal Spincemaille, Ajay Gupta, Susan A Gauthier and Yi Wang in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported in part by NIH K99 NS123229, R01 NS090464, R01 NS104283, R01 NS105144, S10 OD021782, and NMSS RR-1602-07671.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contribution
All authors (JC, TN, WH, ES, XL, IK, SZ, KG, PS, AG, SG, and YW) made substantial contribution to the concept and design, data acquisition, or data analysis and interpretation. All authors assisted in drafting or revising the article. Final approval of the submitted manuscript was provided by all authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
