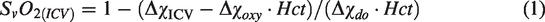Abstract
Studying the relationship between cerebral oxygen utilization and cognitive impairment is essential to understanding neuronal functional changes in the disease progression of multiple sclerosis (MS). This study explores the potential of using venous susceptibility in internal cerebral veins (ICVs) as an imaging biomarker for cognitive impairment in relapsing-remitting MS (RRMS) patients. Quantitative susceptibility mapping derived from fully flow-compensated MRI phase data was employed to directly measure venous blood oxygen saturation levels (SvO2) in the ICVs. Results revealed a significant reduction in the susceptibility of ICVs (212.4 ± 30.8 ppb vs 239.4 ± 25.9 ppb) and a significant increase of SvO2 (74.5 ± 1.89% vs 72.4 ± 2.23%) in patients with RRMS compared with age- and sex-matched healthy controls. Both the susceptibility of ICVs (r = 0.508, p = 0.031) and the SvO2 (r = −0.498, p = 0.036) exhibited a moderate correlation with cognitive decline in these patients assessed by the Paced Auditory Serial Addition Test, while no significant correlation was observed with clinical disability measured by the Expanded Disability Status Scale. The findings suggest that venous susceptibility in ICVs has the potential to serve as a specific indicator of oxygen metabolism and cognitive function in RRMS.
Keywords
Introduction
Multiple sclerosis (MS) is an inflammatory and neurodegenerative disease characterized by neural tissue atrophy and multifocal inflammatory and demyelinating lesions in the central nervous system. 1 The cause and course of MS lesions are not fully elucidated, with neurodegeneration emerging as the primary driving force behind both disease progression and cognitive decline. 2 Several hypotheses underlie progressive neurodegeneration in MS. One explanation revolves around energy deficiency in lesions, termed virtual (also known as metabolic or histotoxic) hypoxia. 3 In contrast to “true” hypoxia, virtual hypoxia does not necessarily involve reduced oxygen availability; rather, it is characterized by diminished oxygen utilization with mitochondrial dysfunction. Virtual hypoxia likely plays a significant role in the pathophysiology of neurodegeneration in MS. 4 This effect is not just localized to lesions; reduced oxygen extraction fraction (OEF), a marker of cell degeneration or dysfunction, has been described in normal-appearing tissue, including in deep and cortical gray matter of MS patients.5,6
Furthermore, a reduced cerebral metabolic rate of oxygen (CMRO2), a more complete measure of cerebral oxygen metabolism proportional to the product of OEF and cerebral blood flow (CBF), has long been related to symptomatology of MS, particularly cognitive impairment.6 –9 Cognitive impairment in MS can arise from decreased oxygen metabolism manifesting either as direct damage to brain tissue or as a decline in neuronal/axonal activity.10 –12 Specifically, cognitive dysfunction is linked to damage in deep gray matter nuclei,11,13,14 with the thalamus being identified as the primary contributor.15,16 Therefore, measurements of OEF in deep cerebral regions could serve as a specific imaging biomarker of cognitive decline in MS.
Studies on cerebral oxygen metabolism have traditionally relied on positron emission tomography (PET), the gold-standard, for assessment of CMRO2 in MS.7,9,17 However, PET is disadvantaged clinically by exposure to radiation and relatively limited availability. Recently, non-invasive MRI-based methods have emerged as a major focus for the quantification of OEF and CMRO2.18 –20 Various techniques measuring cerebral OEF have been proposed, including phase or quantitative susceptibility mapping (QSM) based methods21 –23 and a T2-relaxation-under-spin-tagging (TRUST) method 24 for measuring macrovascular OEF; respiratory challenge calibrated blood-oxygen-level-dependent (BOLD) methods25 –27; calibration-free quantitative BOLD based methods28 –30; QSM-based voxel-wise OEF mappings31 –33; as well as methods combining quantitative BOLD and QSM. 34 – 36 Ge et al. observed a significantly decreased OEF in MS patients measured in the superior sagittal sinus (SSS) using the TRUST method. 8 The reduction of OEF was correlated with disease severity and total lesion load. 8 Fan et al. also reported a reduction of cortical OEF in MS, using a phase-based method measuring pial veins parallel to the main field. 6 More recently, Cho et al. utilized a voxel-wise OEF method combining QSM and quantitative BOLD, observing a whole brain region reduced OEF in MS patients. 5 Common among these various methods is the use of venous blood susceptibility as a direct measure of venous oxygen saturation levels (SvO2) and OEF of the veins draining the cerebral tissue. QSM also eliminates the orientation-based errors in phase-based methods, which can accurately measure susceptibility predominantly for veins parallel to the main field. 37 While gradient echo data needed for QSM-based OEF can be acquired on most clinical scanners, the post-processing needed to compute QSM-based OEF from this gradient echo data is not and requires additional software tools.37,38 To date, it remains largely unknown whether the susceptibility of cerebral veins can be used as an imaging biomarker of neuronal activity in MS. Deep cerebral veins such as internal cerebral veins (ICVs) drain the periventricular white matter (WM) where most MS lesions including the slowly expanding lesions occur, 39 as well as subcortical grey matter nuclei which are associated with cognitive function. 11
The objective of this study was to investigate the susceptibility of ICVs and the subsequent SvO2 in patients with relapsing-remitting MS (RRMS), and their underlying correlations with a clinical measure of cognitive function and disease severity.
Material and methods
Subjects and clinical assessments
Patients with RRMS and age-matched healthy controls (HC) were recruited from the Neurology clinic and approved by the local institutional review board at Wayne State University following the Declaration of Helsinki. Written consent was obtained from each participant. The inclusion and exclusion criteria were described in a previous publication using the quantitative MRI data acquired from the same cohorts. 40 A neurologist performed clinical assessment for MS patients, including the Expanded Disability Status Scale (EDSS) for disease severity 41 and the Paced Auditory Serial Addition Test (PASAT) for cognitive function. 42
Data acquisition and processing
The QSM data presented in this paper were acquired with an interleaved flow-rephased and flow-dephased gradient echo sequence. 43 The data used in this retrospective study was not published previously. The imaging parameters were as follows: TR = 20 ms, flip angle = 12°, number of slices = 80, slice thickness =2 mm, field of view = 256 × 192 mm2, matrix size =384 × 144, leading to an acquisition resolution of 0.67 × 1.34 × 2.0 mm3, two echoes of 12.5 ms acquired with interleaved TRs, and a GRAPPA factor of 2 in the phase encoding direction. The scan time was 4 min 53 sec. The interleaved sequence had a fully flow-compensated echo of 12.5 ms for a bright blood image, and a flow-dephased echo of 12.5 ms given by a VENC value of 1.84 cm/s for a dark blood image (Figure 1(a) and (b)). The subtraction of these two magnitude images yielded an MR angiogram and venogram (MRAV) with no background tissue remaining (Figure 1(c)). 43 The phase of the flow-compensated echo was used for the reconstruction of the QSM data as described (Figure 1(d)). 43 In short, a quality-guided 3D best path algorithm, 3DSRNCP, 44 was used for phase unwrapping followed by background field removal using the SHARP algorithm with a kernel size of 8 pixels and a deconvolution threshold of 0.05. 45 Then, a truncated k-space division (TKD) was used to solve the inverse problem and to generate the susceptibility map. 21 Finally, an iterative algorithm with 4 iterations and a threshold-based geometry mask indicating major veins and high-susceptibility deep grey matter nuclei was used to reduce the streaking artifacts on the TKD-QSM. 46 The final iterated QSM data improves the accuracy of the susceptibility for major veins, such as the ICVs. 46
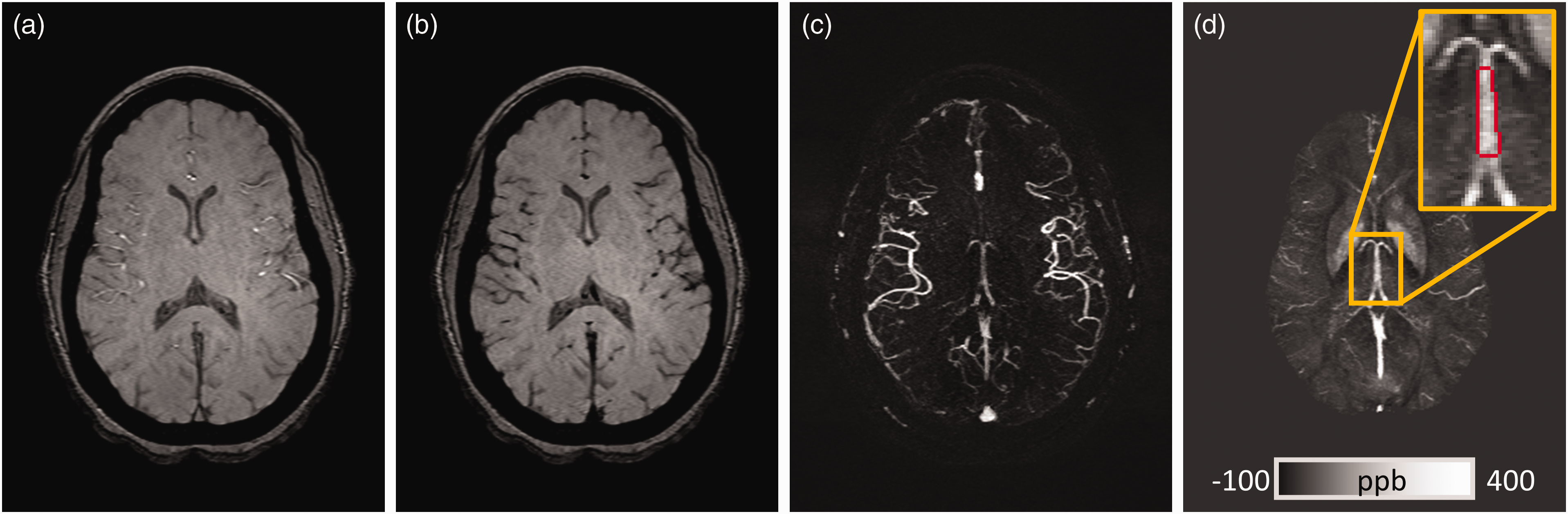
Representative images and regions-of-interest (ROI) for the internal cerebral veins (ICVs). The sequence provides a fully flow-compensated bright blood image (a) and a flow-dephased dark blood image (b). The subtraction of the two magnitude images gives MRAV data (c). The phase of the flow-compensated echo was used for reconstructing the QSM data (d). The ROI of the ICVs is shown as the red boundary in the inset (d).
Data analysis
Two observers independently traced the ICVs using a semi-automated region-growing tool in SPIN software (V1.5, MR Innovations Inc, Detroit, MI) on a maximum intensity projection (MIP) over 8 slices of the QSM data (Figure 1(d)). To investigate the inter-rater and intra-rater reliability, each observer delineated the region-of-interest (ROI) of the ICVs again on all data a week after the initial delineation. To minimize the partial volume effect (PVE), the ROI was placed at the middle segment of the ICVs between the thalamostriate veins and medial atrial veins (Figure 1(d)). Patient disease status was blinded to the observers during ROI delineation. The median susceptibility value of all pixels within the ROI (ΔχICV) was used for each subject. Subsequently, the χICV was converted to SvO2 using equation (1).
Statistical analysis was performed using MATLAB (R2017b, MathWorks, Natick, MA) with in-house developed scripts. Data distribution was tested for normality using the Shapiro-Wilk test. Except for the EDSS scores of the MS group, all other data including ΔχICV, SvO2, and PASAT were in normal distribution. Given the small sample size, we used the non-parametric Wilcoxon rank sum test to compare the age, gender percentage, ΔχICV, and SvO2 between the two cohorts without including covariates. Inter-rater reliability of ΔχICV was assessed by the intraclass correlation coefficient (ICC) analysis 49 and linear regression. Pearson’s correlation was used to correlate ΔχICV with PASAT and EDSS scores in MS patients without including covariates. P < 0.05 was considered statistically significant.
Results
The study enrolled 20 patients with RRMS and 10 age-matched HCs. Nineteen out of the twenty patients are on various disease modifying medications as reported in a previous publication on the same patient population. 40 Two MS patients’ data were excluded from the present study due to strong susceptibility artifacts caused by calcification in the pineal gland (around which the ICVs bifurcate from the great cerebral vein). The demographics and clinical assessments are listed in Table 1. The age and gender percentage were comparable between MS and HC. The reliability analysis demonstrated an excellent intra-rater (ICC = 0.97, r2 = 0.97, p < 0.001) and inter-rater (ICC = 0.96, r2 = 0.97, p < 0.001) agreement of the measuring method. Compared with HCs, patients with MS had a significantly reduced ΔχICV (212.4 ± 30.8 ppb in MS vs 239.4 ± 25.9 ppb in HC), and a significantly increased SvO2 (74.5 ± 1.89% in MS vs 72.4 ± 2.23% in HC), as shown in Figure 2(a) and (b). The PASAT score, a measure of cognitive function, was moderately correlated with the ΔχICV (r = 0.508, p = 0.031) and the SvO2 (r = −0.498, p = 0.036) in MS patients (Figure 2(c) and (d)). In contrast, the disease severity score, EDSS, had no correlation effect with ΔχICV (r = −0.295, p = 0.236), nor SvO2 (r = 0.279, p = 0.260) in MS patients.
Demographics and data.
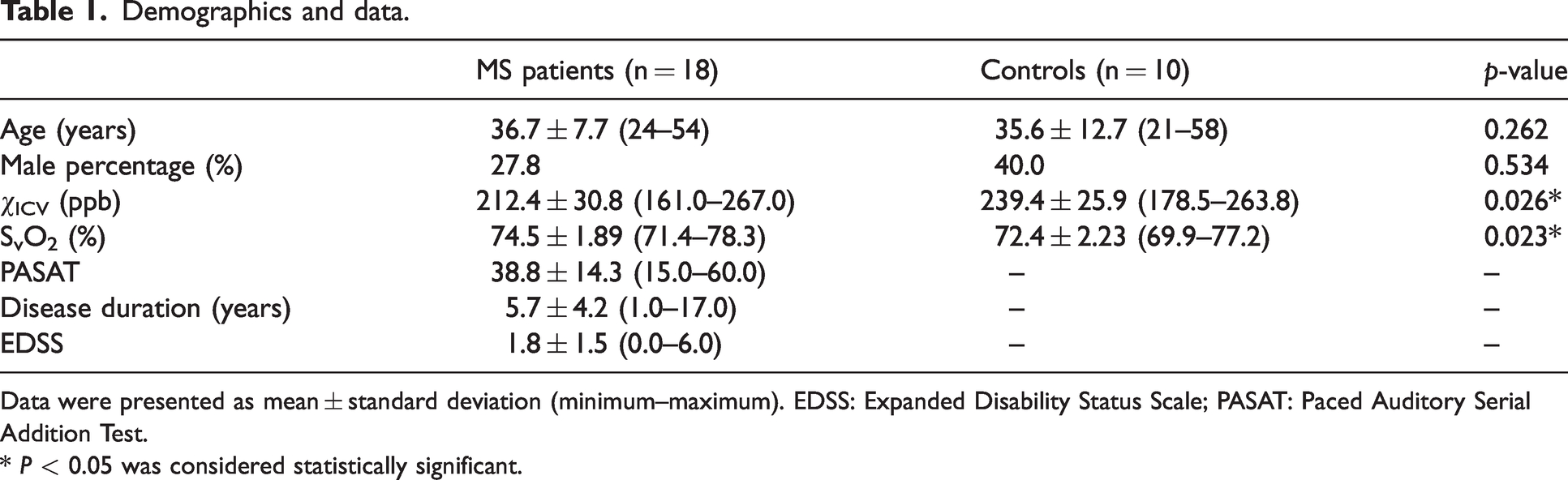
Data were presented as mean ± standard deviation (minimum–maximum). EDSS: Expanded Disability Status Scale; PASAT: Paced Auditory Serial Addition Test.
* P < 0.05 was considered statistically significant.
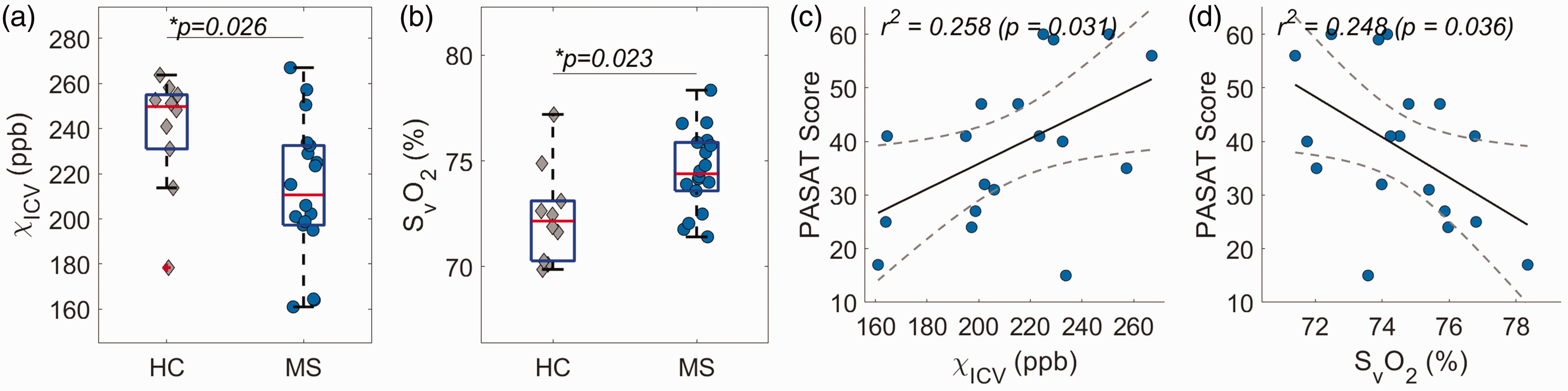
Increased deep cerebral venous blood oxygenation was correlated with cognitive decline. The box plot analysis shows a reduced ΔχICV (a) and increased SvO2 (b) in MS compared with healthy controls (HC). The PASAT score was correlated with ΔχICV (c) and SvO2 (D) in MS patients.
Discussion
In this study, we observed a reduced susceptibility of ICVs in RRMS patients compared to age- and sex-matched controls, indicating decreased deoxyhemoglobin levels in venous blood in ICVs. Consequently, there was an increased SvO2 in these patients, implicating a decreased OEF in the corresponding cerebral regions draining into the ICVs. These imaging biomarkers were correlated with cognitive impairment in these MS patients, as measured by PASAT. The high intra- and inter-rater ICC scores affirm the repeatability and reproducibility of the method.
Oxygen molecules bind to hemoglobin by eliminating unpaired electrons, forming diamagnetic oxyhemoglobin. Upon the release of oxygen from oxyhemoglobin in neuronal tissue, deoxyhemoglobin in venous blood becomes paramagnetic, resulting in relatively strong magnetic field perturbations in MRI. Therefore, the susceptibility of venous blood can serve as a direct measure of SvO2 given the negligible effect of other sources of susceptibility changes in the blood.
48
The phase accumulation as measured for a given echo time in a gradient echo sequence is proportional to the susceptibility changes. Indeed, phase images have been used to directly measure the SvO2 of pial veins parallel to the main field, where there is no magnetic dipole inversion.6,21,22 In this regard, QSM reconstructed from phase data makes it possible to measure venous blood oxygenation in all orientations.21,37,38 However, venous QSM based oxygen saturation measurement requires full flow compensation on all the echoes to reduce blood flow induced phase error.37,50 Uncompensated blood flow thereby becomes the major source of error in venous-QSM based OEF measurement.
23
In addition, gradient echo sequences in clinical MRI scanners only have flow compensation options for the first echo, but not for the second and subsequent echoes. To overcome these issues, we used a single-echo based QSM method along with flow compensation in all gradient directions to minimize blood flow induced phase errors.43,46 Furthermore, we used an optimal echo time of 12.5 ms, which is a fat-water in-phase echo time close to
The observed increased SvO2 in the ICVs is likely due to reduced oxygen consumption in the deep cerebral regions that drain into the ICV, which include periventricular WM and deep gray matter regions. The superior thalamostriate veins, which drain the corpus striatum and thalamus, unite with the superior choroid veins, which drain the hippocampus, fornix, and corpus callosum, to become the ICVs. 51 Damage to any of these deep gray matter nuclei are implicated in cognitive impairment in MS.11,13 –16 The association found in this study between increased SvO2 and cognitive impairment suggests that reduced oxygen consumption, possibly due to virtual hypoxia, is a factor in the damage to these regions. This further aligns with previous studies that found reduced CMRO2 in deep gray matter nuclei.5,6 In addition, reduced oxygen consumption in deep cerebral gray matter may play a key role in cognitive function in other neurodegenerative disorders such as Alzheimer’s disease. 52 WM damage is also of note, as the subependymal veins, which drain the periventricular WM regions through medullary veins, in turn drain into the ICVs. The periventricular region where MS lesions are most commonly seen may also play a role in explaining SvO2 increases in the ICVs. Previous studies have explored the role of WM and reported a contribution from WM to MS-related cognitive impairment.53,54 This is also in line with a previous study demonstrating diminished medullary vein visibility in periventricular WM territories in MS patients. 55 There was no significant correlation observed between the disease severity score (EDSS) and the SvO2 of ICVs in these patients. This is likely because most of the weight of an EDSS score is given to motor functions related to ambulation, and only one component of EDSS is cerebral function. In addition, RRMS patients enrolled in this study have a relatively mild disease severity, which further increases the uncertainty of correlation analysis. Taken together, the findings of this study emphasize the utility of deep cerebral venous susceptibility as a potential imaging biomarker of cognitive decline in RRMS as well as normal aging and other neurodegenerative disorders.
There are several limitations to this study. First, this retrospective study includes only a small cohort of RRMS patients. While RRMS patients constitute most MS patients, 56 limiting the cohort to a single MS subtype may miss an opportunity to explore varying profiles of pathology, given that the prevalence of cognitive impairment varies widely among subtypes. 57 Second, while χICV and SvO2 are good measures of venous deoxyhemoglobin and oxygenation, they fall short of being a complete stand-in for OEF as understood by Fick’s principle, which also requires a measurement of arterial blood oxygen saturation supplying the same tissue. Although arterial oxygen saturation levels can be measured by a finger pulse oximeter, 8 this data was not collected from the present cohort when they underwent MRI. Importantly, true hypoxia plays an equally prevalent role in MS, 4 and previous studies have found lowered deep gray matter blood oxygen saturation in MS using infrared spectroscopy—reasonably, oxygen saturation in the finger may vary from that in the cerebrum. 58 Further studies would benefit from measuring arterial blood oxygen saturation, ideally in the counterpart arteries to the ICVs. In addition, other sources of error in venous QSM-based OEF measurement include the assumptions of Δχdo and hematocrit values which could be subject dependent. Although it is common to use an assumption value,5,6,34 a blood draw hematocrit test at the time of the MRI could increase the reliability of the QSM based SvO2 measurement. Blood draw hematocrit data was not available for the participants in this study. Of note, the interpretation of reduced oxygen consumption by the observed OEF reduction was assuming a constant CBF in these subject as reported in a previous study. 8 Other studies59,60 have reported deep grey matter hypoperfusion in RRMS patients which could further decrease the regional CMRO2. However, the decoupling of neural and vascular deficits would need a complete measure of CMRO2 which was not available from the present study. Third, QSM reconstruction has a step of brain extraction which typically discards cerebral edge regions including most of the cortical veins and the SSS. 37 Thus, the current method is limited to measuring only from veins available in the QSM data. This could be circumvented in future studies by using whole brain QSM methods.61,62 Fourth, PASAT alone may not be the most reliable indicator of our participants’ cognitive function. Despite its high test-retest reliability and internal consistency, it is influenced by patients’ mathematical abilities, intelligence quotient, and age. 42 Finally, although we selected the central segment of ICV to minimize the PVE in the measurement, significant PVE of the ICVs was observed by the generally lower venous susceptibility compared to the theoretical value of 450 ppb. 37 The PVE could be reduced by using a higher imaging resolution. Practically speaking, acquiring a single echo with full flow compensation and a resolution of 0.5 × 0.5 × 1.5 mm³ could be achieved in less than 5 minutes on clinical MRI scanners. This could be accomplished by using a TE of 12.5 ms, a TR of 20 ms, a low bandwidth of 150 Hz/pixel to ensure the SNR of the data, a parallel imaging acceleration factor of 2, and 80 axial slices covering most of the brain. Further studies should consider the correction of PVE in QSM reconstruction. Nevertheless, the findings of this study are endorsed by the excellent agreement of the inter-rater and intra-rater analyses, and the factor that the exact same method was used on patients and controls.
In summary, this study demonstrates the feasibility of using deep cerebral venous susceptibility as an imaging biomarker of cognitive impairment in MS. This pilot study warrants further investigation of cerebral venous QSM as a clinically feasible tool in assessing cerebral oxygen metabolism in large-scale prospective clinical studies in MS and other neurodegenerative disorders.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was partially supported by the NIH/NINDS under award number R61NS119434. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. This work was also partially supported by the Office of the Vice President for Research at Wayne State University through their support of the MR Research Facility. The research data collection was supported by AbbVie Inc through grant award number C91205.
Acknowledgements
We thank Fahad Malik for his contribution to the inter-rater reliability analysis. We thank Dr. Scott Millis for his advice on statistical analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Y.C., and Y.G. designed the study. H.S., S.B., E.B., E.M.H., and Y.C. contributed to the data acquisition. H.S., C.L., and Y.C. contributed to the data anlyses, visualization, and interpretation. H.S. and Y.C. drafted the manuscript. All authors were involved in final interpretation and revising the manuscript.